Abstract
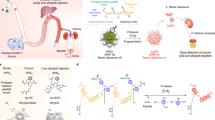
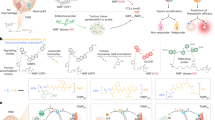
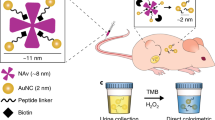
The early detection of the onset of transplant rejection is critical for the long-term survival of patients. The diagnostic gold standard for detecting transplant rejection involves a core biopsy, which is invasive, has limited predictive power and carries a morbidity risk. Here, we show that nanoparticles conjugated with a peptide substrate specific for the serine protease granzyme B, which is produced by recipient T cells during the onset of acute cellular rejection, can serve as a non-invasive biomarker of early rejection. When administered systemically in mouse models of skin graft rejection, these nanosensors preferentially accumulate in allograft tissue, where they are cleaved by granzyme B, releasing a fluorescent reporter that filters into the recipient’s urine. Urinalysis then discriminates the onset of rejection with high sensitivity and specificity before features of rejection are apparent in grafted tissues. Moreover, in mice treated with subtherapeutic levels of immunosuppressive drugs, the reporter signals in urine can be detected before graft failure. This method may enable routine monitoring of allograft status without the need for biopsies.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$99.00 per year
only $8.25 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All data supporting the findings of this study are available within the manuscript and its Supplementary Information. Raw data are available from the corresponding authors.
References
Mas, V. R., Mueller, T. F., Archer, K. J. & Maluf, D. G. Identifying biomarkers as diagnostic tools in kidney transplantation. Expert. Rev. Mol. Diagn. 11, 183–196 (2011).
Gwinner, W. Renal transplant rejection markers. World J. Urol. 25, 445 (2007).
Cornell, L. D., Smith, R. N. & Colvin, R. B. Kidney transplantation: mechanisms of rejection and acceptance. Annu. Rev. Pathol. Mech. Dis. 3, 189–220 (2008).
Nankivell, B. J. & Alexander, S. I. Rejection of the kidney allograft. N. Engl. J. Med. 363, 1451–1462 (2010).
Sijpkens, Y. W. J. et al. Early versus late acute rejection episodes in renal transplantation. Transplantation 75, 204 (2003).
Moreau, A., Varey, E., Anegon, I. & Cuturi, M.-C. Effector mechanisms of rejection. Cold Spring Harb. Perspect. Med. 3, a015461 (2013).
Furness, P. N., Taub, N. & Convergenge of European Renal Transplant Pathology Assessment Procedures (CERTPAP) Project). International variation in the interpretation of renal transplant biopsies: report of the CERTPAP Project. Kidney Int. 60, 1998–2012 (2001).
Piovesan, A. C. et al. Multifocal renal allograft biopsy: impact on therapeutic decisions. Transplant. Proc. 40, 3397–3400 (2008).
Jaffa, M. A. et al. Analyses of renal outcome following transplantation adjusting for informative right censoring and demographic factors: a longitudinal study. Ren. Fail. 32, 691–698 (2010).
Josephson, M. A. Monitoring and managing graft health in the kidney transplant recipient. Clin. J. Am. Soc. Nephrol. 6, 1774–1780 (2011).
Vlaminck, I. D. et al. Circulating cell-free DNA enables noninvasive diagnosis of heart transplant rejection. Sci. Transl. Med. 6, 241ra77–241ra77 (2014).
Choy, J. C. Granzymes and perforin in solid organ transplant rejection. Cell Death Differ. 17, 567–576 (2010).
Wagrowska-Danilewicz, M. & Danilewicz, M. Immunoexpression of perforin and granzyme B on infiltrating lymphocytes in human renal acute allograft rejection. Nefrologia 23, 538–544 (2003).
Rowshani, A. T. et al. Hyperexpression of the granzyme B inhibitor PI-9 in human renal allografts: a potential mechanism for stable renal function in patients with subclinical rejection. Kidney Int. 66, 1417–1422 (2004).
Kummer, J. A. et al. Expression of granzyme A and B proteins by cytotoxic lymphocytes involved in acute renal allograft rejection. Kidney Int. 47, 70–77 (1995).
Suthanthiran, M. et al. Urinary-cell mRNA profile and acute cellular rejection in kidney allografts. N. Engl. J. Med. 369, 20–31 (2013).
Simon, T., Opelz, G., Wiesel, M., Ott, R. C. & Süsal, C. Serial peripheral blood perforin and granzyme B gene expression measurements for prediction of acute rejection in kidney graft recipients. Am. J. Transplant. 3, 1121–1127 (2003).
Calafiore, R. & Basta, G. Clinical application of microencapsulated islets: actual prospectives on progress and challenges. Adv. Drug Deliv. Rev. 67–68, 84–92 (2014).
Li, B. et al. Noninvasive diagnosis of renal-allograft rejection by measurement of messenger RNA for perforin and granzyme B in urine. N. Engl. J. Med. 344, 947–954 (2001).
Sun, J. et al. A cytosolic granzyme B inhibitor related to the viral apoptotic regulator cytokine response modifier A is present in cytotoxic lymphocytes. J. Biol. Chem. 271, 27802–27809 (1996).
Edgington, L. E., Verdoes, M. & Bogyo, M. Functional imaging of proteases: recent advances in the design and application of substrate-based and activity-based probes. Curr. Opin. Chem. Biol. 15, 798–805 (2011).
Sanman, L. E. & Bogyo, M. Activity-based profiling of proteases. Annu. Rev. Biochem. 83, 249–273 (2014).
Konishi, M. et al. Imaging granzyme B activity assesses immune-mediated myocarditis. Circ. Res. 117, 502–512 (2015).
Larimer, B. M. et al. Granzyme B PET imaging as a predictive biomarker of immunotherapy response. Cancer Res. 77, 2318–2327 (2017).
Whitley, M. J. et al. A mouse-human phase 1 co-clinical trial of a protease-activated fluorescent probe for imaging cancer. Sci. Transl. Med. 8, 320ra4–320ra4 (2016).
Olson, E. S. et al. In vivo fluorescence imaging of atherosclerotic plaques with activatable cell-penetrating peptides targeting thrombin activity. Integr. Biol. 4, 595–605 (2012).
Kwong, G. A. et al. Mass-encoded synthetic biomarkers for multiplexed urinary monitoring of disease. Nat. Biotechnol. 31, 63–70 (2013).
Lin, K. Y., Kwong, G. A., Warren, A. D., Wood, D. K. & Bhatia, S. N. Nanoparticles that sense thrombin activity as synthetic urinary biomarkers of thrombosis. ACS Nano 7, 9001–9009 (2013).
Warren, A. D., Kwong, G. A., Wood, D. K., Lin, K. Y. & Bhatia, S. N. Point-of-care diagnostics for noncommunicable diseases using synthetic urinary biomarkers and paper microfluidics. Proc. Natl Acad. Sci. USA 111, 3671–3676 (2014).
Kwong, G. A. et al. Mathematical framework for activity-based cancer biomarkers. Proc. Natl Acad. Sci. USA 112, 12627–12632 (2015).
Holt, B. A., Mac, Q. D. & Kwong, G. A. Nanosensors to detect protease activity in vivo for noninvasive diagnostics. J. Vis. Exp. 137, e57937 (2018).
Fosgerau, K. & Hoffmann, T. Peptide therapeutics: current status and future directions. Drug Discov. Today 20, 122–128 (2015).
Anselmo, A. C. & Mitragotri, S. Nanoparticles in the clinic. Bioeng. Transl. Med. 1, 10–29 (2016).
Arami, H., Khandhar, A., Liggitt, D. & Krishnan, K. M. In vivo delivery, pharmacokinetics, biodistribution and toxicity of iron oxide nanoparticles. Chem. Soc. Rev. 44, 8576–8607 (2015).
Park, J.-H. et al. Magnetic iron oxide nanoworms for tumor targeting and imaging. Adv. Mater. 20, 1630–1635 (2008).
Jokerst, J. V., Lobovkina, T., Zare, R. N. & Gambhir, S. S. Nanoparticle PEGylation for imaging and therapy. Nanomed. 6, 715–728 (2011).
Harris, J. L., Peterson, E. P., Hudig, D., Thornberry, N. A. & Craik, C. S. Definition and redesign of the extended substrate specificity of granzyme B. J. Biol. Chem. 273, 27364–27373 (1998).
Waugh, S. M., Harris, J. L., Fletterick, R. & Craik, C. S. The structure of the pro-apoptotic protease granzyme B reveals the molecular determinants of its specificity. Nat. Struct. Mol. Biol. 7, 762–765 (2000).
Ruggles, S. W., Fletterick, R. J. & Craik, C. S. Characterization of structural determinants of granzyme B reveals potent mediators of extended substrate specificity. J. Biol. Chem. 279, 30751–30759 (2004).
Casciola-Rosen, L. et al. Mouse and human granzyme B have distinct tetrapeptide specificities and abilities to recruit the bid pathway. J. Biol. Chem. 282, 4545–4552 (2007).
Huppa, J. B. & Davis, M. M. T-cell-antigen recognition and the immunological synapse. Nat. Rev. Immunol. 3, 973 (2003).
Dustin, M. L. & Long, E. O. Cytotoxic immunological synapses. Immunol. Rev. 235, 24–34 (2010).
Balaji, K. N., Schaschke, N., Machleidt, W., Catalfamo, M. & Henkart, P. A. Surface cathepsin B protects cytotoxic lymphocytes from self-destruction after degranulation. J. Exp. Med. 196, 493–503 (2002).
Locke, F. L. et al. Phase 1 results of ZUMA-1: a multicenter study of KTE-C19 anti-CD19 CAR T cell therapy in refractory aggressive lymphoma. Mol. Ther. 25, 285–295 (2017).
Goldbach-Mansky, R. et al. Raised granzyme B levels are associated with erosions in patients with early rheumatoid factor positive rheumatoid arthritis. Ann. Rheum. Dis. 64, 715–721 (2005).
Clarke, S. Rm et al. Characterization of the ovalbumin-specific TCR transgenic line OT-I: MHC elements for positive and negative selection. Immunol. Cell Biol. 78, 110–117 (2000).
Kurschus, F. C., Fellows, E., Stegmann, E. & Jenne, D. E. Granzyme B delivery via perforin is restricted by size, but not by heparan sulfate-dependent endocytosis. Proc. Natl Acad. Sci. USA 105, 13799–13804 (2008).
Adrain, C., Duriez, P. J., Brumatti, G., Delivani, P. & Martin, S. J. The cytotoxic lymphocyte protease, granzyme B, targets the cytoskeleton and perturbs microtubule polymerization dynamics. J. Biol. Chem. 281, 8118–8125 (2006).
Giesübel, U., Dälken, B., Mahmud, H. & Wels, W. S. Cell binding, internalization and cytotoxic activity of human granzyme B expressed in the yeast Pichia pastoris. Biochem. J. 394, 563–573 (2006).
Mori, D. N., Kreisel, D., Fullerton, J. N., Gilroy, D. W. & Goldstein, D. R. Inflammatory triggers of acute rejection of organ allografts. Immunol. Rev. 258, 132–144 (2014).
LaRosa, D. F., Rahman, A. H. & Turka, L. A. The innate immune system in allograft rejection and tolerance. J. Immunol. 178, 7503–7509 (2007).
Haas, M. et al. The Banff 2017 Kidney Meeting Report: revised diagnostic criteria for chronic active T cell–mediated rejection, antibody-mediated rejection, and prospects for integrative endpoints for next-generation clinical trials. Am. J. Transplant. 18, 293–307 (2018).
Maeda, H., Wu, J., Sawa, T., Matsumura, Y. & Hori, K. Tumor vascular permeability and the EPR effect in macromolecular therapeutics: a review. J. Control. Release 65, 271–284 (2000).
Fredman, G. et al. Targeted nanoparticles containing the proresolving peptide Ac2-26 protect against advanced atherosclerosis in hypercholesterolemic mice. Sci. Transl. Med. 7, 275ra20 (2015).
Wilhelm, S. et al. Analysis of nanoparticle delivery to tumours. Nat. Rev. Mater. 1, 16014 (2016).
Choi, H. S. et al. Renal clearance of nanoparticles. Nat. Biotechnol. 25, 1165–1170 (2007).
Loupy, A. et al. The Banff 2015 Kidney Meeting Report: current challenges in rejection classification and prospects for adopting molecular pathology. Am. J. Transplant. 17, 28–41 (2017).
Vincenti, F., Jensik, S. C., Filo, R. S., Miller, J. & Pirsch, J. A long-term comparison of tacrolimus (FK506) and cyclosporine in kidney transplantation: evidence for improved allograft survival at five years. Transplantation 73, 775–782 (2002).
Vincenti, F. et al. Belatacept and long-term outcomes in kidney transplantation. N. Engl. J. Med. 374, 333–343 (2016).
Jeansson, M. & Haraldsson, B. Glomerular size and charge selectivity in the mouse after exposure to glucosaminoglycan-degrading enzymes. J. Am. Soc. Nephrol. 14, 1756–1765 (2003).
Hsauyry, P. et al. The inflammatory mechanisms of allograft rejection.Immunol. Rev. 77, 85–142 (1984).
Wood, K. J. & Goto, R. Mechanisms of rejection. Curr. Persp. Transplant. 93, 1–10 (2012).
Staquicini, F. I. et al. Vascular ligand-receptor mapping by direct combinatorial selection in cancer patients. Proc. Natl Acad. Sci. USA 108, 18637–18642 (2011).
Hua, S. Targeting sites of inflammation: intercellular adhesion molecule-1 as a target for novel inflammatory therapies. Front. Pharmacol. 4, 127 (2013).
Kwon, E. J., Dudani, J. S. & Bhatia, S. N. Ultrasensitive tumour-penetrating nanosensors of protease activity. Nat. Biomed. Eng. 1, 0054 (2017).
Han, D. et al. Assessment of cytotoxic lymphocyte gene expression in the peripheral blood of human islet allograft recipients: elevation precedes clinical evidence of rejection. Diabetes 53, 2281–2290 (2004).
Jaffer, F. A. & Weissleder, R. Molecular imaging in the clinical arena. JAMA 293, 855–862 (2005).
Weissleder, R., Tung, C.-H., Mahmood, U. & Bogdanov, J. In vivo imaging of tumors with protease-activated near-infrared fluorescent probes. Nat. Biotechnol. 17, 375–378 (1999).
Olson, E. S. et al. Activatable cell penetrating peptides linked to nanoparticles as dual probes for in vivo fluorescence and MR imaging of proteases. Proc. Natl Acad. Sci. USA 107, 4311–4316 (2010).
Sugahara, K. N. et al. Tissue-penetrating delivery of compounds and nanoparticles into tumors. Cancer Cell. 16, 510–520 (2009).
Hori, S. S. & Gambhir, S. S. Mathematical model identifies blood biomarker–based early cancer detection strategies and limitations. Sci. Transl. Med. 3, 109ra116–109ra116 (2011).
Lutz, A. M., Willmann, J. K., Cochran, F. V., Ray, P. & Gambhir, S. S. Cancer screening: a mathematical model relating secreted blood biomarker levels to tumor sizes. PLoS. Med. 5, e170 (2008).
Dharnidharka, V. R., Kwon, C. & Stevens, G. Serum cystatin C is superior to serum creatinine as a marker of kidney function: a meta-analysis. Am. J. Kidney. Dis. 40, 221–226 (2002).
Kaplan, B., Schold, J. & Meier-Kriesche, H.-U. Poor predictive value of serum creatinine for renal allograft loss. Am. J. Transplant. 3, 1560–1565 (2003).
Slocum, J. L., Heung, M. & Pennathur, S. Marking renal injury: can we move beyond serum creatinine? Transl. Res. 159, 277–289 (2012).
Haase, M. et al. Accuracy of neutrophil gelatinase-associated lipocalin (NGAL) in diagnosis and prognosis in acute kidney injury: a systematic review and meta-analysis. Am. J. Kidney. Dis. 54, 1012–1024 (2009).
Mischak, H. et al. Recommendations for biomarker identification and qualification in clinical proteomics. Sci. Transl. Med. 2, 46ps42–46ps42 (2010).
Prendergast, M. B. & Gaston, R. S. Optimizing medication adherence: an ongoing opportunity to improve outcomes after kidney tansplantation. Clin. J. Am. Soc. Nephrol. 5, 1305–1311 (2010).
Alangaden, G. J. et al. Infectious complications after kidney transplantation: current epidemiology and associated risk factors. Clin. Transplant. 20, 401–409 (2006).
Sellarés, J. et al. Understanding the causes of kidney transplant failure: the dominant role of antibody-mediated rejection and nonadherence. Am. J. Transplant. 12, 388–399 (2012).
Palmacci, S. Synthesis of polysaccharide covered superparamagnetic oxide colloids. US patent 5,262,176 (1993).
Presolski Stanislav, I., Hong Vu, Phong. & Finn, M. G. Copper‐catalyzed azide–alkyne click chemistry for bioconjugation. Curr. Protoc. Chem. Biol. 3, 153–162 (2011).
Acknowledgements
This work was funded by an NIH Director’s New Innovator Award DP2HD091793 awarded to G.A.K. and National Institutes of Health U01 AI132904 awarded to A.B.A. Q.D.M. is supported by the NSF Graduate Research Fellowships Program (Grant No. DGE-1650044). D.V.M. is supported by National Institutes of Health F30 award number DK109665. B.A.H is supported by the National Institutes of Health GT BioMAT Training Grant under Award Number 5T32EB006343. G.A.K. holds a Career Award at the Scientific Interface from the Burroughs Wellcome Fund. The content is solely the responsibility of the authors and does not necessarily represent the official views of the National Institutes of Health.
Author information
Authors and Affiliations
Contributions
Q.D.M., D.V.M., A.B.A. and G.A.K. conceived of the idea, designed experiments, and interpreted results. Q.D.M., D.V.M., J.A.K., C.M.S., O.M.D. and B.A.H. carried out the experiments. Q.D.M., D.V.M., B.A.H., A.B.A. and G.A.K. wrote the manuscript.
Corresponding authors
Ethics declarations
Competing interests
Q.D.M., D.V.M., A.B.A. and G.A.K. are listed as inventors on a patent application pertaining to the results of the paper. G.A.K. is co-founder of and serves as consultant to Glympse Bio, which is developing products related to the research described in this paper. This study could affect his personal financial status. The terms of this arrangement have been reviewed and approved by Georgia Tech in accordance with its conflict of interest policies.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary figures
Rights and permissions
About this article
Cite this article
Mac, Q.D., Mathews, D.V., Kahla, J.A. et al. Non-invasive early detection of acute transplant rejection via nanosensors of granzyme B activity. Nat Biomed Eng 3, 281–291 (2019). https://doi.org/10.1038/s41551-019-0358-7
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41551-019-0358-7
This article is cited by
-
Artificial urinary biomarker probes for diagnosis
Nature Reviews Bioengineering (2024)
-
Activatable near-infrared probes for the detection of specific populations of tumour-infiltrating leukocytes in vivo and in urine
Nature Biomedical Engineering (2023)
-
Challenges with Cell-based Therapies for Type 1 Diabetes Mellitus
Stem Cell Reviews and Reports (2023)
-
Dual-ratiometric magnetic resonance tunable nanoprobe with acidic-microenvironment-responsive property to enhance the visualization of early tumor pathological changes
Nano Research (2023)
-
Urinary detection of early responses to checkpoint blockade and of resistance to it via protease-cleaved antibody-conjugated sensors
Nature Biomedical Engineering (2022)