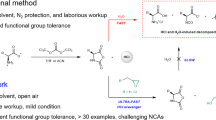
Abstract
Amide bond formation and aromatic/heteroaromatic nitro-group reductions represent two of the most commonly used transformations in organic synthesis. Unfortunately, such processes can be especially wasteful and hence environmentally harmful, and may present safety hazards as well, given the reaction conditions involved. The two protocols herein describe alternative technologies that offer solutions to these issues. Polypeptides can now be made in water at ambient temperatures using small amounts of the designer surfactant TPGS-750-M, thereby eliminating the use of organic solvents as the reaction medium. Likewise, a safe, inexpensive and efficient procedure is outlined for nitro-group reductions, using industrial iron in the form of carbonyl iron powder (CIP), an inexpensive item of commerce. The peptide synthesis will typically take, overall, 3–4 h for a simple coupling and 8 h for a two-step deprotection/coupling process. The workup usually consists of a simple extraction and acidic/basic aqueous washings. The nitro reduction procedure will typically take 6–8 h to complete, including setup, reaction time and workup.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout















Similar content being viewed by others
Data availability
The authors declare that all data supporting the findings of this study are available within the article and its Supplementary Information files.
References
Brown, D. G. & Boström, J. Analysis of past and present synthetic methodologies on medicinal chemistry: where have all the new reactions gone? J. Med. Chem. 59, 4443–4458 (2016).
European Chemicals Agency. Member state committee support document for identification of N,N-dimethylformamide as a substance of very high concern because of its CMR properties. ECHA https://echa.europa.eu/documents/10162/13638/supdoc_n_n-dimethylformamide_3204_en.pdf/3f575bea-5a96-4f18-a110-d294d37422c7 (2012).
US Environmental Protection Agency. Toxicological review of dichloromethane (methylene chloride). EPA.gov https://cfpub.epa.gov/ncea/iris/iris_documents/documents/toxreviews/0070tr.pdf (2011).
Datta, S., Sood, A. & Török, M. A step toward green peptide synthesis. Curr. Org. Synth. 8, 262–280 (2011).
Sabatini, M. T., Boulton, L. T. & Sheppard, T. D. Borate esters: simple catalysts for the sustainable synthesis of complex amides. Sci. Adv. 3, e1701028 (2017).
Sabatini, M. T. et al. Protecting-group-free amidation of amino acids using lewis acid catalysts. Chem. Eur. J. 24, 7033–7043 (2018).
Jad, Y. E. et al. Peptide synthesis beyond DMF: THF and ACN as excellent and friendlier alternatives. Org. Biomol. Chem. 13, 2393–2398 (2015).
Lopez, J., Pletscher, S., Aemissegger, A., Bucher, C. & Gallou, F. N-butylpyrrolidinone as alternative solvent for solid-phase peptide synthesis. Org. Process Res. Dev. 22, 494–503 (2018).
Lawrenson, S. B., Arav, R. & North, M. The greening of peptide synthesis. Green Chem. 19, 1685–1691 (2017).
Wilson, K. L., Murray, J., Jamieson, C. & Watson, A. J. B. Cyrene as a bio-based solvent for HATU mediated amide coupling. Org. Biomol. Chem. 16, 2851–2854 (2018).
Declerck, V., Nun, P., Martinez, J. & Lamaty, F. Solvent‐free synthesis of peptides. Angew. Chem. Int. Ed. Engl. 48, 9318–9321 (2009).
Métro, T.-X. et al. Mechanosynthesis of amides in the total absence of organic solvent from reaction to product recovery. Chem. Commun. 48, 11781–11783 (2012).
Bonnamour, J., Métro, T.-X., Martinez, J. & Lamaty, F. Environmentally benign peptide synthesis using liquid-assisted ball-milling: application to the synthesis of Leu-enkephalin. Green Chem. 15, 1116–1120 (2013).
Hojo, K., Maeda, M. & Kawasaki, K. 2-(4-Sulfophenylsulfonyl)ethoxycarbonyl group: a new water-soluble N-protecting group and its application to solid phase peptide synthesis in water. Tetrahedron Lett. 45, 9293–9295 (2004).
Hojo, K. et al. Aqueous microwave-assisted solid-phase peptide synthesis using Fmoc strategy. III: racemization studies and water-based synthesis of histidine-containing peptides. Amino Acids 46, 2347 (2014).
Gabriel, C. M., Keener, M., Gallou, F. & Lipshutz, B. H. Amide and peptide bond formation in water at room temperature. Org. Lett. 17, 3968–3971 (2015).
Lipshutz, B. H. When does organic chemistry follow nature’s lead and “make the switch”? J. Org. Chem. 82, 2806–2816 (2017).
Wehrstedt, K. D., Wandrey, P. A. & Heitkamp, D. Explosive properties of 1-hydroxybenzotriazoles. Hazard. Mater. 126, 1–7 (2005).
Cortes-Clerget, M., Berthon, J.-Y., Krolikiewicz-Renimel, I., Chaisemartin, L. & Lipshutz, B. H. Tandem deprotection/coupling for peptide synthesis in water at room temperature. Green Chem. 19, 4263–4267 (2017).
Orlandi, M., Brenna, D., Harms, R., Jost, S. & Benaglia, M. Recent developments in the reduction of aromatic and aliphatic nitro compounds to amines. Org. Process Res. Dev. 22, 430–445 (2018).
K. Kadam, H. & G. Tilve, S. Advancement in methodologies for reduction of nitroarenes. RSC Adv. 101, 83391–83407 (2015).
Lee, N. R., Bikovtseva, A. A., Cortes-Clerget, M., Gallou, F. & Lipshutz, B. H. Carbonyl iron powder: a reagent for nitro group reductions under aqueous micellar catalysis conditions. Org. Lett. 19, 6518–6521 (2017).
Macaluso, A. et al. Antiinflammatory influences of alpha-MSH molecules: central neurogenic and peripheral actions. J. Neurosci. 14, 2377–2382 (1994).
Author information
Authors and Affiliations
Contributions
M.C.-C., N.R.L. and B.H.L. wrote, reviewed and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Related links
Key references using this protocol
Gabriel, C. M., Keener, M., Gallou, F. & Lipshutz, B. H. Org. Lett. 17, 3968–3971 (2015): https://pubs.acs.org/doi/abs/10.1021/acs.orglett.5b01812
Cortes-Clerget, M., Berthon, J.-Y., Krolikiewicz-Renimel, I., Chaisemartin, L. & Lipshutz, B. H. Green Chem. 19, 4263−4267 (2017): https://pubs.rsc.org/en/content/articlelanding/2017/gc/c7gc01575e#!divAbstract
Lee, N. R., Bikovtseva, A. A., Cortes-Clerget, M., Gallou, F. & Lipshutz, B. H. Org. Lett. 19, 6518–6521 (2017): https://pubs.acs.org/doi/abs/10.1021/acs.orglett.7b03216
Video tutorials
Peptide synthesis in water—episode 1: coupling: https://youtu.be/WvRVfaR7dwM
Peptide synthesis in water—episode 2: 1-pot deprotection/coupling: https://youtu.be/eNqan3L4LrQ
Supplementary information
Rights and permissions
About this article
Cite this article
Cortes-Clerget, M., Lee, N.R. & Lipshutz, B.H. Synthetic chemistry in water: applications to peptide synthesis and nitro-group reductions. Nat Protoc 14, 1108–1129 (2019). https://doi.org/10.1038/s41596-019-0130-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41596-019-0130-1
This article is cited by
-
Coupling photocatalytic water oxidation with reductive transformations of organic molecules
Nature Communications (2022)
-
Environmentally Conscious In-Water Peptide Synthesis Using Boc Strategy
International Journal of Peptide Research and Therapeutics (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.