Abstract
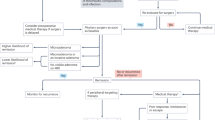
Pituitary tumors, usually adenomas, account for about 10–15% of all intracranial tumors. Their treatment, which includes surgery, medicine or radiotherapy, either isolated or in combination, aims to halt tumor growth or achieve tumor shrinkage, as well as control hormone hypersecretion or ensure hormone replacement. Such approaches have made pregnancy possible for women with pituitary adenomas. Medical therapy with dopamine agonists is the treatment of choice for most patients with prolactinomas, with surgery reserved for individuals resistant to drugs. On the other hand, surgery before conception is indicated as a first-line approach in patients with acromegaly, Cushing disease or clinically nonfunctioning pituitary macroadenomas. In these patient populations, medical therapy with somatostatin analogues (acromegaly) or drugs that target the adrenal glands, such as metyrapone and ketoconazole (Cushing disease), should be reserved for those in whom surgery is unsuccessful or contraindicated.
Key Points
-
Improvements in medical and surgical treatment strategies have increased the numbers of pregnancies occurring in women with pituitary tumors
-
The overall management goals in pregnancy are effective hormonal and tumor control, avoidance of potential risks to the mother, as well as immediate and long-term consequences to the fetus
-
In patients with microprolactinomas, dopamine agonists are effective and safe for pregnancy induction; in those with macroprolactinomas, the decision for medical or surgical treatment depends on tumor characteristics and drug response
-
Once pregnancy is confirmed, dopamine agonists should be discontinued in patients with prolactinomas, except in selected cases, and reintroduced if tumor re-expansion occurs, whereas surgery is indicated in nonresponsive women
-
In pregnant women with acromegaly, somatostatin analogues should not be used unless absolutely necessary, for example, in cases of recurrent symptoms of substantial tumor growth (headaches or visual field defects)
-
Given the high rates of maternal and fetal morbidities in Cushing disease, pituitary surgery is the first choice, followed by medical treatment that targets the adrenal glands (metyrapone or ketoconazole)
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Bronstein, M. D. in Endocrinology 6th edn Vol. 1 Ch. 18 (eds DeGroot, L. J. & Jameson, J. L.) 333–357 (Elsevier, Philadelphia, 2010).
Klibanski, A. Clinical practice. Prolactinomas. N. Engl. J. Med. 362, 1219–1226 (2010).
Sharma, J. B. et al. Pregnancy outcome in pituitary tumors. Arch. Gynecol. Obstet. 280, 401–404 (2009).
Bronstein, M. D. Prolactinomas and pregnancy. Pituitary 8, 31–38 (2005).
Melmed, S. & Kleinberg, D. in Williams Textbook of Endocrinology 11th edn Ch. 8 (eds Kronenberg, H. M., Melmed, S., Polonsky, K. S. & Larsen, P. R.), 255–261 (Elsevier, 2007).
Okafor, U. V., Onwuekwe, I. O. & Ezegwui, H. U. Management of pituitary adenoma with mass effect in pregnancy: a case report. Cases J. 2, 6350 (2009).
Karaca, Z., Tanriverdi, F., Unluhizarci, K. & Kelestimur, F. Pregnancy and pituitary disorders. Eur. J. Endocrinol. 162, 453–475 (2010).
Molitch, M. E. Pituitary disorders during pregnancy. Endocrinol. Metab. Clin. North Am. 35, 99–116 (2006).
Musolino, N. R. C. & Paraiba, D. B. in Clínica Médica 1st edn Vol. 5 Ch. 8 (eds Martins, M. A. et al.) 96–107 (Manole, 2009).
Casanueva, F. F. et al. Guidelines of the Pituitary Society for the diagnosis and management of prolactinomas. Clin. Endocrinol. (Oxf.) 65, 265–273 (2006).
Gillam, M. P., Molitch, M. E., Lombardi, G. & Colao, A. Advances in the treatment of prolactinomas. Endocr. Rev. 27, 485–534 (2006).
Colao, A. Pituitary tumours: the prolactinoma. Best Pract. Res. Clin. Endocrinol. Metab. 23, 575–596 (2009).
Kars, M., Dekkers, O. M., Pereira, A. M. & Romijn, J. A. Update in prolactinomas. Neth. J. Med. 68, 104–112 (2010).
Gemzell, C. & Wang, C. F. Outcome of pregnancy in women with pituitary adenoma. Fertil. Steril. 31, 363–372 (1979).
Molitch, M. E. Pregnancy and the hyperprolactinemic woman. N. Engl. J. Med. 312, 1364–1370 (1985).
Musolino, N. R. C. & Bronstein, M. D. in Pituitary Tumors in Pregnancy 2nd edn Ch. 4 (ed. Bronstein, M. D.) 91–108 (Kluwer Academic Publishers, 2001).
Molitch, M. E. Prolactinomas and pregnancy. Clin. Endocrinol. (Oxf.) 73, 147–148 (2010).
Lebbe, M., Hubinont, C., Bernard, P. & Maiter, D. Outcome of 100 pregnancies initiated under treatment with cabergoline in hyperprolactinaemic women. Clin. Endocrinol. (Oxf.) 73, 236–242 (2010).
Holmgren, U., Bergstrand, G., Hagenfeldt, K. & Werner, S. Women with prolactinoma—effect of pregnancy and lactation on serum prolactin and on tumour growth. Acta Endocrinol. 111, 452–459 (1986).
Robert, E., Musatti, L., Piscitelli, G. & Ferrari, C. I. Pregnancy outcome after treatment with the ergot derivative, cabergoline. Reprod. Toxicol. 10, 333–337 (1996).
Krupp, P. & Monka, C. Bromocriptine in pregnancy: safety aspects. Klin. Wochenschr. 65, 823–827 (1987).
Ricci, E. et al. Pregnancy outcome after cabergoline treatment in early weeks of gestation. Reprod. Toxicol. 16, 791–793 (2002).
Colao, A. et al. Pregnancy outcomes following cabergoline treatment: extended results from a 12-year observational study. Clin. Endocrinol. (Oxf.) 68, 66–71 (2008).
Ono, M. et al. Individualized high-dose cabergoline therapy for hyperprolactinemic infertility in women with micro- and macroprolactinomas. J. Clin. Endocrinol. Metab. 95, 2672–2679 (2010).
Webster, J. A comparative review of the tolerability profiles of dopamine agonists in the treatment of hyperprolactinaemia and inhibition of lactation. Drug Saf. 14, 228–238 (1996).
Mancini, T., Casanueva, F. F. & Giustina, A. Hyperprolactinemia and prolactinomas. Endocrinol. Metab. Clin. North Am. 37, 67–99 (2008).
Rasmussen, C., Bergh, T., Nillius, S. J. & Wide, L. Return of menstruation and normalization of prolactin in hyperprolactinemic women with bromocriptine-induced pregnancy. Fertil. Steril. 44, 31–34 (1985).
Bergh, T., Nillius, S. J. & Wide, L. Clinical course and outcome of pregnancies in amenorrhoeic women with hyperprolactinaemia and pituitary tumors. Br. Med. J. 1, 875–880 (1978).
Crosignani, P. G. et al. Long-term effects of time, medical treatment and pregnancy in 176 hyperprolactinemic women. Eur. J. Obstet. Gynecol. Reprod. Biol. 44, 175–180 (1992).
Badawy, S. Z., Marziale, J. C., Rosenbaum, A. E., Chang, J. K. & Joy, S. E. The long-term effects of pregnancy and bromocriptine treatment on prolactinomas—the value of radiologic studies. Early Pregnancy 3, 306–311 (1997).
Yamada, M. et al. Spontaneous pregnancy after a pregnancy induced by treatment in hyperprolactinemic women. Eur. J. Obstet. Gynecol. Reprod. Biol. 35, 125–129 (1990).
Christin-Maître, S., Delemer, B., Touraine, P. & Young, J. Prolactinoma and estrogens: pregnancy, contraception and hormonal replacement therapy. Ann. Endocrinol. (Paris) 68, 106–112 (2007).
Peillon, F. et al. Prolactin secretion and synthesis in short- and long-term organ culture of pituitary tumours from acromegalic patients. Acta Endocrinol. 87, 701–715 (1978).
Freeman, R. et al. Pregnancy-associated subacute hemorrhage into a prolactinoma resulting in diabetes insipidus. Fertil. Steril. 58, 427–429 (1992).
Freeman, M. E., Kanyicska, B., Lerant, A. & Nagy, G. Prolactin: structure, function, and regulation of secretion. Physiol. Rev. 80, 1523–1631 (2000).
George, D. L., Phillips, J. A. 3rd, Francke, U. & Seeburg, P. H. The genes for growth hormone and chorionic somatomammotropin are on the long arm of human chromosome 17 in region q21 to qter. Hum. Genet. 57, 138–141 (1981).
MacLeod, J. N., Lee, A. K., Liebhaber, S. A. & Cooke, N. E. Developmental control and alternative splicing of the placentally expressed transcripts from the human growth hormone gene cluster. J. Biol. Chem. 267, 14219–14226 (1992).
Chen, E. Y. et al. The human growth hormone locus nucleotidic sequence, biology, and evolution. Genomics 4, 479–497 (1989).
Swenne, I., Hill, D. J., Strain, A. J. & Milner, R. D. Effects of human placental lactogen and growth hormone on the production of insulin and somatomedin C/insulin-like growth factor I by human fetal pancreas in tissue culture. J. Endocrinol. 113, 297–303 (1987).
Frankenne, F. et al. The physiology of growth hormones (GHs) in pregnant women and partial characterization of the placental GH variant. J. Clin. Endocrinol. Metab. 66, 1171–1180 (1988).
Eriksson, L., Frankenne, F., Edèn, S., Hennen, G. & Von Schoultz, B. Growth hormone 24-h serum profiles during pregnancy—lack of pulsatility for the secretion of the placental variant. Br. J. Obstet. Gynaecol. 9 6, 949 (1989).
Eriksson, L., Edén, S., Fröhlander, N., Bengtsson, B. A. & von Schoultz, B. Continuous 24-hour secretion of growth hormone during late pregnancy. A regulator of maternal metabolic adjustment? Acta Obstet. Gynecol. Scand. 67, 543–547 (1988).
McIntyre, H. D. et al. Placental growth hormone (GH), GH-binding protein, and insulin-like growth factor axis in normal, growth-retarded, and diabetic pregnancies: correlations with fetal growth. J. Clin. Endocrinol. Metab. 85, 1143–1150 (2000).
Caufriez, A. et al. Placental growth hormone as a potential regulator of maternal IGF-1 during human pregnancy. Am. J. Physiol. 258 (Pt. 1), E1014–E1019 (1990).
Caufriez, A., Frankenne, F., Hennen, G. & Copinschi, G. Regulation of maternal IGF-1 by placental GH in normal and abnormal human pregnancies. Am. J. Physiol. 265 (Pt. 1), E572–E577 (1993).
Alsat, E., Guibourdenche, J., Luton, D., Frankenne, F. & Evain-Brion, D. Human placental growth hormone. Am. J. Obstet. Gynecol. 177, 1526 (1997).
Daughaday, W. H., Trivedi, B., Winn, H. N. & Yan, H. Hypersomatotropism in pregnant women, as measured by a human liver radioreceptor assay. J. Clin. Endocrinol. Metab. 70, 215–221 (1990).
Mirlesse, V. et al. Placental growth hormone levels in normal pregnancy and in pregnancies with intrauterine growth retardation. Pediatr. Res. 34, 439–442 (1993).
Kaltsas, G. A. et al. Menstrual irregularity in women with acromegaly. J. Clin. Endocrinol. Metab. 84, 2731–2735 (1999).
Katznelson, L. et al. Hypogonadism in patients with acromegaly: data from the multi-centre acromegaly registry pilot study. Clin. Endocrinol. (Oxf.) 54, 183–188 (2001).
Kaltsas, G. A. et al. Polycystic ovaries and the polycystic ovary syndrome phenotype in women with active acromegaly. Clin. Endocrinol. (Oxf.). 67, 917–922 (2007).
Grynberg, M., Salenave, S., Young, J. & Chanson, P. Female gonadal function before and after treatment of acromegaly. J. Clin. Endocrinol. Metab. 95, 4518–4525 (2010).
Jamnongjit, M., Gill, A. & Hammes, S. R. Epidermal growth factor receptor signaling is required for normal ovarian steroidogenesis and oocyte maturation. Proc. Natl Acad. Sci. USA 102, 16257–16261 (2005).
Caron, P. et al. Acromegaly and Pregnancy: A retrospective multicenter study of 59 pregnancies in 46 women. J. Clin. Endocrinol. Metab. 95, 4680–4687 (2010).
Beckers, A., Stevenaert, A., Foidart, J. M., Hennen, G. & Frankenne, F. Placental and pituitary growth hormone secretion during pregnancy in acromegalic women. J. Clin. Endocrinol. Metab. 71, 725–731 (1990).
Serri, O. & Lanoie, G. Successful pregnancy in a woman with acromegaly treated with octreotide long-acting release. Endocrinologist 13, 17–19 (2003).
Lau, S. L., McGrath, S., Evain-Brion, D. & Smith, R. Clinical and biochemical improvement in acromegaly during pregnancy. J. Endocrinol. Invest. 31, 255–261 (2008).
Bétéa, D., Valdes Socin, H., Hansen, I., Stevenaert, A. & Beckers, A. Acromegaly and pregnancy [French]. Ann. Endocrinol. (Paris) 63, 457–463 (2002).
Lunardi, P., Rizzo, A., Missori, P. & Fraioli, B. Pituitary apoplexy in an acromegalic woman operated on during pregnancy by transphenoidal approach. Int. J. Gynaecol. Obstet. 34, 71–74 (1991).
Atmaca, A., Dagdelen, S. & Erbas, T. Follow-up of pregnancy in acromegalic women: different presentations and outcomes. Exp. Clin. Endocrinol. Diabetes 114, 135–139 (2006).
Cozzi, R., Attanasio, R. & Barausse, M. Pregnancy in acromegaly: a one-center experience. Eur. J. Endocrinol. 155(2), 279–284 (2006).
Leung, K. C., Johannsson, G., Leong, G. M. & Ho, K. K. Estrogen regulation of growth hormone action. Endocr. Rev. 25, 693–721 (2004).
Obuobie, K. et al. McCune–Albright syndrome: growth hormone dynamics in pregnancy. J. Clin. Endocrinol. Metab. 86, 2456–2458 (2001).
Herman-Bonert, V., Seliverstov, M. & Melmed, S. Pregnancy in acromegaly: Successful therapeutic outcome. J. Clin. Endocrinol. Metab. 83, 727–731 (1998).
Abelove, W. A., Rupp, J. J. & Paschkis, K. E. Acromegaly and pregnancy. J. Clin. Endocrinol. Metab. 14, 32–44 (1954).
Qureshi, A. et al. IVF/ICSI in a woman with active acromegaly: successful outcome following treatment with pegvisomant. J. Assist. Reprod. Genet. 23, 439–442 (2006).
Guven, S. et al. A case of acromegaly in pregnancy: concomitant transphenoidal adenomectomy and cesarean section. J. Matern. Fetal Neonatal Med. 19, 69–71 (2006).
Shnider, S. M. & Levinson, G. in Anesthesia for Obstetrics 4th edn Vol. 2 Ch. 29 (ed. Miller, R. D.) 509–528 (Lippincott Williams & Wilkins, 2002).
Caron, P., Gerbeau, C. & Pradayrol, L. Maternal-fetal transfer of octreotide. N. Engl. J. Med. 333, 601–602 (1995).
Caron, P., Gerbeau, C., Pradayrol, L., Cimonetta, C. & Bayard, F. Successful pregnancy in an infertile woman with a thyrotropin-secreting macroadenoma treated with somatostatin analog (octreotide). J. Clin. Endocrinol. Metab. 81, 1164–1168 (1996).
Caron, P. et al. Expression of somatostatin receptor SST4 in human placenta and absence of octreotide effect on human placental growth hormone concentration during pregnancy. J. Clin. Endocrinol. Metab. 82, 3771–3776 (1997).
Maffei, P. et al. Effects of octreotide exposure during pregnancy in acromegaly. Clin. Endocrinol. (Oxf.) 72, 668–677 (2010).
Tsalikian, E., Foley, T. P. Jr & Becker, D. J. Characterization of somatostatin specific binding in plasma membranes of human placenta. Pediatr. Res. 18, 953–957 (1984).
Fassnacht, M., Capeller, B., Arlt, W., Steck, T. & Allolio, B. Octreotide LAR treatment throughout pregnancy in acromegalic woman. Clin. Endocrinol. (Oxf.) 5 5, 411–415 (2001).
Bigazzi, M. et al. A pregnancy in an acromegalic woman during bromocriptine treatment: effects on growth hormone and prolactin in the maternal, fetal and amniotic compartments. J. Clin. Endocrinol. Metab. 48, 9–12 (1979).
Aono, T., Shioji, T., Kohno, M., Ueda, G. & Kurachi, K. Pregnancy following 2-bromo-alpha-ergocryptine (CB-154)-induced ovulation in an acromegalic patient with galactorrhea and amenorrhea. Fertil. Steril. 27, 341–344 (1976).
Espersen, T. & Ditzel, J. Pregnancy and delivery under bromocriptine therapy. Lancet 2, 985–986 (1977).
Luboshitzky, R., Dickstein, G. & Barzilai, D. Bromocriptine induced pregnancy in an acromegalic patient. JAMA 244, 584–586 (1980).
Hisano, M. et al. An acromegalic woman first diagnosed in pregnancy. Arch. Gynecol. Obstet. 274, 171–173 (2005).
Brian, S. R., Bidlingmaier, M., Wajnrajch, M. P., Weinzimer, S. A. & Inzucchi, S. E. Treatment of acromegaly with pegvisomant during pregnancy: maternal and fetal effects. J. Clin. Endocrinol. Metab. 92, 3374–3377 (2007).
Vilar, L., Freitas, Mda. C., Lima, L. H., Lyra, R. & Kater, C. E. Cushing's syndrome in pregnancy: an overview. Arq. Bras. Endocrinol. Metabol. 51, 1293–1302 (2007).
Hunt, A. B. & McConahey, W. M. Pregnancy associated with disease of the adrenal glands. Am. J. Obstet. Gynecol. 66, 970–987 (1953).
Buescher, M. A., McClamrock, H. D. & Adashi, E. Y. Cushing syndrome in pregnancy. Obstet. Gynecol. 79, 130–137 (1992).
Pickard, J., Jochen, A. L., Sadur, C. N. & Hofeldt, F. D. Cushing's syndrome in pregnancy. Obstet. Gynecol. Surv. 45, 87–93 (1990).
Klibanski, A., Stephen, A. E., Greene, M. F., Blake, M. A. & Wu, C. L. Case records of the Massachusetts General Hospital. Case 36–2006. A 35-year-old pregnant woman with new hypertension. N. Engl. J. Med. 355, 2237–2245 (2006).
Cohade, C. et al. Ectopic Cushing's syndrome due to a pheochromocytoma: a new case in the post-partum and review of literature. Gynecol. Endocrinol. 25, 624–627 (2009).
Chui, M. H. et al. Case report: Adrenal LH/hCG receptor overexpression and gene amplification causing pregnancy-induced Cushing's syndrome. Endocr. Pathol. 20, 256–261 (2009).
Aron, D. C., Schnall, A. M. & Sheeler, L. R. Cushing's syndrome and pregnancy. Am. J. Obstet. Gynecol. 162, 244–252 (1990).
Lindsay, J. R., Jonklaas, J., Oldfield, E. H. & Nieman, L. K. Cushing's syndrome during pregnancy: personal experience and review of the literature. J. Clin. Endocrinol. Metab. 90, 3077–3083 (2005).
Lindsay, J. R. & Nieman, L. K. The hypothalamic–pituitary–adrenal axis in pregnancy: challenges in disease detection and treatment. Endocr. Rev. 26, 775–799 (2005).
Bronstein, M. D., Salgado, L. R. & de Castro Musolino, N. R. Medical management of pituitary adenomas: the special case of management of the pregnant woman. Pituitary 5, 99–107 (2002).
Cabezón, C., Bruno, O. D., Cohen, M., García, S. & Gutman, R. A. Twin pregnancy in a patient with Cushing's disease. Fertil. Steril. 72, 371–372 (1999).
Ross, R. J. et al. Diagnosis and selective cure of Cushing's disease during pregnancy by transsphenoidal surgery. Eur. J. Endocrinol. 132, 722–726 (1995).
Mellor, A., Harvey, R. D., Pobereskin, L. H. & Sneyd, J. R. Cushing's disease treated by trans-sphenoidal selective adenomectomy in mid-pregnancy. Br. J. Anaesth. 80, 850–852 (1998).
Lo, C. Y., Lo, C. M. & Lam, K. Y. Cushing's syndrome secondary to adrenal adenoma during pregnancy. Surg. Endosc. 16, 219–220 (2002).
Bertagna, X., Guignat, L., Groussin, L. & Bertherat, J. Cushing's disease. Best Pract. Res. Clin. Endocrinol. Metab. 23, 607–623 (2009).
Connell, J. M. et al. Pregnancy complicated by Cushing syndrome: potential hazard of metyrapone therapy (case report). Br. J. Obstet. Gynaecol. 92, 1192–1195 (1985).
Hána, V., Dokoupilová, M., Marek, J. & Plavka, R. Recurrent ACTH independent Cushing's syndrome in multiple pregnancies and its treatment with metyrapone. Clin. Endocrinol. (Oxf.). 54, 277–281 (2001).
Wallace, C., Toth, E. L., Lewanczuk, R. Z. & Siminoski, K. Pregnancy-induced Cushing's syndrome in multiple pregnancies. J. Clin. Endocrinol. Metab. 81, 15–21 (1996).
Amado, J. A. et al. Successful treatment with ketoconazole of Cushing's syndrome in pregnancy. Postgrad. Med. J. 66, 221–223 (1990).
Berwaerts, J., Verhelst, J., Mahler, C. & Abs, R. Cushing's syndrome in pregnancy treated by ketoconazole: case report and review of the literature. Gynecol. Endocrinol. 1 3, 175–182 (1999).
Serafini, P., Motta, E. L. A. & White, J. S. in Pituitary Tumors in Pregnancy 2nd edn Ch. 4 (ed Bronstein, M. D) 173–194 (Kluwer Academic Publishers, 2001).
Masding, M. G., Lees, P. D., Gawne-Cain, M. L. & Sandeman, D. D. Visual field compression by a non-secreting pituitary tumour during pregnancy. J. R. Soc. Med. 96, 27–28 (2003).
Caron, P., Gerbeau, C., Pradayrol, L., Simonetta, C. & Bayard, F. Successful pregnancy in an infertile woman with a thyrotropin-secreting macroadenoma treated with somatostatin analog (octreotide). J. Clin. Endocrinol. Metab. 81, 1164–1168 (1996).
Blackhurst, G. et al. The treatment of a thyrotropin-secreting pituitary macroadenoma with octreotide in twin pregnancy. Clin. Endocrinol. (Oxf.) 57, 401–404 (2002).
Chaiamnuay, S., Moster, M., Katz, M. R. & Kim, Y. N. Successful management of a pregnant woman with a TSH secreting pituitary adenoma with surgical and medical therapy. Pituitary 6, 109–113 (2003).
Gonzalez, J. G. et al. Pituitary gland growth during normal pregnancy: an in vivo study using magnetic resonance imaging. Am. J. Med. 85, 217–220 (1988).
Glezer, A. et al. Human macroprolactin displays low biological activity via its homologous receptor in a new sensitive bioassay. J. Clin. Endocrinol. Metab. 91, 1048–1055 (2006).
Author information
Authors and Affiliations
Contributions
All authors researched the data for the article, provided substantial contributions to discussions of the content, wrote the article and reviewed and/or edited the manuscript before submission.
Corresponding author
Ethics declarations
Competing interests
M. D. Bronstein declares an association with the following companies: Ipsen, Novartis, Pfizer (consultant, speaker and grant/research support). D. B. Paraiba declares an association with the following company: Ipsen (grant/research support). R. S. Jallad declares an association with the following company: Pfizer (speaker, grant/research support).
Rights and permissions
About this article
Cite this article
Bronstein, M., Paraiba, D. & Jallad, R. Management of pituitary tumors in pregnancy. Nat Rev Endocrinol 7, 301–310 (2011). https://doi.org/10.1038/nrendo.2011.38
Published:
Issue Date:
DOI: https://doi.org/10.1038/nrendo.2011.38
This article is cited by
-
Maternal Signatures of Cortisol in First Trimester Small-for-Gestational Age
Reproductive Sciences (2022)
-
Management of sellar and parasellar tumors becoming symptomatic during pregnancy: a practical algorithm based on multi-center experience and systematic literature review
Pituitary (2021)
-
Surgical indications for pituitary tumors during pregnancy: a literature review
Pituitary (2020)