Abstract
Reactive arthritis (ReA), an inflammatory arthritic condition that is commonly associated with Chlamydia infections, represents a significant health burden, yet is poorly understood. The enigma of this disease is reflected in its problematic name and in its ill-defined pathogenesis. The existence of persistent pathogens in the arthritic joint is acknowledged, but their relevance remains elusive. Progress is being made in understanding the underlying mechanisms of ReA, whereby an imbalance between type 1 and type 2 immune responses seems to be critical in determining susceptibility to disease. Such an imbalance occurs prior to the initiation of an adaptive immune response, suggesting that innate cellular and molecular mechanisms in ReA should be prioritized as fruitful areas for investigation.
Similar content being viewed by others
Introduction
Reactive arthritis (ReA), previously known as Reiter's syndrome, is an inflammatory arthritis preceded by infection with enteric pathogens, such as Salmonella spp., Yersinia spp., Shigella spp. and Campylobacter spp., or the oculogenital pathogens, Chlamydia spp. By virtue of HLA-B27 association, the pattern of joint involvement, and the absence of rheumatoid factor, ReA is classified as part of the spectrum of spondyloarthritis (SpA). However, there are clear distinctions between ReA and classic SpA diseases, such as ankylosing spondylitis: notably, a weaker association with HLA-B271,2 and an established link between a previous infection with the aforementioned bacteria and the onset of arthritis. Moreover, it is becoming clear that post-enteric and post-chlamydial ReA are distinct entities, in that viable organisms can be detected in the joints of patients with Chlamydia-induced ReA (CiReA) but not in those with post-enteric ReA.3
Chlamydia trachomatis represents the most common single cause of ReA, whilst Chlamydophila pneumoniae has been implicated in ReA, albeit far less frequently than C. trachomatis.4 As little study has been done in the field of C. pneumoniae-associated ReA, this article will focus on C. trachomatis-associated arthritis. Approximately 4–15% of those with genital C. trachomatis infections subsequently develop arthritis.2 Given the high prevalence of chlamydial genital tract infections, it has been proposed that the incidence of CiReA might rival that of rheumatoid arthritis (RA).3 Furthermore, C. pneumoniae and/or C. trachomatis are detectable in 62% of patients with undifferentiated SpA, suggesting that ReA is hugely underdiagnosed.5 Thus, CiReA represents a significantly understudied disease with a measurable physical and economic burden.6
In this article, we outline recent advances in our understanding of CiReA, with respect to both Chlamydia and host biology, and a focus on factors that might mediate CiReA susceptibility. We also introduce the field of macrophage polarization, an alteration of which is associated with susceptibility to both rheumatic and infectious diseases. The role of chlamydial persistence in the arthritic joint is also discussed, as is the indeterminate area occupied by CiReA between SpA and septic arthritis.
ReA: noncanonical septic arthritis
The term 'reactive' in ReA describes how this disease was traditionally viewed as a nonseptic, autoimmune reaction occurring in the joint in response to an extra-articular bacterial infection.7 This notion was based on the fact that no pathogen could be cultured from the arthritic joint, despite detection of arthritis-associated bacteria at other sites in the body. Surprisingly, extensive searching for the bacterial antigens responsible for adaptive-immunity-mediated pathology has not yielded conclusive results to date. To further confound the autoimmunity-mediated hypotheses for ReA, it was discovered through the use of nucleic acid detection and electron microscopy that Chlamydia is present in the joint, albeit in an aberrant but viable state.2
The possibility of CiReA being driven not by a reactive autoimmune process, but rather by the infection of nonimmune cells in situ, has paralleled a paradigm shift in establishing the mechanism of tissue damage during chlamydial infections in general. Initially, the immunological paradigm proposed that tissue damage results from the aberrant activation of the adaptive immune system through delayed-type hypersensitivity or autoimmunity.8 After a decade of research failed to prove this theory, an alternative cellular paradigm was proposed, postulating that chlamydial pathogenesis is driven by an inflammatory response propagated by sustained infection of nonimmune cells, whereby ongoing release of inflammatory mediators and subsequent recruitment of inflammatory cells causes tissue damage.9 As C. trachomatis is known to infect both macrophage-like and fibroblast-like synoviocytes,10 could the cellular paradigm hold true in CiReA?
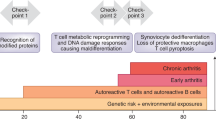
With the recognition of viable Chlamydia in the joint, a contradiction arises regarding the classification and nomenclature of CiReA relative to underlying clinical and pathological features. This concept is illustrated in Figure 1, with ReA occupying a midpoint between SpA and septic arthritis. To support the concept of CiReA as a septic arthritis, a clinical trial in 2010 demonstrated that combination antibiotics could alter the course of CiReA.11 In addition, the immunopathogenesis of CiReA recapitulates that seen in experimental septic arthritis with regard to mediators of susceptibility and the role of the immune system.12,13
Reactive arthritis shares clinical and immunopathogenic features with both spondyloarthritis, of which it is considered a subset, and septic arthritis. Enteric-pathogen-associated reactive arthritis better represents spondyloarthritis, whereas Chlamydia-associated reactive arthritis represents a noncanonical septic arthritis. Abbreviations: CiReA, Chlamydia-induced reactive arthritis; ReA, reactive arthritis.
The classification of CiReA as a septic arthritis necessitates a better understanding of bacterial colonization of the joint. After the primary infection, in both humans and animals, Chlamydia is known to spread throughout the body via monocytic cells14 to a diverse range of tissues, including the spleen, liver, peritoneum and lungs.15 Chlamydia is often detected long after the initial infection in selected tissues, such as the genital tract and synovium. In respect of the ability of Chlamydia to evade clearance in the synovium, the traditional hypothesis has been that the joint is a site of immune privilege; however, immune cells can be found in the joint in both healthy and arthritic states. One extension of this theory proposes that it is the hypoxic environment of the inflamed joint16 that creates an immunoprivileged microenvironment. This idea stems from the fact that the natural site of infection for Chlamydia—the genital tract—is relatively hypoxic, and that Chlamydia thrives under hypoxic conditions.17 Chlamydia might have adapted to growth under such conditions, given the organism's active manipulation of hypoxia inducible factor 1 (HIF-1).18 Additionally, bacteriocidal cytokines, such as interferon γ (IFN-γ),17 and antibiotics19 have decreased efficacy under hypoxic conditions, providing the rationale for chlamydial escape from these control mechanisms within the joint.
Chlamydial persistence in joints
Another incompletely explained aspect of the host–pathogen interaction during CiReA is the observation that Chlamydia adopts an atypical, persistent state within the joint. Chlamydia spp. are obligate intracellular pathogens that exist in two distinct states during their lifecycle: the extracellular, infectious elementary body, and the intracellular, replicative reticulate body. When appropriate stress is applied during the intracellular stage, such as IFN-γ exposure, antibiotic treatment or infection of monocytic cells, Chlamydia enters a nonreplicative and unculturable, yet viable, persistent state.20 The persistent state differs from the normal intracellular state in that the reticulate bodies fail to divide or differentiate into infectious particles, have a reduced metabolism, and are immuno-evasive.21
Persistent Chlamydia were originally identified in CiReA through their detection using microscopy or PCR coupled with an inability to be cultured from synovial biopsy.10 Accurate identification of Chlamydia cells with a persistent phenotype in CiReA is now possible using real-time PCR, as they display a unique gene expression profile.22 The recognition of persistent Chlamydia in ReA represents an important advance in our understanding of this disease, but leaves key unanswered questions with respect to its pathogenesis.
Is persistent Chlamydia the cause of ReA, or the effect of host–pathogen adaptation? A mechanistic hypothesis to address Chlamydia as the instigator of ReA postulates that chlamydial persistence provides a continuous source of bacterial components that stimulate the immune system, resulting in chronic inflammation and tissue damage.4 These components could be pathogen-associated molecular patterns (PAMPs) that stimulate host innate receptors, or microbial antigens that elicit adaptive immune responses. On the other hand, it is equally plausible that persistence represents the host's best attempt at controlling Chlamydia, which itself has developed specialized mechanisms to avoid the immune response, particularly the establishment of the immuno-evasive persistent state. In support of this theory, chronic chlamydial infection elicits little immune stimulation relative to that seen during acute infection.23 This latter theory also provides the rationale for the arthritic flares that are seen commonly in ReA, in which chlamydial escape from persistence could result in an acute inflammatory event. The basic understanding of chlamydial persistence is in its early stages, and an emphasis must be placed on understanding the role of this phenomenon in the pathogenesis of CiReA. Thus, clinical ReA studies need to address the chlamydial state concurrently with joint inflammation, and animal models are needed to assess the temporal relationship between persistence and arthritis.
Mediators of CiReA susceptibility
Up to 80% of chlamydial infections are asymptomatic.24 This statistic poses considerable hurdles in attempting to link clinical evidence of infection with subsequent ReA, but clearly, for reasons not yet defined, only a small proportion of infected individuals go on to develop ReA.
Factors that contribute to the increased virulence of an infection include pathogen, host and environmental variation. The most commonly observed variability of Chlamydia relates to the outer membrane proteins, which dictate its biovar. This variation is associated with tissue tropism rather than virulence; however, a recent study did detect ocular biovars more commonly than genital biovars in ReA.25 A limited number of studies have addressed non-biovar-related variance as a determinant of pathogenicity,26 but the effect of this variance on human infection or CiReA remains to be determined. From a broader perspective, a stable host–pathogen interaction is indicated by the consistent rates of Chlamydia-related disease27 in comparison to the fluctuating temporal incidence of highly mutable pathogens, such as influenza. This stability suggests that alterations in chlamydial pathogenicity are uncommon—an observation that is typical of the relationship seen during prolonged co-evolution of host and microbe, in which an increase in virulence would be disadvantageous to the long-term survival of an obligate intracellular pathogen.
Environmental factors are also known to affect chlamydial infections. Repeated chlamydial infections have been associated with increased genital tract pathology,28 but whether repeated infections are required for the development of CiReA remains unknown. This issue is rarely addressed in practice, as diagnostic tests for Chlamydia do not distinguish between repeat infections or chronic infections with the same pathogen.29 Another potential environmental factor is heavy-metal exposure, which has been shown in animal studies to modify the cytokine balance.30 Experimentally, we have shown that heavy metals sensitize animals that are otherwise resistant to CiRea;31 however, clinical studies are required to validate this effect in humans. Heavy-metal-induced sensitization correlates with the suppression of the inflammatory cytokines IFN-γ and TNF, which raises the question of whether an immune imbalance acts as a mediator of CiReA susceptibility.
Most studies examining mediators of susceptibility to chlamydial sequelae focus on host genetic variability, in particular those determining the immune response. A robust type 1 (classical) inflammatory response is crucial for the control of Chlamydia infection, whereas an enhanced type 2 (alternative) inflammatory response is associated with susceptibility to infection (Figure 2). This distinction has been demonstrated in animal models, whereby a deficiency of type 1 cytokines, such as IL-12 and IFN-γ, influences susceptibility to chlamydial infection, whereas a deficiency of type-2-associated cytokines, such as IL-10, mediates resistance.32 Furthermore, we have shown that high synovial levels of IFN-γ and TNF correlate with resistance to experimental CiReA.33 The protective effect of a type 1 inflammatory response during chlamydial infection in animals has been recapitulated in human studies, in that the sequelae are associated with stronger type 2 cytokine responses.34 Additionally, an attenuated type 1 response is seen in patients with SpA,35 manifested as an impaired IFN-γ-induced gene expression profile.36 Although few such studies have been performed in CiReA, these trends seem to be consistent in this disease: those with prolonged ReA express lower levels of IFN-γ than those who rapidly overcome ReA.37 Another study demonstrated the IL-10–IL-12 balance to be crucial in preventing ReA.38
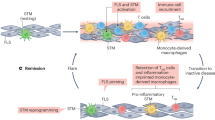
These figures highlight the potential role of the macrophage, whereby a type 1 response may result in early activation of M1 macrophages and a type 2 response may reinforce M2 polarization. a | A type 1 skew is required for effective control of Chlamydia through early suppression of chlamydial growth and removal of extracellular chlamydial EBs during the innate phase. Subsequent effective control, which may involve the induction of persistence, requires adaptive immunity. b | A type 2 skew results in excessive chlamydial growth through an ineffective innate response and subsequently ineffective adaptive immune response. Abbreviations: EB, elementary body; IFN, interferon; NK cell, natural killer cell; iNKT cell, invariant natural killer T cell; TH1, type 1 T helper (cell); TH2, type 2 T helper (cell); TH17, type 17 T helper (cell).
The importance of innate immunity
If susceptibility to CiReA is determined by a disruption in the yin–yang balance of type 1 and type 2 inflammatory responses, at what point during an inflammatory response does such an imbalance have an effect? The immune system comprises two distinct arms: the germline-encoded, rapidly responding innate immune response; and the delayed, but highly specific, adaptive immune response. Both systems can be polarized during a type 1 or type 2 dominant response. CD4+ T cells can be polarized into the well-defined T-helper type 1 (TH1) and type 2 (TH2) phenotypes, which have traditionally defined the type 1 and type 2 responses. TH1 cells are crucial for eliminating chlamydial infections;39 however, an effective immune response to C. trachomatis, and resistance to CiReA, is associated with a robust type 1 cytokine response as early as 3 days after infection.33,40 These temporal kinetics exclude the adaptive immune system as the defining process in early resistance versus susceptibility to Chlamydia. But which cells of the innate immune response might cause a disrupted inflammatory balance, and how might such cells be subsequently affected?
Research into the innate immune response during Chlamydia infection has been somewhat neglected, as the primary focus has been on vaccine development. Nevertheless, many innate immune cells have been shown to have important roles. Natural killer cells have been implicated in providing an early protective effect through IFN-γ release,41 an effect that is also mediated by natural killer T cells during CiReA.42 Neutrophils, on the other hand, seem to have a dual role, in that they reduce early excessive chlamydial growth,43 yet excessive activation can itself contribute to tissue damage, including CiReA.44 Another innate cell type shown to be important in chlamydial infection is the macrophage; macrophage depletion greatly enhances infection progression.45
Macrophages are a heterogeneous group of related cells ranging from aggressive, proinflammatory macrophages to passive, tissue-resident macrophages. Tissue-resident macrophages comprise up to 15% of all cells in healthy tissue, and up to 20% of synoviocytes.46 Thus, the primary role of the macrophage should be viewed as that of a homeostatic local resident cell rather than an aggressive innate immune effector. Tissue-resident macrophages tend to be anti-inflammatory in nature, and have recently been shown to maintain their numbers by local proliferation.47 Inflammatory macrophage populations, on the other hand, rarely exist in healthy tissue, and appear on the scene through recruitment and activation of blood monocytes.47
A classification system has been devised to delineate polarized macrophages on the basis of phenotype and function, encompassing a spectrum ranging from the classically activated or inflammatory M1 macrophage to the homeostatic or alternatively activated M2 macrophage.48 This classification system is based on analogy with the TH1/TH2 paradigm, whereby polarization to M1 is induced by a type 1 inflammatory environment, such as the presence of IFN-γ, and M2 is induced by an environment containing IL-10 and IL-4. Furthermore, the existence of FOXP3+ regulatory macrophages has recently been demonstrated, akin to the identification of FOXP3+ regulatory T cells.49 A key distinction exists between macrophage and TH cell polarization, in that macrophage polarization is highly plastic relative to the somewhat rigid differentiation of T cells.50 This plasticity assists host defense, considering the role of the innate immune system, which is to rapidly respond to alterations in the immediate environment.
In many biological systems, a dynamic balance exists between opposing mechanisms, as is the case with macrophage polarization. Alterations to this balance, either endogenous or exogenous, are associated with certain pathological processes, but it can often be difficult to ascertain whether the polarized state is the cause or the effect. Differential macrophage polarization can be demonstrated in rheumatic diseases; for example, RA is associated with an M1 dominance, and SpA shows an M2 dominance.35,51 Furthermore, a robust M1 response is essential for the control of intracellular pathogens, and it is recognized that a deficient M1 response facilitates uncontrolled acute intracellular bacterial infections.52 Certain intracellular bacteria, such as Francisella tularensis, have been shown to induce M2 polarization in macrophages, resulting in prolonged bacterial survival.53 Chlamydia is known to infect macrophages, but how Chlamydia can survive in differentially polarized macrophages or whether Chlamydia is capable of such immune subversion remains unknown.
A role for macrophages in CiReA?
Given the importance of the macrophage in chlamydial infections, and the association between susceptibility and a deficient early type 1 response, it is surprising that little is known about macrophage polarization in the setting of chlamydial infections, especially in the microenvironment of the joint during CiReA. However, there are indicators that M1 macrophages control Chlamydia infection,54 and that M2 macrophages are permissive to chlamydial growth (Figure 2).55 However, control of Chlamydia has traditionally been measured as a reduction in culturable organisms, which is also the hallmark of persistence.
An important goal is to validate the observation of M2 macrophage dominance in SpA and to determine whether this alteration in macrophage phenotype is also present in CiReA. It is conceivable that individuals with an M2 dominance would be less able to control chlamydial infections, and thus be more susceptible to Chlamydia-associated inflammation of the joints. Additionally, it is plausible that joints are particularly sensitive to M2 polarization, given that IFN-γ has reduced activity under hypoxic conditions.
Conclusions
Current evidence suggests that CiReA is a noncanonical septic arthritis with viable, but nonculturable, persistent Chlamydia infection. Thus, our research in this disease should be tailored accordingly toward a better understanding of the articular immune response. As susceptibility to CiReA seems to be established during the innate immune phase, early cellular and molecular immune mediators should be examined closely. In addition, the ability of ocular chlamydial biovars to cause arthritis needs to be studied in more detail. Perturbations in the type 1–type 2 immune balance, which can be caused by both environmental and genetic factors, are likely to affect innate immunity. As macrophages have gained prominence as key gatekeepers of innate immunity, it might be that this role has also conferred on the macrophages a critical role in defining susceptibility to CiReA.
References
Rich, E., Hook, E. W. 3rd, Alarcón, G. S. & Moreland, L. W. Reactive arthritis in patients attending an urban sexually transmitted diseases clinic. Arthritis Rheum. 39, 1172–1177 (1996).
Sieper, J. Pathogenesis of reactive arthritis. Curr. Rheumatol. Rep. 3, 412–418 (2001).
Carter, J. D. & Hudson, A. P. The evolving story of Chlamydia-induced reactive arthritis. Curr. Opin. Rheumatol. 22, 424–430 (2010).
Gérard, H. C., Whittum-Hudson, J. A., Carter, J. D. & Hudson, A. P. The pathogenic role of Chlamydia in spondyloarthritis. Curr. Opin. Rheumatol. 22, 363–367 (2010).
Carter, J. D. et al. Chlamydiae as etiologic agents in chronic undifferentiated spondylarthritis. Arthritis Rheum. 60, 1311–1316 (2009).
Söderlin, M. K., Kautiainen, H., Skogh, T. & Leirisalo-Repo, M. Quality of life and economic burden of illness in very early arthritis. A population based study in Southern Sweden. J. Rheumatol. 31, 1717–1722 (2004).
Keat, A. et al. Chlamydia trachomatis and reactive arthritis: the missing link. Lancet 1, 72–74 (1987).
Brunham, R. C. & Peeling, R. W. Chlamydia trachomatis antigens: role in immunity and pathogenesis. Infect. Agents Dis. 3, 218–233 (1994).
Stephens, R. S. The cellular paradigm of chlamydial pathogenesis. Trends Microbiol. 11, 44–51 (2003).
Nanagara, R., Li, F., Beutler, A., Hudson, A. & Schumacher, H. R. Jr. Alteration of Chlamydia trachomatis biologic behavior in synovial membranes. Suppression of surface antigen production in reactive arthritis and Reiter's syndrome. Arthritis Rheum. 38, 1410–1417 (1995).
Carter, J. D. et al. Combination antibiotics as a treatment for chronic Chlamydia-induced reactive arthritis: a double-blind, placebo-controlled, prospective trial. Arthritis Rheum. 62, 1298–1307 (2010).
Tarkowski, A. Infection and musculoskeletal conditions: infectious arthritis. Best Pract. Res. Clin. Rheumatol. 20, 1029–1044 (2006).
Mathews, C. J., Weston, V. C., Jones, A., Field, M. & Coakley, G. Bacterial septic arthritis in adults. Lancet 375, 846–855 (2010).
Moazed, T. C., Kuo, C. C., Grayston, J. T. & Campbell, L. A. Evidence of systemic dissemination of Chlamydia pneumoniae via macrophages in the mouse. J. Infect. Dis. 177, 1322–1325 (1998).
Cotter, T. W., Ramsey, K. H., Miranpuri, G. S., Poulsen, C. E. & Byrne, G. I. Dissemination of Chlamydia trachomatis chronic genital tract infection in gamma interferon gene knockout mice. Infect. Immun. 65, 2145–2152 (1997).
Ng, C. T. et al. Synovial tissue hypoxia and inflammation in vivo. Ann. Rheum. Dis. 69, 1389–1395 (2010).
Roth, A. et al. Hypoxia abrogates antichlamydial properties of IFN-γ in human fallopian tube cells in vitro and ex vivo. Proc. Natl Acad. Sci. USA 107, 19502–19507 (2010).
Rupp, J. et al. Chlamydia pneumoniae directly interferes with HIF-1α stabilization in human host cells. Cell. Microbiol. 9, 2181–2191 (2007).
Shima, K., Szaszák, M., Solbach, W., Gieffers, J. & Rupp, J. Impact of a low-oxygen environment on the efficacy of antimicrobials against intracellular Chlamydia trachomatis. Antimicrob. Agents Chemother. 55, 2319–2324 (2011).
Wyrick, P. B. Chlamydia trachomatis persistence in vitro: an overview. J. Infect. Dis. 201 (Suppl. 2), S88–S95 (2010).
Belland, R. J. et al. Genomic transcriptional profiling of the developmental cycle of Chlamydia trachomatis. Proc. Natl Acad. Sci. USA. 100, 8478–8483 (2003).
Gérard, H. C., Whittum-Hudson, J. A., Schumacher, H. R. Jr & Hudson, A. P. Differential expression of three Chlamydia trachomatis hsp60-encoding genes in active vs. persistent infections. Microb. Pathog. 36, 35–39 (2004).
Droemann, D. et al. Disparate innate immune responses to persistent and acute Chlamydia pneumoniae infection in chronic obstructive pulmonary disease. Am. J. Respir. Crit. Care Med. 175, 791–797 (2007).
Zimmerman, H. L. et al. Epidemiologic differences between chlamydia and gonorrhea. Am. J. Public Health 80, 1338–1342 (1990).
Gerard, H. C. et al. Patients with Chlamydia-associated arthritis have ocular (trachoma), not genital, serovars of C. trachomatis in synovial tissue. Microb. Pathog. 48, 62–68 (2010).
Kari, L. et al. Pathogenic diversity among Chlamydia trachomatis ocular strains in nonhuman primates is affected by subtle genomic variations. J. Infect. Dis. 197, 449–456 (2008).
World Health Organization. Global Prevalence and Incidence of Selected Curable Sexually Transmitted Infections: Overviews and Estimates [online] (2001).
Darville, T. & Hiltke, T. J. Pathogenesis of genital tract disease due to Chlamydia trachomatis. J. Infect. Dis. 201 (Suppl. 2), S114–S125 (2010).
Gottlieb, S. L., Martin, D. H., Xu, F., Byrne, G. I. & Brunham, R. C. Summary: The natural history and immunobiology of Chlamydia trachomatis genital infection and implications for Chlamydia control. J. Infect. Dis. 201 (Suppl. 2), S190–S204 (2010).
Heo, Y., Parsons, P. J. & Lawrence, D. A. Lead differentially modifies cytokine production in vitro and in vivo. Toxicol. Appl. Pharmacol. 138, 149–157 (1996).
Inman, R. D. & Chiu, B. Heavy metal exposure reverses genetic resistance to Chlamydia-induced arthritis. Arthritis Res. Ther. 11, R19 (2009).
Rottenberg, M. E., Gigliotti-Rothfuchs, A. & Wigzell, H. The role of IFN-γ in the outcome of chlamydial infection. Curr. Opin. Immunol. 14, 444–451 (2002).
Inman, R. D. & Chiu, B. Early cytokine profiles in the joint define pathogen clearance and severity of arthritis in chlamydia-induced arthritis in rats. Arthritis Rheum. 54, 499–507 (2006).
Holland, M. J. et al. T helper type-1 (Th1)/Th2 profiles of peripheral blood mononuclear cells (PBMC); responses to antigens of Chlamydia trachomatis in subjects with severe trachomatous scarring. Clin. Exp. Immunol. 105, 429–435 (1996).
Vandooren, B. et al. Absence of a classically activated macrophage cytokine signature in peripheral spondylarthritis, including psoriatic arthritis. Arthritis Rheum. 60, 966–975 (2009).
Smith, J. A. et al. Gene expression analysis of macrophages derived from ankylosing spondylitis patients reveals interferon-γ dysregulation. Arthritis Rheum. 58, 1640–1649 (2008).
Bas, S., Kvien, T. K., Buchs, N., Fulpius, T. & Gabay, C. Lower level of synovial fluid interferon-γ in HLA-B27-positive than in HLA-B27-negative patients with Chlamydia trachomatis reactive arthritis. Rheumatology (Oxford) 42, 461–467 (2003).
Yin, Z. et al. Crucial role of interleukin-10/interleukin-12 balance in the regulation of the type 2 T helper cytokine response in reactive arthritis. Arthritis Rheum. 40, 1788–1797 (1997).
Morrison, S. G., Su, H., Caldwell, H. D. & Morrison, R. P. Immunity to murine Chlamydia trachomatis genital tract reinfection involves B cells and CD4+ T cells but not CD8+ T cells. Infect. Immun. 68, 6979–6987 (2000).
Jiang, X., Shen, C., Yu, H., Karunakaran, K. P. & Brunham, R. C. Differences in innate immune responses correlate with differences in murine susceptibility to Chlamydia muridarum pulmonary infection. Immunology 129, 556–566 (2010).
Nagarajan, U. M. et al. MyD88 deficiency leads to decreased NK cell gamma interferon production and T cell recruitment during Chlamydia muridarum genital tract infection, but a predominant Th1 response and enhanced monocytic inflammation are associated with infection resolution. Infect. Immun. 79, 486–498 (2011).
Bharhani, M. S., Chiu, B., Na, K. S. & Inman, R. D. Activation of invariant NKT cells confers protection against Chlamydia trachomatis-induced arthritis. Int. Immunol. 21, 859–870 (2009).
Barteneva, N., Theodor, I., Peterson, E. M. & De La Maza, L. M. Role of neutrophils in controlling early stages of a Chlamydia trachomatis infection. Infect. Immun. 64, 4830–4833 (1996).
Zhang, X. et al. Innate immunity and arthritis: Neutrophil Rac and Toll-like receptor 4 expression define outcomes in infection-triggered arthritis. Arthritis Rheum. 52, 1297–1304 (2005).
Qiu, H. et al. Type I IFNs enhance susceptibility to Chlamydia muridarum lung infection by enhancing apoptosis of local macrophages. J. Immunol. 181, 2092–2102 (2008).
Iwanaga, T., Shikichi, M., Kitamura, H., Yanase, H. & Nozawa-Inoue, K. Morphology and functional roles of synoviocytes in the joint. Arch. Histol. Cytol. 63, 17–31 (2000).
Jenkins, S. J. et al. Local macrophage proliferation, rather than recruitment from the blood, is a signature of TH2 inflammation. Science 332, 1284–1288 (2011).
Mantovani, A. et al. The chemokine system in diverse forms of macrophage activation and polarization. Trends Immunol. 25, 677–686 (2004).
Manrique, S. Z. et al. Foxp3-positive macrophages display immunosuppressive properties and promote tumor growth. J. Exp. Med. 208, 1485–1499 (2011).
Murray, P. J. & Wynn, T. A. Obstacles and opportunities for understanding macrophage polarization. J. Leukoc. Biol. 89, 557–563 (2011).
Baeten, D. et al. Infiltration of the synovial membrane with macrophage subsets and polymorphonuclear cells reflects global disease activity in spondyloarthropathy. Arthritis Res. Ther. 7, R359–R369 (2005).
Benoit, M., Desnues, B. & Mege, J. L. Macrophage polarization in bacterial infections. J. Immunol. 181, 3733–3739 (2008).
Shirey, K. A., Cole, L. E., Keegan, A. D. & Vogel, S. N. Francisella tularensis live vaccine strain induces macrophage alternative activation as a survival mechanism. J. Immunol. 181, 4159–4167 (2008).
Chen, B., Stout, R. & Campbell, W. F. Nitric oxide production: a mechanism of Chlamydia trachomatis inhibition in interferon-γ-treated RAW264.7 cells. FEMS Immunol. Med. Microbiol. 14, 109–120 (1996).
Kuo, C. C., Puolakkainen, M., Lin, T. M., Witte, M. & Campbell, L. A. Mannose-receptor positive and negative mouse macrophages differ in their susceptibility to infection by Chlamydia species. Microb. Pathog. 32, 43–48 (2002).
Author information
Authors and Affiliations
Contributions
E. Gracey researched data for the article. Both authors contributed equally to discussing the content, writing the article and performing review/editing of the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Gracey, E., Inman, R. Chlamydia-induced ReA: immune imbalances and persistent pathogens. Nat Rev Rheumatol 8, 55–59 (2012). https://doi.org/10.1038/nrrheum.2011.173
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrrheum.2011.173
This article is cited by
-
Reaktive Arthritis – ein Update
Monatsschrift Kinderheilkunde (2021)
-
Time to revisit the concept of reactive arthritis
Nature Reviews Rheumatology (2017)
-
Chronic Chlamydial Diseases: From Atherosclerosis to Urogenital Infections
Current Clinical Microbiology Reports (2014)
-
Chlamydia pneumoniae and osteoporosis-associated bone loss: a new risk factor?
Osteoporosis International (2013)
-
Polymorphism of HLA-B27: 105 Subtypes Currently Known
Current Rheumatology Reports (2013)