Abstract
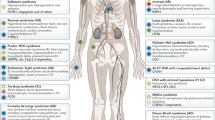
Formerly known as 'intersex' conditions, disorders of sex development (DSDs) are congenital conditions in which chromosomal, gonadal or anatomical sex is atypical. A complete revision of the nomenclature and classification of DSDs has been undertaken, which emphasizes the genetic aetiology of these disorders and discards pejorative terms. Uptake of the new terminology is widespread. DSDs affecting gonadal development are perhaps the least well understood. Unravelling the molecular mechanisms underlying gonadal development has revealed new causes of DSDs, although a specific molecular diagnosis is made in only ∼20% of patients. Conversely, identification of the molecular causes of DSDs has provided insight into the mechanisms of gonadal development. Studies of N-ethyl-N-nitrosourea mutagenesis in the mouse, and multigene diagnostic screening and genome-wide approaches, such as array-comparative genomic hybridization and next-generation sequencing, in patients with DSDs are accelerating the discovery of genes involved in gonadal development and DSDs. Furthermore, long-range gene regulatory mutations and multiple gene mutations are emerging as new causes of DSDs. Patients with DSDs, their parents and medical staff are confronted with challenging decisions regarding gender assignment, genital surgery and lifelong care. These advances are refining prognostic prediction and systematically improving the diagnosis and long-term management of children with DSDs.
Key Points
-
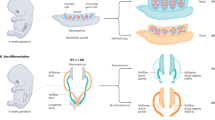
The development of a testis or an ovary from common gonadal primordia is governed by complex molecular networks of gene expression
-
The products of male-specific and female-specific genes promote testis and ovary development, and antagonize each other; disturbance of this fine balance can lead to disorders of sex development (DSDs)
-
A specific molecular diagnosis is currently made in only ∼20% of patients with DSDs; however, advanced genetic technologies could identify new causes of DSDs and their molecular basis
-
DSD diagnosis and management are complex; an experienced multi-disciplinary team is required to work with the patient and their family, placing the patient's quality of life at the forefront
-
New terminology for and classification of DSDs, built around genetic concepts and systematic approaches to lifelong care, have been embraced by health professionals, families and the academic literature
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Hughes, I. A. et al. Consensus statement on management of intersex disorders. Arch. Dis. Child. 91, 554–563 (2006).
Vilain, E. et al. We used to call them hermaphrodites. Genet. Med. 9, 65–66 (2007).
Pasterski, V., Prentice, P. & Hughes, I. A. Impact of the consensus statement and the new DSD classification system. Best Pract. Res. Clin. Endocrinol. Metab. 24, 187–195 (2010).
Biason-Lauber, A. Control of sex development. Best Pract. Res. Clin. Endocrinol. Metab. 24, 163–186 (2010).
Achermann, J. C. & Hughes, I. A. in Williams Textbook of Endocrinology 12th edn Ch. 23 (eds Melmed, S. et al.) 869–934 (Saunders, Philadelphia, 2011).
Barbaro, M., Wedell, A. & Nordenström, A. Disorders of sex development. Semin. Fetal Neonatal Med. 16, 119–127 (2011).
Rey, R. A. & Grinspon, R. P. Normal male sexual differentiation and aetiology of disorders of sex development. Best Pract. Res. Clin. Endocrinol. Metab. 25, 221–238 (2011).
Eggers, S. & Sinclair, A. Mammalian sex determination—insights from humans and mice. Chromosome Res. 20, 215–238 (2012).
Kim, Y. & Capel, B. Balancing the bipotential gonad between alternative organ fates: a new perspective on an old problem. Dev. Dyn. 235, 2292–2300 (2006).
Sekido, R. & Lovell-Badge, R. Sex determination and SRY: down to a wink and a nudge? Trends Genet. 25, 19–29 (2009).
Wilhelm, D., Palmer, S. & Koopman, P. Sex determination and gonadal development in mammals. Physiol. Rev. 87, 1–28 (2007).
Miller, W. L. & Auchus, R. J. The molecular biology, biochemistry, and physiology of human steroidogenesis and its disorders. Endocr. Rev. 32, 81–151 (2011).
Knower, K. C. et al. Failure of SOX9 regulation in 46XY disorders of sex development with SRY, SOX9 and SF1 mutations. PLoS One 6, e17751 (2011).
Knower, K. C. et al. Characterisation of urogenital ridge gene expression in the human embryonal carcinoma cell line NT2/D1. Sex. Dev. 1, 114–126 (2007).
Koopman, P., Münsterberg, A., Capel, B., Vivian, N. & Lovell-Badge, R. Expression of a candidate sex-determining gene during mouse testis differentiation. Nature 348, 450–452 (1990).
Hacker, A., Capel, B., Goodfellow, P. & Lovell-Badge, R. Expression of Sry, the mouse sex determining gene. Development 121, 1603–1614 (1995).
Bullejos, M. & Koopman, P. Spatially dynamic expression of Sry in mouse genital ridges. Dev. Dyn. 221, 201–205 (2001).
Wilhelm, D. et al. Sertoli cell differentiation is induced both cell-autonomously and through prostaglandin signaling during mammalian sex determination. Dev. Biol. 287, 111–124 (2005).
Hanley, N. A. et al. SRY, SOX9, and DAX1 expression patterns during human sex determination and gonadal development. Mech. Dev. 91, 403–407 (2000).
Hammes, A. et al. Two splice variants of the Wilms' tumor 1 gene have distinct functions during sex determination and nephron formation. Cell 106, 319–329 (2001).
Hossain, A. & Saunders, G. F. The human sex-determining gene SRY is a direct target of WT1. J. Biol. Chem. 276, 16817–16823 (2001).
de Santa Barbara, P. et al. Steroidogenic factor-1 contributes to the cyclic-adenosine monophosphate down-regulation of human SRY gene expression. Biol. Reprod. 64, 775–783 (2001).
Tevosian, S. G. et al. Gonadal differentiation, sex determination and normal Sry expression in mice require direct interaction between transcription partners GATA4 and FOG2. Development 129, 4627–4634 (2002).
Katoh-Fukui, Y. et al. Male-to-female sex reversal in M33 mutant mice. Nature 393, 688–692 (1998).
Katoh-Fukui, Y. et al. Cbx2, a polycomb group gene, is required for Sry gene expression in mice. Endocrinology 153, 913–924 (2012).
Warr, N. et al. Gadd45γ and Map3k4 interactions regulate mouse testis determination via p38 MAPK-mediated control of Sry expression. Dev. Cell 23, 1020–1031 (2012).
Gierl, M. S., Gruhn, W. H., von Seggern, A., Maltry, N. & Niehrs, C. GADD45G functions in male sex determination by promoting p38 signaling and Sry expression. Dev. Cell 23, 1032–1042 (2012).
Miyamoto, N., Yoshida, M., Kuratani, S., Matsuo, I. & Aizawa, S. Defects of urogenital development in mice lacking Emx2. Development 124, 1653–1664 (1997).
Nef, S. et al. Testis determination requires insulin receptor family function in mice. Nature 426, 291–295 (2003).
Birk, O. S. et al. The LIM homeobox gene Lhx9 is essential for mouse gonad formation. Nature 403, 909–913 (2000).
Morais da Silva, S. et al. Sox9 expression during gonadal development implies a conserved role for the gene in testis differentiation in mammals and birds. Nat. Genet. 14, 62–68 (1996).
Sekido, R. & Lovell-Badge, R. Sex determination involves synergistic action of SRY and SF1 on a specific Sox9 enhancer. Nature 453, 930–934 (2008).
Barrionuevo, F. et al. Homozygous inactivation of Sox9 causes complete XY sex reversal in mice. Biol. Reprod. 74, 195–201 (2006).
Moniot, B. et al. The PGD2 pathway, independently of FGF9, amplifies SOX9 activity in Sertoli cells during male sexual differentiation. Development 136, 1813–1821 (2009).
Jameson, S. A., Lin, Y. T. & Capel, B. Testis development requires the repression of Wnt4 by Fgf signaling. Dev. Biol. 370, 24–32 (2012).
Oh, C. D. et al. Identification of SOX9 interaction sites in the genome of chondrocytes. PLoS One 5, e10113 (2010).
Chassot, A. A. et al. Activation of β-catenin signaling by Rspo1 controls differentiation of the mammalian ovary. Hum. Mol. Genet. 17, 1264–1277 (2008).
Tomaselli, S. et al. Human RSPO1/R-spondin1 is expressed during early ovary development and augments β-catenin signaling. PloS ONE 6, e16366 (2011).
Tomizuka, K. et al. R-spondin1 plays an essential role in ovarian development through positively regulating Wnt-4 signaling. Hum. Mol. Genet. 17, 1278–1291 (2008).
Yao, H. H. et al. Follistatin operates downstream of Wnt4 in mammalian ovary organogenesis. Dev. Dyn. 230, 210–215 (2004).
Kashimada, K. et al. FOXL2 and BMP2 act cooperatively to regulate follistatin gene expression during ovarian development. Endocrinology 152, 272–280 (2011).
Blount, A. L. et al. FoxL2 and Smad3 coordinately regulate follistatin gene transcription. J. Biol. Chem. 284, 7631–7645 (2009).
Kim, Y. et al. Fgf9 and Wnt4 act as antagonistic signals to regulate mammalian sex determination. PLoS Biol. 4, e187 (2006).
Ottolenghi, C. et al. Loss of Wnt4 and Foxl2 leads to female-to-male sex reversal extending to germ cells. Hum. Mol. Genet. 16, 2795–2804 (2007).
Liu, C. F., Bingham, N., Parker, K. & Yao, H. H. Sex-specific roles of β-catenin in mouse gonadal development. Hum. Mol. Genet. 18, 405–417 (2009).
Maatouk, D. M. et al. Stabilization of β-catenin in XY gonads causes male-to-female sex-reversal. Hum. Mol. Genet. 17, 2949–2955 (2008).
Bernard, P. et al. Wnt signaling in ovarian development inhibits Sf1 activation of Sox9 via the Tesco enhancer. Endocrinology 153, 901–912 (2012).
Lavery, R. et al. XY Sox9 embryonic loss-of-function mouse mutants show complete sex reversal and produce partially fertile XY oocytes. Dev. Biol. 354, 111–122 (2011).
Uhlenhaut, N. H. et al. Somatic sex reprogramming of adult ovaries to testes by FOXL2 ablation. Cell 139, 1130–1142 (2009).
Matson, C. K. et al. DMRT1 prevents female reprogramming in the postnatal mammalian testis. Nature 476, 101–104 (2011).
Ohira, R. et al. Human ARX gene: genomic characterization and expression. Mol. Genet. Metab. 77, 179–188 (2002).
Kitamura, K. et al. Mutation of ARX causes abnormal development of forebrain and testes in mice and X-linked lissencephaly with abnormal genitalia in humans. Nat. Genet. 32, 359–369 (2002).
Gibbons, R. J., Picketts, D. J., Villard, L. & Higgs, D. R. Mutations in a putative global transcriptional regulator cause X-linked mental retardation with α-thalassemia (ATR-X syndrome). Cell 80, 837–845 (1995).
Ion, A. et al. A novel mutation in the putative DNA helicase XH2 is responsible for male-to-female sex reversal associated with an atypical form of the ATR-X syndrome. Am. J. Hum. Genet. 58, 1185–1191 (1996).
Bagheri-Fam, S. et al. Defective survival of proliferating Sertoli cells and androgen receptor function in a mouse model of the ATR-X syndrome. Hum. Mol. Genet. 20, 2213–2224 (2011).
Biason-Lauber, A., Konrad, D., Meyer, M., DeBeaufort, C. & Schoenle, E. J. Ovaries and female phenotype in a girl with 46,XY karyotype and mutations in the CBX2 gene. Am. J. Hum. Genet. 84, 658–663 (2009).
Clark, A. M., Garland, K. K. & Russell, L. D. Desert hedgehog (Dhh) gene is required in the mouse testis for formation of adult-type Leydig cells and normal development of peritubular cells and seminiferous tubules. Biol. Reprod. 63, 1825–1838 (2000).
Parmantier, E. et al. Schwann cell-derived desert hedgehog controls the development of peripheral nerve sheaths. Neuron 23, 713–724 (1999).
Canto, P., Soderlund, D., Reyes, E. & Mendez, J. P. Mutations in the desert hedgehog (DHH) gene in patients with 46,XY complete pure gonadal dysgenesis. J. Clin. Endocrinol. Metab. 89, 4480–4483 (2004).
Umehara, F. et al. A novel mutation of desert hedgehog in a patient with 46,XY partial gonadal dysgenesis accompanied by minifascicular neuropathy. Am. J. Hum. Genet. 67, 1302–1305 (2000).
Canto, P., Vilchis, F., Söderlund, D., Reyes, E. & Méndez, J. P. A heterozygous mutation in the desert hedgehog gene in patients with mixed gonadal dysgenesis. Mol. Hum. Reprod. 11, 833–836 (2005).
Muroya, K. et al. Sex-determining gene(s) on distal 9p: clinical and molecular studies in six cases. J. Clin. Endocrinol. Metab. 85, 3094–3100 (2000).
Ottolenghi, C. et al. The region on 9p associated with 46,XY sex reversal contains several transcripts expressed in the urogenital system and a novel doublesex-related domain. Genomics 64, 170–178 (2000).
Barbaro, M. et al. Characterization of deletions at 9p affecting the candidate regions for sex reversal and deletion 9p syndrome by MLPA. Eur. J. Hum. Genet. 17, 1439–1447 (2009).
Ledig, S., Hiort, O., Wünsch, L. & Wieacker, P. Partial deletion of DMRT1 causes 46,XY ovotesticular disorder of sexual development. Eur. J. Endocrinol. 167, 119–124 (2012).
Raymond, C. S., Murphy, M. W., O'Sullivan, M. G., Bardwell, V. J. & Zarkower, D. Dmrt1, a gene related to worm and fly sexual regulators, is required for mammalian testis differentiation. Genes Dev. 14, 2587–2595 (2000).
Garg, V. et al. GATA4 mutations cause human congenital heart defects and reveal an interaction with TBX5. Nature 424, 443–447 (2003).
Lourenço, D. et al. Loss-of-function mutation in GATA4 causes anomalies of human testicular development. Proc. Natl Acad. Sci. USA 108, 1597–1602 (2011).
Molkentin, J. D., Lin, Q., Duncan, S. A. & Olson, E. N. Requirement of the transcription factor GATA4 for heart tube formation and ventral morphogenesis. Genes Dev. 11, 1061–1072 (1997).
Kuo, C. T. et al. GATA4 transcription factor is required for ventral morphogenesis and heart tube formation. Genes Dev. 11, 1048–1060 (1997).
Fukami, M. et al. CXorf6 is a causative gene for hypospadias. Nat. Genet. 38, 1369–1371 (2006).
Fukami, M. et al. Mastermind-like domain-containing 1 (MAMLD1 or CXorf6) transactivates the Hes3 promoter, augments testosterone production, and contains the SF1 target sequence. J. Biol. Chem. 283, 5525–5532 (2008).
Nakamura, M. et al. Mamld1 knockdown reduces testosterone production and Cyp17a1 expression in mouse Leydig tumor cells. PLoS ONE 6, e19123 (2011).
Miyado, M. et al. Mamld1 deficiency significantly reduces mRNA expression levels of multiple genes expressed in mouse fetal Leydig cells but permits normal genital and reproductive development. Endocrinology 153, 6033–6040 (2012).
Pearlman, A. et al. Mutations in MAP3K1 cause 46,XY disorders of sex development and implicate a common signal transduction pathway in human testis determination. Am. J. Hum. Genet. 87, 898–904 (2010).
Warr, N. et al. Minor abnormalities of testis development in mice lacking the gene encoding the MAPK signalling component, MAP3K1. PLoS ONE 6, e19572 (2011).
Bogani, D. et al. Loss of mitogen-activated protein kinase kinase kinase 4 (MAP3K4) reveals a requirement for MAPK signalling in mouse sex determination. PLoS Biol. 7, e1000196 (2009).
Zanaria, E. et al. An unusual member of the nuclear hormone receptor superfamily responsible for X-linked adrenal hypoplasia congenita. Nature 372, 635–641 (1994).
Muscatelli, F. et al. Mutations in the DAX-1 gene give rise to both X-linked adrenal hypoplasia congenita and hypogonadotropic hypogonadism. Nature 372, 672–676 (1994).
Bardoni, B. et al. A dosage sensitive locus at chromosome Xp21 is involved in male to female sex reversal. Nat. Genet. 7, 497–501 (1994).
Swain, A., Narvaez, V., Burgoyne, P., Camerino, G. & Lovell-Badge, R. Dax1 antagonizes Sry action in mammalian sex determination. Nature 391, 761–767 (1998).
Meeks, J. J., Weiss, J. & Jameson, J. L. Dax1 is required for testis determination. Nat. Genet. 34, 32–33 (2003).
Ludbrook, L. M. & Harley, V. R. Sex determination: a 'window' of DAX1 activity. Trends Endocrinol. Metab. 15, 116–121 (2004).
Ludbrook, L. M. et al. Excess DAX1 leads to XY ovotesticular disorder of sex development (DSD) in mice by inhibiting steroidogenic factor-1 (SF1) activation of the testis enhancer of SRY-box-9 (Sox9). Endocrinology 153, 1948–1958 (2012).
Shen, W. H., Moore, C. C., Ikeda, Y., Parker, K. L. & Ingraham, H. A. Nuclear receptor steroidogenic factor 1 regulates the Müllerian inhibiting substance gene: A link to the sex determination cascade. Cell 77, 651–661 (1994).
Luo, X., Ikeda, Y. & Parker, K. L. A cell-specific nuclear receptor is essential for adrenal and gonadal development and sexual differentiation. Cell 77, 481–490 (1994).
Bland, M. L. et al. Haploinsufficiency of steroidogenic factor-1 in mice disrupts adrenal development leading to an impaired stress response. Proc. Natl Acad. Sci. USA 97, 14488–14493 (2000).
Achermann, J. C. et al. Gonadal determination and adrenal development are regulated by the orphan nuclear receptor steroidogenic factor-1, in a dose-dependent manner. J. Clin. Endocrinol. Metab. 87, 1829–1833 (2002).
Achermann, J. C., Ito, M., Ito, M., Hindmarsh, P. C. & Jameson, J. L. A mutation in the gene encoding steroidogenic factor-1 causes XY sex reversal and adrenal failure in humans. Nat. Genet. 22, 125–126 (1999).
Allali, S. et al. Mutation analysis of NR5A1 encoding steroidogenic factor 1 in 77 patients with 46, XY disorders of sex development (DSD) including hypospadias. PLoS ONE 6, e24117 (2011).
Ferraz-de-Souza, B., Lin, L. & Achermann, J. C. Steroidogenic factor-1 (SF-1, NR5A1) and human disease. Mol. Cell. Endocrinol. 336, 198–205 (2011).
Foster, J. W. et al. Campomelic dysplasia and autosomal sex reversal caused by mutations in an SRY-related gene. Nature 372, 525–530 (1994).
Wagner, T. et al. Autosomal sex reversal and campomelic dysplasia are caused by mutations in and around the SRY-related gene SOX9. Cell 79, 1111–1120 (1994).
Mansour, S., Hall, C. M., Pembrey, M. E. & Young, I. D. A clinical and genetic study of campomelic dysplasia. J. Med. Genet. 32, 415–420 (1995).
Akiyama, H. et al. Essential role of Sox9 in the pathway that controls formation of cardiac valves and septa. Proc. Natl Acad. Sci. USA 101, 6502–6507 (2004).
Chaboissier, M.C. et al. Functional analysis of Sox8 and Sox9 during sex determination in the mouse. Development 131, 1891–1901 (2004).
Benko, S. et al. Disruption of a long distance regulatory region upstream of SOX9 in isolated disorders of sex development. J. Med. Genet. 48, 825–830 (2011).
Cox, J. J., Willatt, L., Homfray, T. & Woods, C. G. A SOX9 duplication and familial 46,XX developmental testicular disorder. N. Engl. J. Med. 364, 91–93 (2011).
Vetro, A. et al. XX males SRY negative: a confirmed cause of infertility. J. Med. Genet. 48, 710–712 (2011).
Georg, I. et al. Mutations of the SRY-responsive enhancer of SOX9 are uncommon in XY gonadal dysgenesis. Sex Dev. 4, 321–325 (2010).
Harley, V. R. The molecular action and regulation of the testis-determining factors, SRY (sex-determining region on the Y chromosome) and SOX9 [SRY-related high-mobility group (HMG) box 9]. Endocr. Rev. 24, 466–487 (2003).
Sinclair, A. H. et al. A gene from the human sex-determining region encodes a protein with homology to a conserved DNA-binding motif. Nature 346, 240–244 (1990).
Berta, P. et al. Genetic evidence equating SRY and the testis-determining factor. Nature 348, 448–450 (1990).
Harley, V. R. et al. DNA binding activity of recombinant SRY from normal males and XY females. Science 255, 453–456 (1992).
Harley, V. R. et al. Defective importin β recognition and nuclear import of the sex-determining factor SRY are associated with XY sex-reversing mutations. Proc. Natl Acad. Sci. USA 100, 7045–7050 (2003).
Sim, H. et al. Inhibition of SRY-calmodulin complex formation induces ectopic expression of ovarian cell markers in developing XY gonads. Endocrinology 152, 2883–2893 (2011).
Jordan, B. K. et al. Up-regulation of WNT-4 signaling and dosage-sensitive sex reversal in humans. Am. J. Hum. Genet. 68, 1102–1109 (2001).
Jordan, B. K., Shen, J. H., Olaso, R., Ingraham, H. A. & Vilain, E. Wnt4 overexpression disrupts normal testicular vasculature and inhibits testosterone synthesis by repressing steroidogenic factor 1/β-catenin synergy. Proc. Natl Acad. Sci. USA 100, 10866–10871 (2003).
Pritchard-Jones, K. et al. The candidate Wilms' tumour gene is involved in genitourinary development. Nature 346, 194–197 (1990).
Kreidberg, J. A. et al. WT-1 is required for early kidney development. Cell 74, 679–691 (1993).
Haber, D. A. et al. An internal deletion within an 11p13 zinc finger gene contributes to the development of Wilms' tumor. Cell 61, 1257–1269 (1990).
Pelletier, J. et al. WT1 mutations contribute to abnormal genital system development and hereditary Wilms' tumour. Nature 353, 431–434 (1991).
Pelletier, J. et al. Germline mutations in the Wilms' tumor suppressor gene are associated with abnormal urogenital development in Denys–Drash syndrome. Cell 67, 437–447 (1991).
Barbaux, S. et al. Donor splice-site mutations in WT1 are responsible for Frasier syndrome. Nat. Genet. 17, 467–470 (1997).
White, S. et al. A multi-exon deletion within WWOX is associated with a 46,XY disorder of sex development. Eur. J. Hum. Genet. 20, 348–351 (2012).
Aqeilan, R. I. et al. Targeted ablation of the WW domain-containing oxidoreductase tumor suppressor leads to impaired steroidogenesis. Endocrinology 150, 1530–1535 (2009).
Ludes-Meyers, J. H. et al. WWOX hypomorphic mice display a higher incidence of B-cell lymphomas and develop testicular atrophy. Genes Chromosomes Cancer 46, 1129–1136 (2007).
Brandao, M. P. et al. MAMLD1 (mastermind-like domain containing 1) homozygous gain-of-function missense mutation causing 46,XX disorder of sex development in a virilized female. Adv. Exp. Med. Biol. 707, 129–131 (2011).
Jeyasuria, P. et al. Cell-specific knockout of steroidogenic factor 1 reveals its essential roles in gonadal function. Mol. Endocrinol. 18, 1610–1619 (2004).
Lourenco, D. et al. Mutations in NR5A1 associated with ovarian insufficiency. N. Engl. J. Med. 360, 1200–1210 (2009).
Fonseca, D. J. et al. CITED2 mutations potentially cause idiopathic premature ovarian failure. Transl. Res. 160, 384–388 (2012).
Parma, P. et al. R-spondin1 is essential in sex determination, skin differentiation and malignancy. Nat. Genet. 38, 1304–1309 (2006).
Tomaselli, S. et al. Syndromic true hermaphroditism due to an R-spondin1 (RSPO1) homozygous mutation. Hum. Mutat. 29, 220–226 (2008).
Houmard, B. et al. Global gene expression in the human fetal testis and ovary. Biol. Reprod. 81, 438–443 (2009).
Sutton, E. et al. Identification of SOX3 as an XX male sex reversal gene in mice and humans. J. Clin. Invest. 121, 328–341 (2011).
Foster, J. W. & Graves, J. A. An SRY-related sequence on the marsupial X chromosome: implications for the evolution of the mammalian testis-determining gene. Proc. Natl Acad. Sci. USA 91, 1927–1931 (1994).
Vidal, V. P., Chaboissier, M. C., de Rooij, D. G. & Schedl, A. Sox9 induces testis development in XX transgenic mice. Nat. Genet. 28, 216–217 (2001).
Huang, B., Wang, S., Ning, Y., Lamb, A. N. & Bartley, J. Autosomal XX sex reversal caused by duplication of SOX9. Am. J. Med. Genet. 87, 349–353 (1999).
Koopman, P., Gubbay, J., Vivian, N., Goodfellow, P. & Lovell-Badge, R. Male development of chromosomally female mice transgenic for Sry. Nature 351, 117–121 (1991).
Vainio, S., Heikkilä, M., Kispert, A., Chin, N. & McMahon, A. P. Female development in mammals is regulated by Wnt-4 signalling. Nature 397, 405–409 (1999).
Biason-Lauber, A., Konrad, D., Navratil, F. & Schoenle, E. J. A WNT4 mutation associated with Müllerian-duct regression and virilization in a 46,XX woman. N. Engl. J. Med. 351, 792–798 (2004).
Mandel, H. et al. SERKAL syndrome: an autosomal-recessive disorder caused by a loss-of-function mutation in WNT4. Am. J. Hum. Genet. 82, 39–47 (2008).
Hersmus, R. et al. A 46,XY female DSD patient with bilateral gonadoblastoma, a novel SRY missense mutation combined with a WT1 KTS splice-site mutation. PLoS ONE 7, e40858 (2012).
Arboleda, V. A. et al. Targeted massively parallel sequencing provides comprehensive genetic diagnosis for patients with disorders of sex development. Clin. Genet. http://dx.doi.org/10.1111/j.1399-0004.2012.01879.x.
Nef, S. et al. Gene expression during sex determination reveals a robust female genetic program at the onset of ovarian development. Dev. Biol. 287, 361–377 (2005).
Beverdam, A. & Koopman, P. Expression profiling of purified mouse gonadal somatic cells during the critical time window of sex determination reveals novel candidate genes for human sexual dysgenesis syndromes. Hum. Mol. Genet. 15, 417–431 (2006).
Wolf, S. M. et al. Managing incidental findings in human subjects research: analysis and recommendations. J. Law Med. Ethics 36, 219–248 (2008).
Biesecker, L. G. Opportunities and challenges for the integration of massively parallel genomic sequencing into clinical practice: lessons from the ClinSeq project. Genet. Med. 14, 393–398 (2012).
Russell, W. L. et al. Specific-locus test shows ethylnitrosourea to be the most potent mutagen in the mouse. Proc. Natl Acad. Sci. USA 76, 5818–5819 (1979).
Gondo, Y., Fukumura, R., Murata, T. & Makino, S. ENU-based gene-driven mutagenesis in the mouse: a next-generation gene-targeting system. Exp. Anim. 59, 537–548 (2010).
Weiss, J. et al. ENU mutagenesis in mice identifies candidate genes for hypogonadism. Mamm. Genome 23, 346–355 (2012).
Bagheri-Fam, S. et al. Loss of Fgfr2 leads to partial XY sex reversal. Dev. Biol. 314, 71–83 (2008).
Buganim, Y. et al. Direct reprogramming of fibroblasts into embryonic sertoli-like cells by defined factors. Cell Stem Cell 11, 373–386 (2012).
Ahmed, S. F. et al. UK guidance on the initial evaluation of an infant or an adolescent with a suspected disorder of sex development. Clin. Endocrinol. (Oxf.) 75, 12–26 (2011).
Crissman, H. P. et al. Children with disorders of sex development: a qualitative study of early parental experience. Int. J. Pediatr. Endocrinol. 2011, 10 (2011).
Cools, M., Wolffenbuttel, K. P., Drop, S. L., Oosterhuis, J. W. & Looijenga, L. H. Gonadal development and tumor formation at the crossroads of male and female sex determination. Sex. Dev. 5, 167–180 (2011).
Looijenga, L. H. et al. Gonadal tumours and DSD. Best Pract. Res. Clin. Endocrinol. Metab. 24, 291–310 (2010).
University of Glasgow. The I-DSD Network [online], (2012).
Ahmed, S. F., Rodie, M., Jiang, J. & Sinnott, R. O. The European disorder of sex development registry: a virtual research environment. Sex. Dev. 4, 192–198 (2010).
University of California San Francisco. Disorders of Sex Development—Translational Research Network [online], (2012).
Murdock Childrens Research Institute. DSD Network Online Registry [online], (2012).
Gillam, L. H., Hewitt, J. K. & Warne, G. L. Ethical principles for the management of infants with disorders of sex development. Horm. Res. Paediatr. 74, 412–418 (2010).
Vidal, I. et al. Surgical options in disorders of sex development (DSD) with ambiguous genitalia. Best Pract. Res. Clin. Endocrinol. Metab. 24, 311–324 (2010).
Wiesemann, C., Ude-Koeller, S., Sinnecker, G. H. & Thyen, U. Ethical principles and recommendations for the medical management of differences of sex development (DSD)/intersex in children and adolescents. Eur. J. Pediatr. 169, 671–679 (2010).
Acknowledgements
The authors thank P. Bernard, S. Bagheri-Fam, R. Lavery, J. Ryan, D. Alankarage and R. Sreenivasan at Prince Henry's Institute, Melbourne, Australia for critical reading of this manuscript, and A. Greenfield of the Medical Research Council, Harwell, UK and E. Turbitt at Murdoch Children's Research Institute, Melbourne, Australia for fruitful discussions of its content. The authors' research work was supported by National Health and Medical Research Council Program Grant 546517 and Fellowship 1020034 to V. R. Harley and the State of Victoria Operational Infrastructure Support Program, Australia.
Author information
Authors and Affiliations
Contributions
Both authors contributed equally to researching data for the article, discussing the content, writing the manuscript, and reviewing and/or editing the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary information Table 1
Classification of DSDs (DOC 44 kb)
Supplementary information Table 2
Genes and proteins implicated in disorders of sex development (DOC 94 kb)
Rights and permissions
About this article
Cite this article
Ono, M., Harley, V. Disorders of sex development: new genes, new concepts. Nat Rev Endocrinol 9, 79–91 (2013). https://doi.org/10.1038/nrendo.2012.235
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2012.235
This article is cited by
-
Yolk sac tumor and dysgerminoma in the left gonad following gonadoblastoma in the right gonad in a 46,XY DSD with a novel SRY missense mutation: a case report
BMC Pregnancy and Childbirth (2023)
-
Pathological variants in genes associated with disorders of sex development and central causes of hypogonadism in a whole-genome reference panel of 8380 Japanese individuals
Human Genome Variation (2022)
-
Clinical and molecular spectrum of 46,XY disorders of sex development that harbour MAMLD1 variations: case series and review of literature
Orphanet Journal of Rare Diseases (2020)
-
Efficacy of intelligent diagnosis with a dynamic uncertain causality graph model for rare disorders of sex development
Frontiers of Medicine (2020)
-
Identification of novel candidate genes for 46,XY disorders of sex development (DSD) using a C57BL/6J-Y POS mouse model
Biology of Sex Differences (2018)