Abstract
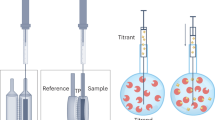
The activity of many biomolecules and drugs crucially depends on whether they bind to biological membranes and whether they translocate to the opposite lipid leaflet and trans aqueous compartment. A general strategy to measure membrane binding and permeation is the uptake and release assay, which compares two apparent equilibrium situations established either by the addition or by the extraction of the solute of interest. Only solutes that permeate the membrane sufficiently fast do not show any dependence on the history of sample preparation. This strategy can be pursued for virtually all membrane-binding solutes, using any method suitable for detecting binding. Here, we present in detail one example that is particularly well developed, namely the nonspecific membrane partitioning and flip-flop of small, nonionic solutes as characterized by isothermal titration calorimetry. A complete set of experiments, including all sample preparation procedures, can typically be accomplished within 2 days. Analogous protocols for studying charged solutes, virtually water-insoluble, hydrophobic compounds or specific ligands are also considered.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Seelig, J. & Ganz, P. Nonclassical hydrophobic effect in membrane binding equilibria. Biochemistry 30, 9354–9359 (1991).
Seelig, J. Titration calorimetry of lipid–peptide interactions. Biochim. Biophys. Acta 1331, 103–116 (1997).
Heerklotz, H. & Seelig, J. Titration calorimetry of surfactant-membrane partitioning and membrane solubilization. Biochim. Biophys. Acta 1508, 69–85 (2000).
Lin, L.N., Li, J.Y., Brandts, J.F. & Weiss, R.M. The serine receptor of bacterial chemotaxis exhibits half-site saturation for serine binding. Biochemistry 33, 6564–6570 (1994).
Heerklotz, H., Lantzsch, G., Binder, H., Klose, G. & Blume, A. Thermodynamic characterization of dilute aqueous lipid/detergent mixtures of POPC and C12EO8 by means of isothermal titration calorimetry. J. Phys. Chem. 100, 6764–6774 (1996).
Ladbury, J.E. Application of isothermal titration calorimetry in the biological sciences: things are heating up!. Biotechniques 37, 885–887 (2004).
Ladbury, J. E. & Doyle, M. L. (eds.) Biocalorimetry Vol. 2 (Wiley-VCH, Weinheim, Germany, 2004).
Heerklotz, H. Microcalorimetry of lipid membranes. J. Phys.: Condens. Matter 16, R441–R467 (2004).
Heerklotz, H.H., Binder, H. & Epand, R.M. A “release” protocol for isothermal titration calorimetry. Biophys. J. 76, 2606–2613 (1999).
Heerklotz, H., Szadkowska, H., Anderson, T. & Seelig, J. The sensitivity of lipid domains to small perturbations demonstrated by the effect of triton. J. Mol. Biol. 329, 793–799 (2003).
Tsamaloukas, A., Szadkowska, H. & Heerklotz, H. Nonideal mixing in multicomponent lipid/detergent systems. J. Phys.: Condens. Matter 18 (2006).
Hagen, V. et al. Coumarinylmethyl esters for ultrafast release of high concentrations of cyclic nucleotides upon one- and two-photon photolysis. Angew. Chem. Int. Ed. Engl. 44, 7887–7891 (2005).
Heerklotz, H. Membrane stress and permeabilization induced by asymmetric incorporation of compounds. Biophys. J. 81, 184–195 (2001).
Keller, S., Heerklotz, H. & Blume, A. Monitoring lipid membrane translocation of sodium dodecyl sulfate by isothermal titration calorimetry. J. Am. Chem. Soc. 128, 1279–1286 (2006).
Montich, G., Scarlata, S., McLaughlin, S., Lehrmann, R. & Seelig, J. Thermodynamic characterization of the association of small basic peptides with membranes containing acidic lipids. Biochim. Biophys. Acta 1146, 17–24 (1993).
Murray, D. et al. Electrostatic properties of membranes containing acidic lipids and adsorbed basic peptides: theory and experiment. Biophys. J. 77, 3176–3188 (1999).
Cambridge, S.B., Geissler, D., Keller, S. & Curten, B. A caged doxycycline analogue for photoactivated gene expression. Angew. Chem. Int. Ed. Engl. 45, 2229–2231 (2006).
Gilbert, D. et al. Caged Capsaicins: new tools for the examination of TRPV1 channels in somatosensory neurons. ChemBioChem 8, 89–97 (2007).
Bárány-Wallje, E. et al. A critical reassessment of penetratin translocation across lipid membranes. Biophys. J. 89, 2513–2521 (2005).
Keller, S., Böthe, M., Bienert, M., Dathe, M. & Blume, A. A simple fluorescence-spectroscopic membrane translocation assay. ChemBioChem DOI: 10.1002/cbic.200600553.
Tsamaloukas, A., Szadkowska, H., Slotte, P.J. & Heerklotz, H. Interactions of cholesterol with lipid membranes and cyclodextrin characterized by calorimetry. Biophys. J. 89, 1109–1119 (2005).
Murphy, K.P. Predicting binding energetics from structure: looking beyond ΔG0 . Med. Res. Rev. 19, 333–339 (1999).
Spolar, R.S., Ha, J.H. & Record, M.T. Jr. Hydrophobic effect in protein folding and other noncovalent processes involving proteins. Proc. Natl. Acad. Sci. USA 86, 8382–8385 (1989).
Kresheck, G.C. & Hargraves, W.A. Thermometric titration studies of the effect of head group, chain length, solvent, and temperature on the thermodynamics of micelle formation. J. Coll. Interf. Sci. 48, 481–493 (1974).
Hope, M.J., Bally, M.B., Webb, G. & Cullis, P.R. Production of large unilamellar vesicles by a rapid extrusion procedure. Characterization of size distribution, trapped volume and ability to maintain a membrane potential. Biochim. Biophys. Acta 812, 55–65 (1985).
Record, M.T. Jr., Lohman, T.M. & De Haseth, P. Ion effects on ligand nucleic acid interactions. J. Mol. Biol. 107, 145–158 (1976).
Mascotti, D.P. & Lohman, T.M. Thermodynamic extent of counterion release upon binding oligolysines to single-stranded nucleic acids. Proc. Natl. Acad. Sci. USA 87, 3142–3146 (1990).
Patel, D.R. et al. Calcium-dependent binding of annexin 12 to phospholipid bilayers: stoichiometry and implications. Biochemistry 40, 7054–7060 (2001).
Chellani, M. Isothermal titration calorimetry: biological applications. Am. Biotechnol. Lab. 17, 14–18 (1999).
MacDonald, R.C. et al. Small-volume extrusion apparatus for preparation of large, unilamellar vesicles. Biochim. Biophys. Acta 1061, 297–303 (1991).
Wieprecht, T., Apostolov, O., Beyermann, M. & Seelig, J. Thermodynamics of the alpha-helix–coil transition of amphipathic peptides in a membrane environment: implications for the peptide-membrane binding equilibrium. J. Mol. Biol. 294, 785–794 (1999).
Tsamaloukas, A., Szadkowska, H. & Heerklotz, H. Thermodynamic comparison of the interactions of cholesterol with unsaturated phospholipid and sphingomyelins. Biophys. J. 90, 4479–4487 (2006).
Keller, M., Kerth, A. & Blume, A. Thermodynamics of interaction of octyl glucoside with phosphatidylcholine vesicles: partitioning and solubilization as studied by high sensitivity titration calorimetry. Biochim. Biophys. Acta 1326, 178–192 (1997).
Heerklotz, H., Binder, H., Lantzsch, G., Klose, G. & Blume, A. Lipid/detergent interaction thermodynamics as a function of molecular shape. J. Phys. Chem. B 101, 639–645 (1997).
Wenk, M.R., Alt, T., Seelig, A. & Seelig, J. Octyl-β-D-glucopyranoside partitioning into lipid bilayers: thermodynamics of binding and structural changes of the bilayer. Biophys. J. 72, 1719–1731 (1997).
Acknowledgements
We dedicate this publication to Professor Joachim Seelig of the Biozentrum of the University of Basel, on the occasion of his 65th birthday. He has played a seminal role in the development of membrane microcalorimetry. We are indebted to him for the friendly uptake into his laboratory, his invaluable advice and support and the generous release into the freedom to pursue our own projects and plans. We thank Natalie Bordag for help with compiling the fitting routine. This work was supported by a fellowship within the Postdoc Program of the German Academic Exchange Service (DAAD) to A.D.T., by grant no. QLK3-CT-2002-01989 of the European Commission to S.K. and by grant no. 31-67216.01 of the Swiss National Science foundation to H.H.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Data
Uptake & Release for Nonionics, © Tsamaloukas, Keller, and Heerklotz (November 2006) (XLS 256 kb)
Rights and permissions
About this article
Cite this article
Tsamaloukas, A., Keller, S. & Heerklotz, H. Uptake and release protocol for assessing membrane binding and permeation by way of isothermal titration calorimetry. Nat Protoc 2, 695–704 (2007). https://doi.org/10.1038/nprot.2007.98
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2007.98
This article is cited by
-
Fluorescent artificial receptor-based membrane assay (FARMA) for spatiotemporally resolved monitoring of biomembrane permeability
Communications Biology (2020)
-
Integration and global analysis of isothermal titration calorimetry data for studying macromolecular interactions
Nature Protocols (2016)
-
Nonlinear least-squares data fitting in Excel spreadsheets
Nature Protocols (2010)
-
Monitoring detergent-mediated solubilization and reconstitution of lipid membranes by isothermal titration calorimetry
Nature Protocols (2009)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.