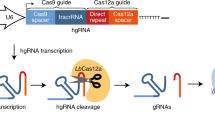
Abstract
Systematic mapping of genetic interactions (GIs) and interrogation of the functions of sizable genomic segments in mammalian cells represent important goals of biomedical research. To advance these goals, we present a CRISPR (clustered regularly interspaced short palindromic repeats)-based screening system for combinatorial genetic manipulation that employs coexpression of CRISPR-associated nucleases 9 and 12a (Cas9 and Cas12a) and machine-learning-optimized libraries of hybrid Cas9–Cas12a guide RNAs. This system, named Cas Hybrid for Multiplexed Editing and screening Applications (CHyMErA), outperforms genetic screens using Cas9 or Cas12a editing alone. Application of CHyMErA to the ablation of mammalian paralog gene pairs reveals extensive GIs and uncovers phenotypes normally masked by functional redundancy. Application of CHyMErA in a chemogenetic interaction screen identifies genes that impact cell growth in response to mTOR pathway inhibition. Moreover, by systematically targeting thousands of alternative splicing events, CHyMErA identifies exons underlying human cell line fitness. CHyMErA thus represents an effective screening approach for GI mapping and the functional analysis of sizable genomic regions, such as alternative exons.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The hgRNA sequences and contents of all libraries are included in supplementary tables. The datasets generated and analyzed in this study are included in the manuscript and deposited at the website http://crispr.ccbr.utoronto.ca/chymera. Raw fastq files for all sequencing data are available upon request and at the Gene Expression Omnibus: RBM26 and RBM27 RNA-seq data, GSE144078; CRISPR screens sequencing read archive, GSE144281.
Code availability
Descriptions of analyses, tools and algorithms are provided in Methods. Custom code for generation of hgRNA counts from fastq files, code for CHyMErA-Net and code for analysis of screens with the dual-targeting/paralog and exon-targeting libraries are available at Github (https://github.com/BlencoweLab/chymeranet) and at http://crispr.ccbr.utoronto.ca/chymera.
References
Cong, L. et al. Multiplex genome engineering using CRISPR/Cas systems. Science 339, 819–823 (2013).
Jinek, M. et al. A programmable dual-RNA-guided DNA endonuclease in adaptive bacterial immunity. Science 337, 816–821 (2012).
Mali, P. et al. RNA-guided human genome engineering via Cas9. Science 339, 823–826 (2013).
Wright, A. V., Nuñez, J. K. & Doudna, J. A. Biology and applications of CRISPR systems: harnessing nature’s toolbox for genome engineering. Cell 164, 29–44 (2016).
Doench, J. G. Am I ready for CRISPR? A user’s guide to genetic screens. Nat. Rev. Genet. 19, 67–80 (2018).
Hart, T. et al. High-resolution CRISPR screens reveal fitness genes and genotype-specific cancer liabilities. Cell 163, 1515–1526 (2015).
Shalem, O. et al. Genome-scale CRISPR-Cas9 knockout screening in human cells. Science 343, 84–87 (2014).
Wang, T. et al. Identification and characterization of essential genes in the human genome. Science 350, 1096–1101 (2015).
Wang, T., Wei, J. J., Sabatini, D. M. & Lander, E. S. Genetic screens in human cells using the CRISPR-Cas9 system. Science 343, 80–84 (2014).
Liu, G. et al. Data independent acquisition analysis in ProHits 4.0. J. Proteomics 149, 64–68 (2016).
Patel, S. J. et al. Identification of essential genes for cancer immunotherapy. Nature 548, 537–542 (2017).
Park, R. J. et al. A genome-wide CRISPR screen identifies a restricted set of HIV host dependency factors. Nat. Genet. 49, 193–203 (2016).
Haney, M. S. et al. Identification of phagocytosis regulators using magnetic genome-wide CRISPR screens. Nat. Genet. 50, 1716–1727 (2018).
Gonatopoulos-Pournatzis, T. et al. Genome-wide CRISPR-Cas9 interrogation of splicing networks reveals a mechanism for recognition of autism-misregulated neuronal microexons. Mol. Cell 72, 510–524.e12 (2018).
Shifrut, E. et al. Genome-wide CRISPR screens in primary human T cells reveal key regulators of immune function. Cell 175, 1958–1971.e15 (2018).
Mair, B. et al. Essential gene profiles for human pluripotent stem cells identify uncharacterized genes and substrate dependencies. Cell Rep. 27, 599–615.e12 (2019).
Lynch, M. & Conery, J. S. The evolutionary fate and consequences of duplicate genes. Science 290, 1151–1155 (2000).
Han, K. et al. Synergistic drug combinations for cancer identified in a CRISPR screen for pairwise genetic interactions. Nat. Biotechnol. 35, 463–474 (2017).
Najm, F. J. et al. Orthologous CRISPR–Cas9 enzymes for combinatorial genetic screens. Nat. Biotechnol. 36, 179–189 (2017).
Zhu, S. et al. Genome-scale deletion screening of human long non-coding RNAs using a paired-guide RNA CRISPR–Cas9 library. Nat. Biotechnol. 34, 1279–1286 (2016).
Wong, A. S. L. et al. Multiplexed barcoded CRISPR-Cas9 screening enabled by CombiGEM. Proc. Natl Acad. Sci. USA 113, 2544–2549 (2016).
Shen, J. P. et al. Combinatorial CRISPR–Cas9 screens for de novo mapping of genetic interactions. Nat. Methods 14, 573–576 (2017).
Brake, Oter et al. Lentiviral vector design for multiple shRNA expression and durable HIV-1 inhibition. Mol. Ther. 16, 557–564 (2008).
Vidigal, J. A. & Ventura, A. Rapid and efficient one-step generation of paired gRNA CRISPR–Cas9 libraries. Nat. Commun. 6, 8083 (2015).
Sack, L. M., Davoli, T., Xu, Q., Li, M. Z. & Elledge, S. J. Sources of error in mammalian genetic screens. G3 (Bethesda) 6, 2781–2790 (2016).
Adamson, B. et al. A multiplexed single-cell CRISPR screening platform enables systematic dissection of the unfolded protein response. Cell 167, 1867–1882.e21 (2016).
Zetsche, B. et al. Cpf1 is a single RNA-guided endonuclease of a class 2 CRISPR-cas system. Cell 163, 759–771 (2015).
Zetsche, B. et al. Multiplex gene editing by CRISPR–Cpf1 using a single crRNA array. Nat. Biotechnol. 35, 31–34 (2016).
Fonfara, I., Richter, H., Bratovič, M., Le Rhun, A. & Charpentier, E. The CRISPR-associated DNA-cleaving enzyme Cpf1 also processes precursor CRISPR RNA. Nature 532, 517–521 (2016).
Chow, R. D. et al. In vivo profiling of metastatic double knockouts through CRISPR–Cpf1 screens. Nat. Methods 16, 405 (2019).
Xu, H. et al. Sequence determinants of improved CRISPR sgRNA design. Genome Res. 25, 1147–1157 (2015).
Doench, J. G. et al. Optimized sgRNA design to maximize activity and minimize off-target effects of CRISPR–Cas9. Nat. Biotechnol. 34, 184–191 (2016).
Hart, T. et al. Evaluation and design of genome-wide CRISPR/SpCas9 knockout screens. G3 (Bethesda) 7, 2719–2727 (2017).
Listgarten, J. et al. Prediction of off-target activities for the end-to-end design of CRISPR guide RNAs. Nat. Biomed. Eng. 2, 38–47 (2018).
Kim, H. K. et al. In vivo high-throughput profiling of CRISPR–Cpf1 activity. Nat. Methods 14, 153–159 (2017).
Kim, H. K. et al. Deep learning improves prediction of CRISPR–Cpf1 guide RNA activity. Nat. Biotechnol. 36, 239–241 (2018).
Moder, M. et al. Parallel genome-wide screens identify synthetic viable interactions between the BLM helicase complex and Fanconi anemia. Nat. Commun. 8, 1238 (2017).
Bertomeu, T. et al. A high-resolution genome-wide CRISPR/Cas9 viability screen reveals structural features and contextual diversity of the human cell-essential proteome. Mol. Cell. Biol. 38, e00302–17 (2017).
Morgens, D. W. et al. Genome-scale measurement of off-target activity using Cas9 toxicity in high-throughput screens. Nat. Commun. 8, 15178 (2017).
Meyers, R. M. et al. Computational correction of copy number effect improves specificity of CRISPR–Cas9 essentiality screens in cancer cells. Nat. Genet. 49, 1779–1784 (2017).
Slovackova, J., Smarda, J. & Smardova, J. Roscovitine-induced apoptosis of H1299 cells depends on functional status of p53. Neoplasma 59, 606–612 (2012).
Gu, Z. et al. Role of duplicate genes in genetic robustness against null mutations. Nature 421, 63–66 (2003).
Ewen-Campen, B., Mohr, S. E., Hu, Y. & Perrimon, N. Accessing the phenotype gap: enabling systematic investigation of paralog functional complexity with CRISPR. Dev. Cell 43, 6–9 (2017).
Costanzo, M. et al. A global genetic interaction network maps a wiring diagram of cellular function. Science 353, aaf1420 (2016).
Bassik, M. C. et al. A systematic mammalian genetic interaction map reveals pathways underlying ricin susceptibility. Cell 152, 909–922 (2013).
Viswanathan, S. R. et al. Genome-scale analysis identifies paralog lethality as a vulnerability of chromosome 1p loss in cancer. Nat. Genet. 50, 937–943 (2018).
Meyer, C. et al. The TIA1 RNA-binding protein family regulates EIF2AK2-mediated stress response and cell cycle progression. Mol. Cell 69, 622–635.e6 (2018).
Saxton, R. A. & Sabatini, D. M. mTOR signaling in growth, metabolism, and disease. Cell 168, 960–976 (2017).
Valvezan, A. J. & Manning, B. D. Molecular logic of mTORC1 signalling as a metabolic rheostat. Nat. Metab. 1, 321–333 (2019).
Thoreen, C. C. et al. An ATP-competitive mammalian target of rapamycin inhibitor reveals rapamycin-resistant functions of mTORC1. J. Biol. Chem. 284, 8023–8032 (2009).
Koo, J., Yue, P., Deng, X., Khuri, F. R. & Sun, S.-Y. mTOR complex 2 stabilizes Mcl-1 protein by suppressing its glycogen synthase kinase 3-dependent and SCF-FBXW7-mediated degradation. Mol. Cell. Biol. 35, 2344–2355 (2015).
Koo, J., Yue, P., Gal, A. A., Khuri, F. R. & Sun, S.-Y. Maintaining glycogen synthase kinase-3 activity is critical for mTOR kinase inhibitors to inhibit cancer cell growth. Cancer Res. 74, 2555–2568 (2014).
Shu, L. & Houghton, P. J. The mTORC2 complex regulates terminal differentiation of C2C12 myoblasts. Mol. Cell. Biol. 29, 4691–4700 (2009).
Martin, T. D. et al. Ral and Rheb GTPase activating proteins integrate mTOR and GTPase signaling in aging, autophagy, and tumor cell invasion. Mol. Cell 53, 209–220 (2014).
Sakamuro, D., Elliott, K. J., Wechsler-Reya, R. & Prendergast, G. C. BIN1 is a novel MYC-interacting protein with features of a tumour suppressor. Nat. Genet. 14, 69–77 (1996).
Pineda-Lucena, A. et al. A structure-based model of the c-Myc/Bin1 protein interaction shows alternative splicing of Bin1 and c-Myc phosphorylation are key binding determinants. J. Mol. Biol. 351, 182–194 (2005).
Ge, K. et al. Mechanism for elimination of a tumor suppressor: aberrant splicing of a brain-specific exon causes loss of function of Bin1 in melanoma. Proc. Natl Acad. Sci. USA 96, 9689–9694 (1999).
Ashworth, A., Lord, C. J. & Reis-Filho, J. S. Genetic interactions in cancer progression and treatment. Cell 145, 30–38 (2011).
Costanzo, M. et al. Global genetic networks and the genotype-to-phenotype relationship. Cell 177, 85–100 (2019).
Horlbeck, M. A. et al. Mapping the genetic landscape of human cells. Cell 174, 953–967.e22 (2018).
Dvinge, H., Kim, E., Abdel-Wahab, O. & Bradley, R. K. RNA splicing factors as oncoproteins and tumour suppressors. Nat. Rev. Cancer 16, 413–430 (2016).
Quesnel-Vallières, M., Weatheritt, R. J., Cordes, S. P. & Blencowe, B. J. Autism spectrum disorder: insights into convergent mechanisms from transcriptomics. Nat. Rev. Genet. 20, 51–63 (2019).
Scotti, M. M. & Swanson, M. S. RNA mis-splicing in disease. Nat. Rev. Genet. 17, 19–32 (2016).
Zhao, D. et al. Combinatorial CRISPR-Cas9 metabolic screens reveal critical redox control points dependent on the KEAP1-NRF2 regulatory axis. Mol. Cell 69, 699–708.e7 (2018).
Gasperini, M. et al. CRISPR/Cas9-mediated scanning for regulatory elements required for HPRT1 expression via thousands of large, programmed genomic deletions. Am. J. Hum. Genet. 101, 192–205 (2017).
Boettcher, M. et al. Dual gene activation and knockout screen reveals directional dependencies in genetic networks. Nat. Biotechnol. 36, 170–178 (2018).
Liu, J. et al. Pooled library screening with multiplexed Cpf1 library. Nat. Commun. 10, 3144 (2019).
Campa, C. C., Weisbach, N. R., Santinha, A. J., Incarnato, D. & Platt, R. J. Multiplexed genome engineering by Cas12a and CRISPR arrays encoded on single transcripts. Nat. Methods 16, 887–893 (2019).
Sanson, K. R. et al. Optimization of AsCas12a for combinatorial genetic screens in human cells. Preprint at bioRxiv https://doi.org/10.1101/747170 (2019).
Kleinstiver, B. P. et al. Engineered CRISPR–Cas12a variants with increased activities and improved targeting ranges for gene, epigenetic and base editing. Nat. Biotechnol. 37, 276–282 (2019).
Joung, J. et al. Genome-scale CRISPR–Cas9 knockout and transcriptional activation screening. Nat. Protoc. 12, 828–863 (2017).
Dang, Y. et al. Optimizing sgRNA structure to improve CRISPR-Cas9 knockout efficiency. Genome Biol. 16, 280 (2015).
Guschin, D. Y. et al. A rapid and general assay for monitoring endogenous gene modification. Methods Mol. Biol. 649, 247–256 (2010).
Singh, P. P., Arora, J. & Isambert, H. Identification of ohnolog genes originating from whole genome duplication in early vertebrates, based on synteny comparison across multiple genomes. PLoS Comput. Biol. 11, e1004394 (2015).
Hubbard, K. S., Gut, I. M., Lyman, M. E. & McNutt, P. M. Longitudinal RNA sequencing of the deep transcriptome during neurogenesis of cortical glutamatergic neurons from murine ESCs. F1000Res. 2, 35 (2013).
Raj, B. et al. A global regulatory mechanism for activating an exon network required for neurogenesis. Mol. Cell 56, 90–103 (2014).
Tapial, J. et al. An atlas of alternative splicing profiles and functional associations reveals new regulatory programs and genes that simultaneously express multiple major isoforms. Genome Res. 27, 1759–1768 (2017).
Hanna, R. E. & Doench, J. G. A case of mistaken identity. Nat. Biotechnol. 36, 802–804 (2018).
Feldman, D., Singh, A., Garrity, A. J. & Blainey, P. C. Lentiviral co-packaging mitigates the effects of intermolecular recombination and multiple integrations in pooled genetic screens. Preprint at bioRxiv https://doi.org/10.1101/262121 (2018).
Lorenz, R. et al. ViennaRNA Package 2.0. Algorithms Mol. Biol. 6, 26 (2011).
Cock, P. J. A. et al. Biopython: freely available Python tools for computational molecular biology and bioinformatics. Bioinformatics 25, 1422–1423 (2009).
ENCODE Project Consortium. An integrated encyclopedia of DNA elements in the human genome. Nature 489, 57–74 (2012).
Pedregosa, F. et al. Scikit-learn: machine learning in python. J. Mach. Learn. Res. 12, 2825–2830 (2011).
Abadi, M. et al. TensorFlow: large-scale machine learning on heterogeneous distributed systems. Preprint at arXiv https://arxiv.org/abs/1603.04467 (2016).
Bergstra, J. & Bengio, Y. Random search for hyper-parameter optimization. J. Mach. Learn. Res. 13, 281–305 (2012).
Eden, E. et al. GOrilla: a tool for discovery and visualization of enriched GO terms in ranked gene lists. BMC Bioinformatics 10, 48 (2009).
Berriz, G. F., King, O. D., Bryant, B., Sander, C. & Roth, F. P. Characterizing gene sets with FuncAssociate. Bioinformatics 19, 2502–2504 (2003).
Acknowledgements
We thank Q. Morris for advice on machine learning implementation, F. Zhang and B. Zetsche (Broad Institute, MIT) for providing Cas12a reagents and insights, and members of the Moffat, Blencowe and Myers laboratories for helpful discussions. G. O’Hanlon, Q. Huang, C. Sheene and S. Sidhu are gratefully acknowledged for assistance with sequencing and molecular biology experiments. T.G.-P. was supported by postdoctoral fellowships from the European Molecular Biology Organization, Ontario Institute of Regenerative Medicine (OIRM) and Canadian Institutes for Health Research. M.A. was supported by a Swiss National Science Foundation fellowship. H.N.W. was supported by a National Institutes of Health (NIH) Biotechnology Training Grant. This research was funded by grants from Canadian Institutes for Health Research (to B.J.B. and grant no. MOP-142375 to J.M.), Medicine-by-Design Canada First Research Excellence Fund (grant no. CITPA-2016-11 to B.J.B. and J.M.), OIRM (to B.J.B.), Genome Canada (grant no. OGI-157 to J.M.), NIH (to C.L.M.) and the National Science Foundation (to C.L.M.). J.M. is a Canadian Research Chair in Functional Genomics. B.J.B. holds the Banbury Chair of Medical Research at the University of Toronto.
Author information
Authors and Affiliations
Contributions
T.G.P., M.A., J.M. and B.J.B. conceptualized the study and study design. Initial conceptualization, development and demonstration of efficacy of the hybrid Cas9–Cas12a system were performed by T.G.-P. and S.F. Experimental investigation was carried out by T.G.-P., M.A., S.F. and A.W. Guide library design and data analysis were performed by K.R.B., U.B., H.N.W., M.A., T.G.-P., T.D., M.B. and J.M. Machine learning implementation was carried out by K.C.H. The original draft was written by T.G.-P., M.A., J.M. and B.J.B. with input from K.R.B. Writing, reviewing and editing were carried out by T.G.-P., M.A., K.R.B., J.M. and B.J.B. with input from the other authors. Supervision was done by B.J.B., J.M., C.L.M. and T.G.-P. Funding was acquired by B.J.B., J.M., C.L.M. and T.G.-P.
Corresponding authors
Ethics declarations
Competing interests
A patent application (no. GB 1907733.8) describing the development and applications of CHyMErA, to the University of Toronto and T.G.-P., M.A., K.R.B., S.F., J.M. and B.J.B., is pending.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated Supplementary Information
Supplementary Figure 1 Generation of dual Cas9 gRNA expression vectors for exon deletion.
(a) Schematic of targeting Ptbp1 exon 8 for deletion (top panel) and of dual Cas9 gRNA expression cassettes (bottom panel). (b) PCR monitoring of Ptbp1 exon 8 deletion in CGR8 cells transiently transfected (top panel) or transduced (bottom panel) with dual Cas9 guides (see Supplementary Fig. 1a). Representative data from two independent experiments. (c) Immunofluorescence analysis of N2A cells transiently transfected or stably transduced with lenti-Lb- or As-Cas12a containing a single nuclear localization signal (left panel). Immunofluorescence analysis of stably transduced N2A cells with lenti-Lb- or As-Cas12a containing two nuclear localization signals (right panel). Scale corresponds to 27 µm. Representative data from two independent experiments. (d) Western blot analysis of Cas9 and Cas12a in stably transduced N2A, CGR8, HAP1 and RPE1 cells, as indicated. Asterisk indicates non-specific signal. Representative data from two independent experiments. (e) hgRNA pre-crRNA processing based on qRT-PCR analysis. The strategy used for the quantification is indicated below the panel. All data are represented as mean ± SD (n = 3 replicates). (f) PCR monitoring of exon deletion from Parp6 and HPRT1 genes in the indicated cell lines using CHyMErA. Independent pLCHKO constructs expressing Cas9 and Cas12a gRNAs targeting flanking intronic sites for exon deletions or controls were used as indicated. Representative data from two independent experiments. (g) Relative cell viability following sequential drug treatments (thymidine and 6-thioguanine) of HAP1 cells transduced with pLCHKO vectors expressing hgRNAs targeting TK1 and HPRT1, as indicated in the schematic on the left. As described in Fig. 1c, for all hgRNA constructs, the first and last positions encode a TK1-targeting Cas9 and HPRT1-targeting Cas12a gRNA, respectively, while the intervening positions encode intergenic Cas12a gRNAs. After subjecting cells to the first drug treatment, cells were passaged at an equal ratio and challenged with the second drug treatment. Cell viability was assessed following both treatments using an AlamarBlue assay. Data represented as mean ± SD, n = 3 independent biological replicates. (h) Enrichment of intergenic, exonic and intronic HPRT1 targeting hgRNAs in non-treated (NT) or 6-TG treated HAP1 cells (pairwise two-tailed Mann-Whitney U test with Holm multiple testing correction). Data derived from n = 3 independent technical replicates. Boxes show the interquartile range (IQR - 25th to 75th percentile), with the median indicated by a line. The whiskers extend to the quartile +/- 1.5 x IQR. (i) Enrichment of paired guides targeting an exon in TK1 for deletion (red), or gene knockout (black = Cas9, blue = Cas12a) in double-thymidine block-treated (y-axis) vs. non-treated (x-axis) HAP1 cells at the end time point (T18). Intronic/intergenic control hgRNAs are indicated in yellow and non-TK1 targeting constructs detectable in at least one Thymidine-treated replicate (n=43,117 sequences) are shown in grey. Screens performed with Lb-Cas12a and As-Cas12a are depicted in the top and bottom panel, respectively.
Supplementary Figure 2 Feature analysis of Cas12a guides.
(a) Schematic of exon-targeting hgRNA approach with CHyMErA. (b) HgRNA screening libraries were generated by performing two rounds of Golden Gate assembly. During the first step the synthesized 113-nt oligos containing both Cas9 and Cas12a guides were introduced into pLHCKO. During the second step, the spacer sequence between the two oligos was replaced with a hybrid scaffold consisting of the Cas9 tracrRNA followed by the Lb- or As-Cas12a direct repeat (DR). Schematic of Cas9 and Cas12a guide length, PAM sequence and double-stranded DNA cutting pattern is indicated at the bottom. (c) Dropout of Cas9 or Cas12a guides targeting essential genes in CGR8 cells. (d) Exonic Lb-Cas12a guides were grouped based on LFC cut-offs in the HAP1 and CGR8 optimization screens. Strongly depleting guides were use as positive, and neutral guides as negative cases. (e) Precision-recall (left panel) and receiver operating characteristic (right panel) curves of different machine-learning approaches for predicting Cas12a guide performance in HAP1 and CGR8 cells. CNN: convolutional neural networks; L1Logit: lasso regularized logistic regression. (f) Weblogo of filters learned by CHyMErA-Net in the convolutional layer. (g) Performance of exonic Lb-Cas12a hgRNAs grouped according to their PAM sequence. (n = 1,237 TTTA, 1,882 TTTC, 1,547 TTTG). Data derived from n = 3 independent technical replicates. Boxes show the interquartile range (IQR - 25th to 75th percentile), with the median indicated by a line. The whiskers extend to the quartile +/- 1.5 x IQR. (h) Enrichment analysis of active and inactive Lb-Cas12a guides based on chromatin accessibility from K562 cells.
Supplementary Figure 3 Second-generation CHyMErA libraries provide enhanced gene targeting.
(a) Correlation between the mean LFC of hgRNA-targeted genes in HAP1 and RPE1 cells at the T18 timepoint. HgRNAs targeting core essential genes (CEG2) are indicated in red and all other hgRNAs are indicated in blue. (b) LFC distribution of hgRNAs targeting intergenic regions and/or non-targeting (NT) regions in HAP1 and RPE1 cells. Each box comprises 92 hgRNAs except the intergenic:intergenic controls, which comprise 4,993 constructs. Distributions compared by two-tailed Wilcoxon rank-sum tests with Benjamini-Hochberg multiple testing correction. Data derived from n = 3 independent technical replicates. Boxes show the interquartile range (IQR, 25th to 75th percentile), with the median indicated by horizontal line. Whiskers extend to the quartile ±1.5 IQR. (c) Enhanced depletion of genes using the dual-targeting hgRNAs compared to single-targeting Cas9 guides. (d) Dropout profiles of dual-targeting hgRNAs, as measured by the LFC at T18 in the HAP1 cell line, were binned into ten equal sized bins (n = 1,093 – 1,097) according to the distance between Cas9 and Cas12a target sites. Data derived from n = 3 independent technical replicates. Boxes show the interquartile range (IQR - 25th to 75th percentile), with the median indicated by a line. The whiskers extend to the quartile +/- 1.5 x IQR. (e) Western blot depicting p53, pRb and p21 protein levels following camptothecin treatment in RPE1 CHyMErA cells transduced or not with hgRNA constructs. Representative data of two independent experiments. (f) CERES scores from the DepMap CRISPR screens are shown for CEG2 (red) and non-essential (black) genes, genes discovered by both single- (ST) and dual-targeting (DT) (yellow), or genes discovered only through dual-targeting by CHyMErA (blue). Lower CERES scores correspond to greater depletion through the screens. CERES scores for each gene set across all 558 screens were aggregated together for plotting: red – 367,164 scores corresponding to 658 genes, yellow – 990,450 scores from 1,775 genes, blue – 313,038 scores from 561 genes, black – 435,798 scores from 781 genes. CERES score distributions for CHyMErA DT-only genes (n = 313,038) and non-essential genes (n = 435,798) were compared using a two-tailed Wilcoxon rank-rum test.
Supplementary Figure 4 CHyMErA reveals widespread non-additive fitness phenotypes upon combinatorial perturbation of paralogous genes.
(a-b) Enrichment or depletion of single or combinatorial gene ablations as indicated. The expected combinatorial effect size based on single perturbation is indicated with dotted bars. All data are represented as mean ± 2*SEM. Number of hgRNAs: MDM4, MCL1, KDM6B – 8; TP53, BCL2L1, BRD4 – 16; paralog pairs – 30. (c-d) Expected vs observed LFC of paralog pairs in HAP1 (c) or RPE1 (d) cells. Paralogs displaying significant genetic interaction at both time points, or only at the late time point, are highlighted in red and orange, respectively. Two-tailed Wilcoxon rank-sum test with Benjamini–Hochberg multiple testing correction, n = 3 independent technical replicates. (e-f) Difference (residual) between the mean observed and expected LFCs for orientation 1 against orientation 2 of paralog pairs in HAP1 (e) or RPE1 (f) cells (left panel). Paralogs with significant genetic interactions at the early or both time points are highlighted in yellow and red, respectively. Two-tailed Wilcoxon rank-sum test, Benjamini–Hochberg multiple testing correction, n = 3 independent technical replicates. The density of FDR values for all gene pairs in both orientations are also displayed, and the significance threshold of 0.1 is indicated as a dashed line (right panel). (g-h) Enrichment or depletion of single or combinatorial gene ablations in HAP1 (g) or RPE1 (h), as indicated. LFC represented as mean ± 2*SEM from n = 3 independent technical replicates. (i) Cell viability of WT and single-knockout HAP1 clones was measured by AlamarBlue staining 6 days post-transduction of two independent lentiCRISPRv2 gRNA expression cassettes targeting the indicated genes. Cell viability was normalized to intergenic-targeting control gRNAs. All data are represented as mean ± SD (n = 3 independent biological replicates; two-tailed t test). (j) Real-time RT-PCR was performed to quantify RBM26 and RBM27 knock-down efficiency in HAP1 cells. All data are represented as mean ± SD (n = 3 independent biological replicates; two-tailed t test). (k) Cell viability of HAP1 and RPE1 cells was measured by AlamarBlue staining 3 days post-transfection of siRNAs targeting RBM26, RBM27 or both. All data are represented as mean ± SD (n = 5 and 3 independent biological replicates for HAP1 and RPE1, respectively; two-tailed t test). (l) Gene ontology enrichment analysis for genes with significantly decreased expression upon co-depletion of RBM26 and RBM27 following siRNA treatment. (n = 2 independent biological replicates. FDR was calculated using FuncAssociate (Berriz et al., Bioinformatics, 2003).
Supplementary Figure 5 CHyMErA outperforms single Cas9 targeting chemo-genetic screens.
(a) Differential LFC of genes perturbed by single- (left panel) and dual-targeting (right panel) hgRNAs upon Torin1 treatment in HAP1 cells at the early time point (T12). Sensitizer (blue) and suppressor gene hits (yellow) are highlighted (FDR < 0.01, two-tailed Wilcoxon rank-sum test with Benjamini–Hochberg multiple testing correction, n = 3 independent technical replicates) and the top 10 as well as selected genes from the top 20 significant hits are listed. (b) Torin1 IC50 values (drug concentration resulting in 50% reduction of cell viability) in HAP1 WT and EED knockout cell clones. IC50 values were calculated based on dose response curves in the respective HAP1 cell lines. Data represented as mean ± SD (n = 4 independent biological replicates; p=0.026, two-tailed t test). (c) Differential LFC of paralogs perturbed by single- (left panel) and combinatorial-targeting (right panel) hgRNAs upon Torin1 treatment in HAP1 cells at the early time point (T12). Sensitizer (blue) and suppressor gene hits (yellow) are highlighted (FDR < 0.01, two-tailed Wilcoxon rank-sum test with Benjamini–Hochberg multiple testing correction, n = 3 independent samples) and the top 10 as well as selected genes from the top 20 significant hits are listed. (d) Gene ontology enrichment of sensitizer (upper panel) or suppressor hits (lower panel) identified at an FDR < 0.1 across both time points (n = 3 independent technical replicates). FDR was calculated using GOrilla (Eden et al., BMC Bioinformatics, 2009). (e) Differential LFC of diphthamide biosynthesis genes perturbed by single- or dual-targeting hgRNAs as indicated. Two-tailed Wilcoxon rank-sum test with Benjamini–Hochberg multiple testing correction, n = 3 independent technical replicates.
Supplementary Figure 6 CHyMErA is suitable for exon-deletion phenotypic screens.
(a) The LFC of exon-deletion hgRNAs (intronic/intronic) vs. control hgRNAs in which only the Cas9 (left) or Cas12a guide (right) is targeting an intronic region, while the other nuclease is targeting an intergenic region. The red dots represent exon-deletion hgRNAs that are significantly depleted, while grey dots represent all other exon-deletion hgRNAs. Significant depletion was scored against the empirical null distribution of 1,647 intergenic-intergenic control pairs (refer to Methods for details). Marginal histograms indicate the density distribution of control guide pairs corresponding to significant and non-significant exon-deletion pairs, respectively. (b) The density of exonic ‘hits’ (orange) compared to all other exons (grey) from the exon-deletion screen as a function of PSI (percent spliced in). p-value is from a two-tailed Mann-Whitney U test (n = 91 for hits, 1,514 for background). (c) Length distribution of the alternative exons targeted by CHyMErA exon-deletion library. p-value is from a two-tailed Mann-Whitney U test (n = 91 for hits, 1,514 for background). (d) Percentage of alternative exons that overlap a modular protein domain. (e) PCR monitoring of exon deletion from PDPR, MDM4 and SRFS7 genes in RPE1 cells using hgRNAs guides with different phenotypic scores. Representative data of two independent experiments. (f) Representative examples of hgRNA constructs targeting frame-disruptive exons in BIN1, FUZ, FHOD3, MEGF8, TNRC6A or C1orf77 are shown above the gene model (x-axis), with the observed LFC value for each hgRNA (y-axis). Exon deletion (that is intronic-intronic) and single-targeting control (that is intronic-intergenic) hgRNAs are indicated in different colours, while significantly depleted hgRNAs are highlighted and numbers of significant and total exon deletion pairs are indicated in the top right corner. Significant depletion was scored against the empirical null distribution of 1,647 intergenic-intergenic control pairs (refer to Methods for details).
Supplementary Figure 7 Comparison of CHyMErA with other dual-targeting screening systems.
(a) PCR monitoring of exon deletion from Ptbp1 and HPRT1 genes in the indicated cell lines using CHyMErA or BigPapi. Independent pLCHKO and pPapi constructs expressing Sp-Cas9 and Cas12a (CHyMErA) or Sa-Cas9 (BigPapi) gRNAs targeting flanking intronic sites for exon deletions or controls were used as indicated. Representative data of two independent experiments. (b) Schematic of combinatorial gene targeting by CHyMErA (left panel) or BigPapi (middle panel). Comparison between CHyMErA and the BigPapi system for the combinatorial targeting of TK1 and HPRT1, as determined by resistance to thymidine and 6-thioguanine treatments, respectively (right panel). The same Cas9 guide targeting TK1 was used for CHyMErA and all BigPapi constructs. Data represented as mean ± SD, n = 3 independent biological replicates. (c) Table summarizing the key characteristics and applications of dual-targeting CRISPR screening systems.
Supplementary information
Supplementary Figures
Supplementary Figs. 1–8.
Supplementary Table 1
Human hgRNA optimization library. Human first-generation CHyMErA optimization library. Columns include the following information: hgRNA ID (A); type of hgRNA (that is, dual-targeting of individual genes or paralog targeting) (B); targeted gene name (C); targeted exon coordinates (D); targeted exon number (E); Cas9 guide coordinates (F); Cas9 guide sequence (G); type of targeted site (that is, intronic, exonic, intergenic) by Cas9 guide (H); Cas12a guide coordinates (I); Cas12a guide sequence (J); type of targeted site (that is, intronic, exonic, intergenic) by Cas12a guide (K); Cas12a PAM sequence (L).
Supplementary Table 2
Mouse hgRNA optimization library. Mouse first-generation CHyMErA optimization library. Columns include the following information: hgRNA ID (B); type of hgRNA (that is, dual-targeting of individual genes or paralog targeting) (C); targeted gene name (D); targeted exon coordinates (E); targeted exon number (F); Cas9 guide coordinates (G); Cas9 guide sequence (H); type of targeted site (that is, intronic, exonic, intergenic) by Cas9 guide (I); Cas12a guide coordinates (J); Cas12a guide sequence (K); type of targeted site (that is, intronic, exonic, intergenic) by Cas12a guide (L); Cas12a PAM sequence (M).
Supplementary Table 3
Human hgRNA optimization library screening results. HAP1 optimization screen. Screens were performed in technically independent triplicates (single transduction at T0, split into three arms). Normalized read counts for each time point and across the three replicates are shown. HgRNAs that failed to exceed CPM = 2 in at least two samples were removed (n = 3,548). Mean LFC summary of each time point (T6, T12 and T18) and statistical comparison performed using two-tailed exactTest in edgeR package. Benjamini–Hochberg multiple testing correction FDRs are provided.
Supplementary Table 4
Mouse hgRNA optimization library screening results. CGR8 optimization screen. Screens were performed in technically independent triplicates (single transduction at T0, split into three arms). Normalized read counts for each time point and across three replicates are shown. HgRNAs that failed to exceed CP = 2 in at least two samples were removed (n = 4,162). Mean LFC summary of each time point (T6, T12 and T18) and statistical comparison performed using two-tailed exactTest in edgeR package. Benjamini–Hochberg multiple testing correction FDRs are provided.
Supplementary Table 5
Human second-generation library (including RNA-seq data across five cell lines). Sheet 1: human second-generation CRISPR dual-targeting and paralog library. Columns include the following information: hgRNA ID (A); type of hgRNA (that is, dual-targeting of individual genes or paralog targeting) (B); targeted gene name (C,D); Cas9 guide coordinates (E); source of Cas9 guide (F); Cas9 guide sequence (G); type of site targeted by Cas9 guide (that is, exonic, intergenic, nontargeting) (H); Cas12a guide coordinates (I); Cas12a guide sequence (J); type of site targeted by Cas12a guide (that is, exonic, intergenic, nontargeting) (K); Cas12a guide CHyMErA-Net score (L). Sheet 2: RNA-seq expression data used to select expressed paralog pairs. Columns include the following information: gene name and IDs (A–C); paralog symbol and family (D,E); cell lines where paralogs are expressed (F); log2(FPKM) of genes across different cell lines and media conditions (G–Q).
Supplementary Table 6
Human second-generation library screening results. Human second-generation dual-targeting and paralog CRISPR screen data. Normalized read counts for each replicate. Screen performed in technically independent triplicates (single transduction T0, split into three independent arms). Mean LFC values for each hgRNA present in the library (see Supplementary Table 5) computed for each condition represented.
Supplementary Table 7
Fitness genes identified by single- versus dual-targeting of individual genes. List of genes found to be essential in dual-targeting screens at different time points as indicated. Screen performed in technically independent triplicates (common transduction at T0, split into three independent arms). Essential genes identified by MAGeCK with beta score < 0 and FDR < 0.05.
Supplementary Table 8
Paralog scoring. Effect size and statistical evaluation of the human paralog screen in HAP1 and RPE1 cells. Screens performed in technically independent triplicates (common transduction at T0, split into three independent arms). Effect size represents mean LFC at early (T12 for HAP1, T18 for RPE1) or late (T18 for HAP1, T24 for RPE1) time points. Columns in the summary sheet describe: targeted gene pairs (A,B); type of observed GI across the two time points (none, shared, unique-early, unique-late) (C–E); early and late effect size (F–K); overlap of observed GIs between nontreated (–Torin) and Torin-treated (+Torin) HAP1 cells (L). The other sheets include detailed information for each cell line and time point: targeted gene pairs (A,B); expected LFC for combinatorial gene targeting based on addition of single-targeting phenotypes in both orientations (C,D); observed LFC for combinatorial gene targeting in both orientations (E,F); FDR for differences between observed and expected LFC for both orientations, calculated using two-tailed Wilcoxon rank-sum test and Benjamini–Hochberg multiple testing correction (G,H); mean expected and observed LFC for both orientations (K,L); significant FDR in both interactions at early and late time points (M,N); early and late effect size accounting for the difference between observed and expected LFC (O,P); significant FDR at both time points (Q); observed type of GI (S,T); gene expression of gene pairs (R); observed LFC of gene 1 single-targeted by Cas9 or Cas12a (W); mean LFC of single-targeted gene 1 (U,V); observed LFC of gene 2 single-targeted by Cas9 or Cas12a (X,Y); mean LFC of single-targeted gene 1 (Z); observed LFC for combinatorial gene targeting in both orientations (AA,AB); mean LFC for combinatorial gene targeting in both orientations (AC).
Supplementary Table 9
Gene expression analysis following RBM26/RBM27 knockdown in HAP1 cells. RNA-seq analysis of bulk gene expression following RBM26 and/or RBM27 knockdown in HAP1 cells. Columns include Ensembl gene ID (A) and gene symbol (B), and RPKM values, LFC, P values and FDR of control (siNT, nontargeting) and single- or double-knockdowns, as indicated (C–O). Comparisons performed using the two-tailed exactTest function in the Bioconductor package edgeR, followed by Benjamini–Hochberg multiple testing correction.
Supplementary Table 10
Gene Ontology enrichment analysis of differentially expressed genes following RBM26 and/or RBM27 knockdown. Gene Ontology enrichment analysis of differentially expressed genes (upregulated, sheet 1; downregulated, sheet 2) using FuncAssociate. Each column is defined on Sheet 3 in the workbook.
Supplementary Table 11
Torin1 drug sensitivity scoring. The HAP1 Torin1 drug sensitivity screen was performed in technically independent triplicates (common transduction at T0, split into three independent arms). Effect size represents mean LFC at early (T12) or late (T18) time points. Data are displayed separately for single-, dual- and combinatorial (paralog) gene-targeting hgRNAs. Columns include the following: targeted gene name (A); observed LFC of hgRNAs in treated and nontreated cells (B,C); P values for residual LFC (D); residual LFC representing difference between the two treatments (E); FDR (F); significance of residual effect size (G); category of screen hit (none, sensitizer, suppressor) (H); and hit ranking (I). Data were analyzed using a two-tailed Wilcoxon rank-sum test followed by Benjamini–Hochberg multiple testing correction (see Methods).
Supplementary Table 12
Human exon-targeting library. Human exon-deletion CHyMErA library designs. Columns include the following information: event ID (A); hgRNA purpose (B); gene symbol (C); exon coordinates (D); Cas9 guide coordinates (E); Cas9 guide sequence (F); Cas9 guide position (UP: upstream of exon, DN: downstream of exon) and targeted genetic segment type (intronic/exonic/ intergenic) (G); Cas12a guide coordinates (H); Cas12a guide sequence (I); Cas12a guide position (UP: upstream of exon, DN: downstream of exon) and targeted genetic segment type (intronic/exonic/ intergenic) (J); Cas12a guide CHyMErA-Net score (K).
Supplementary Table 13
Human exon-targeting library screen results. LFC scores of exons targeted by CHyMErA in RPE1 cells during a fitness/dropout screen. Columns include the following information: Ensembl ID (A); gene symbol (B); exon ID (C); exon coordinates (D); exon length (E); exon PSI (percentage spliced in) in RPE1 cells (F); transcript cRPKM in RPE1 cells (G); gene fitness score (LFC) in RPE1 cells (H); exon characteristics (for example, frame-preserving or frame-disruptive (I,J); indication of regulation during cell cycle (K); number of hgRNAs in the library designed for exon deletion (L); number of hgRNAs with significant LFC (M); number of single-targeting control hgRNAs (N); number of single-targeting control hgRNAs with significant LFC (O); number of hgRNAs targeting exonic sequences (P); number of hgRNAs targeting exonic sequences with significant LFC (Q); fraction of hgRNAs with significant LFC (R); hit classification (S).
Supplementary Table 14
Primers and oligos. List of primers and oligos used for cloning, PCR and high-throughput sequencing.
Rights and permissions
About this article
Cite this article
Gonatopoulos-Pournatzis, T., Aregger, M., Brown, K.R. et al. Genetic interaction mapping and exon-resolution functional genomics with a hybrid Cas9–Cas12a platform. Nat Biotechnol 38, 638–648 (2020). https://doi.org/10.1038/s41587-020-0437-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41587-020-0437-z
This article is cited by
-
An Update on the Application of CRISPR Technology in Clinical Practice
Molecular Biotechnology (2024)
-
The function of alternative splicing in the proteome: rewiring protein interactomes to put old functions into new contexts
Nature Structural & Molecular Biology (2023)
-
Chromatin complex dependencies reveal targeting opportunities in leukemia
Nature Communications (2023)
-
CRISPR screens for functional interrogation of immunity
Nature Reviews Immunology (2023)
-
Optimized metrics for orthogonal combinatorial CRISPR screens
Scientific Reports (2023)