Abstract
Aging involves the systemic deterioration of all known cell types in most eukaryotes. Several recently discovered compounds that extend the healthspan and lifespan of model organisms decelerate pathways that govern the aging process. Among these geroprotectors, spermidine, a natural polyamine ubiquitously found in organisms from all kingdoms, prolongs the lifespan of fungi, nematodes, insects and rodents. In mice, it also postpones the manifestation of various age-associated disorders such as cardiovascular disease and neurodegeneration. The specific features of spermidine, including its presence in common food items, make it an interesting candidate for translational aging research. Here, we review novel insights into the geroprotective mode of action of spermidine at the molecular level, as we discuss strategies for elucidating its clinical potential.
Similar content being viewed by others
Main
Life expectancy has steadily increased during the past decades, while healthspan lags behind, leading to an increased risk of age-related disease. Fasting and reducing calorie intake without malnutrition (caloric restriction (CR)) are the best-studied nongenetic interventions to promote healthspan and lifespan across species borders and are comprehensively investigated in clinical settings1,2,3,4,5. Like any harsh nutritional intervention, they are afflicted by several problems precluding their broad implementation. Few individuals succeed in complying with long-term CR or regular fasting. Also, CR is contraindicated in certain conditions such as infection or sarcopenia5. This spurred the search for pharmacological CR mimetics, which ignite (gero)protective pathways similarly to CR6,7,8,9. CR mimetics, by definition, act as antiaging agents, thus prolonging healthspan or lifespan. At the forefront, the polyamine spermidine has been characterized as a natural autophagy inducer10.
In eukaryotes, the diamine putrescine and the polyamines spermidine and spermine are essential metabolites regulating important cellular functions, including growth and proliferation, RNA and DNA stability, RNA-to-protein translation, autophagy and immune responses11. In mammalian tissues, spermidine and spermine are more abundant than putrescine12. Still, their comprehensive molecular targets remain incompletely understood. We will review how the polyamine pathway is affected by aging and how spermidine supplementation prolongs healthspan and lifespan in model organisms. In recent years, the notion has been confirmed that the antiaging effects of spermidine involve autophagy induction. However, its action may extend to other geroprotective mechanisms. Here, we will discuss its impact on each hallmark of aging13 and scrutinize the potential of spermidine for combating human diseases.
The polyamine pathway during aging
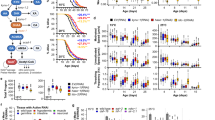
Polyamine metabolism is evolutionarily conserved among eukaryotes and deeply integrated into cellular bioenergetic pathways (Fig. 1). Experimental deletion or hereditary defects in the polyamine pathway cause severe cellular and organismal defects or compromise viability. Research into the antiaging properties of spermidine has been encouraged by reports on its age-dependent decline (Table 1). For example, spermidine concentrations decrease in chronologically aging Saccharomyces cerevisiae, a model for postmitotic aging14. Similarly, spermidine declines in the heads of middle-aged Drosophila15.
Due to strain-specific, sex-specific and tissue-specific differences and dietary preferences, the time-dependent trajectories of spermidine are more heterogeneous in mammals. In female C57BL/6 mice, Nishimura et al. measured a decrease in most tissues with few exceptions, including the brain, during the first 6 months of age16. Several studies investigated polyamines in aging rats: in 1964, a study reported reduced spermidine in several tissues during the first 9 months17, a tendency confirmed for selected tissues in longer-lasting studies18,19. Interestingly, in male rats, spermidine sharply increased in kidneys at the age of 3 years, after an initial decline20. This exemplifies the need for a systematic study of polyamines throughout the entire lifespan of mammals. One study found no effect or even a slight increase in several brain areas from Sprague-Dawley rats21, similarly to the aforementioned mouse study16. However, this contrasts with another report showing a constant decline of spermidine in cerebral cortex samples from aging rats19. Finally, circulating spermidine levels in serum from 4- and 13-month-old mice exhibited a 40% decrease when normalized to serum protein22. Given the animals’ relatively young age in several of these studies, it is important to separate developmental from actual aging effects in future studies.
Two studies reported an age-dependent decline of spermidine in human blood23,24. One of these found that healthy nonagenarians and centenarians retained levels comparable to those of middle-aged (45-year-old) participants24, but this has not yet been confirmed. Spermidine declined with age in circulating peripheral blood mononuclear cells (PBMCs)25,26 and red blood cells27. By contrast, a recent Japanese study reported an increase in whole-blood from aging humans28. Postmortem analyses of polyamines are rare, but two studies found stable levels throughout adulthood in human liver29 and cortex30 samples. Of note, polyamines were reported to rise in some aging-associated diseases, for example, renal failure31, cardiovascular disease32 and neurodegeneration33,34, potentially as an adaptive response to cellular stress.
Spermidine (but not spermine) is utilized for the posttranslational hypusination of eukaryotic initiation factor 5A (eIF5A, a translation factor mainly involved in elongation and termination, despite its name35), which stimulates autophagy25,36,37. eIF5A hypusination declines with aging in vivo38 and can be restored by dietary spermidine supplementation38,39. Mechanistically, the depletion of spermidine and reduced eIF5A hypusination probably result from reduced activity of ornithine decarboxylase (ODC1), the rate-limiting step in polyamine biosynthesis. Indeed, ODC1 activity declines in several tissues of male rats19, which has been confirmed for liver40 and heart41, while increased spermidine/spermine-N1-acetyltransferase (SAT1) activity suggested activated catabolism upon aging42. Postmortem analysis of human liver samples revealed decreased ODC1, but upregulated spermine oxidase, perhaps accounting for relatively stable spermidine levels29.
In-depth studies of preclinical and human cohorts are missing to fully comprehend the impact of aging on polyamine metabolism. Since polyamines occur in soluble and bound forms (for example, complexed to nucleic acids or ribosomes), which are usually not distinguished by the current methods, it remains elusive whether aging affects these pools differently. Moreover, most studies do not consider the circadian aspect of sampling time points, or fail to report them. Polyamines and their metabolic enzymes vary with feeding status and circadian rhythms, with the amplitude of this nychthemeron-related fluctuation reduced in aged mice22. At the same time, spermidine may modulate cellular circadian phases, as shown in a mouse fibroblast cell line43. Thus, when choosing heterogeneous sampling time points, age effects may be overlooked. Moreover, the dietary intake of polyamine and their precursors can be highly variable, especially from human diets, complicating conclusions about age-dependent changes in humans. Thus, the decline in polyamines observed in Westernized countries may be avoided in Asian regions owing to traditionally polyamine-rich diets44. Therefore, variations in dietary habits and polyamine intake should be considered in clinical trials. Finally, many studies lack the statistical power to detect meaningful biological trends in a reproducible fashion. There is no consensus on the normalization of polyamines, especially for fluid biospecimens, for which it is not clear whether absolute or relative concentrations of analytes (for example, per volume or protein content) or ratios of analytes would be preferable (Table 1). Likewise, extraction and detection methods differ across studies, rendering their interpretation and cross comparison difficult.
Thus, future studies addressing the impact of aging on polyamines should consider the aforementioned points and perform systematic meta-analyses requiring methodological standardization and statistical normalization.
The molecular basis of geroprotection and autophagy induction by spermidine
Many cellular effects caused by spermidine have been mechanistically linked to autophagy14 (Fig. 2), a homeostatic, intracellular degradation and recycling mechanism. Autophagy enhancement harbors broad health-promoting potential and constitutes the molecular basis for several geroprotective strategies5,9,45. Dysfunctional autophagy is linked to various age-related diseases46,47, and the activation of autophagy by genetic (for example, overexpression of Atg5 in mice48 or Atg1 in flies49) or pharmacological (for example, via rapamycin50,51 or spermidine14,52) means is sufficient for extending lifespan in model organisms, including flies and mice53.
Based on our current knowledge of spermidine signaling, we attempted to classify the geroprotective mechanisms elicited by spermidine into three hierarchic layers. (1) Spermidine serves as a substrate for the hypusination reaction, activating eIF5A, hence facilitating the translation and synthesis of the pro-autophagic transcription factor TFEB and mitochondrial biogenesis. Many of the downstream effects of spermidine have been convincingly linked to its capacity to stimulate eIF5A hypusination. (2) Besides downstream mediators of hypusinated eIF5A (exemplified by ATG3, mitochondrial proteins, and TFEB), several factors and pathways have been described that are directly or indirectly affected. These include acetyltransferases such as EP300, deacetylases (probably including HDACs and sirtuins), transcription factors (perhaps including FOXOs) and energy sensors (such as mTORC1 and AMPK), some of which regulate autophagy and cellular effects as described elsewhere in more detail10. (3) The cellular outcome is governed by increased autophagic flux, improved mitochondrial function and epigenetic reprogramming, accounting for the cellular, organismal and systemic antiaging effects. Note that the summarized effects have been identified in various cell types and organisms. Molecular effects that potentially contribute to autophagy induction and lifespan extension are depicted in dashed boxes with gray connectors. Ac, acetylated.
While evidence for autophagy’s causal role in spermidine-mediated geroprotection emerged from several in vitro and in vivo studies, future work using in vitro cell culture models should carefully distinguish effects caused by mechanistic targets of spermidine and exogenously produced oxidative stress. Fetal bovine serum (a standard ingredient of many cell culture media) contains an amine oxidase capable of oxidizing polyamines and producing hydrogen peroxide, which itself promotes autophagy and might thus be partly responsible for some effects in vitro54. A solution to this methodological problem is the use of the serum amine oxidase inhibitor aminoguanidine or the replacement of bovine serum by amine oxidase-free serum (for example, from human origin55). Notwithstanding this caveat of in vitro experiments, the effects observed in vivo in animal models and clinical trials should remain valid. This also applies to the crucial autophagy enhancement in spermidine-mediated geroprotection.
Spermidine and acetylation
Spermidine modifies the epigenetic landscape by reducing histone acetylation and affecting the acetylation status of many cytoplasmic proteins14,56 (Box 1). In aging yeast cells, spermidine triggers deacetylation of histone H3 by inhibiting histone acetyltransferases (HATs), thereby affecting autophagic gene transcription14. Autophagy induction by spermidine seemed independent of sirtuin 1 (SIRT1), a deacetylase, in one study57 but dependent on the inhibition of the acetyltransferase E1A binding protein P300 (EP300)56. However, two studies reported a spermidine-induced upregulation of SIRT1 and a functional SIRT1 dependency for its cellular effects42,58. Nonetheless, autophagy was not characterized in those latter studies, and spermidine-mediated lifespan extension seemed independent of SIRT1 orthologs in yeast and worms57.
EP300 acetylates and inactivates several autophagy-related genes (ATGs), including microtubule-associated proteins 1A/1B light chain 3B (known as LC3), adding further complexity to the autophagy-stimulating properties of spermidine56. Spermidine also stimulates the translocation of histone deacetylase (HDAC) 4 to the nucleus, preventing the deacetylation of cytosolic microtubule-associated protein 1S (MAP1S)52 and promoting autophagic flux59. Additional HDACs might be involved in the effects of spermidine60,61. Activated polyamine metabolism can reduce protein acetylation by depleting acetyl coenzyme A (acetyl-CoA) pools62,63,64. For instance, overexpression of the acetyl-CoA-catabolizing enzyme SAT1 is sufficient to reduce acetyl-CoA pools in vivo62,63,64, favoring protein deacetylation and autophagy56,65. Contrariwise, impaired polyamine synthesis and reduced hypusination alter the tricarboxylic acid cycle, hence increasing acetyl-CoA levels and HAT-mediated histone acetylation66. Thus, polyamine metabolism acts as an important nexus connecting cellular energy status and epigenetic modifications that govern cellular processes, including autophagy.
Interestingly, deacetylation of the transcription factor EB (TFEB), a target of eIF5A, favors its activity67, suggesting a possible link between spermidine-induced protein deacetylation and TFEB activation. Similarly, eIF5A itself is regulated by acetylation events68,69,70, with deacetylation promoting its cytosolic location and activity70. Whether EP300 is involved in this acetylation-based regulation is currently elusive. Future studies should address these unknowns to understand the cytoplasmic and epigenetic events linking high spermidine levels to the stimulation of autophagic flux. In a hypothetical scenario, spermidine rapidly stimulates autophagy through direct effects on cytoplasmic effectors but then mediates additional epigenetic, transcriptional and translational adaptations to sustain autophagic flux.
Spermidine, hypusination and autophagy: an intimate relationship
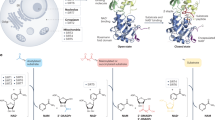
Many effects of spermidine can be traced to its use for posttranslational hypusination of eIF5A, a small, highly conserved and abundant protein containing 157 amino acids25,38,39,66,71 (Box 2). At least two isoforms of eIF5A (>80% sequence identity) have been identified in mice and humans. Both can be hypusinated and are partly tissue specific, with eIF5A2 mainly expressed in neuronal tissue. Hypusine (N6-(4-amino-2-hydroxybutyl)lysine) is a rare and unusual amino acid formed via a two-step enzymatic reaction: deoxyhypusine synthase (DHS) transfers the 4-aminobutyl moiety from spermidine to the ε-amino group of a lysine residue in eIF5A (in humans Lys50) in an NAD+-dependent reaction. DHS is thought to be eIF5A exclusive, as no other protein is known to be hypusinated. The amino acid sequence surrounding the critical lysine residue is conserved across species. Then, deoxyhypusine hydroxylase (DOHH) transforms deoxyhypusine into hypusine by adding a hydroxyl group, which activates eIF5A. The high conservation of the DHS–DOHH–eIF5A pathway reflects its critical role in cellular growth and translational regulation. In yeast, the knockouts of HYP2, the eIF5A homolog, or DYS1 (DHS) are lethal, and Eif5a-null or Dhps-null mice die during early embryogenesis. Postnatal conditional knockouts in mice compromise pancreas development72 and hematopoiesis25 and cause major neurodevelopmental problems73, underscoring the pathway’s paramount ontological importance.
Hypusinated eIF5A regulates protein synthesis at translation initiation elongation, and termination stages. It helps to overcome ribosomal stalls, for instance, at poly-proline motifs and specific non-proline tripeptide sequences (a recent review details the molecular and physiological implications of eIF5A69). Among others, it contributes to mitochondrial protein translation and, consequently, mitochondrial function66,74,75. Accordingly, blockade of polyamine synthesis and consequent hypo-hypusination increases the tolerance to hypoxia by downregulating mitochondrial complexes and remodeling the mitochondrial network, which protects tissues from ischemia-induced damage76. Blocking hypusination has also been tested in diabetes models, showing improvements in glucose tolerance69, among other mechanisms, by causing a switch from glutaminolysis to glycolysis77. This is corroborated by a recent study showing exacerbated diabetic pathophysiology in mice fed high doses of spermidine, although hypusination was not assessed in this study78. Moreover, T lymphocyte differentiation critically relies on a functional polyamine–hypusine axis66,79, which thus has an important role in shaping the immune system.
Importantly, hypusinated eIF5A facilitates the translation of certain ATGs36,38, providing a direct link between increased hypusination and autophagy stimulation. eIF5A depletion in cell lines and Caenorhabditis elegans causes the rarefaction of autophagosomes (critical membranous structures formed to transport degradable cargo to lysosomes) and limits the lipidation of LC3, a key autophagosomal adaptor protein, and marker for autophagy36,37. Similar effects, including the depletion of ATG3, can be achieved by pharmacologically blocking DHS by the spermidine-analog N1-guanyl-1,7-diaminoheptane (GC7)36. A recent study corroborated these findings in vivo by showing that spermidine increases ATG3 levels in Drosophila brains, depending on eIF5A hypusination38.
Hypusinated eIF5A is also important for TFEB translation, the master inducer of lysosomal biogenesis and autophagy, in B and T lymphocytes. TFEB contains triproline motifs (one in mice and two in humans) and is decreased in immune cells from older adults25. TFEB drives the expression of genes with a CLEAR (coordinated lysosome expression and regulation) element and acts as a major transactivator of autophagy-relevant genes. TFEB is subjected to inhibitory phosphorylation by mechanistic target of rapamycin complex 1 (mTORC1), a major negative regulator of autophagy80,81,82. Importantly, externally supplied spermidine elevates hypusination in mouse39 and Drosophila38 brains, in which aging is typically coupled to the decline of spermidine and hypusination15,38. Cultured PBMCs from older (but not young) adult human donors also show increased hypusination upon in vitro spermidine treatment, alongside increased TFEB expression and autophagic flux26. This suggests the functional incorporation of external spermidine into the hypusination–TFEB–autophagy axis both in vitro and in vivo.
The spermidine-mediated, eIF5A-driven increase of TFEB levels is probably one of the pathways explaining polyamine-mediated autophagy activation, at least in some tissues. The age-associated enfeeblement of this axis may compromise the function of immune cells, including B and T lymphocytes, some of which heavily depend on mitochondrial function25,26,66. This probably explains the rate-limiting role of the polyamine pathway in neuronal tissues (‘Human diseases and defects in the polyamine metabolism’), which is particularly energy consuming.
Interestingly, a self-amplifying loop in which spermidine enhances autophagic flux by EP300 inhibition, and autophagy favors polyamine synthesis, was recently described in kidney podocytes83. The authors identified the transcription factor MAFB as an upstream regulator of polyamine metabolism in kidney cells83. It remains to be studied whether this feedforward loop is active in other cell types. Beyond eIF5A, polyamines are known to directly modulate mRNA-to-protein translation, as exemplified by the autoregulation of polyamine enzymes that are translated less efficiently at high polyamine levels84. As far as we know, this remarkable feature of translational control85,86 has not been investigated for aging-relevant and autophagy-relevant genes. Of note, aging per se leads to decreased translation efficiency and increased ribosomal pausing, thus favoring the collapse of proteostasis87, while the age-associated decline of polyamine levels and hypusination is elusive in this context. At this point, it appears likely that polyamines affect multiple genes and proteins by intersecting transcriptional, translational or proteostatic pathways, requiring further mechanistic studies.
At the organelle level, mitochondria seem especially important for spermidine effects, as shown in cell cultures88, worms89, fruit flies38,39 and mice39,42,90,91,92. Valuable lessons have been learned about the cellular consequences of impaired polyamine biosynthesis and reduced hypusination by using pharmacological and genetic strategies to target DHS or DOHH. For instance, genetic tools to reduce hypusination in Drosophila compromise mitochondrial respiration and ATP production alongside a general decrease in mitochondria-related factors38. Data from mammalian cell cultures corroborate the essential role of this axis in ensuring optimal mitochondrial function66. Possibly, the upregulation of TFEB levels via eIF5A facilitates mitophagy, the autophagic removal of defective mitochondria, for enhanced mitochondrial quality control and optimization of cellular bioenergetics. As an alternative but nonexclusive possibility, spermidine may trigger mitochondrial biogenesis, potentially a necessary response to mitophagy. Indeed, oral spermidine increases the relative mitochondrial volume per myocyte90 and the number of mitochondria in cardiac tissues from aged mice93 and PGC-1α in aged rat hearts42. Mechanistically, this may involve the SIRT1-dependent deacetylation and activation of PGC-1α42.
Both core autophagy genes and mitophagy-specific factors are required for the positive effects of spermidine on mitochondria and lifespan in vitro88 and in flies38,39, causally implicating mitophagy. TFEB and its homolog TFE3 are important stimulators of mitochondrial degradation processes94 and metabolic flexibility95, inter alia by increasing the expression of genes involved in mitochondrial biogenesis, oxidative phosphorylation and fatty acid oxidation. Thus, TFEB-like transcription factors also contribute to the activation and promotion of mitophagy, and loss of TFEB and TFE3 results in autophagic dysfunctions96.
Other factors involved in geroprotection by spermidine
Several other mechanisms have been implicated in spermidine-induced autophagy, some of which may act independently of the abovementioned molecular cascade. Many autophagy triggers inhibit the negative regulator mTORC1 or activate the energy-sensing and autophagy-promoting AMP-activated kinase (AMPK). Spermidine caused a decrease in mTOR phosphorylation and increased AMPK activation in rat models of cardiac dysfunction in vivo and in vitro97, but this does not apply to other systems25,56,57. The importance of mTORC1 and other energy sensors might be time dependent, as suggested by a report on a transient, reversible decrease in mTORC1 activity upon spermidine supplementation in vitro79. Of note, activation of mTORC1 has recently been shown to promote polyamine synthesis in cancer cells via S-adenosylmethionine decarboxylase 1 (AMD1), which generates the N-propylamine residue required for polyamine synthesis98,99. mTORC1 also phosphorylates TFEB, inhibiting its translocation from the cytoplasm to the nucleus and preventing it from transactivating pro-autophagic genes100,101. This provides another control layer of TFEB-mediated autophagy that may be influenced by spermidine. Hence, the interactions between spermidine, mTORC1 and TFEB remain to be elucidated in different contexts, and may depend on age, cell and tissue types, as well as on diets, as recently demonstrated for mTOR signaling after spermidine injections into mice102. Future studies should compare effects on primary rather than cancer cells, which often display rewired mTORC1 signaling.
Additionally, spermidine may suppress caspase 3-mediated Beclin1 cleavage, avoiding the destruction of an essential factor for autophagy initiation, which has been reported in a model of neuronal injury103. Moreover, it elicits anti-inflammatory effects in dendritic cells by activating the transcription factor forkhead box O3 (FOXO3)104 and recently spermidine treatment was shown to ameliorate neuroinflammation in an Alzheimer’s disease mouse model105. Whether FOXOs or other factors involved in inflammation regulation intersect with autophagy activation by spermidine remains unknown.
Interestingly, amine oxidase copper-containing 1 (AOC1) is downregulated in prostate cancer and its stimulation leads to increased spermidine oxidation, reactive oxygen species (ROS) production and anticancer effects106. However, the implications of spermidine oxidation by amine oxidases in vivo remain largely unexplored.
Of note, autophagy-independent effects might additionally contribute to geroprotection by spermidine. Polyamines are potent scavengers of ROS, thus are able to prevent oxidative damage. This has been studied in various settings, including inflammatory conditions in vivo107. In fact, it remains elusive whether the anti-inflammatory action of spermidine observed in several studies is autophagy dependent10,108. In a less explored line of research, spermidine might feed back on associated biosynthetic pathways. For example, methionine can be converted into S-adenosylmethionine (SAM) and decarboxylated SAM (dcSAM), an essential co-factor required for polyamine synthesis. SAM, the sole donor of methyl groups for the methylation of nucleic acids and proteins, may connect spermidine to autophagy-promoting, but also autophagy-independent, epigenetic changes. SAM inhibits methionine-starvation-induced autophagy via mTORC1 activation109,110. Hence, increased spermidine synthesis could result in SAM consumption, thus alleviating SAM-mediated autophagy inhibition. However, whether externally supplied spermidine modulates SAM levels has not been formally established. Additionally, increased spermidine synthesis can reduce arginine bioavailability, which is used by arginase 1 (ARG1) for ornithine production. If external spermidine supply reduced the ARG1 activity, arginine would become available for its conversion into citrulline, generating nitric oxide (NO), which in turn has immunomodulatory and cardioprotective effects. Indeed, a spermidine-elicited NO augmentation has marked protective effects in models of arterial aging111. NO has important functions, including signaling, vasodilation, metabolic regulation and stimulation of mitochondrial biogenesis112, and its progressive decline might be involved in the pathogenesis of aging113. Finally, the aforementioned pathways have not been systematically studied in the context of spermidine-induced eIF5A hypusination, autophagy and geroprotection.
Since spermidine, hypusination of eIF5A, TFEB levels, autophagic capacity and mitochondrial function decline with aging, it appears plausible that their restoration via spermidine supplementation accounts for the extension of healthspan and longevity. Notwithstanding the heuristic value of the three-layer hierarchy of spermidine effects (Fig. 2), it will be important to perform careful epistatic analyses to understand whether combined interventions on specific elements of the hypothetical cascade will yield redundant effects (as this would occur if all elements act in a linear pathway) or rather may have additive antiaging effects (as this would happen if they were not arranged hierarchically). Hence, for theoretical and practical reasons, the model exposed in Fig. 2 should be subjected to further experimentation.
The effects of spermidine on the hallmarks of aging
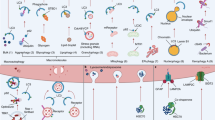
Spermidine might be considered for both the prevention and treatment of (i) diseases caused by decreased autophagy or lysosomal function, (ii) metabolic disorders, (iii) diseases of energy-demanding tissues (for example, brain and heart) and (iv) inflammatory conditions and immune defects. Instead of discussing each disease individually, we refer interested readers to extensive reviews of this field by us10 and others114. Here, we outline the effects of spermidine on the hallmarks of aging13 (Fig. 3).
For details and references see the main text. This figure was inspired by ref. 13. HFD, high-fat diet.
Genomic instability
During aging, DNA damage accumulates despite sophisticated repair mechanisms. It is largely unexplored whether polyamine supplementation impacts genomic stability, DNA repair or mutation frequency during aging. In a modified Ames test, measuring compounds’ antimutagenic properties in bacteria, putrescine and spermidine were found to protect from DNA mutations115. Likewise, polyamines were crucial for homology-directed DNA repair, at least in hair follicles with induced DNA double-strand breaks116. ROS subserve important signaling functions, but excessive levels may cause DNA or membrane damage117. Polyamines can act as ROS scavengers118,119, while their enzymatic degradation produces ROS120,121,122, suggesting a dual role in oxidative homeostasis123. Intriguingly, conditions tied to elevated polyamine levels (for example, some cancers, Snyder-Robinson syndrome (SRS)), as outlined below, experience oxidative damage. Along these premises and considering that most intracellular polyamines are bound to nucleic acids, influencing their stability and structure124, this calls for a more detailed investigation of genomic instability and polyamines.
Telomere attrition
Telomeres are damage-susceptible, protective regions at chromosome ends and have been suspected of eroding with aging. Specialized DNA polymerases, telomerases, replicate these DNA ends but are absent or insufficiently expressed in many somatic cells, causing continuous telomere shortening. Protecting telomeres via genetic means is geroprotective in vivo but may come as a double-edged sword, potentially with carcinogenic effects. Spermidine supplementation preserves telomere length in aging mice125. The exact mechanism is yet to be revealed and could be due to anti-ROS effects of polyamines and autophagy126. It remains elusive whether spermidine directly modulates telomerases.
Epigenetic alterations
Aging is accompanied by epigenetic alterations (reversible DNA and histone modifications), probably reducing the flexibility of the transcriptome. Spermidine affects the epigenetic landscape in yeast and cell culture, which could be specific for certain promoter and gene regions, upregulating the transcription of autophagy-relevant genes14. In human dermal fibroblasts, spermidine inhibits HATs but stimulates HDAC1 and SIRT1 (ref. 60). Similarly, in vascular smooth muscle cells, spermidine upregulates SIRT1, which mediates the inhibition of calcification58. Spermidine also promotes the nuclear translocation of HDAC4 in cell lines and mice, preventing the deacetylation of cytosolic MAP1S, thereby promoting autophagy52. Conversely, in rat brains, spermidine was shown to promote phosphorylation and acetylation of non-histone chromosomal proteins127, suggesting a more complex interplay of polyamines and acetylation than previously thought. Of note, (de)acetylation of polyamines and proteins might be executed by the same enzymes. For instance, HDAC10, an autophagy promoter128, deacetylates N8-acetylspermidine for conversion to spermidine61. A concentration-dependent bimodal model has been suggested to explain how, in Drosophila, low spermidine concentrations promote, while higher concentrations reduce, HAT activity129. Whether such a model may be applied to mammalian systems remains an open question.
Loss of proteostasis
Protein quality-control mechanisms include degradation and recycling pathways, for example, autophagy or the proteasome, and mechanisms to ensure proper folding and stabilization of proteins. Once proteostasis is out of balance, especially during aging, molecular protein errors, including accumulation of unfolded or misfolded peptides/proteins, inclusion bodies and dysfunctional protein complexes, lead to cellular and organismal perturbations. Improving proteostasis harbors great potential to ameliorate age-associated deteriorations and prolong lifespan and healthspan. Accordingly, autophagy is required for the lifespan-prolonging effects of spermidine14,15,52. Likewise, in cardiac tissues from mice, properly functioning autophagy is needed for cardioprotection by spermidine90. It can therefore be assumed that spermidine improves aging-associated proteostasis. However, this conjecture should be experimentally verified.
Deregulated nutrient sensing
Nutrient and energy states must be properly sensed for cellular and organismal functions. Aging leads to a disbalance in master regulators of nutrient sensing, including insulin-like growth factor 1 (IGF-1), mTORC1, AMPK and NAD+-dependent sirtuins. Inhibiting the trophic insulin/IGF-1 pathway and mTORC1 or stimulating low-energy-sensing AMPK and sirtuins elicits antiaging effects. Polyamines are abundantly present in pancreatic tissue of mice and rats130, and putrescine and spermidine regulate insulin synthesis and release in pancreatic islets131,132. Conversely, in mice fed a high-fat diet but not in controls, spermidine reversed hyperinsulinemia and insulin resistance, thus improving insulin signaling in the liver and white adipose tissue133. In a mouse model of streptozotocin-induced type 1 diabetes, spermidine attenuated impaired testicular function134. Conversely, high doses of spermidine (10 mM via drinking water) aggravated the diabetic phenotype of nonobese diabetic mice78, highlighting the possibility that, in specific settings, spermidine may mediate toxic effects. The dose dependency of such effects has not yet been investigated. Interestingly, lifespan extension of a long-lived Drosophila strain with reduced insulin signaling was mediated by elevated spermidine levels135, suggesting an interconnection between insulin signaling and polyamines during aging.
Moreover, polyamines regulate IGF–PI3K–Akt signaling in tumor-bearing mice136, and spermidine suppresses Akt phosphorylation in skeletal muscle137, suggesting another link between polyamines and nutrient signaling relevant to aging. Conflicting effects of in vitro spermidine treatment on mTOR activity were found, but most evidence points to mTORC1 inhibition56,57,138,139, especially in the short term98. In mouse cardiomyocytes subjected to ischemia–reperfusion injury, spermidine reduced mTOR activity and activated AMPK97. The latter was also shown in a mouse model of neurodegeneration140. Because mTORC1 activation promotes polyamine synthesis in cancer cells via AMD1 (refs. 98, 99), there may be bidirectional links between nutrient sensing and polyamine metabolism. However, the relationship between mTOR and the polyamine pathway is insufficiently understood in non-neoplastic cells.
Mitochondrial dysfunction
Mitochondrial integrity and function are compromised during aging. Mitochondrial dysfunction can accelerate aging, even independently from ROS levels141, by reducing cellular energy supply, engaging in inflammatory processes and igniting cell death pathways. Conversely, spermidine upregulates mitophagic subroutines39,89,90,142 and improves mitochondrial function in cell culture, flies and mice, partly depending on hypusination, autophagy and mitophagy38,39,42,88,90,142. While these effects depend on mitophagic turnover and hence mitochondrial quality control (which eliminates dysfunctional organelles), the polyamine pathway also stimulates the biogenesis of mitochondrial proteins66. Thus, in the fungus Ganoderma lucidum, the abundance of mitochondrial proteins is reduced upon genetic inhibition of spermidine synthesis or hypusination143.
Cellular senescence
The state of cellular senescence is characterized by an arrest of the cell cycle, along with molecular alterations, including a specific secretome enriched in pro-inflammatory factors. One study showed that spermidine could suppress the induction of senescence markers by bleomycin in lung cells in vitro and in vivo138. Accordingly, in female germline stem cells, spermidine reduced tumor protein 53 (TP53) levels and cyclin-dependent kinase inhibitor 2A (p16/CDKN2A)139, markers of cellular senescence144. Yet another study showed a reduction of senescence markers in a cellular interleukin-1β-induced senescence model and a mouse model of intervertebral disk degeneration145. However, how spermidine treatment affects senescent cells in other tissues and, importantly, during physiological aging, remains elusive.
Stem cell exhaustion
During aging, the regenerative potential of tissues declines, including most stem cell compartments. Restoring stem cell function in aged organisms harbors the potential for reducing tissue damage and inflammation. In female germline stem cells, spermidine induced autophagy and reduced induced cellular senescence139. Similarly, spermidine-induced autophagy restored the regenerative function of muscle stem cells146 and increased myogenesis in vivo147, suggesting its utility for reversing sarcopenia. Spermidine seems essential for hair growth and stem cell function of hair follicles in vitro and in vivo148,149. Furthermore, high levels of AMD1 are required for embryonic stem cell pluripotency150 and spermidine mediated the reprogramming of mouse embryonic fibroblast somatic cells to induced pluripotent stem cells151. Nonetheless, its potential to prolong stem cell function in different compartments in vivo in wild-type aging organisms has not been addressed.
Altered intercellular communication
Age-associated intercellular communication perturbations comprise disturbed nutrient sensing (‘Deregulated nutrient sensing’), hormonal regulation alterations, chronic inflammatory reactions and reduced immunosurveillance. Little is known about how spermidine affects these hallmarks. Spermidine has anti-inflammatory properties in several mouse models10,104,152. In ex vivo experiments, spermidine treatment improved the function of immune cells from aged human donors26 and reduced viral propagation of severe acute respiratory syndrome coronavirus 2 in human lung cells153. Spermidine, polyamine metabolism and eIF5A are critical factors for T cell development and differentiation26,79,154. In mice, spermidine increased anticancer immunosurveillance through effects on both cancer and T cells155,156. Conversely, polyamine-lowering approaches are being investigated in immune-invasive cancer types that rely on high polyamine levels157. Combining polyamine transport and synthesis inhibitors reverses local immunosuppression in mouse tumors and increases T cell-mediated anticancer immune responses158,159, a notion that is currently put forward in the first clinical trials (NCT03536728). In sum, spermidine may affect the cancer-immunity dialogue in a context-dependent fashion.
Interest in the microbiome as both a disease contributor and a therapeutic target has recently risen160 and microbially produced polyamines are absorbed from the gut lumen, contributing to the systemic pool161. In mouse models of diet-induced obesity, spermidine feeding improved insulin resistance133,162, and one study concluded that this could be due to gut microbiota changes133. Furthermore, oligofructose treatment-associated changes in microbiota, leading to increased spermidine production, were found to be involved in reduced bacterial infection163. In a mouse model of abdominal aortic aneurysm, spermidine feeding improved intestinal dysbiosis164 and inflammation108. Altogether this indicates an interdependent relationship between microbiota-produced polyamines, microbial composition and systemic health.
Spermidine-mediated lifespan extension
For potential geroprotective compounds or interventions, measurable increases in lifespan or healthspan are crucial benchmarks. Here, we summarize the effects of spermidine supplementation on lifespan in different model organisms (Table 2).
Unicellular and invertebrate models
The unicellular model organism yeast was at the forefront of basic biology achievements, including autophagy165,166,167. In 2009, we found that spermidine supplementation reversed its cellular decline, induced autophagy and prolonged survival in an ATG7-dependent manner14. This principle was conserved for C. elegans, Drosophila melanogaster and human PBMCs14. Consistent with the notion that autophagy induction by pharmacological or genetic means can extend lifespan168, this places enhanced proteostasis at the mechanistic core of spermidine-mediated geroprotection. Several studies corroborated the lifespan extension in different fly strains, showing additional mechanistic dependencies on glycine N‐methyltransferase135 and DHS38. Glycine N‐methyltransferase is an important enzyme in the methionine cycle, providing the precursor SAM for polyamine biosynthesis. This suggests a possible link between methionine metabolism and increased lifespan by spermidine. As outlined before, spermidine-mediated longevity in flies depends on the hypusination pathway38. In a fly model of Parkinson’s disease, spermidine increased survival169, suggesting potential effects against neurodegeneration. Initial reports of spermidine-mediated longevity in worms14,57 were recently corroborated by data showing that the shortened lifespan due to B12 deficiency could be partially reversed by spermidine170.
Lifespan extension in mammals
Lifelong spermidine supplementation via drinking water is sufficient to prolong the lifespan of adult female and male mice90. Similar effects were observed for spermine but not for putrescine90. Spermidine also sufficed to prolong the lifespan of pre-aged 18-month-old mice90 and short-lived male mice with progeria (Zmpste24−/− mice)171. Likewise, daily injections late in life caused a nonsignificant lifespan extension in mice102 and polyamine-enriched food prolonged the lifespan of aging mice172, suggesting that the mode of administration is of little importance for the longevity effects. Interestingly, the upregulation of intestinal polyamine production in 14-month-old mice by feeding the prebiotic arginine combined with the probiotic bifidobacteria LKM512 (equipped with the enzymatic machinery to convert arginine into spermidine) is sufficient to prolong lifespan and reduce the incidence of age-related diseases173,174. While the cardioprotective effects of spermidine could mechanistically be linked to autophagy90, only one mechanistic lifespan study has been conducted in mammalian models. In the study by Yue et al., lifespan extension and autophagy induction by spermidine feeding were abolished in autophagy-deficient male Map1s-knockout mice52. Fewer studies have been carried out on rats: in salt-sensitive Dahl rats, spermidine exerts systemic cardioprotective effects90. One study found no impact on life expectancy but a significant healthspan-extending effect in middle-aged male Sprague-Dawley rats175.
Several recent studies revealed protective effects of spermidine against high-fat or high-calorie diets, including reduced body weight gain or obesity, improved insulin/IGF-1 signaling and delayed metabolic syndrome102,133,152,162,176. It will be interesting to test whether these benefits also translate to lifespan extension on such diets.
As a noteworthy control, in several mouse studies, orally or parenterally administered spermidine did not change food or water intake39,90,102,108, excluding reduced caloric intake as a trivial reason for lifespan extension. Importantly, despite its tightly regulated metabolism, in mice, exogenous supplementation increases the levels of spermidine in plasma90,108 and cardiac tissue90. Moreover, dietary spermidine passes the blood–brain barrier and integrates into the polyamine pools of neuronal tissues39. A one-year-long clinical study detected increased spermine but constant spermidine plasma levels upon feeding polyamine-rich natto to healthy male volunteers28. Data on the impact of spermidine supplementation on tissue levels and cellular polyamine metabolism are still scarce. A systematic investigation of these unknowns is warranted. Capturing aging-specific, sex-specific and organ-specific effects upon spermidine administration probably requires high spatiotemporal resolution and flux measurements, as steady-state quantifications might fail to detect changes in metabolic dynamics.
To date, there is a lack of systematic research to determine the optimal dose, required onset (whether there is a point of no return?) and timing (continuous or intermittent) of spermidine treatments. Moreover, there is insufficient information on possible gender-specific effects and mosaic aging. Does spermidine equally decelerate all hallmarks of aging in all organs, or does it preferentially affect specific features and tissues? And finally, would spermidine improve the quality of life and delay aging in nonhuman primates similar to rodents?
Spermidine in clinical trials and human diseases
Geroprotective compounds have been identified en masse in recent years177, but the translation to human trials has been slow. Given its natural occurrence, spermidine seems an ideal candidate for clinical evaluation. The consideration that mammals probably have adapted to varying concentrations of dietary polyamine intake supports this idea. Of note, Janssens and Houtkooper compared the likelihood of side effects for putative geroprotectors by querying databases. They concluded that spermidine possessed the lowest chance of causing adverse effects178, which is in line with our research regarding the safety of low-level spermidine supplementation179 and suggestions by others180. Accordingly, Partridge and colleagues ranked spermidine among the most promising interventions that may slow aging177. Here, we discuss the current state of clinical and epidemiological research and disorders in polyamine metabolism (Box 3).
Spermidine in epidemiological studies
In humans, no interventional studies of polyamines on age-associated parameters or biomarkers (for example, epigenetic clocks or frailty-based aging estimates) have been reported. However, correlative studies suggest positive associations between spermidine intake and life expectancy in Asian countries181. Moreover, dietary spermidine is associated with reduced mortality in two non-interventional European cohorts182, and lower incidence of cardiovascular disease90,183, cancer182 and cognitive impairment39, withstanding potentially confounding factors. Spermidine intake is subjected to large variations and estimated as 5–15 mg per day6,161,184,185,186,187,188,189. The highest spermidine intake had a lower estimated mortality risk corresponding to a 5.7-year younger age182. Interestingly, one study found spermidine and spermine in healthy nonagenarians and centenarians at comparable levels to the young- to middle-aged group (31–56 years), while the aged group (60–80 years) had significantly decreased levels24. Hence, ‘successful aging’ might be linked to conserving high polyamine levels. This conjecture is supported by positive correlations between dietary spermidine intake and hippocampal volume and cortical thickness, which decline with old age190.
However, the interpretation of epidemiological data requires care and caution, as underlying problems may blur these associations. For instance, food polyamine content varies notably between databases6,44,184,191 and the impact of seasons, food storage and preparation techniques on polyamine levels is insufficiently understood44. Dietary assessments via questionnaires are afflicted by intrinsic problems, including subjectivity and compliance. Despite these limitations, it appears reassuring that the epidemiological correlations are endorsed by preclinical experimentation.
Randomized clinical trials using spermidine
The expanding preclinical literature on spermidine has spurred the design of clinical trials. In a 3-month pilot trial, older adults were supplemented with a wheat germ extract containing concentrated polyamines, leading to subtle amelioration of memory performance192. The same extract was tested for safety in aged persons and no safety issues were detected179. Based on these results, a larger 12-month trial193 was conducted on 100 participants aged 60–90 years with subjective cognitive decline. In this trial, the wheat germ extract failed to produce effects on memory function or other secondary outcomes, while there was no detectable cognitive decline in the placebo group during the study duration. However, subgroup analyses focusing on participants with high compliance suggested beneficial effects on selected memory parameters and soluble intercellular adhesion molecule 1 levels194, a marker for vascular inflammation and injury195. Another small-scale study used wheat-germ-containing bread rolls to elevate daily spermidine intake by 3.3 mg in older adults in Austrian nursing homes. This study reported mild improvements in cognitive function in a subgroup affected by mild dementia196. In line with accumulating preclinical data15,38,39,197,198, this implies that spermidine might be useful for preventing or treating age-associated cognitive decline.
To the best of our knowledge, the longest interventional study was reported by Soda et al.28: A one-year-long polyamine supplementation using natto, a polyamine-rich Japanese dish based on fermented soybeans (daily increases of 14.5 mg spermidine and 4.5 mg spermine) elicited decreased lymphocyte function-associated antigen 1 levels, suggesting anti-inflammatory effects. Another strategy for increasing polyamine availability consists in administering yogurt containing the precursor arginine and Bifidobacterium animalis subsp. lactis, which generates spermidine in the intestinal tract. This synbiotic combination of prebiotic and probiotic caused increased putrescine and spermidine levels in blood, decreased heart rates, improved endothelial function and improvements in the blood lipid profile199,200. Moreover, spermidine supplementation reportedly prolongs the anagen phase of hair follicles in a randomized, placebo-controlled, double-blind trial201, confirming similar results from preclinical studies148,202.
Given the dependency of many cancers on polyamines, concerns have been raised that elevated dietary spermidine might favor oncogenesis203,204. However, some epidemiological studies suggest a decreased cancer risk in people with naturally elevated spermidine intake182,205. Preclinical experiments show that oral spermidine administration reduces the risk of inflammation-induced colon carcinogenesis and other cancers156,206. Nevertheless, caution regarding increased polyamine intake should prevail in individuals with existing neoplastic diseases because spermidine reportedly has an ambiguous impact on cancer: under some circumstances, spermidine might reduce oncogenesis (possibly via the induction of oncosuppressive autophagy and the stimulation of cancer immunosurveillance). On the contrary, decreasing polyamine synthesis in neoplastic cells harbors the potential for halting tumorous growth and rendering cancer cells vulnerable to immune effectors157,207. Hence, both polyamine-lowering therapies and spermidine supplementation could be considered for the prevention and treatment of cancer, calling for a more profound understanding before oncological clinical trials investigating spermidine supplementation are launched.
Several clinical trials (3–6 mg spermidine per day) are ongoing to improve hypertension in combination with first-line antihypertensive drugs (NCT04405388) or to explore effects on depression and sleep quality (NCT04823806). One study combines spermidine with exercise (along with shiitake powder, vitamins C, B1 and zinc) to treat aged patients with heart failure with preserved ejection fraction (NCT05128331). Another small study plans to explore escalating doses of single postprandial spermidine supplementations (5, 10 and 15 mg), in combination with three different compounds, on inflammation (NCT05017428).
Human diseases and defects in the polyamine metabolism
Genetically determined polyamine pathway defects account for several human diseases. Faundes et al. noted that heterozygous EIF5A variants causing translational defects explain syndromes including developmental problems, cognitive disabilities, facial dysmorphisms and congenital microcephaly208. In an eIF5A-knockdown zebrafish model recapitulating the human phenotypes, spermidine attenuated the severity of the pathology, suggesting a potential therapeutic avenue208. A genetic defect in spermine synthase leading to increased cellular spermidine and an enhanced spermidine/spermine ratio has been identified as the cause of the SRS209,210. This X-linked, recessive mutation causes mental retardation and facial asymmetry, among other problems including osteoporosis and muscle weakness211. Surprisingly, SRS is characterized by lysosomal and mitochondrial damage, as well as by exacerbated oxidative stress and decreased autophagic flux212. The provision of stable spermine analogs rebalancing the polyamine system increases spermidine catabolism and ameliorates these disbalances, constituting a viable treatment option for SRS213. The exact mechanistic relationship between excessive spermidine and SRS pathogenesis remains enigmatic. Neuronal cells are especially susceptible to defects in spermine synthase, perhaps due to the importance of spermidine-mediated hypusination for eIF5A function in mRNA-to-protein translation. Mutations in other translation factors (for example, EF2 and EF1a2) have also been linked to neurological, neurodegenerative and neurodevelopmental disorders214.
Rare heterozygous gain-of-function mutations of ODC1 cause the Bachmann-Bupp syndrome, leading to accumulating putrescine levels, developmental delay and macrocephaly (in contrast to loss-of-function mutations that cause microcephaly)215,216,217,218. Repurposing the ODC1 inhibitor difluoromethylornithine (DFMO) has been suggested as a potential therapy219, which was originally investigated as a putative treatment for cancers characterized by high polyamines, although with limited effects.
Disbalances in polyamine levels might be involved in a series of human diseases and have been suggested as potential biomarkers220,221. Amin et al. summarized various conditions linked to dysregulated polyamine levels222. Elevated polyamine levels and increased biosynthetic capacities have been identified in human cancers and were investigated as potential drug targets and biomarkers98,222,223. Thus far, despite numerous preclinical studies, polyamine-targeted agents have not been successful as anticancer therapies. Furthermore, deregulated polyamines have been intensively investigated in neurodegenerative diseases, such as Alzheimer’s224 and Parkinson’s225. A recent study suggested that higher serum spermidine in patients with mild cognitive impairment could be predictive of developing Alzheimer’s disease226. This, however, stands at odds with another study showing the opposite association227. A comprehensive review of altered polyamine metabolism in Alzheimer’s disease has recently been published228.
Interestingly, despite accumulating data on the cardioprotective effects of spermidine in preclinical models and the positive association of dietary intake with reduced cardiovascular risk factors, a recent Chinese study reported higher serum levels to be associated with increased stroke risk229. Conversely, serum spermidine levels were positively related to good prognosis of patients with myocardial infarction230. This interpretation is supported by a preclinical study showing that exogenous spermidine protects rats against acute myocardial infarctions231. In sum, the effects of blood spermidine levels on the human cardiovascular system are still controversial.
Despite many studies investigating polyamines as biomarkers, technical limitations, small-scale trials and nonsystematic evaluation of the pathway have hindered a comprehensive understanding of polyamines in diseases. By the nature of such biomarker studies, an increase or disbalance of polyamine levels in a disease state does not necessarily mean a causal involvement in that condition and instead could reflect a homeostatic stress response. Moreover, to understand pathologic alterations in the polyamine pathway, flux studies should be performed.
Conclusion
The intricate relationship between polyamines and human disorders implies a delicate balance of the polyamine system to maintain homeostasis. As can be inferred from the pathogenic effect of gain-of-function ODC1 mutations, the accumulation of polyamines in SRS and the reliance of many cancers on elevated polyamines may mediate toxic or pro-tumorigenic effects, calling for caution in dose-escalation studies. The same degree of caution needs to be applied for long-term spermidine supplementation. Its clinical evaluation must go hand in hand with rigorous safety evaluations. The variety of diseases linked to disturbed polyamine levels warrants further research considering state-of-the-art techniques232 to quantify polyamines, their precursors and degradation products reliably. A systematic assessment of polyamines as biomarkers in the aforementioned medical conditions, in combination with metabolic flux analysis of the pathway, may help to distinguish physiological and pathophysiological changes in aging and diseases in future studies and could provide a better understanding of pathogenesis and disease progression. In this context, disbalances in the polyamine pathway that contribute to normal or progeroid aging should be investigated.
Outlook
Spermidine emerges as a bona fide geroprotector that naturally occurs in food and is vital for cellular and organismal function through a highly conserved pathway. Seven lines of evidence support its antiaging properties: (i) spermidine levels decrease in most, but not all, investigated tissues and species during aging. (ii) Spermidine supplementation elongates the healthspan and lifespan of multiple species. (iii) Epidemiological data support the notion that elevated dietary intake alleviates, delays or halts age-associated deteriorations, including cancer, cardiovascular disease and cognitive dysfunction. (iv) Metabolic processes within and adjacent to the polyamine pathway are well understood, pharmacologically targetable and highly conserved across species, allowing the informed use of models and the development of targeted interventions. (v) Spermidine affects most of the hallmarks of aging. (vi) Spermidine seems safe for humans. (vii) The bioavailability of spermidine seems reasonably good in mammals.
Due to its intricate interactions with cellular metabolism, spermidine probably exhibits multiple modes of action and molecular targets that may vary in a cell-type-specific, organ-specific and context-specific manner. This may be advantageous for challenging aging as a multifactorial process, but the lack of one unique mode of action may constitute an obstacle to the targeted pharmacological use of the compound. Alternatively, the synthetic design of polyamine-related compounds could overcome some limitations that are intrinsically present for these bioactive body-endogenous substances. Recently, this concept was successfully tested: the polyamine-related compound 1,8-diaminooctane mimicked or even exceeded spermidine-mediated geroprotection in worms. This compound also activated mitophagy and conferred cytoprotection to human cell lines233. Important questions remain to be answered for the future clinical application of spermidine. These concern its uptake by human tissues, the monitoring of autophagy or specific signaling pathways as pharmacodynamic markers in humans, optimal dosing and scheduling, and the effects of externally supplied spermidine on intestinal, cellular and systemic polyamine metabolism. We hope that further research into these questions will improve the design of future registration trials that evaluate the antiaging properties of spermidine.
References
Fontana, L. The scientific basis of caloric restriction leading to longer life. Curr. Opin. Gastroenterol. 25, 144–150 (2009).
Lee, C. & Longo, V. Dietary restriction with and without caloric restriction for healthy aging. F1000Res 5, F1000 (2016).
Green, C. L., Lamming, D. W. & Fontana, L. Molecular mechanisms of dietary restriction promoting health and longevity. Nat. Rev. Mol. Cell. Biol. https://doi.org/10.1038/s41580-021-00411-4 (2021).
Longo, V. D., Di Tano, M., Mattson, M. P. & Guidi, N. Intermittent and periodic fasting, longevity and disease. Nat. Aging 1, 47–59 (2021).
Hofer, S. J., Carmona-Gutierrez, D., Mueller, M. I. & Madeo, F. The ups and downs of caloric restriction and fasting: from molecular effects to clinical application. EMBO Mol. Med. https://doi.org/10.15252/emmm.202114418 (2021).
Hofer, S. J., Davinelli, S., Bergmann, M., Scapagnini, G. & Madeo, F. Caloric restriction mimetics in nutrition and clinical trials. Front. Nutr. 8, 717343 (2021).
Ingram, D. K. & Roth, G. S. Glycolytic inhibition as a strategy for developing calorie restriction mimetics. Exp. Gerontol. 46, 148–154 (2011).
Ingram, D. K. et al. Calorie restriction mimetics: an emerging research field. Aging Cell 5, 97–108 (2006).
Madeo, F., Carmona-Gutierrez, D., Hofer, S. J. & Kroemer, G. Caloric restriction mimetics against age-associated disease: targets, mechanisms, and therapeutic potential. Cell Metab. 29, 592–610 (2019).
Madeo, F., Eisenberg, T., Pietrocola, F. & Kroemer, G. Spermidine in health and disease. Science 359, eaan2788 (2018).
Pegg, A. E. Mammalian polyamine metabolism and function. IUBMB Life 61, 880–894 (2009).
Teixeira, D., Santaolaria, M. L., Meneu, V. & Alonso, E. Dietary arginine slightly and variably affects tissue polyamine levels in male swiss albino mice. J. Nutr. 132, 3715–3720 (2002).
López-Otín, C., Blasco, M. A., Partridge, L., Serrano, M. & Kroemer, G. The hallmarks of aging. Cell 153, 1194–1217 (2013).
Eisenberg, T. et al. Induction of autophagy by spermidine promotes longevity. Nat. Cell Biol. 11, 1305–1314 (2009).
Gupta, V. K. et al. Restoring polyamines protects from age-induced memory impairment in an autophagy-dependent manner. Nat. Neurosci. 16, 1453–1460 (2013).
Nishimura, K., Shiina, R., Kashiwagi, K. & Igarashi, K. Decrease in polyamines with aging and their ingestion from food and drink. J. Biochem. 139, 81–90 (2006).
Jänne, J., Raina, A. & Siimes, M. Spermidine and spermine in rat tissues at different ages. Acta Physiol. Scand. 62, 352–358 (1964).
Ferioli, M. E. & Comolli, R. Changes of liver and kidney polyamine levels during ageing. Exp. Gerontol. 10, 13–15 (1975).
Das, R. & Kanungo, M. S. Activity and modulation of ornithine decarboxylase and concentrations of polyamines in various tissues of rats as a function of age. Exp. Gerontol. 17, 95–103 (1982).
Ferioli, M. E., Sessa, A., Tunici, P., Pinotti, O. & Perin, A. Aging and polyamine acetylation in rat kidney. Biochim. Biophys. Acta 1317, 15–18 (1996).
Liu, P., Gupta, N., Jing, Y. & Zhang, H. Age-related changes in polyamines in memory-associated brain structures in rats. Neuroscience 155, 789–796 (2008).
Zwighaft, Z. et al. Circadian clock control by polyamine levels through a mechanism that declines with age. Cell Metab. 22, 874–885 (2015).
Pekar, T. et al. Spermidine in dementia: relation to age and memory performance. Wien. Klin. Wochenschr. 132, 42–46 (2020).
Pucciarelli, S. et al. Spermidine and spermine are enriched in whole blood of nona/centenarians. Rejuvenation Res. 15, 590–595 (2012).
Zhang, H. et al. Polyamines control eIF5A hypusination, TFEB translation and autophagy to reverse B cell senescence. Mol. Cell 76, 110–125 (2019).
Alsaleh, G. et al. Autophagy in T cells from aged donors is maintained by spermidine and correlates with function and vaccine responses. eLife 9, e57950 (2020).
Elworthy, P. & Hitchcock, E. Polyamine levels in red blood cells from patient groups of different sex and age. Biochim Biophys. Acta 993, 212–216 (1989).
Soda, K., Uemura, T., Sanayama, H., Igarashi, K. & Fukui, T. Polyamine-rich diet elevates blood spermine levels and inhibits pro-inflammatory status: an interventional study. Med. Sci. 9, 22 (2021).
Uemura, T., Akasaka, Y. & Ikegaya, H. Correlation of polyamines, acrolein-conjugated lysine and polyamine metabolic enzyme levels with age in human liver. Heliyon 6, e05031 (2020).
Morrison, L. D., Becker, L., Ang, L. C. & Kish, S. J. Polyamines in human brain: regional distribution and influence of aging. J. Neurochemistry 65, 636–642 (1995).
Igarashi, K. & Kashiwagi, K. Use of polyamine metabolites as markers for stroke and renal failure. in Polyamines (eds. A. E. Pegg & R. A. Casero) vol. 720, 395–408 (Humana Press, 2011).
Cheng, M.-L. et al. Metabolic disturbances identified in plasma are associated with outcomes in patients with heart failure: diagnostic and prognostic value of metabolomics. J. Am. Coll. Cardiol. 65, 1509–1520 (2015).
Pan, X. et al. Alzheimer’s disease-like pathology has transient effects on the brain and blood metabolome. Neurobiol. Aging 38, 151–163 (2016).
Graham, S. F. et al. Untargeted metabolomic analysis of human plasma indicates differentially affected polyamine and l-arginine metabolism in mild cognitive impairment subjects converting to Alzheimer’s disease. PLoS ONE 10, e0119452 (2015).
Schuller, A. P., Wu, C. C., Dever, T. E., Buskirk, A. R. & Green, R. eIF5A functions globally in translation elongation and termination. Mol. Cell 66, 194–205 (2017).
Lubas, M. et al. eIF5A is required for autophagy by mediating ATG3 translation. EMBO Rep. 19, e46072 (2018).
Frankel, L. B. EIF5A mediates autophagy via translation of ATG3. Autophagy 14, 1288–1289 (2018).
Liang, Y. et al. eIF5A hypusination, boosted by dietary spermidine, protects from premature brain aging and mitochondrial dysfunction. Cell Rep. 35, 108941 (2021).
Schroeder, S. et al. Dietary spermidine improves cognitive function. Cell Rep. 35, 108985 (2021).
Beyer, H. S., Ellefson, M., Sherman, R. & Zieve, L. Aging alters ornithine decarboxylase and decreases polyamines in regenerating rat liver but putrescine replacement has no effect. J. Lab. Clin. Med. 119, 38–47 (1992).
Wang, W. et al. Exercise training preserves ischemic preconditioning in aged rat hearts by restoring the myocardial polyamine pool. Oxid. Med. Cell. Longev. 2014, 457429 (2014).
Wang, J. et al. Spermidine alleviates cardiac aging by improving mitochondrial biogenesis and function. Aging 12, 650–671 (2020).
Yang, D., Oike, H., Furuse, M. & Yasuo, S. Spermidine resets circadian clock phase in NIH3T3 cells. Biomed. Res 42, 221–227 (2021).
Madeo, F. et al. Nutritional aspects of spermidine. Annu. Rev. Nutr. 40, 135–159 (2020).
Yin, Z., Pascual, C. & Klionsky, D. J. Autophagy: machinery and regulation. Micro. Cell 3, 588–596 (2016).
Klionsky, D. J. et al. Autophagy in major human diseases. EMBO J. 40, e108863 (2021).
Aman, Y. et al. Autophagy in healthy aging and disease. Nat. Aging 1, 634–650 (2021).
Pyo, J.-O. et al. Overexpression of Atg5 in mice activates autophagy and extends lifespan. Nat. Commun. 4, 2300 (2013).
Bjedov, I. et al. Fine-tuning autophagy maximises lifespan and is associated with changes in mitochondrial gene expression in Drosophila. PLoS Genet 16, e1009083 (2020).
Schinaman, J. M., Rana, A., Ja, W. W., Clark, R. I. & Walker, D. W. Rapamycin modulates tissue aging and lifespan independently of the gut microbiota in Drosophila. Sci. Rep. 9, 7824 (2019).
Bjedov, I. et al. Mechanisms of lifespan extension by rapamycin in the fruit fly Drosophila melanogaster. Cell Metab. 11, 35–46 (2010).
Yue, F. et al. Spermidine prolongs lifespan and prevents liver fibrosis and hepatocellular carcinoma by activating MAP1S-mediated autophagy. Cancer Res. 77, 2938–2951 (2017).
Hansen, M., Rubinsztein, D. C. & Walker, D. W. Autophagy as a promoter of longevity: insights from model organisms. Nat. Rev. Mol. Cell Biol. 19, 579–593 (2018).
Holbert, C. E. et al. Autophagy induction by exogenous polyamines is an artifact of bovine serum amine oxidase activity in culture serum. J. Biol. Chem. 295, 9061–9068 (2020).
Wang, L. et al. Oxidative degradation of polyamines by serum supplement causes cytotoxicity on cultured cells. Sci. Rep. 8, 10384 (2018).
Pietrocola, F. et al. Spermidine induces autophagy by inhibiting the acetyltransferase EP300. Cell Death Differ. 22, 509–516 (2015).
Morselli, E. et al. Spermidine and resveratrol induce autophagy by distinct pathways converging on the acetylproteome. J. Cell Biol. 192, 615–629 (2011).
Liu, X. et al. Spermidine inhibits vascular calcification in chronic kidney disease through modulation of SIRT1 signaling pathway. Aging Cell 20, e13377 (2021).
Liu, L., McKeehan, W. L., Wang, F. & Xie, R. MAP1S enhances autophagy to suppress tumorigenesis. Autophagy 8, 278–280 (2012).
Park, I.-H. & Kim, M.-M. Spermidine inhibits MMP-2 via modulation of histone acetyltransferase and histone deacetylase in HDFs. Int. J. Biol. Macromol. 51, 1003–1007 (2012).
Hai, Y., Shinsky, S. A., Porter, N. J. & Christianson, D. W. Histone deacetylase 10 structure and molecular function as a polyamine deacetylase. Nat. Commun. 8, 15368 (2017).
Jell, J. et al. Genetically altered expression of spermidine/spermine-N1-acetyltransferase affects fat metabolism in mice via Acetyl-CoA. J. Biol. Chem. 282, 8404–8413 (2007).
Kee, K. et al. Activated polyamine catabolism depletes acetyl-CoA pools and suppresses prostate tumor growth in TRAMP mice. J. Biol. Chem. 279, 40076–40083 (2004).
Pegg, A. E. Spermidine/spermine-N1-acetyltransferase: a key metabolic regulator. Am. J. Physiol. Endocrinol. Metab. 294, E995–1010 (2008).
Mariño, G. et al. Regulation of autophagy by cytosolic acetyl-coenzyme A. Mol. Cell 53, 710–725 (2014).
Puleston, D. J. et al. Polyamines and eIF5A hypusination modulate mitochondrial respiration and macrophage activation. Cell Metab. 30, 352–363 (2019).
Zhang, J. et al. Importance of TFEB acetylation in control of its transcriptional activity and lysosomal function in response to histone deacetylase inhibitors. Autophagy 14, 1043–1059 (2018).
Klier, H. et al. Isolation and structural characterization of different isoforms of the hypusine-containing protein eIF-5A from HeLa cells. Biochemistry 34, 14693–14702 (1995).
Tauc, M. et al. The eukaryotic initiation factor 5A (eIF5A1), the molecule, mechanisms and recent insights into the pathophysiological roles. Cell Biosci. 11, 219 (2021).
Ishfaq, M. et al. Acetylation regulates subcellular localization of eukaryotic translation initiation factor 5A (eIF5A). FEBS Lett. 586, 3236–3241 (2012).
Hofer, S. J. et al. Spermidine-induced hypusination preserves mitochondrial and cognitive function during aging. Autophagy 17, 2037–2039 (2021).
Padgett, L. R. et al. Deoxyhypusine synthase, an essential enzyme for hypusine biosynthesis, is required for proper exocrine pancreas development. FASEB J. 35, e21473 (2021).
Kar, R. K. et al. Neuron-specific ablation of eIF5A or deoxyhypusine synthase leads to impairments in growth, viability, neurodevelopment, and cognitive functions in mice. J. Biol. Chem. 297, 101333 (2021).
Barba-Aliaga, M. et al. Yeast translation elongation factor eIF5A expression is regulated by nutrient availability through different signalling pathways. Int. J. Mol. Sci. 22, 219 (2020).
Barba-Aliaga, M. & Alepuz, P. The activator/repressor Hap1 binds to the yeast eIF5A-encoding gene TIF51A to adapt its expression to the mitochondrial functional status. FEBS Lett. 596, 1809–1826 (2022).
Melis, N. et al. Targeting eIF5A hypusination prevents anoxic cell death through mitochondrial silencing and improves kidney transplant outcome. J. Am. Soc. Nephrol. 28, 811–822 (2017).
Cougnon, M. et al. Inhibition of eIF5A hypusination reprogrammes metabolism and glucose handling in mouse kidney. Cell Death Dis. 12, 283 (2021).
Karacay, C. et al. The effect of spermidine on autoimmunity and beta cell function in NOD mice. Sci. Rep. 12, 4502 (2022).
Carriche, G. M. et al. Regulating T cell differentiation through the polyamine spermidine. J. Allergy Clin. Immunol. 147, 335–348 (2021).
Sha, Y., Rao, L., Settembre, C., Ballabio, A. & Eissa, N. T. STUB1 regulates TFEB-induced autophagy–lysosome pathway. EMBO J. 36, 2544–2552 (2017).
Zhang, W., Li, X., Wang, S., Chen, Y. & Liu, H. Regulation of TFEB activity and its potential as a therapeutic target against kidney diseases. Cell Death Discov. 6, 32 (2020).
Zhao, E. & Czaja, M. J. TFEB: a central regulator of both the autophagosome and lysosome. Hepatol. 55, 1632–1634 (2012).
Liang, W. et al. A reciprocal regulation of spermidine and autophagy in podocytes maintains the filtration barrier. Kidney Int. 98, 1434–1448 (2020).
Dever, T. E. & Ivanov, I. P. Roles of polyamines in translation. J. Biol. Chem. 293, 18719–18729 (2018).
Igarashi, K. & Kashiwagi, K. Modulation of protein synthesis by polyamines. IUBMB Life 67, 160–169 (2015).
Mandal, S., Mandal, A., Johansson, H. E., Orjalo, A. V. & Park, M. H. Depletion of cellular polyamines, spermidine and spermine, causes a total arrest in translation and growth in mammalian cells. Proc. Natl Acad. Sci. USA 110, 2169–2174 (2013).
Stein, K. C., Morales-Polanco, F., van der Lienden, J., Rainbolt, T. K. & Frydman, J. Ageing exacerbates ribosome pausing to disrupt cotranslational proteostasis. Nature 601, 637–642 (2022).
Jing, Y.-H. et al. Spermidine ameliorates the neuronal aging by improving the mitochondrial function in vitro. Exp. Gerontol. 108, 77–86 (2018).
Yang, X. et al. Spermidine inhibits neurodegeneration and delays aging via the PINK1–PDR1-dependent mitophagy pathway in C. elegans. Aging 12, 16852–16866 (2020).
Eisenberg, T. et al. Cardioprotection and lifespan extension by the natural polyamine spermidine. Nat. Med. 22, 1428–1438 (2016).
Maglione, M. et al. Spermidine protects from age-related synaptic alterations at hippocampal mossy fiber-CA3 synapses. Sci. Rep. 9, 19616 (2019).
Chai, N. et al. Spermidine prevents heart injury in neonatal rats exposed to intrauterine hypoxia by inhibiting oxidative stress and mitochondrial fragmentation. Oxid. Med. Cell. Longev. 2019, 5406468 (2019).
Messerer, J. et al. Spermidine supplementation influences mitochondrial number and morphology in the heart of aged mice. J. Anat. https://doi.org/10.1111/joa.13618 (2021).
Sass, F. et al. TFEB deficiency attenuates mitochondrial degradation upon brown adipose tissue whitening at thermoneutrality. Mol. Metab. 47, 101173 (2021).
Mansueto, G. et al. Transcription factor EB controls metabolic flexibility during exercise. Cell Metab. 25, 182–196 (2017).
Wang, S. et al. Emerging role of transcription factor EB in mitochondrial quality control. Biomed. Pharmacother. 128, 110272 (2020).
Yan, J. et al. Spermidine-enhanced autophagic flux improves cardiac dysfunction following myocardial infarction by targeting the AMPK/mTOR signalling pathway. Br. J. Pharmacol. 176, 3126–3142 (2019).
Gomes, A. P., Schild, T. & Blenis, J. Adding polyamine metabolism to the mTORC1 toolkit in cell growth and cancer. Dev. Cell 42, 112–114 (2017).
Zabala-Letona, A. et al. mTORC1-dependent AMD1 regulation sustains polyamine metabolism in prostate cancer. Nature 547, 109–113 (2017).
Martina, J. A., Chen, Y., Gucek, M. & Puertollano, R. MTORC1 functions as a transcriptional regulator of autophagy by preventing nuclear transport of TFEB. Autophagy 8, 903–914 (2012).
Napolitano, G. et al. mTOR-dependent phosphorylation controls TFEB nuclear export. Nat. Commun. 9, 3312 (2018).
Liao, C.-Y. et al. The autophagy inducer spermidine protects against metabolic dysfunction during overnutrition. J. Gerontol. A Biol. Sci. Med Sci. 76, 1714–1725 (2021).
Yang, Y. et al. Induction of autophagy by spermidine is neuroprotective via inhibition of caspase 3-mediated Beclin 1 cleavage. Cell Death Dis. 8, e2738 (2017).
Li, G. et al. Spermidine suppresses Inflammatory DC function by activating the FOXO3 pathway and counteracts autoimmunity. iScience 23, 100807 (2020).
Freitag, K. et al. Spermidine reduces neuroinflammation and soluble amyloid beta in an Alzheimer’s disease mouse model. J. Neuroinflammation 19, 172 (2022).
Ding, Y. et al. SOX15 transcriptionally increases the function of AOC1 to modulate ferroptosis and progression in prostate cancer. Cell Death Dis. 13, 673 (2022).
Jeong, J.-W. et al. Spermidine protects against oxidative stress in inflammation models using macrophages and zebrafish. Biomol. Ther. 26, 146–156 (2018).
Liu, S. et al. Spermidine suppresses development of experimental abdominal aortic aneurysms. J. Am. Heart Assoc. 9, e014757 (2020).
Sutter, B. M., Wu, X., Laxman, S. & Tu, B. P. Methionine inhibits autophagy and promotes growth by inducing the SAM-responsive methylation of PP2A. Cell 154, 403–415 (2013).
Ouyang, Y., Wu, Q., Li, J., Sun, S. & Sun, S. S-adenosylmethionine: a metabolite critical to the regulation of autophagy. Cell Prolif. 53, e12891 (2020).
LaRocca, T. J., Gioscia-Ryan, R. A., Hearon, C. M. & Seals, D. R. The autophagy enhancer spermidine reverses arterial aging. Mech. Ageing Dev. 134, 314–320 (2013).
Knott, A. B. & Bossy-Wetzel, E. Impact of nitric oxide on metabolism in health and age-related disease. Diabetes Obes. Metab. 12, 126–133 (2010).
Valerio, A. & Nisoli, E. Nitric oxide, interorganelle communication, and energy flow: a novel route to slow aging. Front Cell Dev. Biol. 3, 6 (2015).
Ni, Y.-Q. & Liu, Y.-S. New insights into the roles and mechanisms of spermidine in aging and age-related diseases. Aging Dis. 12, 16 (2021).
Pillai, S. P. & Shankel, D. M. Polyamines and their potential to be antimutagens. Mutat. Res. 377, 217–224 (1997).
Lee, C. -Y. et al. Promotion of homology-directed DNA repair by polyamines. Nat. Commun. 10, 65 (2019).
Cooke, M. S., Evans, M. D., Dizdaroglu, M. & Lunec, J. Oxidative DNA damage: mechanisms, mutation, and disease. FASEB J. 17, 1195–1214 (2003).
Ha, H. C. et al. The natural polyamine spermine functions directly as a free radical scavenger. Proc. Natl Acad. Sci. USA 95, 11140–11145 (1998).
Khan, A. U., Mei, Y. H. & Wilson, T. A proposed function for spermine and spermidine: protection of replicating DNA against damage by singlet oxygen. Proc. Natl Acad. Sci. USA 89, 11426–11427 (1992).
Vujcic, S., Diegelman, P., Bacchi, C. J., Kramer, D. L. & Porter, C. W. Identification and characterization of a novel flavin-containing spermine oxidase of mammalian cell origin. Biochemical J. 367, 665–675 (2002).
Vujcic, S., Liang, P., Diegelman, P., Kramer, D. L. & Porter, C. W. Genomic identification and biochemical characterization of the mammalian polyamine oxidase involved in polyamine back-conversion. Biochemical J. 370, 19–28 (2003).
Wang, Y. et al. Cloning and characterization of a human polyamine oxidase that is inducible by polyamine analogue cxposure. Cancer Res. 61, 5370–5373 (2001).
Murray Stewart, T., Dunston, T. T., Woster, P. M. & Casero, R. A. Polyamine catabolism and oxidative damage. J. Biol. Chem. 293, 18736–18745 (2018).
Pegg, A. E. Functions of polyamines in mammals. J. Biol. Chem. 291, 14904–14912 (2016).
Wirth, A. et al. Novel aspects of age-protection by spermidine supplementation are associated with preserved telomere length. GeroScience 43, 673–690 (2021).
Scherz-Shouval, R. & Elazar, Z. Regulation of autophagy by ROS: physiology and pathology. Trends Biochemical Sci. 36, 30–38 (2011).
Bose, R. & Kanungo, M. S. Polyamines modulate phosphorylation and acetylation of non-histone chromosomal proteins of the cerebral cortex of rats of various ages. Arch. Gerontol. Geriatrics 1, 339–348 (1982).
Oehme, I. et al. Histone deacetylase 10 promotes autophagy-mediated cell survival. Proc. Natl Acad. Sci. USA 110, E2592–2601 (2013).
Burgio, G., Corona, D. F. V., Nicotra, C. M. A., Carruba, G. & Taibi, G. P/CAF-mediated spermidine acetylation regulates histone acetyltransferase activity. J. Enzyme Inhib. Med. Chem. 31, 75–82 (2016).
Hougaard, D. M., Nielsen, J. H. & Larsson, L. I. Localization and biosynthesis of polyamines in insulin-producing cells. Biochem J. 238, 43–47 (1986).
Welsh, N. & Sjöholm, A. Polyamines and insulin production in isolated mouse pancreatic islets. Biochem J. 252, 701–707 (1988).
Sjoholm, A. Role of polyamines in the regulation of proliferation and hormone production by insulin-secreting cells. Am. J. Physiol. 264, C501–C518 (1993).
Ma, L. et al. Spermidine improves gut barrier integrity and gut microbiota function in diet-induced obese mice. Gut Microbes 12, 1832857 (2020).
Wang, J. -Y. et al. Effect of spermidine on ameliorating spermatogenic disorders in diabetic mice via regulating glycolysis pathway. Reprod. Biol. Endocrinol. 20, 45 (2022).
Tain, L. S. et al. Longevity in response to lowered insulin signaling requires glycine N-methyltransferase-dependent spermidine production. Aging Cell 19, e13043 (2020).
Rajeeve, V., Pearce, W., Cascante, M., Vanhaesebroeck, B. & Cutillas, P. R. Polyamine production is downstream and upstream of oncogenic PI3K signalling and contributes to tumour cell growth. Biochemical J. 450, 619–628 (2013).
Chrisam, M. et al. Reactivation of autophagy by spermidine ameliorates the myopathic defects of collagen VI-null mice. Autophagy 11, 2142–2152 (2015).
Baek, A. R. et al. Spermidine attenuates bleomycin-induced lung fibrosis by inducing autophagy and inhibiting endoplasmic reticulum stress-induced cell death in mice. Exp. Mol. Med 52, 2034–2045 (2020).
Yuan, X. et al. Spermidine induces cytoprotective autophagy of female germline stem cells in vitro and ameliorates aging caused by oxidative stress through upregulated sequestosome-1/p62 expression. Cell Biosci. 11, 107 (2021).
Xu, T.-T. et al. Spermidine and spermine delay brain aging by inducing autophagy in SAMP8 mice. Aging 12, 6401–6414 (2020).
Green, D. R., Galluzzi, L. & Kroemer, G. Mitochondria and the autophagy-inflammation-cell death axis in organismal aging. Science 333, 1109–1112 (2011).
Qi, Y., Qiu, Q., Gu, X., Tian, Y. & Zhang, Y. ATM mediates spermidine-induced mitophagy via PINK1 and Parkin regulation in human fibroblasts. Sci. Rep. 6, 24700 (2016).
Han, X. et al. Spermidine regulates mitochondrial function by enhancing eIF5A hypusination and contributes to reactive oxygen species production and ganoderic acids biosynthesis in Ganoderma lucidum. Appl. Environ. Microbiol. https://doi.org/10.1128/AEM.02037-21 (2022).
Rayess, H., Wang, M. B. & Srivatsan, E. S. Cellular senescence and tumor suppressor gene p16. Int. J. Cancer 130, 1715–1725 (2012).
Che, H. et al. Rebalance of the polyamine metabolism suppresses oxidative stress and delays senescence in nucleus pulposus cells. Oxid. Med. Cell. Longev. 2022, 8033353 (2022).
García-Prat, L. et al. Autophagy maintains stemness by preventing senescence. Nature 529, 37–42 (2016).
Balnis, J. et al. Deaccelerated myogenesis and autophagy in genetically induced pulmonary emphysema. Am. J. Respir. Cell Mol. Biol. 66, 623–637 (2022).
Ramot, Y. et al. Polyamines and hair: a couple in search of perfection. Exp. Dermatol. 19, 784–790 (2010).
Ramot, Y. et al. Spermidine promotes human hair growth and is a novel modulator of human epithelial stem cell functions. PLoS ONE 6, e22564 (2011).
Zhang, D. et al. AMD1 is essential for ESC self-renewal and is translationally down-regulated on differentiation to neural precursor cells. Genes Dev. 26, 461–473 (2012).
Chen, T. et al. Rapamycin and other longevity-promoting compounds enhance the generation of mouse induced pluripotent stem cells. Aging Cell 10, 908–911 (2011).
Ma, L. et al. Spermidine ameliorates high-fat diet-induced hepatic steatosis and adipose tissue inflammation in preexisting obese mice. Life Sci. 265, 118739 (2021).
Gassen, N. C. et al. SARS-CoV-2-mediated dysregulation of metabolism and autophagy uncovers host-targeting antivirals. Nat. Commun. 12, 3818 (2021).
Wagner, A. et al. Metabolic modeling of single TH17 cells reveals regulators of autoimmunity. Cell 184, 4168–4185 (2021).
Pietrocola, F., Bravo-San Pedro, J. M., Galluzzi, L. & Kroemer, G. Autophagy in natural and therapy-driven anticancer immunosurveillance. Autophagy 13, 2163–2170 (2017).
Pietrocola, F. et al. Caloric restriction mimetics enhance anticancer immunosurveillance. Cancer Cell 30, 147–160 (2016).
Holbert, C. E., Cullen, M. T., Casero, R. A. & Stewart, T. M. Polyamines in cancer: integrating organismal metabolism and antitumour immunity. Nat. Rev. Cancer 22, 467–480 (2022).
Alexander, E. T., Minton, A., Peters, M. C., Phanstiel, O. & Gilmour, S. K. A novel polyamine blockade therapy activates an anti-tumor immune response. Oncotarget 8, 84140–84152 (2017).
Hayes, C. S. et al. Polyamine-blocking therapy reverses immunosuppression in the tumor microenvironment. Cancer Immunol. Res. 2, 274–285 (2014).
Fan, Y. & Pedersen, O. Gut microbiota in human metabolic health and disease. Nat. Rev. Microbiol. 19, 55–71 (2021).
Ramos-Molina, B., Queipo-Ortuño, M. I., Lambertos, A., Tinahones, F. J. & Peñafiel, R. Dietary and gut microbiota polyamines in obesity- and age-related diseases. Front. Nutr. 6, 24 (2019).
Wang, D. et al. Oral spermidine targets brown fat and skeletal muscle to mitigate diet-induced obesity and metabolic disorders. Mol. Nutr. Food Res. 65, 2100315 (2021).
Bui, T. I., Gill, A. L., Mooney, R. A. & Gill, S. R. Modulation of gut microbiota metabolism in obesity-related type 2 diabetes reduces osteomyelitis severity. Microbiol. Spectr. 10, e0017022 (2022).
Liu, S. et al. Effects of spermidine on gut microbiota modulation in experimental abdominal aortic aneurysm mice. Nutrients 14, 3349 (2022).
Reggiori, F. & Klionsky, D. J. Autophagic processes in yeast: mechanism, machinery and regulation. Genetics 194, 341–361 (2013).
Zimmermann, A. et al. Autophagy: one more Nobel Prize for yeast. Microb. Cell 3, 579–581 (2016).
Carmona-Gutierrez, D. et al. Guidelines and recommendations on yeast cell death nomenclature. Microb. Cell 5, 4–31 (2018).
Yamaguchi, O. & Otsu, K. Role of autophagy in aging. J. Cardiovasc Pharmacol. 60, 242–247 (2012).
Büttner, S. et al. Spermidine protects against α-synuclein neurotoxicity. Cell Cycle 13, 3903–3908 (2014).
Bito, T. et al. Involvement of spermidine in the reduced lifespan of Caenorhabditis elegans during vitamin B12 deficiency. Metabolites 9, 192 (2019).
Ao, Y. et al. Lamin A buffers CK2 kinase activity to modulate aging in a progeria mouse model. Sci. Adv. https://doi.org/10.1126/sciadv.aav5078 (2019).
Soda, K., Dobashi, Y., Kano, Y., Tsujinaka, S. & Konishi, F. Polyamine-rich food decreases age-associated pathology and mortality in aged mice. Exp. Gerontol. 44, 727–732 (2009).
Matsumoto, M., Kurihara, S., Kibe, R., Ashida, H. & Benno, Y. Longevity in mice is promoted by probiotic-induced suppression of colonic senescence dependent on upregulation of gut bacterial polyamine production. PLoS ONE 6, e23652 (2011).
Kibe, R. et al. Upregulation of colonic luminal polyamines produced by intestinal microbiota delays senescence in mice. Sci. Rep. 4, 4548 (2014).
Filfan, M. et al. Long-term treatment with spermidine increases healthspan of middle-aged Sprague-Dawley male rats. GeroScience 42, 937–949 (2020).
Fernández, Á. F. et al. Autophagy couteracts weight gain, lipotoxicity and pancreatic β-cell death upon hypercaloric pro-diabetic regimens. Cell Death Dis. 8, e2970 (2017).
Partridge, L., Fuentealba, M. & Kennedy, B. K. The quest to slow ageing through drug discovery. Nat. Rev. Drug Discov. 19, 513–532 (2020).
Janssens, G. E. & Houtkooper, R. H. Identification of longevity compounds with minimized probabilities of side effects. Biogerontology 21, 709–719 (2020).
Schwarz, C. et al. Safety and tolerability of spermidine supplementation in mice and older adults with subjective cognitive decline. Aging 10, 19–33 (2018).
Moskalev, A. et al. Developing criteria for evaluation of geroprotectors as a key stage toward translation to the clinic. Aging Cell 15, 407 (2016).
Binh, P. N. T., Soda, K., Maruyama, C. & Kawakami, M. Relationship between food polyamines and gross domestic product in association with longevity in Asian countries. Health 2, 1390–1396 (2010).
Kiechl, S. et al. Higher spermidine intake is linked to lower mortality: a prospective population-based study. Am. J. Clin. Nutr. 108, 371–380 (2018).
Soda, K., Kano, Y. & Chiba, F. Food polyamine and cardiovascular disease—an epidemiological study. Glob. J. Health Sci. 4, 170–178 (2012).
Zoumas-Morse, C. et al. Development of a polyamine database for assessing dietary intake. J. Am. Diet. Assoc. 107, 1024–1027 (2007).
Muñoz-Esparza, N. C. et al. Polyamines in Food. Front. Nutr. 6, 108 (2019).
Buyukuslu, N., Hizli, H., Esin, K. & Garipagaoglu, M. A cross-sectional study: nutritional polyamines in frequently consumed foods of the Turkish population. Foods 3, 541–557 (2014).
Binh, P. N. T., Soda, K. & Kawakami, M. Mediterranean diet and polyamine intake: possible contribution of increased polyamine intake to inhibition of age-associated disease. NDS 3, 1–7 (2010).
Bardócz, S. Polyamines in food and their consequences for food quality and human health. Trends Food Sci. Technol. 6, 341–346 (1995).
Ali, M. A., Poortvliet, E., Strömberg, R. & Yngve, A. Polyamines: total daily intake in adolescents compared to the intake estimated from the Swedish Nutrition Recommendations Objectified (SNO). Food Nutr. Res. 55 (2011).
Schwarz, C. et al. Spermidine intake is associated with cortical thickness and hippocampal volume in older adults. NeuroImage 221, 117132 (2020).
Atiya Ali, M., Poortvliet, E., Strömberg, R. & Yngve, A. Polyamines in foods: development of a food database. Food Nutr. Res. 55 (2011).
Wirth, M. et al. The effect of spermidine on memory performance in older adults at risk for dementia: a randomized controlled trial. Cortex 109, 181–188 (2018).
Wirth, M. et al. Effects of spermidine supplementation on cognition and biomarkers in older adults with subjective cognitive decline (SmartAge)-study protocol for a randomized controlled trial. Alzheimers Res Ther. 11, 36 (2019).
Schwarz, C. et al. Effects of spermidine supplementation on cognition and biomarkers in older adults with subjective cognitive decline: a randomized clinical trial. JAMA Netw. Open 5, e2213875 (2022).
Witkowska, A. M. Soluble ICAM-1: a marker of vascular inflammation and lifestyle. Cytokine 31, 127–134 (2005).
Pekar, T. et al. The positive effect of spermidine in older adults suffering from dementia: first results of a 3-month trial. Wien. Klin. Wochenschr. 133, 484–491 (2021).
Huang, J. et al. Spermidine exhibits protective effects against traumatic brain injury. Cell Mol. Neurobiol. 40, 927–937 (2020).
Guerra, G. P., Rubin, M. A. & Mello, C. F. Modulation of learning and memory by natural polyamines. Pharm. Res. 112, 99–118 (2016).
Matsumoto, M. & Benno, Y. Consumption of Bifidobacterium lactis LKM512 yogurt reduces gut mutagenicity by increasing gut polyamine contents in healthy adult subjects. Mutat. Res. 568, 147–153 (2004).
Matsumoto, M., Kitada, Y. & Naito, Y. Endothelial function is improved by inducing microbial polyamine production in the gut: a randomized placebo-controlled trial. Nutrients 11, 1188 (2019).
Rinaldi, F., Marzani, B., Pinto, D. & Ramot, Y. A spermidine-based nutritional supplement prolongs the anagen phase of hair follicles in humans: a randomized, placebo-controlled, double-blind study. Dermatol. Pract. Concept. 7, 17–21 (2017).
Chai, M. et al. Stimulation of hair growth by small molecules that activate autophagy. Cell Rep. 27, 3413–3421 (2019).
Vargas, A. J. et al. Dietary polyamine intake and risk of colorectal adenomatous polyps. Am. J. Clin. Nutr. 96, 133–141 (2012).
Vargas, A. J. et al. Dietary polyamine intake and colorectal cancer risk in postmenopausal women. Am. J. Clin. Nutr. 102, 411–419 (2015).
Pietrocola, F. et al. Spermidine reduces cancer-related mortality in humans. Autophagy 15, 362–365 (2019).
Gobert, A. P. et al. Protective role of spermidine in colitis and colon carcinogenesis. Gastroenterology 162, 813–827 (2022).
Chin, A., Bieberich, C. J., Stewart, T. M. & Casero, R. A. Polyamine depletion strategies in cancer: remodeling the tumor immune microenvironment to enhance anti-tumor responses. Med. Sci. 10, 31 (2022).
Faundes, V. et al. Impaired eIF5A function causes a Mendelian disorder that is partially rescued in model systems by spermidine. Nat. Commun. 12, 833 (2021).
Murray-Stewart, T., Dunworth, M., Foley, J. R., Schwartz, C. E. & Casero, R. A. Polyamine homeostasis in Snyder-Robinson syndrome. Med. Sci. 6, 112 (2018).
Larcher, L. et al. The complete loss of function of the SMS gene results in a severe form of Snyder-Robinson syndrome. Eur. J. Med. Genet. 63, 103777 (2020).
Lauren Cason, A. et al. X-linked spermine synthase gene (SMS) defect: the first polyamine deficiency syndrome. Eur. J. Hum. Genet 11, 937–944 (2003).
Li, C. et al. Spermine synthase deficiency causes lysosomal dysfunction and oxidative stress in models of Snyder-Robinson syndrome. Nat. Commun. 8, 1257 (2017).
Murray Stewart, T. et al. (R,R)-1,12-Dimethylspermine can mitigate abnormal spermidine accumulation in Snyder-Robinson syndrome. J. Biol. Chem. 295, 3247–3256 (2020).
Kapur, M. & Ackerman, S. L. mRNA translation gone awry: translation fidelity and neurological disease. Trends Genet. 34, 218–231 (2018).
Schultz, C. R., Bupp, C. P., Rajasekaran, S. & Bachmann, A. S. Biochemical features of primary cells from a pediatric patient with a gain-of-function ODC1 genetic mutation. Biochemical J. 476, 2047–2057 (2019).
VanSickle, E. A. et al. Expanding the phenotype: four new cases and hope for treatment in Bachmann-Bupp syndrome. Am. J. Med. Genet. A 185, 3485–3493 (2021).
Bupp, C. P., Schultz, C. R., Uhl, K. L., Rajasekaran, S. & Bachmann, A. S. Novel de novo pathogenic variant in the ODC1 gene in a girl with developmental delay, alopecia, and dysmorphic features. Am. J. Med. Genet. A 176, 2548–2553 (2018).
Rodan, L. H. et al. Gain-of-function variants in the ODC1 gene cause a syndromic neurodevelopmental disorder associated with macrocephaly, alopecia, dysmorphic features, and neuroimaging abnormalities. Am. J. Med. Genet. A 176, 2554–2560 (2018).
Rajasekaran, S. et al. Repurposing eflornithine to treat a patient with a rare ODC1 gain-of-function variant disease. Elife 10, e67097 (2021).
Mayeux, R. Biomarkers: potential uses and limitations. NeuroRx 1, 182–188 (2004).
Strimbu, K. & Tavel, J. A. What are biomarkers? Curr. Opin. HIV AIDS 5, 463–466 (2010).
Amin, M. et al. Polyamine biomarkers as indicators of human disease. Biomarkers 26, 77–94 (2021).
Maksymiuk, A. W. et al. Spermidine/spermine-N1-acetyltransferase-1 as a diagnostic biomarker in human cancer. Future Sci. OA 4, FSO345 (2018).
Morrison, L. D. & Kish, S. J. Brain polyamine levels are altered in Alzheimer’s disease. Neurosci. Lett. 197, 5–8 (1995).
Saiki, S. et al. A metabolic profile of polyamines in Parkinson disease: a promising biomarker. Ann. Neurol. 86, 251–263 (2019).
Sternberg, Z. et al. Elevated spermidine serum levels in mild cognitive impairment, a potential biomarker of progression to Alzheimer dementia, a pilot study. J. Clin. Neurosci. 100, 169–174 (2022).
Xu, J. et al. Non-linear association between serum spermidine and mild cognitive impairment: results from a cross-sectional and longitudinal study. Front. Aging Neurosci. 14, 924984 (2022).
Polis, B., Karasik, D. & Samson, A. O. Alzheimer’s disease as a chronic maladaptive polyamine stress response. Aging 13, 10770–10795 (2021).
Zheng, L. et al. Serum spermidine in relation to risk of stroke: a multilevel study. Front Nutr. 9, 843616 (2022).
Yu, Z. et al. Effect of serum spermidine on the prognosis in patients with acute myocardial infarction: a cohort study. Nutrients 14, 1394 (2022).
Omar, E. M., Omar, R. S., Shoela, M. S. & El Sayed, N. S. A study of the cardioprotective effect of spermidine: a novel inducer of autophagy. Chin. J. Physiol. 64, 281–288 (2021).
Magnes, C. et al. Polyamines in biological samples: rapid and robust quantification by solid-phase extraction online-coupled to liquid chromatography–tandem mass spectrometry. J. Chromatogr. A 1331, 44–51 (2014).
Srivastava, V. et al. Distinct designer diamines promote mitophagy, and thereby enhance healthspan in C. elegans and protect human cells against oxidative damage. Autophagy 0, 1–31 (2022).
Acknowledgements
All figures were initially created with BioRender.com. S.J.H. acknowledges support from a Longevity Impetus Grant from Norn Group. F.M. is grateful to the Austrian Science Fund FWF (F3012, W1226, P29203, P29262, P27893, P31727) and the Austrian Federal Ministry of Education, Science and Research as well as the University of Graz for grants ‘Unkonventionelle Forschung-InterFast and Fast4Health’ and ‘flysleep’ (BMWFW-80.109/0001-WF/V/3b/2015). T.E. acknowledges support from Austrian Science Fund FWF (P 33957 and TAI 602 1000). We acknowledge the support of the Field of Excellence BioHealth of NAWI Graz and the BioTechMed-Graz flagship project ‘EPIAge’. G.K. is supported by the Ligue contre le Cancer (équipe labellisée); Agence National de la Recherche (ANR)–Projets blancs; AMMICa US23/CNRS UMS3655; Association pour la recherche sur le cancer (ARC); Cancéropôle Ile-de-France; Fondation pour la Recherche Médicale (FRM); a donation by Elior; Equipex Onco-Pheno-Screen; European Joint Programme on Rare Diseases (EJPRD); Gustave Roussy Odyssea, the European Union Horizon 2020 Projects Oncobiome and Crimson; Fondation Carrefour; Institut National du Cancer (INCa); Institut Universitaire de France; LabEx Immuno-Oncology (ANR-18-IDEX-0001); a Cancer Research ASPIRE Award from the Mark Foundation; the RHU Immunolife; Seerave Foundation; SIRIC Stratified Oncology Cell DNA Repair and Tumor Immune Elimination (SOCRATE); and SIRIC Cancer Research and Personalized Medicine (CARPEM). This study contributes to the IdEx Université de Paris ANR-18-IDEX-0001.
Author information
Authors and Affiliations
Contributions
S.J.H. wrote the first draft. S.J.H., G.K. and F.M. conceptualized the review. M.B. and S.J.H. collected information for the tables. All authors proofread, edited and contributed substantially to the final version.
Corresponding authors
Ethics declarations
Competing interests
F.M. and G.K. are scientific cofounders of Samsara Therapeutics, a company that develops new pharmacological autophagy inducers. F.M. and T.E. have equity interests in and are advisors of The Longevity Labs (TLL). A.K.S. is consulting for TLL and Oxford Healthspan. G.K. is a scientific cofounder of everImmune, Osasuna Therapeutics and Therafast Bio. G.K. is holding research contracts with Daiichi Sankyo, Eleor, Osasuna Therapeutics, Samsara Therapeutics, Sanofi, Tollys and Vascage. S.J.H. and M.B. declare no competing interests.
Peer review
Peer review information
Nature Aging thanks Luigi Fontana, Tracy Murray-Stewart and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Hofer, S.J., Simon, A.K., Bergmann, M. et al. Mechanisms of spermidine-induced autophagy and geroprotection. Nat Aging 2, 1112–1129 (2022). https://doi.org/10.1038/s43587-022-00322-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s43587-022-00322-9
This article is cited by
-
The buck stops with spermidine
Nature Chemical Biology (2024)
-
Mitochondria, Autophagy and Inflammation: Interconnected in Aging
Cell Biochemistry and Biophysics (2024)
-
The plasma metabolome is associated with preservation of physiological function following lifelong aerobic exercise in mice
GeroScience (2024)
-
The million-molecule challenge: a moonshot project to rapidly advance longevity intervention discovery
GeroScience (2023)