Abstract
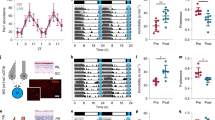
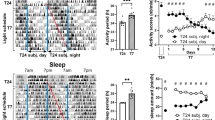
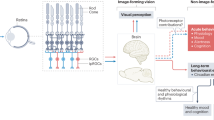
The daily solar cycle allows organisms to synchronize their circadian rhythms and sleep–wake cycles to the correct temporal niche1. Changes in day-length, shift-work, and transmeridian travel lead to mood alterations and cognitive function deficits2. Sleep deprivation and circadian disruption underlie mood and cognitive disorders associated with irregular light schedules2. Whether irregular light schedules directly affect mood and cognitive functions in the context of normal sleep and circadian rhythms remains unclear. Here we show, using an aberrant light cycle that neither changes the amount and architecture of sleep nor causes changes in the circadian timing system, that light directly regulates mood-related behaviours and cognitive functions in mice. Animals exposed to the aberrant light cycle maintain daily corticosterone rhythms, but the overall levels of corticosterone are increased. Despite normal circadian and sleep structures, these animals show increased depression-like behaviours and impaired hippocampal long-term potentiation and learning. Administration of the antidepressant drugs fluoxetine or desipramine restores learning in mice exposed to the aberrant light cycle, suggesting that the mood deficit precedes the learning impairments. To determine the retinal circuits underlying this impairment of mood and learning, we examined the behavioural consequences of this light cycle in animals that lack intrinsically photosensitive retinal ganglion cells. In these animals, the aberrant light cycle does not impair mood and learning, despite the presence of the conventional retinal ganglion cells and the ability of these animals to detect light for image formation. These findings demonstrate the ability of light to influence cognitive and mood functions directly through intrinsically photosensitive retinal ganglion cells.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Reppert, S. M. & Weaver, D. R. Coordination of circadian timing in mammals. Nature 418, 935–941 (2002)
Foster, R. G. & Wulff, K. The rhythm of rest and excess. Nature Rev. Neurosci. 6, 407–414 (2005)
Berson, D. M., Dunn, F. A. & Takao, M. Phototransduction by retinal ganglion cells that set the circadian clock. Science 295, 1070–1073 (2002)
Hattar, S., Liao, H. W., Takao, M., Berson, D. M. & Yau, K. W. Melanopsin-containing retinal ganglion cells: architecture, projections, and intrinsic photosensitivity. Science 295, 1065–1070 (2002)
Güler, A. D. et al. Melanopsin cells are the principal conduits for rod–cone input to non-image-forming vision. Nature 453, 102–105 (2008)
Hattar, S. et al. Central projections of melanopsin-expressing retinal ganglion cells in the mouse. J. Comp. Neurol. 497, 326–349 (2006)
Altimus, C. M. et al. Rods-cones and melanopsin detect light and dark to modulate sleep independent of image formation. Proc. Natl Acad. Sci. USA 105, 19998–20003 (2008)
Lam, R. W. & Levitan, R. D. Pathophysiology of seasonal affective disorder: a review. J. Psychiatry Neurosci. 25, 469–480 (2000)
Nestler, E. J. et al. Neurobiology of depression. Neuron 34, 13–25 (2002)
McEwen, B. S. Protective and damaging effects of stress mediators. N. Engl. J. Med. 338, 171–179 (1998)
Cryan, J. F. & Holmes, A. The ascent of mouse: advances in modelling human depression and anxiety. Nature Rev. Drug Discov. 4, 775–790 (2005)
Baldi, E., Lorenzini, C. A. & Corrado, B. Task solving by procedural strategies in the Morris water maze. Physiol. Behav. 78, 785–793 (2003)
Vorhees, C. V. & Williams, M. T. Morris water maze: procedures for assessing spatial and related forms of learning and memory. Nature Protocols 1, 848–858 (2006)
Bliss, T. V. & Collingridge, G. L. A synaptic model of memory: long-term potentiation in the hippocampus. Nature 361, 31–39 (1993)
McDermott, C. M. et al. Sleep deprivation causes behavioral, synaptic, and membrane excitability alterations in hippocampal neurons. J. Neurosci. 23, 9687–9695 (2003)
Honey, R. C., Watt, A. & Good, M. Hippocampal lesions disrupt an associative mismatch process. J. Neurosci. 18, 2226–2230 (1998)
Dulawa, S. C., Holick, K. A., Gundersen, B. & Hen, R. Effects of chronic fluoxetine in animal models of anxiety and depression. Neuropsychopharmacology 29, 1321–1330 (2004)
Sprouse, J., Braselton, J. & Reynolds, L. Fluoxetine modulates the circadian biological clock via phase advances of suprachiasmatic nucleus neuronal firing. Biol. Psychiatry 60, 896–899 (2006)
Lockley, S. W. et al. Short-wavelength sensitivity for the direct effects of light on alertness, vigilance, and the waking electroencephalogram in humans. Sleep 29, 161–168 (2006)
Vandewalle, G. et al. Spectral quality of light modulates emotional brain responses in humans. Proc. Natl Acad. Sci. USA 107, 19549–19554 (2010)
İyilikci, O., Aydin, E. & Canbeyli, R. Blue but not red light stimulation in the dark has antidepressant effect in behavioral despair. Behav. Brain Res. 203, 65–68 (2009)
Warthen, D. M., Wiltgen, B. J. & Provencio, I. Light enhances learned fear. Proc. Natl Acad. Sci. USA 108, 13788–13793 (2011)
Fonken, L. K. et al. Influence of light at night on murine anxiety- and depressive-like responses. Behav. Brain Res. 205, 349–354 (2009)
Ma, W. P. et al. Exposure to chronic constant light impairs spatial memory and influences long-term depression in rats. Neurosci. Res. 59, 224–230 (2007)
Roybal, K. et al. Mania-like behavior induced by disruption of CLOCK. Proc. Natl Acad. Sci. USA 104, 6406–6411 (2007)
Tataroğlu, O., Aksoy, A., Yilmaz, A. & Canbeyli, R. Effect of lesioning the suprachiasmatic nuclei on behavioral despair in rats. Brain Res. 1001, 118–124 (2004)
Lee, H. K., Min, S. S., Gallagher, M. & Kirkwood, A. NMDA receptor-independent long-term depression correlates with successful aging in rats. Nature Neurosci. 8, 1657–1659 (2005)
Acknowledgements
We would like to thank T. Gould, G. Ball and A. Sawa for their expert advice on the behavioural tests. We would like to thank R. Kuruvilla for her critical reading and advice on this manuscript. We would also like to thank the mouse tri-laboratory for suggestions and advice. This work was supported by the David and Lucile Packard Foundation grant to S.H.
Author information
Authors and Affiliations
Contributions
T.A.L., C.M.A., H.Z., E.T.W. and S.H. designed experiments. T.A.L. and C.M.A. carried out experiments. H.W., H.-K.L., S.Y. and A.K. designed and performed electrophysiological experiments. T.A.L., C.M.A., H.Z., E.T.W. and S.H. wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Information
This file contains Supplementary Text, Supplementary Figures 1-13 and Supplementary References. (PDF 9371 kb)
Rights and permissions
About this article
Cite this article
LeGates, T., Altimus, C., Wang, H. et al. Aberrant light directly impairs mood and learning through melanopsin-expressing neurons. Nature 491, 594–598 (2012). https://doi.org/10.1038/nature11673
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nature11673
This article is cited by
-
The cognitive impact of light: illuminating ipRGC circuit mechanisms
Nature Reviews Neuroscience (2024)
-
Determination of intensity and spread of light to the surrounding in conventional phototherapy and comparison with novel converging photo unit — an observational study
Egyptian Pediatric Association Gazette (2023)
-
Regulation of mouse exploratory behaviour by irradiance and cone-opponent signals
BMC Biology (2023)
-
Reversible suppression of circadian-driven locomotor rhythms in mice using a gradual fragmentation of the day-night cycle
Scientific Reports (2023)
-
Delaying circadian sleep phase under ultradian light cycle causes time-of-day-dependent alteration of cognition and mood
Scientific Reports (2023)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.