Abstract
Hepatocellular carcinoma (HCC) ranks as one of the most common and lethal malignancies worldwide. A better understanding of the mechanism responsible for HCC metastasis will be helpful for the treatment of HCC patients. Thymidine phosphorylase (TP), a key enzyme that catalyzes the conversion of thymidine to thymine and deoxyribose-1-phosphate, was demonstrated to promote the invasion and metastasis of HCC in our study. Clinical retrospective analysis revealed that metastatic HCC tumor tissues have higher TP expression, and TP expression was significantly correlated with matrix metalloproteinase (MMP) 2 and 9 expression. Survival analysis revealed that TP expression was negatively correlated with the prognosis of HCC patients. Moreover, in vitro cell experiments confirmed that TP could promote the migration and invasion of HCC cells. In addition, MMP2 and MMP9 were activated by TP overexpression. Overall, this study suggests that TP promotes metastasis and may serve as a marker of poor prognosis in HCC. Thus, TP is a potential target for the treatment of HCC.
Similar content being viewed by others
Main
Hepatocellular carcinoma (HCC) ranks as one of the top causes of cancer-related deaths worldwide and has a poor prognosis.1, 2 Metastasis of HCC is one of the top reasons for the death of HCC patients.3 Thymidine phosphorylase (TP), an enzyme that catalyzes the reversible phosphorolysis of thymidine and other pyrimidine-2′-deoxyribonucleosides, is more highly expressed in colorectal, pancreatic, breast, lung, and liver cancers than in adjacent non-neoplastic tissues.4, 5, 6, 7 Also, TP plays an important role in tumor angiogenesis, apoptosis, and proliferation.8, 9 Although TP was shown to be associated with the metastasis of some tumors such as gastric and colorectal cancers, whether TP could promote the metastasis of HCC remains unclear; this subject therefore requires further exploration.10, 11
In this study, specimens from 75 cases of clinical HCC were used for clinicopathologic analysis. Results revealed that TP expression was correlated with tumor size, pathologic grading, and metastasis and high TP expression meant a poor prognosis. An in vitro study further confirmed that TP promoted HCC cell invasion and migration. Additionally, TP was able to promote the expression and activity of MMP2 and MMP9. As both TP and MMPs are important components of the metastasis cascade, we hypothesized that TP depends on MMP2 and MMP9 to promote the metastasis of HCC.10, 12
This study reveals the relationship between TP and metastasis, and prognosis of HCC for the first time. Our findings will provide a basis for the treatment of HCC by targeting TP and taking TP as a prognostic marker.
Materials and methods
Patient Selection and Clinical Data
An HCC tissue microarray that contains 75 cases was obtained from US Biomax for IHC. Tissue blocks for creating a tissue microarray were collected within 5 years. Each single tissue spot on every array slide was individually examined by pathologists and certified according to standards of diagnosis, classification, and pathological grade published by the World Health Organization. Each specimen collected from any clinic was consented to by both the hospital and the individual. All tissues were collected under the highest ethical standards with the donor being informed completely and with their consent. All human tissues were collected under HIPAA-approved protocols. In this study, the age of patients ranged from 17 to 79 years. The patients consisted of 14 females and 61 males, and the survival time was between 1 and 116 months.
Cell Culture and Transfection
Human HCC cell lines HepG2 and PLC-PRF-5 were purchased from KeyGen Biotech (Nanjing, China). Cells were separately cultured with DMEM and RPMI-1640 medium (Hyclone) supplemented with 10% fetal bovine serum (Hyclone) and penicillin (50 μg/ml)/streptomycin (50 μg/ml). All cultures were maintained at 37 °C in a humidified atmosphere that contained 5% CO2. When cells reached a density of ~80%, the overexpression and interference plasmids of M02-TYMP and psiU6-TYMP purchased from GeneCopoeia (Guangzhou, China) were transfected into cells according to the instructions of the Roche transfection reagent (Roche, Switzerland).
Western Blot Analysis
Immunoblots were performed with samples that contain total protein (30 μg) through sodium dodecyl sulfate (SDS)-polyacrylamide gel electrophoresis and transferred to polyvinylidene difluoride membranes (Millipore, USA). The membrane was probed with primary antibody of TP (1:1000; Abcam) or MAPK2, extracellular signal-regulated kinase (Erk2), GAPDH purchased from Affinity Bioreagents (Colorado, USA), followed by incubation with a second antibody (1:2000; Santa Cruz Biotechnology). Blots were detected by using an enhanced chemiluminescence detection kit (Millipore). Densitometric analysis was performed using the ImageJ software.
Wound Healing Assay
The transfected cells were seeded into 24-well culture plates at a density of 5 × 105 cells per well. When the cells adhered to the plate, a 200 μl pipette tip was used to scratch the wound and the cells were incubated with serum-free medium. Images of the wounds were taken at the time of 0, 24, and 48 h. Each experiment was repeated in triplicate, and the mean values (mean±s.d.) were presented.
Matrigel Invasion Assay
Cell invasion assay was performed using Transwell cell culture inserts (Corning, USA) and Matrigel (BD, USA). The transfected cells were cultured for 48 h and allowed to invade for another 24 h. The passed cells were fixed in 4% paraformaldehyde, stained with crystal violet solution, and counted with a light microscope (Nikon, Japan).
Gelatin Zymography Assay
The transfected cells were grown in 12-well plates and cultured in serum-free medium at a density of 1 × 106 cells per well for 24 h. The conditioned medium was harvested and subjected to 10% SDS-polyacrylamide gel electrophoresis that contained 0.1% gelatin. The gel was soaked in 2.5% Triton X-100 for 40 min at room temperature and incubated in substrate buffer (50 mM Tris-HCl (pH 7.5), 5 mM CaCl2, 1 μM ZnCl2, and 0.02% NaN3) for 48 h at 37 °C. The gel was stained with Coomassie R250 and destained in water with 10% acetic acid and 30% methanol until the apparent cleared zones appeared associated with MMP activity. The zones were quantified with the ImageJ software.
MMP Activity Assays
The MMP activity assay kit was purchased from Abcam. The collecting medium was incubated with APMA working solution for 10–15 min at room temperature. Then, an equal volume of MMP Green Substrate solution was added to the samples for another 1 h incubation at 37 °C. Fluorescence detection was mediated by the Microplate Reader (Thermo Scientific) at wavelengths of 490 and 525 nm.
Reporter Gene Assays
MMP2 and MMP9 reporter gene plasmids were purchased from GeneCopoeia. After cotransfection, the cells were incubated for another 48 h. Fluorescence detection was performed according to the Dual-Luciferase Assays System (Promega) by using the Luminoskan Ascent reader system (Thermo Scientific).
Animal Studies
Sixteen BALB/c nu/nu mice that were 5–6 weeks old, with an equal ratio of male and female, were divided randomly into four groups. Xenografts of tumors were established by subcutaneous injection of 1 × 107 cells of HepG2 (C/psiU6-TYMP) or PLC-PRF-5 (C/M02-TYMP) in different groups into the flank. The mice were maintained in a specific pathogen-free animal care facility according to institutional guidelines. On the 10th day after tumor inoculation, body weights and tumor diameters were measured every 2 days, and tumor volumes were calculated according to the formula V=ab2/2 (a=length, b=width). Four weeks after treatment, all mice were killed, and xenografts and lungs were collected. Lung tissues were harvested for histologic examination to measure the metastasis, and the xenografts were further analyzed by IHC.
Immunohistochemical Analysis
Tumor tissues from mice were fixed in 4% paraformaldehyde, embedded in paraffin, cut into 4 μm thick slices, and placed on slides. IHC staining was used to verify the expression of different proteins in the HCC tissue microarray and the slides. In the progress, tissues were deparaffinized in xylene prior and then rehydrated by gradient alcohol. Then, 3% hydrogen peroxide in methanol was used to block the endogenous peroxidase activity for 30 min. Then the chips were soaked in 0.01 M citric acid buffer for 10 min at 95°C in a microwave oven. After blocking with 10% normal goat serum for 20 min, the chips were incubated with primary antibodies for 30 min at room temperature and then 4° C overnight. Antibody dilutions were as follows: mouse monoclonal anti-TP (Abcam, dilution 1:100); rabbit polyclonal anti-MMP2 (Abcam, 1:200), and rabbit polyclonal anti-MMP9 (Abcam, 1:100). The next morning, chips were washed with phosphate buffered solution three times and incubated with horseradish peroxidase-conjugated goat anti-rabbit/mouse IgG for 1 h at room temperature. 3, 3′-diaminobenzidine was used for staining. Chips were then counterstained with haematoxylin, dehydrated, and mounted.
Scoring and Statistical Analysis
Immunohistochemical stains were scored based on positive degree and positive rate. The degree was divided into four classes: none for 0, weak brown for 1, moderate brown for 2, and strong brown for 3. The positive rate includes four classes: 0–25% for 1, 25–50% for 2, 50–75% for 3, and 75–100% for 4. Multiplication of the degree and rate scores was utilized as the staining index to judge the results. A staining index >6 was the value of a high-expression immunohistochemical assay.
We tested the relation between TP, MMP2, and MMP9 expression, and clinic pathological parameters using Pearson χ2-test. Overall survival analyses were conducted using the Kaplan–Meier method with the log-rank test. P-values <0.05 were deemed statistically significant. All statistical analyses were performed using IBM SPSS Statistics 22.0 software (Chicago, IL, USA).
Proteomics
The control and M02-TYMP transfected PLC-PRF-5 cell lines were incubated for 48 h. Proteins were digested with trypsin for 20 h at 37 °C. The sample was evaporated and resuspended in water/0.1% formic acid for LC-MS/MS analysis. The database search was performed using Trans-Proteomic Pipeline software, and the results were imported into Microsoft Excel for further DAVID analysis.
Results
Characteristics of Patients and TP Expression in HCC
As shown in Table 1, characteristics of the HCC in the microarray were presented. A total of 75 patients consisted of 14 (18.7%) men and 61 (81.3%) women with ages from 17 to 79 years (median, 50 years). Forty-one (54.7%) patients were diagnosed with lower than stage III HCC, and 34 (45.3%) patients were diagnosed with higher than stage III HCC. Fifty-four (72.0%) patients had lower than grade II HCC, and 21 (28.0%) patients had higher than grade II HCC. Eleven (14.7%) patients had lymph and/or distant metastasis, and 64 (85.3%) patients had no metastasis. In the 75 patients, 16 (21.3%) patients had a family history of the disease, 38 (50.7%) patients had a history of wine consumption, 33 (44.0%) patients had a history of smoking, 28 (37.3%) patients had liver cirrhosis, 40 (53.3%) patients had an AFP higher than 200 μg/l, and 50 (66.7%) patients had an ALT higher than 40 U/l.
By IHC staining of TP, 29 (38.7%) patients were diagnosed as low expression, while 46 (61.3%) patients had high expression.
Association of TP Expression with Tumor Characteristics
To evaluate the clinical significance of TP in HCC, the TP expression in HCC tissues of the microarray was compared on the basis of 12 characteristics, including gender, age, and tumor size (Table 1). TP expression was significantly increased in HCC and cirrhotic liver tissues than in normal liver tissues (Figure 1a). TP was highly expressed in primary tumors with lymph and/or distant metastasis, and it had a significant correlation with metastasis (P=0.002) (Table 1; Figure 1b). Similarly, significant associations among the TP expression and tumor size (P<0.001) and stage (P<0.001) were observed (Table 1; Figure 1b). However, high TP expression had no significant relationship with gender, age, grade, family history, wine consumption, smoking, cirrhosis, and AFP and ALT contents (all P>0.05, Table 1).
Correlation of TP expression with HCC tumor characteristics in clinical samples. (a) TP staining was negative, weakly positive, and strongly positive in normal, cirrhosis, and tumor tissues, respectively. (b) The first column represents staining of negative expression of TP in liver cancers of stage (<III), grade (≤II), and metastasis (−), while the second column indicates that tumors with stage (≥III), grade (>II), and metastasis highly express TP. Images were taken at × 400 magnification. (*P<0.05, **P<0.01). HCC, hepatocellular carcinoma; TP, thymidine phosphorylase.
Association between Survival Status and TP Expression
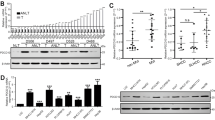
To investigate the correlation between TP expression and survival time, overall survival analysis and Cox proportional hazards analysis were used. The median survival time was 45 months (ranging from 1 to 116 months). Overall survival analysis revealed that patients who suffered from high TP expression had a significantly lower survival time compared with those with a low TP expression (P=0.013, Figure 2). The Cox proportional hazards analysis revealed that the TP expression, tumor size, clinical stage, and metastasis were significantly correlated with survival time in HCC patients with relative risk coefficients of 2.557, 2.332, 1.850, and 3.078, respectively (Table 2).
Overall survival analysis for TP expression. Kaplan–Meier curves for overall survival in 28 patients with high TP expression and 47 patients with low TP expression. Survival rate sharply decreased in patients with high TP expression in the first 4 years when significantly compared with those expressing low TP (P=0.013). TP, thymidine phosphorylase.
Promotion of Migration and Invasion of HCC Cells In vitro by TP
In this study, we selected PLC-PRF-5 cell lines that expressed low levels of TP and HepG2 cell lines with high TP expression as the pairing model to study the relationship between TP expression and HCC migration and invasion. The TP expression upregulated through transfection with the plasmid of M02-TYMP. TP was downregulated through transfection with the plasmid of psiU6-TYMP (Figure 3a). Wound healing assay was performed to verify the relation of TP with cell migration, and results showed that after 24 and 48 h treatment, the scratch wounds were thinner in the high TP expression groups (Figure 3b). In the Matrigel invasion assay, compared with the control groups after incubation of 24 and 48 h, the number of the cells that passed through the chamber significantly increased when TP was overexpressed in PLC-PRF-5 cells and significantly decreased when TP was knocked down in HepG2 cell lines (Figure 3c). PLC-PRF-5 cell lines were transfected with psiU6-TYMP and the HepG2 cell lines were transfected with M02-TYMP as the control group. The results showed that there were no significant differences in TP expression, migration, and invasion compared with the empty vector groups (Supplementary Figure S1).
Effects of TP expression on cell migration and invasion. (a) PLC-PRF-5 and HepG2 cells were overexpressed or knocked down TP with plasmids of M02-TYMP or psiU6-TYMP, separately. After 48 h of transfection, the proteins were examined by western blot. (b) Following the wound healing assay, quantitative analysis showed a significant difference in the speed of wound healing among the TP overexpression or knockingdown groups compared with the negative control (n=3). (c) Cell invasion assay was performed using Transwell filters; when TP was overexpressed, the passed cells increased nearly three-fold compared with the control group in PLC-PRF-5; in the interference group of HepG2, knockingdown of TP significantly reduced the invasion (n=3). Images were taken at × 200 magnification. Results show the means of the three experiments, and the error bars represent s.d. (**P<0.01). TP, thymidine phosphorylase.
Promotion of the Expression and Activity of MMP2 and MMP9 by TP
During cancer invasion and metastasis, the extracellular matrix and basement membrane are degraded by proteolytic enzymes. When TP was overexpressed in KB cells, the expression and activity of MMP9 increased.13 Thus, immunohistochemical analysis was performed to assess the TP, MMP2, and MMP9 expression. Results showed that the MMP2 and MMP9 expression were higher in cancers with high TP expression compared with those with low TP expression, and their expression was significantly correlated with Pearson coefficients of 0.006 and 0.001, respectively (Figure 4a). Gelatin zymography assay showed that when TP was overexpressed, the cancer cells obviously secreted MMP2 and MMP9 at a higher level than control, and with the knockdown of TP, the secreted level of MMP2 and MMP9 decreased (Figure 4b). MMP2 and MMP9 activity were significantly increased when TP was overexpressed in PLC-PRF-5 cell lines and decreased after TP was knocked down in cell lines of HepG2 (Figure 4c). In TP overexpressed cells, the luciferase reporter plasmids of MMP2 and MMP9 were transfected, and TP promoted the transcription of MMP2 and MMP9. Correspondingly, in the interference group, the transcription level of MMP2 and MMP9 decreased (Figure 4d). Taken together, these results indicated that TP is a positive regulator of MMP2 and MMP9.
TP promoted the expression and activity of MMP2 and MMP9. (a) Clinical samples with high TP expression showed strong positive staining of MMP2 (P=0.006) and MMP9 (P=0.001). (b and c) The activities of MMP2 and MMP9 were detected by gelatin zymography assay and activity assay. Both MMP2 and MMP9 were significantly higher in the TP overexpression group in the PLC-PRF-5 cell line compared with the control group. Similarly, in the interference group, the activity of MMP2 and MMP9 was significantly decreased when TP was knocked down. (d) Reporter gene assay results for PLC-PRF-5 and HepG2 cells suggested that TP promoted the transcriptional activity of MMP2 and MMP9. Images were taken at × 200 magnification. Results show the means of the three experiments, and the error bars represent s.d. (*P<0.05, **P<0.01). MMP, matrix metalloproteinase; TP, thymidine phosphorylase.
TP Affects the Tumor Growth in Mouse Xenograft Models
The mice xenograft model was used to measure the relationship between TP and metastasis in vivo. The model revealed that high TP expression promoted tumor growth and significantly increased the tumor formation of lung metastasis from 1.67±0.57 to 12.33±1.52. A higher mortality rate was observed in the TP overexpression group. Consistent with this result, in the HepG2 xenografts, when TP was knocked down, tumor growth was inhibited and lung metastasis was also decreased from 8.67±1.53 to 3.33±0.58 (Figures 5a and b). Further IHC analysis of the xenografts showed that TP significantly promoted the expression of MMP2 and MMP9 in vivo (Figure 5c).
Effects of TP in HCC metastasis in vivo. (a) Overexpression of TP promoted PLC-PRF-5 xenograft growth, and knockingdown of TP inhibited HepG2 xenografts growth. (b) The numbers of tumor formation of lung metastasis in different groups, images were taken at × 40 magnification. (c) TP, MMP2, and MMP9 expression in xenografts; images were taken at × 400 magnification. Results show the means of the three experiments, and the error bars represent s.d. (*P<0.05, **P<0.01). HCC, hepatocellular carcinoma; MMP, matrix metalloproteinase; TP, thymidine phosphorylase.
TP may Promote HCC Progression through MAPK-MMP Pathway
To elucidate the molecular mechanism of TP’s promotion of HCC progression, proteomic analysis was used to compare the differential protein expression when TP was overexpressed. The proteomic analysis revealed that TP overexpression influenced the expression of a series of proteins related to invasion and migration (Figure 6a). The pathway analysis showed that the Ras-MAPK-MMPs signaling pathway was activated by upregulation of the protein of MAPK2 and Erk2. Western blot analysis revealed that the expression of MAPK2 and Erk2 was significantly upregulated with the overexpression of TP (Figure 6b). These results may explain the mechanism of tumor metastasis and invasion mediated by TP (Figure 6c).
Proteomics analysis results. (a) Multidimensional liquid chromatography–tandem mass spectrometry was performed to analyze the differentially expressed proteins of PLC-PRF-5 cells. Among these proteins, 8 proteins were upregulated and 4 were downregulated, which were involved in invasion, and 13 proteins were upregulated and 4 were downregulated, which were involved in migration. (b) MAPK2 and Erk2 were upregulated when PLC-PRF-5 cells were transfected with M02-TYMP by western blot. (c) TP-Ras-MAPK-MMPs signaling pathway. Results show the means of the three experiments, and the error bars represent s.d. (**P<0.01). Erk, extracellular signal-regulated kinase; MMP, matrix metalloproteinase.
Discussion
HCC ranks as the second most common cause of cancer-related mortality with 1.6 million global deaths annually.14 HCC patients are usually diagnosed at advanced stages, and metastasis is a major reason for the high mortality and poor prognosis for HCC patients.15 The mechanism of HCC metastasis remains unclear and requires further explanation. TP, also known as platelet-derived endothelial cell growth factor, is an enzyme involved in pyrimidine nucleoside metabolism, and it catalyzes the reaction of thymidine to thymine and deoxyribose-1-phosphate.5 TP is upregulated in a wide variety of solid tumors and promotes tumor progression by preventing apoptosis.16, 17, 18 Elevated TP levels are associated with tumor aggressiveness and poor prognosis. The presence and overexpression of TP in HCC have been reported. However, whether TP can promote the metastasis of HCC deserves further discussion.7
In this study, our results show that TP was correlated with HCC progression and significantly promoted HCC cell migration and invasion, and that high TP expression was positively correlated with poor prognosis of HCC patients. These results indicate that TP may be used as a marker of HCC. Further results showed that TP regulated HCC metastasis through upregulating the activity of MMPs. MMP2 and MMP9 usually have an upregulated expression in different metastatic tumors; the MMPs could degrade the extracellular matrix, thereby making it easy for the tumor cells to transfer.19, 20, 21 However, TP is not a transcription factor, and the MMPs cannot be directly regulated; how TP influences MMPs needs further explanation.
Erk, a member of the MAPK family, may stimulate cell proliferation and restrain cell apoptosis by activating the Ras-Raf-Mek-Erk signal cascade pathway.22 Erk also plays an important role in the metastasis of different tumors including HCC.23, 24 Phosphorylation of the Erk1/2 signaling pathway could affect the expression of some transcriptional coactivators, which could then regulate the gene transcription of MMPs.25, 26, 27 Our current study does not examine whether TP directly induces the chemokines production or corresponding receptor expression. It is possible that increased invasion and migration might be due to increased generation of chemokines from tumor cells. In this study, proteomic analysis showed that with the overexpression of TP, a series of proteins related to invasion and migration was affected; the expression of the proteins of MAPK2 and Erk2 in the Ras/Raf pathway in particular increased significantly. These results indicate that TP is likely to affect the expression and activity of MMPs by regulating the MAPK pathway, which promotes the metastasis of HCC. However, how TP affects the MAPK pathway is unclear and needs further exploration.
In summary, we provided quantitative data to clarify the important role of TP in the metastasis of HCC. We demonstrated that TP was correlated with MMP2 and MMP9 expression, and that TP mediated the migration of tumor cells through the activity of MMPs in HCC cell lines. TP affected the expression of MMPs through Erk2 in the Ras/Raf pathway. This study provides further understanding of the molecular mechanisms of metastasis in HCC by TP. Moreover, TP may be a potential marker of poor prognosis in HCC.
References
Torre LA, Bray F, Siegel RL et al. Global cancer statistics, 2012. CA Cancer J Clin 2015;65:87–108.
Ferlay J, Soerjomataram I, Dikshit R et al. Cancer incidence and mortality worldwide: sources, methods and major patterns in GLOBOCAN 2012. Int J Cancer 2015;136:E359–E386.
Giannelli G, Koudelkova P, Dituri F et al. Role of epithelial to mesenchymal transition in hepatocellular carcinoma. J Hepatol 2016;65:798–808.
Takebayashi Y, Yamada K, Miyadera K et al. The activity and expression of thymidine phosphorylase in human solid tumours. Eur J Cancer 1996;32 A:1227–1232.
Iltzsch MH, el Kouni MH, Cha S . Kinetic studies of thymidine phosphorylase from mouse liver. Biochemistry 1985;24:6799–6807.
Terranova-Barberio M, Roca MS, Zotti AI et al. Valproic acid potentiates the anticancer activity of capecitabine in vitro and in vivo in breast cancer models via induction of thymidine phosphorylase expression. Oncotarget 2015;7:7715–7731.
Yamamoto A, Dhar DK, El-Assal ON et al. Thymidine phosphorylase (platelet-derived endothelial cell growth factor), microvessel density and clinical outcome in hepatocellular carcinoma. J Hepatol 1998;29:290–299.
Miyatani T, Kurita N, Utsunomiya T et al. Platelet-derived endothelial cell growth factor/thymidine phosphorylase inhibitor augments radiotherapeutic efficacy in experimental colorectal cancer. Cancer Lett 2012;318:199–205.
Schwartz PA, Vetticatt MJ, Schramm VL . Transition state analysis of thymidine hydrolysis by human thymidine phosphorylase. J Am Chem Soc 2010;132:13425–13433.
Yu EJ, Lee Y, Rha SY et al. Angiogenic factor thymidine phosphorylase increases cancer cell invasion activity in patients with gastric adenocarcinoma. Mol Cancer Res 2008;6:1554–1566.
Takebayashi Y, Akiyama S, Akiba S et al. Clinicopathologic and prognostic significance of an angiogenic factor, thymidine phosphorylase, in human colorectal carcinoma. J Natl Cancer Inst 1996;88:1110–1117.
Giannelli G, Falk-Marzillier J, Schiraldi O et al. Induction of cell migration by matrix metalloprotease-2 cleavage of laminin-5. Science 1997;277:225–228.
Nakajima Y, Haraguchi M, Furukawa T et al. 2-Deoxy-L-ribose inhibits the invasion of thymidine phosphorylase-overexpressing tumors by suppressing matrix metalloproteinase-9. Int J Cancer 2006;119:1710–1716.
Loomis D, Huang W, Chen G . The International Agency for Research on Cancer (IARC) evaluation of the carcinogenicity of outdoor air pollution: focus on China. Chin J Cancer 2014;33:189–196.
Stravitz RT, Heuman DM, Chand N et al. Surveillance for hepatocellular carcinoma in patients with cirrhosis improves outcome. Am J Med 2008;121:119–126.
Pula G, Mayr U, Evans C et al. Proteomics identifies thymidine phosphorylase as a key regulator of the angiogenic potential of colony-forming units and endothelial progenitor cell cultures. Circ Res 2009;104:32–40.
Bronckaers A, Gago F, Balzarini J et al. The dual role of thymidine phosphorylase in cancer development and chemotherapy. Med Res Rev 2009;29:903–953.
Vaupel P, Schlenger K, Knoop C et al. Oxygenation of human tumors: evaluation of tissue oxygen distribution in breast cancers by computerized O2 tension measurements. Cancer Res 1991;51:3316–3322.
Jia YL, Shi L, Zhou JN et al. Epimorphin promotes human hepatocellular carcinoma invasion and metastasis through activation of focal adhesion kinase/extracellular signal-regulated kinase/matrix metalloproteinase-9 axis. Hepatology 2011;54:1808–1818.
Jeon H, Kim JH, Lee E et al. Methionine deprivation suppresses triple-negative breast cancer metastasis in vitro and in vivo. Oncotarget 2016;7:67223–67234.
Wells JM, Gaggar A, Blalock JE . MMP generated matrikines. Matrix Biol 2015;44-46:122–129.
Busca R, Pouyssegur J, Lenormand P . ERK1 and ERK2 map kinases: specific roles or functional redundancy? Front Cell Dev Biol 2016;4:53.
Wang Z, Qu L, Deng B et al. STYK1 promotes epithelial-mesenchymal transition and tumor metastasis in human hepatocellular carcinoma through MEK/ERK and PI3K/AKT signaling. Sci Rep 2016;6:33205.
Miller MA, Oudin MJ, Sullivan RJ et al. Reduced proteolytic shedding of receptor tyrosine kinases is a post-translational mechanism of kinase inhibitor resistance. Cancer Discov 2016;6:382–399.
Yeh CM, Lin CW, Yang JS et al. Melatonin inhibits TPA-induced oral cancer cell migration by suppressing matrix metalloproteinase-9 activation through the histone acetylation. Oncotarget 2016;7:21952–21967.
Adhikary S, Kocieda VP, Yen JH et al. Signaling through cannabinoid receptor 2 suppresses murine dendritic cell migration by inhibiting matrix metalloproteinase 9 expression. Blood 2012;120:3741–3749.
Taira N, Mimoto R, Kurata M et al. DYRK2 priming phosphorylation of c-Jun and c-Myc modulates cell cycle progression in human cancer cells. J Clin Invest 2012;122:859–872.
Acknowledgements
This study was supported by the National Natural Science Funds of China (grant nos. 81572838, 81201650, and 81402973); the National Biomedical Special Project of International Innovation Park (grant nos. 11ZCKFSY06900 and 13ZCZDSY03300); the Tianjin Natural Science and Technology Fund (grant no. 15JCYBJC26400); the Foundation for the Author of National Excellent Doctoral Dissertation of China (grant no. 201482); and the Natural Science Foundation of Tianjin (grant no. 16JCQNJC13300).
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Laboratory Investigation website
This study clarifies the important role of thymidine phosphorylase (TP) in metastasis of hepatocellular carcinoma (HCC). High expression of TP is a predictor of high metastasis potential and poor prognosis in HCC patients. The mechanism relies on increasing expression and activity of MMP2 and MMP9, which is regulated by the MAPK pathway under the influence of TP.
Supplementary information
Rights and permissions
About this article
Cite this article
Zhang, Q., Zhang, Y., Hu, X. et al. Thymidine phosphorylase promotes metastasis and serves as a marker of poor prognosis in hepatocellular carcinoma. Lab Invest 97, 903–912 (2017). https://doi.org/10.1038/labinvest.2017.51
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2017.51