Abstract
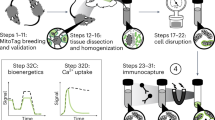
Extracellular vesicles (EVs) are nanoscale vesicles secreted into the extracellular space by all cell types, including neurons and astrocytes in the brain. EVs play pivotal roles in physiological and pathophysiological processes such as waste removal, cell-to-cell communication and transport of either protective or pathogenic material into the extracellular space. Here we describe a detailed protocol for the reliable and consistent isolation of EVs from both murine and human brains, intended for anyone with basic laboratory experience and performed in a total time of 27 h. The method includes a mild extracellular matrix digestion of the brain tissue, a series of filtration and centrifugation steps to purify EVs and an iodixanol-based high-resolution density step gradient that fractionates different EV populations, including mitovesicles, a newly identified type of EV of mitochondrial origin. We also report detailed downstream protocols for the characterization and analysis of brain EV preparations using nanotrack analysis, electron microscopy and western blotting, as well as for measuring mitovesicular ATP kinetics. Furthermore, we compared this novel iodixanol-based high-resolution density step gradient to the previously described sucrose-based gradient. Although the yield of total EVs recovered was similar, the iodixanol-based gradient better separated distinct EV species as compared with the sucrose-based gradient, including subpopulations of microvesicles, exosomes and mitovesicles. This technique allows quantitative, highly reproducible analyses of brain EV subtypes under normal physiological processes and pathological brain conditions, including neurodegenerative diseases such as Alzheimer’s disease and Parkinson’s disease.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data Availability
All data needed to evaluate the conclusions of the paper are present in the paper. No datasets or custom code were generated in this study. All single datapoints are reported in the respective graphs, when possible (Figs. 4,5) and as Excel Source Data files. Raw, uncropped blots for Figs. 2,3 are provided as pdf source data files. Additional data related to this paper may be requested from the authors. Source data are provided with this paper.
References
van Niel, G., D’Angelo, G. & Raposo, G. Shedding light on the cell biology of extracellular vesicles. Nat. Rev. Mol. Cell Biol. 19, 213–228 (2018).
Del Conde, I., Shrimpton, C. N., Thiagarajan, P. & Lopez, J. A. Tissue-factor-bearing microvesicles arise from lipid rafts and fuse with activated platelets to initiate coagulation. Blood 106, 1604–1611 (2005).
D’Acunzo, P. et al. Mitovesicles are a novel population of extracellular vesicles of mitochondrial origin altered in Down syndrome. Sci. Adv. https://doi.org/10.1126/sciadv.abe5085 (2021).
Valadi, H. et al. Exosome-mediated transfer of mRNAs and microRNAs is a novel mechanism of genetic exchange between cells. Nat. Cell Biol. 9, 654–659 (2007).
Perez-Gonzalez, R. et al. Neuroprotection mediated by cystatin C-loaded extracellular vesicles. Sci. Rep. 9, 11104 (2019).
Kaur, G. et al. Cystatin C prevents neuronal loss and behavioral deficits via the endosomal pathway in a mouse model of down syndrome. Neurobiol. Dis. 120, 165–173 (2018).
Liu, S., Hossinger, A., Gobbels, S. & Vorberg, I. M. Prions on the run: how extracellular vesicles serve as delivery vehicles for self-templating protein aggregates. Prion 11, 98–112 (2017).
Guo, B. B., Bellingham, S. A. & Hill, A. F. The neutral sphingomyelinase pathway regulates packaging of the prion protein into exosomes. J. Biol. Chem. 290, 3455–3467 (2015).
Guo, B. B., Bellingham, S. A. & Hill, A. F. Stimulating the release of exosomes increases the intercellular transfer of prions. J. Biol. Chem. 291, 5128–5137 (2016).
Zhang, X. et al. Potential transfer of polyglutamine and CAG-repeat RNA in extracellular vesicles in Huntington’s disease: background and evaluation in cell culture. Cell Mol. Neurobiol. 36, 459–470 (2016).
Guix, F. X. et al. Detection of aggregation-competent tau in neuron-derived extracellular vesicles. Int. J. Mol. Sci. https://doi.org/10.3390/ijms19030663 (2018).
Perez-Gonzalez, R., Gauthier, S. A., Kumar, A. & Levy, E. The exosome secretory pathway transports amyloid precursor protein carboxyl-terminal fragments from the cell into the brain extracellular space. J. Biol. Chem. 287, 43108–43115 (2012).
Perez-Gonzalez, R. et al. A pleiotropic role for exosomes loaded with the amyloid beta precursor protein carboxyl-terminal fragments in the brain of Down syndrome patients. Neurobiol. Aging 84, 26–32 (2019).
Perez-Gonzalez, R. et al. Extracellular vesicles: where the amyloid precursor protein carboxyl-terminal fragments accumulate and amyloid-β oligomerizes. FASEB J. 34, 12922–12931 (2020).
Mathews, P. M. & Levy, E. Exosome production is key to neuronal endosomal pathway integrity in neurodegenerative diseases. Front. Neurosci. 13, 1347 (2019).
Gauthier, S. A. et al. Enhanced exosome secretion in Down syndrome brain—a protective mechanism to alleviate neuronal endosomal abnormalities. Acta Neuropathol. Commun. 5, 65 (2017).
D’Acunzo, P. et al. Enhanced generation of intraluminal vesicles in neuronal late endosomes in the brain of a Down syndrome mouse model with endosomal dysfunction. Dev. Neurobiol. 79, 656–663 (2019).
Peng, K. Y. et al. Apolipoprotein E4 genotype compromises brain exosome production. Brain 142, 163–175 (2019).
Barreto, B. R. et al. Cocaine modulates the neuronal endosomal system and extracellular vesicles in a sex-dependent manner. Neurochem. Res. https://doi.org/10.1007/s11064-022-03612-1 (2022).
Cataldo, A. M. et al. Endocytic pathway abnormalities precede amyloid beta deposition in sporadic Alzheimer’s disease and Down syndrome: differential effects of APOE genotype and presenilin mutations. Am. J. Pathol. 157, 277–286 (2000).
Zhang, Y. et al. Cerebellar Kv3.3 potassium channels activate TANK-binding kinase 1 to regulate trafficking of the cell survival protein Hax-1. Nat. Commun. 12, 1731 (2021).
Nuriel, T. et al. The endosomal–lysosomal pathway is dysregulated by APOE4 expression in vivo. Front. Neurosci. 11, 702 (2017).
Norman, M. et al. L1CAM is not associated with extracellular vesicles in human cerebrospinal fluid or plasma. Nat. Methods 18, 631–634 (2021).
Melki, I. et al. Platelets release mitochondrial antigens in systemic lupus erythematosus. Sci. Transl. Med. https://doi.org/10.1126/scitranslmed.aav5928 (2021).
Thul, P. J. et al. A subcellular map of the human proteome. Science https://doi.org/10.1126/science.aal3321 (2017).
Uhlen, M. et al. Proteomics. Tissue-based map of the human proteome. Science 347, 1260419 (2015).
Uhlen, M. et al. Towards a knowledge-based Human Protein Atlas. Nat. Biotechnol. 28, 1248–1250 (2010).
Kim, K. M. et al. Mitochondrial RNA in Alzheimer’s disease circulating extracellular vesicles. Front. Cell Dev. Biol. 8, 581882 (2020).
Liangsupree, T., Multia, E. & Riekkola, M. L. Modern isolation and separation techniques for extracellular vesicles. J. Chromatogr. A 1636, 461773 (2021).
Perez-Gonzalez, R. et al. A method for isolation of extracellular vesicles and characterization of exosomes from brain extracellular space. Methods Mol. Biol. 1545, 139–151 (2017).
Vella, L. J. et al. A rigorous method to enrich for exosomes from brain tissue. J. Extracell. Vesicles 6, 1348885 (2017).
Ruan, Z. et al. Alzheimer’s disease brain-derived extracellular vesicles spread tau pathology in interneurons. Brain https://doi.org/10.1093/brain/awaa376 (2020).
Crescitelli, R., Lasser, C. & Lotvall, J. Isolation and characterization of extracellular vesicle subpopulations from tissues. Nat. Protoc. 16, 1548–1580 (2021).
Brinkman, J. E. & Sharma, S. Physiology, Body Fluids. In StatPearls (2020).
Qian, X. et al. Timing of CNS cell generation: a programmed sequence of neuron and glial cell production from isolated murine cortical stem cells. Neuron 28, 69–80 (2000).
Brewer, G. J. & Torricelli, J. R. Isolation and culture of adult neurons and neurospheres. Nat. Protoc. 2, 1490–1498 (2007).
Lam, D. M. Biosynthesis of acetylcholine in turtle photoreceptors. Proc. Natl Acad. Sci. USA 69, 1987–1991 (1972).
Miyazawa, T. Enzymatic resolution of amino acids via ester hydrolysis. Amino Acids 16, 191–213 (1999).
Varughese, K. I. et al. Crystal structure of a papain-E-64 complex. Biochemistry 28, 1330–1332 (1989).
Hussain, R. Z. et al. Defining standard enzymatic dissociation methods for individual brains and spinal cords in EAE. Neurol. Neuroimmunol. Neuroinflamm. 5, e437 (2018).
Volovitz, I. et al. A non-aggressive, highly efficient, enzymatic method for dissociation of human brain-tumors and brain-tissues to viable single-cells. BMC Neurosci. 17, 30 (2016).
Bonneh-Barkay, D. & Wiley, C. A. Brain extracellular matrix in neurodegeneration. Brain Pathol. 19, 573–585 (2009).
Crescitelli, R. et al. Subpopulations of extracellular vesicles from human metastatic melanoma tissue identified by quantitative proteomics after optimized isolation. J. Extracell. Vesicles 9, 1722433 (2020).
Pérez-González, R., Gauthier, S. A., Kumar, A. & Levy, E. The exosome-secretory pathway transports amyloid precursor protein carboxyl terminal fragments from the cell into the brain extracellular space. J. Biol. Chem. 287, 43108–43115 (2012).
Corballis, M. C. Left brain, right brain: facts and fantasies. PLoS Biol. 12, e1001767 (2014).
Jeppesen, D. K. et al. Reassessment of exosome composition. Cell 177, 428–445 e418 (2019).
Kugeratski, F. G. et al. Quantitative proteomics identifies the core proteome of exosomes with syntenin-1 as the highest abundant protein and a putative universal biomarker. Nat. Cell Biol. 23, 631–641 (2021).
Rao, L., Perez, D. & White, E. Lamin proteolysis facilitates nuclear events during apoptosis. J. Cell Biol. 135, 1441–1455 (1996).
Thery, C. et al. Minimal information for studies of extracellular vesicles 2018 (MISEV2018): a position statement of the International Society for Extracellular Vesicles and update of the MISEV2014 guidelines. J. Extracell. Vesicles 7, 1535750 (2018).
Schneider, C. A., Rasband, W. S. & Eliceiri, K. W. NIH Image to ImageJ: 25 years of image analysis. Nat. Methods 9, 671–675 (2012).
Chuo, S. T., Chien, J. C. & Lai, C. P. Imaging extracellular vesicles: current and emerging methods. J. Biomed. Sci. 25, 91 (2018).
Acknowledgements
This work was supported by the National Institute on Aging (grant numbers AG017617, AG056732 and AG057517) and the National Institute on Drug Abuse (grant number DA044489). The authors thank M. Pawlik and S. DeRosa for the animal husbandry and G. Ferrari for coordinating and managing our laboratory.
Author information
Authors and Affiliations
Contributions
Conceptualization: P.D. and E.L.; methodology: P.D., Y.K. and R.P.-G.; formal analysis: P.D.; investigation: P.D., C.G. and J.M.U.; writing original and revised drafts: P.D. and E.L.; visualization: P.D. and E.L.; supervision: E.L.; project administration: E.L. and P.D.; funding acquisition: E.L.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Protocols thanks Éric Boilard, Ashok Shetty and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
D’Acunzo, P. et al. Sci. Adv. 7, eabe5085 (2021): https://doi.org/10.1126/sciadv.abe5085
Zhang, Y. et al. Nat. Commun. 12, 1731 (2021): https://doi.org/10.1038/s41467-021-22003-8
Perez-Gonzalez, R. et al. J. Biol. Chem. 287, 43108–43115 (2012): https://doi.org/10.1074/jbc.M112.404467
Extended data
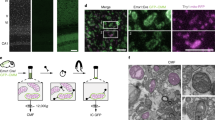
Extended Data Fig. 1 Purified brain EVs fixed with PFA and stained with uranyl acetate show a distinctive cup-shape morphology.
Representative photomicrograph of sucrose fraction c brain EVs isolated from a 12-month-old female mouse and visualized after negative stain. Note the cup shape of fixed EVs and the absence of contaminating material. Scale bar, 200 nm. All animal procedures were performed following the National Institutes of Health guidelines with approval from the Institutional Animal Care and Use Committee at the Nathan S. Kline Institute for Psychiatric Research.
Extended Data Fig. 2 Representative cryo-EM photomicrograph of contaminating crystals during acquisition of mitovesicles (iodixanol fraction 8 EVs).
Crystals are visualized as electron-dense bodies that are either amorphous (orange arrowheads) or polygonal, for instance, hexagonal or cubic (red arrowheads). The white arrow indicates a typical mitovesicle, which is characterized by the presence of a double membrane. The amorphous and polygonal dark structures in this case are the same contaminant, caused by moisture from the air that has frozen during the freezing process. Scale bar, 200 nm. All animal procedures were performed following the National Institutes of Health guidelines with approval from the Institutional Animal Care and Use Committee at the Nathan S. Kline Institute for Psychiatric Research.
Source data
Source Data Fig. 2
Unprocessed western blots.
Source Data Fig. 3
Unprocessed western blots.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Rights and permissions
Springer Nature or its licensor holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
D’Acunzo, P., Kim, Y., Ungania, J.M. et al. Isolation of mitochondria-derived mitovesicles and subpopulations of microvesicles and exosomes from brain tissues. Nat Protoc 17, 2517–2549 (2022). https://doi.org/10.1038/s41596-022-00719-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-022-00719-1
This article is cited by
-
Mitovesicles secreted into the extracellular space of brains with mitochondrial dysfunction impair synaptic plasticity
Molecular Neurodegeneration (2024)
-
The role and applications of extracellular vesicles in osteoporosis
Bone Research (2024)
-
Mechanical overload-induced release of extracellular mitochondrial particles from tendon cells leads to inflammation in tendinopathy
Experimental & Molecular Medicine (2024)
-
Mitochondria-derived vesicles and their potential roles in kidney stone disease
Journal of Translational Medicine (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.