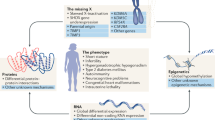
Key Points
-
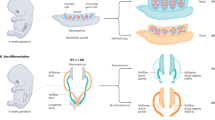
Disorders of sex development (DSDs) include at least 50 different congenital abnormalities of urogenital differentiation
-
The terms 'variations of sex development' and 'differences of sex development' can also be used to describe individuals with DSDs
-
Diagnosis of DSDs requires clinical assessment, morphological determination, endocrine evaluation and genetic studies; however, the pathogenetic mechanisms have only been elucidated in a subset of individuals with DSDs
-
Management of patients with DSDs should involve a group of specialists working in an interdisciplinary team in a dedicated Centre of Expertise
-
Important areas of DSD research include personalized approaches to patient management and the development of diagnostic strategies that combine modern genetic techniques with endocrine evaluations
Abstract
The medical term disorders of sex development (DSDs) is used to describe individuals with an atypical composition of chromosomal, gonadal and phenotypic sex, which leads to differences in the development of the urogenital tract and reproductive system. A variety of genetic factors have been identified that affect sex development during gonadal differentiation or in specific disorders associated with altered androgen biosynthesis or action. The diagnosis of DSDs in individuals and the subsequent management of patients and their families requires a targeted and structured approach, involving a multidisciplinary team with effective communication between the disciplines. This approach includes distinct clinical, imaging, laboratory and genetic evaluations of patients with DSDs. Although treatment of patients with DSDs can include endocrine and surgical options, many patients have concerns that arise from past incorrect treatments that were founded on the traditional binary concept of the sexes. To dispel these concerns, it is necessary to create centres of expertise for DSDs that include physicians, surgeons, psychologists and specialists in diagnostic procedures to manage patients and their families. Additionally, the inclusion of trained peer support in the multidisciplinary DSD team seems to be integral to the supportive management of patients with DSDs. Most importantly, dealing with DSDs requires acceptance of the fact that deviation from the traditional definitions of gender is not necessarily pathologic.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Hughes, I. A., Houk, C., Ahmed, S. F. & Lee, P. A. Consensus statement on management of intersex disorders. Arch. Dis. Child. 91, 554–563 (2006).
Vilain, E. et al. We used to call them hermaphrodites. Genet. Med. 9, 65–66 (2007).
Pasterski, V., Prentice, P. & Hughes, I. A. Consequences of the Chicago consensus on disorders of sex development (DSD): current practices in Europe. Arch. Dis. Child. 95, 618–623 (2010).
Miyagawa, S. et al. Genetic interactions of the androgen and Wnt/β-catenin pathways for the masculinization of external genitalia. Mol. Endocrinol. 23, 871–880 (2009).
Bashamboo, A. & McElreavey, K. Gene mutations associated with anomalies of human gonad formation. Sex. Dev. 7, 126–146 (2013).
Ono, M. & Harley, V. R. Disorders of sex development: new genes, new concepts. Nat. Rev. Endocrinol. 9, 79–91 (2013).
Koboldt, D. C., Steinberg, K. M., Larson, D. E., Wilson, R. K. & Mardis, E. R. The next-generation sequencing revolution and its impact on genomics. Cell 155, 27–38 (2013).
Ledig, S. et al. Array-CGH analysis in patients with syndromic and non-syndromic XY gonadal dysgenesis: evaluation of array CGH as diagnostic tool and search for new candidate loci. Hum. Reprod. 25, 2637–2646 (2010).
Hiort, O. The differential role of androgens in early human sex development. BMC Med. 11, 152 (2013).
Ammerpohl, O. et al. Androgen receptor function links human sexual dimorphism to DNA methylation. PLoS ONE 8, e73288 (2013).
Piferrer, F. Epigenetics of sex determination and gonadogenesis. Dev. Dyn. 242, 360–370 (2013).
Klebs, E. Handbuch der pathologischen Anatomie (August Hirschwald, 1876).
Money, J., Hampson, J. G. & Hampson, J. L. Hermaphroditism: recommendations concerning assignment of sex, change of sex and psychologic management. Bull. Johns Hopkins Hosp. 97, 284–300 (1955).
Diamond, M. & Sigmundson, H. K. Sex reassignment at birth. Long-term review and clinical implications. Arch. Pediatr. Adolesc. Med. 151, 298–304 (1997).
Diamond, M. & Sigmundson, H. K. Management of intersexuality. Guidelines for dealing with persons with ambiguous genitalia. Arch. Pediatr. Adolesc. Med. 151, 1046–1050 (1997).
Diamond, M. & Beh, H. G. Changes in the management of children with intersex conditions. Nat. Clin. Pract. Endocrinol. Metab. 4, 4–5 (2008).
Diamond, M. & Garland, J. Evidence regarding cosmetic and medically unnecessary surgery on infants. J. Pediatr. Urol. 10, 2–6 (2014).
Meyer-Bahlburg, H. F. Gender assignment and reassignment in 46,XY pseudohermaphroditism and related conditions. J. Clin. Endocrinol. Metab. 84, 3455–3458 (1999).
Hines, M. Sex steroids and human behavior: prenatal androgen exposure and sex-typical play behavior in children. Ann. NY Acad. Sci. 1007, 272–282 (2003).
Jurgensen, M., Hiort, O., Holterhus, P. M. & Thyen, U. Gender role behavior in children with XY karyotype and disorders of sex development. Horm. Behav. 51, 443–453 (2007).
Lux, A., Kropf, S., Kleinemeier, E., Jurgensen, M. & Thyen, U. Clinical evaluation study of the German network of disorders of sex development (DSD)/intersexuality: study design, description of the study population, and data quality. BMC Public Health 9, 110 (2009).
Jurgensen, M. et al. Health-related quality of life in children with disorders of sex development (DSD). Eur. J. Pediatr. http://dx.doi.org/10.1007/s00431-014-2264-z.
Jurgensen, M. et al. Psychosexual development in adolescents and adults with disorders of sex development—results from the German clinical evaluation study. J. Sex. Med. 10, 2703–2714 (2013).
Kohler, B. et al. Satisfaction with genital surgery and sexual life of adults with XY disorders of sex development: results from the German clinical evaluation study. J. Clin. Endocrinol. Metab. 97, 577–588 (2012).
Faisal Ahmed, S. et al. UK guidance on the initial evaluation of an infant or an adolescent with a suspected disorder of sex development. Clin. Endocrinol. (Oxf.) 75, 12–26 (2011).
Thyen, U., Lanz, K., Holterhus, P. M. & Hiort, O. Epidemiology and initial management of ambiguous genitalia at birth in Germany. Horm. Res. 66, 195–203 (2006).
Deutscher Ethikrat. Stellungnahme Intersexualität [online], (2012).
The European Union Committee of Experts on Rare Diseases. [online], (2014).
Brain, C. E. et al. Holistic management of DSD. Best Pract. Res. Clin. Endocrinol. Metab. 24, 335–354 (2010).
Ahmed, F., Bryce, J. & Hiort, O. International networks for supporting research & clinical care in the field of DSD. Endocr. Dev. http://dx.doi.org/10.1159/000363676.
Moran, M. E. & Karkazis, K. Developing a multidisciplinary team for disorders of sex development: planning, implementation, and operation tools for care providers. Adv. Urol. 2012, 604135 (2012).
Prader, A. Genital findings in the female pseudo-hermaphroditism of the congenital adrenogenital syndrome; morphology, frequency, development and heredity of the different genital forms. Helv. Paediatr. Acta 9, 231–248 (1954).
Quigley, C. A. et al. Androgen receptor defects: historical, clinical, and molecular perspectives. Endocr. Rev. 16, 271–321 (1995).
Sinnecker, G. H., Hiort, O., Nitsche, E. M., Holterhus, P. M. & Kruse, K. Functional assessment and clinical classification of androgen sensitivity in patients with mutations of the androgen receptor gene. German Collaborative Intersex Study Group. Eur. J. Pediatr. 156, 7–14 (1997).
Ahmed, S. F., Khwaja, O. & Hughes, I. A. The role of a clinical score in the assessment of ambiguous genitalia. BJU Int. 85, 120–124 (2000).
Thankamony, A. et al. Anogenital distance and penile length in infants with hypospadias or cryptorchidism: comparison with normative data. Environ. Health Perspect. 122, 207–211 (2014).
Tanner, J. M. & Eveleth, P. B. in Puberty: Biologic and Psychosocial Components (ed. Berenberg, S. R.) 256–273 (Stenfert Kroese, 1975).
Lloyd, J., Crouch, N. S., Minto, C. L., Liao, L. M. & Creighton, S. M. Female genital appearance: “normality” unfolds. BJOG 112, 643–646 (2005).
Wunsch, L. Checklist for the structural description of the deep phenotype in disorders of sexual development. Int. J. Endocrinol. 2012, 816365 (2012).
Hiort, O., Wunsch, L., Cools, M., Looijenga, L. & Cuckow, P. Requirements for a multicentric multidisciplinary registry on patients with disorders of sex development. J. Pediatr. Urol. 8, 624–628 (2012).
Tzeng, W. Y., Chen, L. H., Cherng, C. G., Tsai, Y. N. & Yu, L. Sex differences and the modulating effects of gonadal hormones on basal and the stressor-decreased newly proliferative cells and neuroblasts in dentate gyrus. Psychoneuroendocrinology 42, 24–37 (2014).
Zhou, J. N., Hofman, M. A., Gooren, L. J. & Swaab, D. F. A sex difference in the human brain and its relation to transsexuality. Nature 378, 68–70 (1995).
Joint LWPES/ESPE CAH Working Group. Consensus statement on 21-hydroxylase deficiency from the Lawson Wilkins Pediatric Endocrine Society and the European Society for Paediatric Endocrinology. J. Clin. Endocrinol. Metab. 87, 4048–4053 (2002).
Handelsman, D. J. & Wartofsky, L. Requirement for mass spectrometry sex steroid assays in the Journal of Clinical Endocrinology and Metabolism. J. Clin. Endocrinol. Metab. 98, 3971–3973 (2013).
Kulle, A. E., Riepe, F. G., Melchior, D., Hiort, O. & Holterhus, P. M. A novel ultrapressure liquid chromatography tandem mass spectrometry method for the simultaneous determination of androstenedione, testosterone, and dihydrotestosterone in pediatric blood samples: age- and sex-specific reference data. J. Clin. Endocrinol. Metab. 95, 2399–2409 (2010).
Koal, T., Schmiederer, D., Pham-Tuan, H., Rohring, C. & Rauh, M. Standardized LC–MS/MS based steroid hormone profile-analysis. J. Steroid Biochem. Mol. Biol. 129, 129–138 (2012).
Courant, F. et al. Assessment of circulating sex steroid levels in prepubertal and pubertal boys and girls by a novel ultrasensitive gas chromatography–tandem mass spectrometry method. J. Clin. Endocrinol. Metab. 95, 82–92 (2010).
Kulle, A. E., Welzel, M., Holterhus, P. M. & Riepe, F. G. Principles and clinical applications of liquid chromatography–tandem mass spectrometry for the determination of adrenal and gonadal steroid hormones. J. Endocrinol. Invest. 34, 702–708 (2011).
Kulle, A. E., Welzel, M., Holterhus, P. M. & Riepe, F. G. Implementation of a liquid chromatography tandem mass spectrometry assay for eight adrenal C-21 steroids and pediatric reference data. Horm. Res. Paediatr. 79, 22–31 (2013).
Hiort, O. et al. Molecular genetic analysis and human chorionic gonadotropin stimulation tests in the diagnosis of prepubertal patients with partial 5α-reductase deficiency. Eur. J. Pediatr. 155, 445–451 (1996).
Ng, K. L., Ahmed, S. F. & Hughes, I. A. Pituitary–gonadal axis in male undermasculinisation. Arch. Dis. Child. 82, 54–58 (2000).
Twesten, W. et al. Clinical, endocrine, and molecular genetic findings in patients with 17β-hydroxysteroid dehydrogenase deficiency. Horm. Res. 53, 26–31 (2000).
Steinmetz, L. et al. Inhibin A production after gonadotropin stimulus: a new method to detect ovarian tissue in ovotesticular disorder of sex development. Horm. Res. 71, 94–99 (2009).
Krone, N. et al. Gas chromatography/mass spectrometry (GC/MS) remains a pre-eminent discovery tool in clinical steroid investigations even in the era of fast liquid chromatography tandem mass spectrometry (LC/MS/MS). J. Steroid Biochem. Mol. Biol. 121, 496–504 (2010).
Wudy, S. A., Hartmann, M. & Homoki, J. Hormonal diagnosis of 21-hydroxylase deficiency in plasma and urine of neonates using benchtop gas chromatography–mass spectrometry. J. Endocrinol. 165, 679–683 (2000).
Krone, N. et al. Genotype–phenotype analysis in congenital adrenal hyperplasia due to P450 oxidoreductase deficiency. J. Clin. Endocrinol. Metab. 97, E257–E267 (2012).
Kamrath, C., Hochberg, Z., Hartmann, M. F., Remer, T. & Wudy, S. A. Increased activation of the alternative “backdoor” pathway in patients with 21-hydroxylase deficiency: evidence from urinary steroid hormone analysis. J. Clin. Endocrinol. Metab. 97, E367–E375 (2012).
Fluck, C. E. et al. Why boys will be boys: two pathways of fetal testicular androgen biosynthesis are needed for male sexual differentiation. Am. J. Hum. Genet. 89, 201–218 (2011).
Rey, R. A. et al. Evaluation of gonadal function in 107 intersex patients by means of serum antimullerian hormone measurement. J. Clin. Endocrinol. Metab. 84, 627–631 (1999).
Josso, N., Rey, R. A. & Picard, J. Y. Anti-Müllerian hormone: A valuable addition to the toolbox of the pediatric endocrinologist. Int. J. Endocrinol. 2013, 674105 (2013).
Kubini, K. et al. Basal inhibin B and the testosterone response to human chorionic gonadotropin correlate in prepubertal boys. J. Clin. Endocrinol. Metab. 85, 134–138 (2000).
Lindhardt Johansen, M. et al. 45,X/46,XY mosaicism: phenotypic characteristics, growth, and reproductive function—a retrospective longitudinal study. J. Clin. Endocrinol. Metab. 97, E1540–E1549 (2012).
Kaufmann, M., Straisfeld, C. & Pinsky, L. Male pseudohermaphroditism presumably due to target organ unresponsiveness to androgens. Deficient 5α-dihydrotestosterone binding in cultured skin fibroblasts. J. Clin. Invest. 58, 345–350 (1976).
Sinnecker, G. & Kohler, S. Sex hormone-binding globulin response to the anabolic steroid stanozolol: evidence for its suitability as a biological androgen sensitivity test. J. Clin. Endocrinol. Metab. 68, 1195–1200 (1989).
Holterhus, P. M. et al. Mosaicism due to a somatic mutation of the androgen receptor gene determines phenotype in androgen insensitivity syndrome. J. Clin. Endocrinol. Metab. 82, 3584–3589 (1997).
Bertelloni, S. et al. Biochemical selection of prepubertal patients with androgen insensitivity syndrome by sex hormone-binding globulin response to the human chorionic gonadotropin test. Pediatr. Res. 41, 266–271 (1997).
Appari, M. et al. Apolipoprotein D (APOD) is a putative biomarker of androgen receptor function in androgen insensitivity syndrome. J. Mol. Med. (Berl.) 87, 623–632 (2009).
Ohnesorg, T., Vilain, E. & Sinclair, A. H. The genetics of disorders of sex development in humans. Sex. Dev. http://dx.doi.org/10.1159/000357956.
Hersmus, R. et al. SRY mutation analysis by next generation (deep) sequencing in a cohort of chromosomal disorders of sex development (DSD) patients with a mosaic karyotype. BMC Med. Genet. 13, 108 (2012).
Tobias, E. & McElreavey, K. Next generation sequencing for disorders of sex development. Endocr. Dev. http://dx.doi.org/10.1159/000363615.
Bashamboo, A. et al. Mutations in the FOG2/ZFPM2 gene are associated with anomalies of human testis determination. Hum. Mol. Genet. http://dx.doi.org/10.1093/hmg/ddu074.
Ledig, S., Hiort, O., Wunsch, L. & Wieacker, P. Partial deletion of DMRT1 causes 46,XY ovotesticular disorder of sexual development. Eur. J. Endocrinol. 167, 119–124 (2012).
Hiort, O. & Gillessen-Kaesbachb, G. Disorders of sex development in developmental syndromes. Endocr. Dev. 14, 174–180 (2009).
Baxter, R. M. & Vilain, E. Translational genetics for diagnosis of human disorders of sex development. Annu. Rev. Genomics Hum. Genet. 14, 371–392 (2013).
Gillam, L. H., Hewitt, J. K. & Warne, G. L. Ethical principles for the management of infants with disorders of sex development. Horm. Res. Paediatr. 74, 412–418 (2010).
Gillam, L. H., Hewitt, J. K. & Warne, G. L. Ethical principles: an essential part of the process in disorders of sex development care. Horm. Res. Paediatr. 76, 367–368 (2011).
Wiesemann, C., Ude-Koeller, S., Sinnecker, G. H. & Thyen, U. Ethical principles and recommendations for the medical management of differences of sex development (DSD)/intersex in children and adolescents. Eur. J. Pediatr. 169, 671–679 (2010).
Hiort, O. et al. Puberty in disorders of somatosexual differentiation. J. Pediatr. Endocrinol. Metab. 16 (Suppl. 2), 297–306 (2003).
Hiort, O., Thyen, U. & Holterhus, P. M. The basis of gender assignment in disorders of somatosexual differentiation. Horm. Res. 64 (Suppl. 2), 18–22 (2005).
Thyen, U., Richter-Appelt, H., Wiesemann, C., Holterhus, P. M. & Hiort, O. Deciding on gender in children with intersex conditions: considerations and controversies. Treat. Endocrinol. 4, 1–8 (2005).
Bertelloni, S. et al. 17β-Hydroxysteroid dehydrogenase-3 deficiency: from pregnancy to adolescence. J. Endocrinol. Invest. 32, 666–670 (2009).
Werner, R. et al. Testosterone synthesis in patients with 17β-hydroxysteroid dehydrogenase 3 deficiency. Sex. Dev. 6, 161–168 (2012).
Hochberg, Z. et al. Clinical, biochemical, and genetic findings in a large pedigree of male and female patients with 5α-reductase 2 deficiency. J. Clin. Endocrinol. Metab. 81, 2821–2827 (1996).
Kang, H. J., Imperato-McGinley, J., Zhu, Y. S. & Rosenwaks, Z. The effect of 5α-reductase-2 deficiency on human fertility. Fertil. Steril. 101, 310–316 (2014).
Jurgensen, M., Hampel, E., Hiort, O. & Thyen, U. “Any decision is better than none” decision-making about sex of rearing for siblings with 17β-hydroxysteroid-dehydrogenase-3 deficiency. Arch. Sex. Behav. 35, 359–371 (2006).
T'Sjoen, G. et al. Male gender identity in complete androgen insensitivity syndrome. Arch. Sex. Behav. 40, 635–638 (2011).
Bundesregierung Deutschland. Gesetz zur Änderung personenstandsrechtlicher Vorschriften (Personenstandsrechts-Änderungsgesetz – PStRÄndG) [online] (2013).
Creighton, S. M., Michala, L., Mushtaq, I. & Yaron, M. Childhood surgery for ambiguous genitalia: glimpses of practice changes or more of the same? Psycho. Sex. 5, 34–43 (2013).
United Nations General Assembly. Resolution 67/146. Intensifying global efforts for the elimination of female genital mutilations [online], (2012).
Yankovic, F., Cherian, A., Steven, L., Mathur, A. & Cuckow, P. Current practice in feminizing surgery for congenital adrenal hyperplasia; a specialist survey. J. Pediatr. Urol. 9, 1103–1107 (2013).
Steven, L. et al. Current practice in paediatric hypospadias surgery; a specialist survey. J. Pediatr. Urol. 9, 1126–1130 (2013).
Wolffenbuttel, K. P. & Crouch, N. S. Timing of feminizing surgery. Endocr. Dev. http://dx.doi.org/10.1159/000363665.
Wunsch, L., Holterhus, P. M., Wessel, L. & Hiort, O. Patients with disorders of sex development (DSD) at risk of gonadal tumour development: management based on laparoscopic biopsy and molecular diagnosis. BJU Int. 110, E958–E965 (2012).
Looijenga, L. H., Van Agthoven, T. & Biermann, K. Development of malignant germ cells—the genvironmental hypothesis. Int. J. Dev. Biol. 57, 241–253 (2013).
Cools, M. et al. Pubertal androgenization and gonadal histology in two 46,XY adolescents with NR5A1 mutations and predominantly female phenotype at birth. Eur. J. Endocrinol. 166, 341–349 (2012).
Kaprova-Pleskacova, J. et al. Complete androgen insensitivity syndrome: factors influencing gonadal histology including germ cell pathology. Mod. Pathol. 27, 721–730 (2013).
Warne, G. L., Grover, S. & Zajac, J. D. Hormonal therapies for individuals with intersex conditions: protocol for use. Treat. Endocrinol. 4, 19–29 (2005).
Bertelloni, S., Dati, E. & Baroncelli, G. I. Disorders of sex development: hormonal management in adolescence. Gynecol. Endocrinol. 24, 339–346 (2008).
Jurgensen, M. et al. Psychosexual development in children with disorder of sex development (DSD)—results from the German Clinical Evaluation Study. J. Pediatr. Endocrinol. Metab. 23, 565–578 (2010).
Han, T. S., Goswami, D., Trikudanathan, S., Creighton, S. M. & Conway, G. S. Comparison of bone mineral density and body proportions between women with complete androgen insensitivity syndrome and women with gonadal dysgenesis. Eur. J. Endocrinol. 159, 179–185 (2008).
Kiess, W. et al. Induction of puberty in the hypogonadal girl—practices and attitudes of pediatric endocrinologists in Europe. Horm. Res. 57, 66–71 (2002).
Drobac, S., Rubin, K., Rogol, A. D. & Rosenfield, R. L. A workshop on pubertal hormone replacement options in the United States. J. Pediatr. Endocrinol. Metab. 19, 55–64 (2006).
Walvoord, E. Sex steroid replacement for induction of puberty in multiple pituitary hormone deficiency. Pediatr. Endocrinol. Rev. 6 (Suppl. 2), 298–305 (2009).
Bertelloni, S., Baroncelli, G. I., Garofalo, P. & Cianfarani, S. Androgen therapy in hypogonadal adolescent males. Horm. Res. Paediatr. 74, 292–296 (2010).
Bertelloni, S., Dati, E., Baroncelli, G. I. & Hiort, O. Hormonal management of complete androgen insensitivity syndrome from adolescence onward. Horm. Res. Paediatr. 76, 428–433 (2011).
DSD-Life [online], (2014).
I-DSD Registry [online], (2014).
Ahmed, S. F., Rodie, M., Jiang, J. & Sinnott, R. O. The European disorder of sex development registry: a virtual research environment. Sex. Dev. 4, 192–198 (2010).
Cox, K. et al. Novel associations in disorders of sex development: findings from the I-DSD registry. J. Clin. Endocrinol. Metab. 99, E348–E355 (2014).
Arboleda, V. A. et al. Targeted massively parallel sequencing provides comprehensive genetic diagnosis for patients with disorders of sex development. Clin. Genet. 83, 35–43 (2013).
Holterhus, P. M. et al. Intrinsic androgen-dependent gene expression patterns revealed by comparison of genital fibroblasts from normal males and individuals with complete and partial androgen insensitivity syndrome. BMC Genomics 8, 376 (2007).
Holterhus, P. M. et al. Disorders of sex development expose transcriptional autonomy of genetic sex and androgen-programmed hormonal sex in human blood leukocytes. BMC Genomics 10, 292 (2009).
dsdfamilies [online], (2014).
Acknowledgements
The authors would like to acknowledge the support of the European Science Foundation (COST Action BM1303).
Author information
Authors and Affiliations
Contributions
O.H., W.B., L.M., L.W., R.W., T.S. and P.-M.H. researched data for the article. O.H., W.B., L.M., L.W., R.W., U.D. and P.-M.H. made substantial contributions to discussions of the content. O.H., W.B., L.W., R.W. and P.-M.H. wrote the article. O.H., W.B., L.M., L.W., R.W., T.S., U.D. and P.-M.H. reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Hiort, O., Birnbaum, W., Marshall, L. et al. Management of disorders of sex development. Nat Rev Endocrinol 10, 520–529 (2014). https://doi.org/10.1038/nrendo.2014.108
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2014.108
This article is cited by
-
Sex difference and risk factors in burden of urogenital congenital anomalies from 1990 to 2019
Scientific Reports (2023)
-
A review of the essential concepts in diagnosis, therapy, and gender assignment in disorders of sexual development
Annals of Pediatric Surgery (2022)
-
Metabolic effects of estradiol versus testosterone in complete androgen insensitivity syndrome
Endocrine (2022)
-
Genital anomalies in newborns
Journal of Perinatology (2021)
-
Rari disordini genetici dello sviluppo sessuale
L'Endocrinologo (2021)