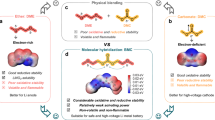
Abstract
Acidic oxygen evolution reaction is crucial for practical proton exchange membrane water splitting electrolysers, which have been hindered by the high catalytic overpotential and high loading of noble metal catalysts. Here we present a torsion-strained Ta0.1Tm0.1Ir0.8O2-δ nanocatalyst with numerous grain boundaries that exhibit a low overpotential of 198 mV at 10 mA cm−2 towards oxygen evolution reaction in 0.5 M H2SO4. Microstructural analyses, X-ray absorption spectroscopy and theoretical calculations reveal that the synergistic effects between grain boundaries that result in torsion-strained Ir–O bonds and the doping induced ligand effect collectively tune the adsorption energy of oxygen intermediates, thus enhancing the catalytic activity. A proton exchange membrane electrolyser using a Ta0.1Tm0.1Ir0.8O2-δ nanocatalyst with a low mass loading of 0.2 mg cm−2 can operate stably at 1.5 A cm−2 for 500 hours with an estimated cost of US$1 per kilogram of H2, which is much lower than the target (US$2 per kg of H2) set by the US Department of Energy.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Data availability
All data needed to evaluate the conclusions in the paper are presented in the paper or the Supplementary Information. Source data are provided with this paper. Extra data are available from the corresponding author upon reasonable request.
References
Stamenkovic, V. R., Strmcnik, D., Lopes, P. P. & Markovic, N. M. Energy and fuels from electrochemical interfaces. Nat. Mater. 16, 57–69 (2017).
Li, F. et al. Molecular tuning of CO2-to-ethylene conversion. Nature 577, 509–513 (2020).
Kibsgaard, J. & Chorkendorff, I. Considerations for the scaling-up of water splitting catalysts. Nat. Energy 4, 430–433 (2019).
Carmo, M., Fritz, D. L., Mergel, J. & Stolten, D. A comprehensive review on PEM water electrolysis. Int. J. Hydrog. Energy 38, 4901–4934 (2013).
Lagadec, M. F. & Grimaud, A. Water electrolysers with closed and open electrochemical systems. Nat. Mater. 19, 1140–1150 (2020).
Nong, H. N. et al. Key role of chemistry versus bias in electrocatalytic oxygen evolution. Nature 587, 408–413 (2020).
Chatti, M. et al. Intrinsically stable in situ generated electrocatalyst for long-term oxidation of acidic water at up to 80 °C. Nat. Catal. 2, 457–465 (2019).
Seitz, L. C. et al. A highly active and stable IrOx/SrIrO3 catalyst for the oxygen evolution reaction. Science 353, 1011 (2016).
Blasco-Ahicart, M., Soriano-López, J., Carbó, J. J., Poblet, J. M. & Galan-Mascaros, J. R. Polyoxometalate electrocatalysts based on earth-abundant metals for efficient water oxidation in acidic media. Nat. Chem. 10, 24–30 (2018).
Yang, L. et al. Efficient oxygen evolution electrocatalysis in acid by a perovskite with face-sharing IrO6 octahedral dimers. Nat. Commun. 9, 5236 (2018).
Ping, Y., Nielsen, R. J. & Goddard, W. A. III The reaction mechanism with free energy barriers at constant potentials for the oxygen evolution reaction at the IrO2 (110) surface. J. Am. Chem. Soc. 139, 149–155 (2017).
Bessarabov, D. et al. Gas crossover mitigation in PEM water electrolysis: hydrogen cross-over benchmark study of 3M’s Ir-NSTF based electrolysis catalyst-coated membranes. ECS Trans. 75, 1165–1173 (2016).
Geiger, S. et al. The stability number as a metric for electrocatalyst stability benchmarking. Nat. Catal. 1, 508–515 (2018).
Man, I. C. et al. Universality in oxygen evolution electrocatalysis on oxide surfaces. Chem. Cat. Chem. 3, 1159–1165 (2011).
Bajdich, M., García-Mota, M., Vojvodic, A., Nørskov, J. K. & Bell, A. T. Theoretical investigation of the activity of cobalt oxides for the electrochemical oxidation of water. J. Am. Chem. Soc. 135, 13521–13530 (2013).
Suen, N. T. et al. Electrocatalysis for the oxygen evolution reaction: recent development and future perspectives. Chem. Soc. Rev. 46, 337–365 (2017).
Grimaud, A. et al. Activation of surface oxygen sites on an iridium-based model catalyst for the oxygen evolution reaction. Nat. Energy 2, 16189 (2016).
Gao, J. et al. Breaking long-range order in iridium oxide by alkali ion for efficient water oxidation. J. Am. Chem. Soc. 141, 3014–3023 (2019).
Diaz-Morales, O. et al. Iridium-based double perovskites for efficient water oxidation in acid media. Nat. Commun. 7, 12363 (2016).
Escudero-Escribano, M. et al. Tuning the activity of Pt alloy electrocatalysts by means of the lanthanide contraction. Science 352, 73–76 (2016).
You, B. et al. Enhancing electrocatalytic water splitting by strain engineering. Adv. Mater. 31, e1807001 (2019).
Li, M. et al. Ultrafine jagged platinum nanowires enable ultrahigh mass activity for the oxygen reduction reaction. Science 354, 1414 (2016).
Luo, M. et al. PdMo bimetallene for oxygen reduction catalysis. Nature 574, 81–85 (2019).
Shan, J. et al. Charge-redistribution-enhanced nanocrystalline Ru@IrOx electrocatalysts for oxygen evolution in acidic media. Chem. 5, 445–459 (2019).
Petrie, J. R., Jeen, H., Barron, S. C., Meyer, T. L. & Lee, H. N. Enhancing perovskite electrocatalysis through strain tuning of the oxygen deficiency. J. Am. Chem. Soc. 138, 7252–7255 (2016).
Wang, H. et al. Direct and continuous strain control of catalysts with tunable battery electrode materials. Science 354, 1031 (2016).
Tobias, R., Hong Nhan, N., Detre, T., Robert, S. & Strasser, P. Electrocatalytic oxygen evolution reaction in acidic environments—reaction mechanisms and catalysts. Adv. Energy Mater. 7, 1601275 (2017).
Zhu, Y. T., Liao, X. Z. & Wu, X. L. Deformation twinning in nanocrystalline materials. Prog. Mater. Sci. 57, 1–62 (2012).
Penn, R. L. & Banfield, J. F. Imperfect oriented attachment: dislocation generation in defect-free nanocrystals. Science 281, 969 (1998).
Mariano, R. G., McKelvey, K., White, H. S. & Kanan, M. W. Selective increase in CO2 electroreduction activity at grain-boundary surface terminations. Science 358, 1187 (2017).
Chhetri, M. et al. Mechanochemical synthesis of free-standing platinum nanosheets and their electrocatalytic properties. Adv. Mater. 27, 4430–4437 (2015).
Bøjesen, E. D. & Iversen, B. B. The chemistry of nucleation. Cryst. Eng. Comm. 18, 8332–8353 (2016).
De Yoreo, J. J. et al. Crystallization by particle attachment in synthetic, biogenic, and geologic environments. Science 349, aaa6760 (2015).
Morin, S. A., Bierman, M. J., Tong, J. & Jin, S. Mechanism and kinetics of spontaneous nanotube growth driven by screw dislocations. Science 328, 476 (2010).
Palumbo, G., Thorpe, S. & Aust, K. On the contribution of triple junctions to the structure and properties of nanocrystalline materials. Scr. Metall. Mater. 24, 1347–1350 (1990).
Hÿtch, M. J., Snoeck, E. & Kilaas, R. Quantitative measurement of displacement and strain fields from HREM micrographs. Ultramicroscopy 74, 131–146 (1998).
Freakley, S. J., Ruiz-Esquius, J. & Morgan, D. J. The X-ray photoelectron spectra of Ir, IrO2 and IrCl3 revisited. Surf. Interf. Anal. 49, 794–799 (2017).
Mesa, C. A. et al. Multihole water oxidation catalysis on haematite photoanodes revealed by operando spectroelectrochemistry and DFT. Nat. Chem. 12, 82–89 (2020).
Pham, H. H., Cheng, M. J., Frei, H. & Wang, L. W. Surface proton hopping and fast-kinetics pathway of water oxidation on Co3O4 (001) Surface. ACS Catal. 6, 5610–5617 (2016).
Anantharaj, S., Noda, S., Driess, M. & Menezes, P. W. The pitfalls of using potentiodynamic polarization curves for tafel analysis in electrocatalytic water splitting. ACS Energy Lett. 6, 1607–1611 (2021).
Cherevko, S., Geiger, S., Kasian, O., Mingers, A. & Mayrhofer, K. J. J. Oxygen evolution activity and stability of iridium in acidic media. Part 2—electrochemically grown hydrous iridium oxide. J. Electroanal. Chem. 774, 102–110 (2016).
Meng, G. et al. Strain regulation to optimize the acidic water oxidation performance of atomic-layer IrOx. Adv. Mater. 31, e1903616 (2019).
Exner, K. S. Design criteria for oxygen evolution electrocatalysts from first principles: introduction of a unifying material-screening approach. ACS Appl. Energy Mater. 2, 7991–8001 (2019).
Wang, M., Wang, Z., Gong, X. & Guo, Z. The intensification technologies to water electrolysis for hydrogen production—a review. Renew. Sustain. Energy Rev. 29, 573–588 (2014).
Bender, G. & Dinh, H. HydroGEN: Low-Temperature Electrolysis (LTE) and LTE/Hybrid Supernode (No. NREL/PR-5900-76549). National Renewable Energy Lab.(NREL), Golden, CO (United States, 2020).
Ravel, B. N. M. & Newville, M. Data analysis for X-ray absorption spectroscopy using IFEFFIT. J. Synchrotron Rad. 12, 537–541 (2005).
Kresse, G. & Furthmüller, J. Efficiency of ab-initio total energy calculations for metals and semiconductors using a plane-wave basis set. Comput. Mater. Sci. 6, 15–50 (1996).
Kresse, G. & Furthmüller, J. Efficient iterative schemes for ab initio total-energy calculations using a plane-wave basis set. Phys. Rev. B. 54, 11169–11186 (1996).
Perdew, J. P., Burke, K. & Ernzerhof, M. Generalized gradient approximation made simple. Phys. Rev. Lett. 77, 3865–3868 (1996).
Kresse, G. & Joubert, D. From ultrasoft pseudopotentials to the projector augmented-wave method. Phys. Rev. B. 59, 1758–1775 (1999).
Blöchl, P. E. Projector augmented-wave method. Phys. Rev. B. 50, 17953–17979 (1994).
Acknowledgements
This work is supported by the Fundamental Research Funds for the Central Universities (2020XZZX002-07), Natural Science Foundation of China (project nos. U1932148, 21872174, 51672057, 21676246 and 21776248) and International Science and Technology Cooperation Program (grant nos. 2018YFE0203400 and 2017YFE0127800). H.S. and S.J. thank the US NSF (grant no. CHE-1955074) for support.
Author information
Authors and Affiliations
Contributions
X.Z. and S.J. conceived the idea. X.Z. designed experiments. S.H. prepared the samples, characterized them and analysed the data. H.S. and J.H. helped to analyse the XPS and electrochemical data. M.L. helped to analyse EXAFS data. G.Z. and Y.H. performed the DFT calculations. F.Z., X.L., Z.S., J.H., Y.Q. and L.Z. assisted in the fabrication of PEM electrolysers and analysed the device efficiency. S.H., H.S., J.H., B.S., L.L., X.Z. and S.J. wrote the paper with input from all authors.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Nanotechnology thanks the anonymous reviewers for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–41, Tables 1–17 and References.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Rights and permissions
About this article
Cite this article
Hao, S., Sheng, H., Liu, M. et al. Torsion strained iridium oxide for efficient acidic water oxidation in proton exchange membrane electrolyzers. Nat. Nanotechnol. 16, 1371–1377 (2021). https://doi.org/10.1038/s41565-021-00986-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41565-021-00986-1
This article is cited by
-
Surface oxidation/spin state determines oxygen evolution reaction activity of cobalt-based catalysts in acidic environment
Nature Communications (2024)
-
Constructing regulable supports via non-stoichiometric engineering to stabilize ruthenium nanoparticles for enhanced pH-universal water splitting
Nature Communications (2024)
-
Distance effect of single atoms on stability of cobalt oxide catalysts for acidic oxygen evolution
Nature Communications (2024)
-
Efficient bubble/precipitate traffic enables stable seawater reduction electrocatalysis at industrial-level current densities
Nature Communications (2024)
-
Locking the lattice oxygen in RuO2 to stabilize highly active Ru sites in acidic water oxidation
Nature Communications (2024)