Abstract
Hypoxia-induced pulmonary hypertension (PH), which is characterized by vasoconstriction and subsequent structural remodeling of blood vessels, is an important event in chronic obstructive pulmonary disease patients and in people living at high altitudes. Hypoxia-inducible factor-1α (HIF-1α) and its regulator four-and-a-half LIM (Lin-11, Isl-1 and Mec-3) domain 1 (Fhl-1) have important roles in hypoxia-induced PH. MicroRNA-206 (miR-206) is critical for myogenesis and related diseases; however, the role of miR-206 in hypoxia-induced PH is unknown. miR-206 expression was evaluated in a hypoxic rat model and in cultured hypoxic pulmonary artery smooth muscle cells (PASMCs) using real-time quantitative PCR (RT-qPCR). HIF-1α and Fhl-1 expression were evaluated using RT-qPCR, western blotting, immunohistochemistry and immunofluorescence. The function of miR-206 was assessed by transfecting miR-206 mimics and inhibitors. Dual-luciferase reporter gene assays and western blotting were performed to validate the target genes of miR-206. siRNA targeted against Fhl-1 was used to investigate the effect of Fhl-1 on miR-206. Flow cytometry was used to detect the cell cycle phase distribution in each group of PASMCs. Significant downregulation of miR-206 in hypoxic lung tissue and PASMCs was identified, whereas HIF-1α and Fhl-1 were upregulated in these samples. The expression of miR-206 in the serum was different from that in the lung tissue. Transfection of pre-miR miR-206 in hypoxic conditions led to increased expression of HIF-1α and Fhl-1 rather than abolishing hypoxia-induced HIF-1α and Fhl-1, as was expected, and promoted the entry of cells into the S phase and enhanced PASMC proliferation. Fhl-1-targeted siRNA in PASMC prevented cell proliferation and led to an increased proportion of cells in the G1 phase without altering miR-206 expression. Bioinformatic analysis and dual-luciferase reporter gene assays revealed direct evidence for miR-206 targeting of HIF-1α. In conclusion, hypoxia-induced downregulation of miR-206 promotes PH by targeting the HIF-1α/Fhl-1 pathway in PASMCs. miR-206 could be a triggering factor of early stage of hypoxia-induced PH.
Similar content being viewed by others
Main
Hypoxia-induced pulmonary hypertension (PH) is a serious disorder characterized by pulmonary vasoconstriction and enhanced proliferation of pulmonary artery smooth muscle cells (PASMCs) that lead to structural remodeling of blood vessel walls. PH is a common clinical manifestation of chronic obstructive pulmonary disease (COPD) and in people who live at high altitude.1 Hypoxia-inducible factor-1α (HIF-1α) has a key role in the pathophysiology of hypoxia-induced PH.
The four-and-a-half LIM domain 1 (Fhl-1) protein, a member of the Fhl family, has also been implicated in the biological pathways of muscle growth and differentiation in PH2, 3, 4 and is regulated by HIF-1α in a feedback loop that serves to limit HIF-1α activity under conditions of prolonged hypoxia.5
MicroRNAs (miRs) are a class of ∼23-nucleotide, noncoding single-stranded RNAs that regulate gene function by identifying the 3′-untranslated regions (3′-UTRs) of target genes, lead to the negative regulation of relevant mRNAs for translation and sometimes repress the expression of the mRNAs themselves.6, 7 miRs have a significant impact on a wide spectrum of cellular biology and pathophysiologic regulatory pathways.8, 9, 10 miRs are also present as cell-free miRs in various biological fluids including serum and urine. Unlike mRNA, cell-free miRs are remarkably stable and resistant to RNase effects,11, 12 which makes peripheral miRs potentially valuable sources of noninvasive biomarkers of various diseases.13, 14, 15, 16, 17 Although the complex biological functions of miRs are not well understood, some miRs are tissue- or cell-specific.18
The miR-1 family, which includes miR-1, -133 and -206, among others, has been proven to be a muscle-specific miR family; they have a critical role in myogenesis and growth.19, 20, 21, 22 The role of the miR-1 family in hypoxia-induced PH is unknown.
Because bioinformatic analysis confirmed that miR-206 carries the 5′-GGAAUG-3′ ‘seed’ region, which is the putative binding site of the 3′-UTR region of HIF-1α, and the aforementioned existence of a feedback loop between Fhl-1 and HIF-1α, we hypothesized that miR-206 contributes to hypoxia-induced PH by targeting HIF-1α and its regulator Fhl-1.
In this study, we focus on the expression of miR-206 in lung tissue and serum in a hypoxia-induced Sprague–Dawley (SD) rat model of PH and in cultured PASMCs, and the possible regulatory mechanisms of miR-206 in the process of hypoxia-induced PH.
MATERIALS AND METHODS
Rat Model of Hypoxia-Induced PH
Adult male specific-pathogen-free SD rats (150–180 g) (acquired from the Center for Animal Experiment of Wuhan University, which is awarded with full accreditation by the Association for Assessment and Accreditation of Laboratory Animal Care International, AAALAC Intl.) were exposed to normobaric hypoxia (FIO2 of 0.10) in an isobaric hypoxic chamber23 for 8 h per day for a period of 1, 4, 7, 14, 21 or 30 days (n=5 each). An oxygen analyzer (XP-3180; New Cosmos Electric, Osaka, Japan) was used to monitor oxygen content in the chamber. Normobaric normoxia (FIO2 of 0.21) control animals (n=5) were maintained in a normal oxygen environment in the same (12:12) light–dark cycle. All experimental protocols were performed in accordance with the Guide for the Care and Use of Laboratory Animals published by the US National Institutes of Health (NIH Publication No. 85-23, revised 1996) and were approved by the Animal Use Subcommittee at the Huazhong University of Science and Technology, People’s Republic of China (Approval ID: 00006384).
Tissue Processing
At the end of each time point of hypoxia exposure, rats were administered an analgesic (a subcutaneous injection of carprofen; 4 mg/kg; Sigma-Aldrich, Shanghai, China) and an anesthetic (an intraperitoneal injection of 2% sodium pentobarbital; 40 mg/kg; Sigma-Aldrich). After collecting adequate amounts of peripheral blood, animals were euthanized humanely. The right lower lobe of the lung was removed and divided into two equal parts. Part one was fixed in 10% neutral-buffered formaldehyde for 12 h. Paraffin-embedded sections of 4 μm in thickness were cut for hematoxylin and eosin and immunohistochemical staining. The other part was snap-frozen in liquid nitrogen and stored at −80 °C for further assays.
Serum Collection and Storage
Peripheral blood samples for miR detection were collected and then coagulated at 37 °C for 0.5 h. Following centrifugation at 3000 g for 15 min at 4 °C, aliquots of the supernatant were transferred to RNase-free tubes and stored at −80 °C.
Preparation of Primary PASMCs
After euthanasia, under a stereomicroscope (Phoenix Optical, Jiangxi, China) primary PASMCs were isolated from the pulmonary arteries of the rat lung lobes and were cultured in M199 (Gibco, Carlsbad, CA, USA) medium supplemented with 10% fetal bovine serum (Gibco) as previously described with a minor modification.24 The second- to third-passage (n=5 each) PASMCs were used.
Groups In Vitro and Transfection
The in vitro transfection experimental groups are summarized in Table 1. For the third through the seventh groups, the miR and plasmid were transfected in parallel with corresponding negative controls at the same final concentration and transfection time. Pre-miR-206, miRNA precursor and its negative control were from Applied Biosystems (Foster City, CA, USA), the miR-206 inhibitor and its negative control were from Ribobio (Guangzhou, China) and the si-Fhl-1 plasmid and GFP-conjugated blank plasmid were from Genechem (Shanghai, China). Three RNAi vectors targeting different sequences of the rat Fhl-1 gene (Table 2) were used. The transient transfection was performed using FuGENE HD transfection reagent (Roche, Shanghai, China) in six-well plates. The efficiency of transfection was confirmed using RT-qPCR.
Hypoxia Treatment of PASMCs
Hypoxia was induced using a chamber equilibrated with a water-saturated gas mixture of 1% O2, 5% CO2 and 94% N2 at 37 °C for 24 h.
Measurement of miRs and mRNAs in Lung Tissue and PASMCs Using RT-qPCR
Total RNA was extracted using TRIzol reagent (Invitrogen, Carlsbad, CA, USA). The quality and quantity of RNA were evaluated, the standard negative controls were established and the reference genes for normalization were chosen based on stringent appropriate criteria.25, 26, 27 U87 snRNA and glyceraldehyde-3-phosphate dehydrogenase (GAPDH) levels were used for normalization of miRNAs and mRNAs, respectively. RT-qPCR assay kits, Taqman primers, miR probes and the control miR U87 were from Applied Biosystems. mRNA expression was quantified by RT-qPCR using the ABI Prism 7900 HT Fast Real-time PCR System (Applied Biosystems). For relative quantification of Fhl-1 and HIF-1α mRNA, total RNA was subjected to cDNA synthesis using the First Strand cDNA Synthesis Kit (Fermentas, Burlington, ON, Canada) according to the manufacturer’s protocol. The qPCR reactions were performed using SYBR-Green I (2 × SYBR Green PCR Master Mix from Applied Biosystems) as the fluorogenic reagent. Primer sequences (5′–3′) and information on the products are summarized in Table 3. Each gene was measured in triplicate in at least three independent experiments. Calculation of relative miR and mRNA expression levels was performed using the ΔΔCT method.
Measurement of miR Levels in Serum Using RT-qPCR
Total RNA was extracted and cDNA was generated by reverse transcription using the miRVana™ PARIS™ Kit (Ambion, Austin, TX, USA) and the TaqMan MiR Reverse Transcription Kit (Applied Biosystems), respectively. The subsequent experimental procedure was the same as described above for the lung tissue, with the exception that the cycle number was modified to 50 for the RT-qPCR assay. Each miR sample was run in triplicate.
Identical conditions were used to detect target miR in each sample because an ideal endogenous control has not been established for the serum miR assay. Experimental data were treated as 50-Ct for the subsequent statistical analysis, as described previously.28
Protein Preparation and Western Blotting
Protease inhibitors (Roche, Shanghai, China) were used during the isolation process. Protein from part of the right lung and from PASMCs (n=5 each group) was isolated using the proteoJET Mammalian Cell Lysis Reagent (Fermentas). Protein concentration was determined using a bicinchoninic acid protein assay kit (Pierce, Rockford, IL, USA). Cell lysates (n=5 per group) were run on a 10% SDS polyacrylamide gel, followed by electrotransfer to 0.45-μm polyvinylidene difluoride membranes (Millipore Corporation, Bedford, MA, USA). The membranes were blocked using 5% nonfat dry milk in Tris-buffered saline containing 0.1% Tween-20 for 1 h, and then were incubated overnight at 4 °C with one of the following primary antibodies: anti-Fhl-1 (Abcam, Cambridge, UK) diluted to 1:500; anti-HIF-1α (Abcam) diluted to 1:400; and anti-GAPDH (Cell Signaling Technology, Beverly, MA, USA) diluted to 1:1000. On the following day, after incubating for 1 h at 37 °C with horseradish-peroxidase-conjugated secondary antibodies (all at 1:2000; Pierce), protein signals were visualized using the ECL detection system (Pierce) and were analyzed using the Quantity One 1-D analysis software, version 4.6.9 (Bio-Rad, Hercules, CA, USA).
Immunohistochemistry
The paraffin-embedded sections were incubated with anti-rat Fhl-1 and α-SMA (both from Abcam) primary antibodies at the same dilution of 1:500. After overnight incubation at 4 °C, slides were washed three times using PBS (pH 7.4–7.6) and then incubated with Real-envision Detection System (Dako, Shanghai, China). Negative controls were performed by omitting the primary antibody. The nuclei were counterstained using hematoxylin.
Indirect Immunofluorescence Staining
PASMCs grown on glass slides were fixed using cold acetone for 10 min, rinsed three times using PBS and permeabilized using 0.5% Triton X-100 solution in PBS for 10 min at 37 °C. After blocking with 5% BSA for 30 min, the cells were incubated with anti-Fhl-1 (1:500), anti-HIF-1α (1:1000) and anti-Ki-67 (1:100; all from Abcam) for 2 h at 37 °C. After three washes with PBS, the cells were incubated in corresponding TRITC-labeled secondary antibodies (Pierce) at 1:50 dilutions for 1 h at 37 °C. Cells were counterstained using DAPI (Pierce) to identify nuclear DNA. For controls, duplicate samples were processed without primary antibodies. Fluorescence was imaged using an Olympus BX-51 microscope (Olympus, Tokyo, Japan).
Flow Cytometry Analysis
The cells were harvested, and single-cell suspensions were generated, which were fixed for 30 min using ice-cold 70% ethanol. The supernatant was discarded after centrifugation (300 g, 5 min), and the cell pellet was washed again using PBS and was then resuspended in 400 μl PBS. PI (final concentration 50 μg/ml) and RNase (final concentration 100 μg/ml) were added to the cell suspensions. The samples were analyzed by flow cytometry (BD, Franklin Lakes, NJ, USA) after incubation for 30 min at 37 °C.
miR Response Elements
The 3′-UTR of the HIF-1α gene was screened for the presence of miR response elements (MREs). The HIF-1α sequence was obtained from the website http://www.ncbi.nlm.nih.gov/mapview/, and the seed region chosen for the MREs was 5′-CAUUCC-3′.
Dual-Luciferase Assays
Dual-luciferase reporter experiments were performed using the PASMCs. Wild-type or mutant 3′-UTRs of HIF-1α (Table 4) were subcloned using standard procedures into the pmiR-RB-REPORT™ promoter vector (Ribobio) downstream of the luciferase gene. PASMCs were plated into 96-well plates at 70% confluence at 24 h before transfection. Using Lipofectamine 2000 (Invitrogen), cells were transfected with 100 ng/ml of either the wild-type or mutant 3′-UTR vector and 50 nM of either the miR-206 mimic or miR-206 negative control (Ribobio Co). At 48 h after transfection, luciferase activity was measured in cell lysates using a dual-luciferase reporter kit (Promega, Madison, WI, USA).
Statistical Analysis
Before statistical analyses, data obtained from technical replicates were averaged. Values are presented as means±s.d. For normally distributed data, Levene’s test of homogeneity of variance was performed. For continuous variables, one-way ANOVAs were employed for data that showed homogeneity of variance and normal distribution and LSD test was used as post hoc test; for data that did not show homogeneous variance, the Kruskal–Wallis test was used. Non-normally distributed data were analyzed using the Mann–Whitney U-test (the Kruskal–Wallis H-test was performed when the group number was more than 2). The relationship between gene expression levels and other parameters was analyzed using Spearman’s correlation test. A two-tailed value of P<0.05 was considered statistically significant. All statistical analyses were performed using SPSS 17.0 (SPSS, Chicago, IL, USA).
RESULTS
Remodeling Changes in Pulmonary Small Arteries and Changes in Expression of miR-206 in Lungs Following Short-Term or Sustained Hypoxic Exposure
It has been well documented that PH can occur within 21 days of hypoxic treatment.29 We previously observed that prolonged exposure to hypoxia could lead to structural changes in small intrapulmonary arteries in rat lungs.23 In this study, a time-dependent increase in neomuscularization and a thickening of small intrapulmonary arteries was observed, the neomuscularization was demonstrated by α-SMA immunohistochemical staining (Figure 1a). Muscularization of pulmonary arteries was fully established in the advanced stage of PH.
Remodeling changes in pulmonary small arteries and changes in the expression of microRNA-206 (miR-206) in lungs following hypoxic exposure. (a) Hematoxylin and eosin staining and α-smooth muscle actin (α-SMA) immunohistochemical staining. Left: Pulmonary arteries from normoxic animals; middle: following prolonged exposure to hypoxia (21 days); right: following 4 weeks of hypoxic exposure. After short-term and sustained hypoxic exposure, consistent downregulation of miR-206 (b) was observed (*P<0.05; **P<0.01 relative to the normoxia group; n=5 per group).
The level of miR-206 in the lungs was significantly lower following 1 day of hypoxia exposure than in a normoxia control. miR-206 showed sustained low levels during long-term hypoxia, and its expression level was not associated with the disease process (Figure 1b).
Serum Levels of miR-206 are High Following Short-Term Hypoxia and Tend to Decrease Over Time
To investigate whether miR-206 could be detected in circulating blood, serum samples were collected at various time points (0, 1, 4, 7, 14, 21 and 30 days after hypoxic exposure). The data on the expression level of miR-206 in the erum are summarized in Figure 2. In contrast to the lung expression model, rats exposed to short-term hypoxia (1–4 days) had higher serum levels of miR-206, which then decreased in rats exposed to more than 4 days of treatment. During prolonged hypoxia (7, 14, 21 and 30 days), there was a tendency to decreased expression over time.
Activation of the HIF-1α/Fhl-1 Pathway in Acute-Stage PH
Following 1 day of hypoxia treatment, western blotting from lung homogenates (Figure 3a) revealed upregulated expression of HIF-1α; this result was confirmed by an immunofluorescence assay in vitro (Figure 3b). Protein levels of HIF-1α in the lungs showed a tendency to decrease with increasing exposure to hypoxia (Figure 3a). RT-qPCR confirmed that the HIF-1α mRNA level in the hypoxia group did not change significantly compared with the control normoxia group in vivo (Figure 3c).
Activation of hypoxia-inducible factor-1α (HIF-1α)/four-and-a-half LIM (Lin-11, Isl-1 and Mec-3) domain 1 (Fhl-1) proteins during early-stage pulmonary hypertension (PH). (a) Increased levels of HIF-1α protein after short-term hypoxia, but significantly decreased levels following sustained hypoxia. (b) Immunofluorescence staining for HIF-1α (red) and nuclear staining using 4′,6-diamidino-2-phenylindole (DAPI) (blue) in pulmonary artery smooth muscle cells (PASMCs) after normoxia treatment or 1 day of hypoxia exposure. (c) Real-time quantitative PCR (RT-qPCR) confirmed that HIF-1α mRNA in the context of hypoxia did not change significantly compared with the control group in vivo. Western blotting assays (d) and RT-qPCR (g) showed that the Fhl-1 protein and mRNA levels were both upregulated after 1 day of hypoxia exposure in vivo. (e) Expression of Fhl-1 in lung small vascular smooth muscle cells using immunohistochemistry. (f) Immunofluorescence staining for Fhl-1 (red) and nuclear staining using DAPI (blue) revealed the cytoplasmic expression of Fhl-1 in PASMCs after 1 day of hypoxia exposure compared with the normoxia group. (*P<0.05; **P<0.01 relative to the normoxia group; n=5 per group). GAPDH, glyceraldehyde 3-phosphate dehydrogenase.
To investigate the expression pattern of Fhl-1, western blotting assay of homogenized lungs was performed, and a higher level of Fhl-1 protein was observed after hypoxia treatment (Figure 3d). To localize Fhl-1, immunohistochemistry of lung tissue and immunofluorescence staining of PASMCs was performed. Expression of Fhl-1 was observed in small vascular SMCs of the lungs (Figure 3e) and also in the cytoplasm of PASMCs following 1 day of hypoxia treatment (Figure 3f). Quantitative mRNA analysis revealed increased Fhl-1 levels in lung tissue exposed to hypoxia for 1 or 4 days, which then decreased at 7, 14, 21 or 30 days of exposure (Figure 3g).
Effect of miR-206 on the HIF-1α/Fhl-1 Pathway
miRs are generally considered to be post-transcriptional negative regulators of targeted genes; however, our results above indicated opposing trends for miR-206 and the expression of HIF-1α/Fhl-1 after hypoxia treatment in both lungs and PASMCs. We thus explored the possible effect of miR-206 on the hypoxic HIF-1α/Fhl-1 pathway. Unexpectedly, compared with the normoxia and pre-miR negative control transfection groups, we found that HIF-1α and Fhl-1 proteins were strongly upregulated in the group transfected with pre-miR-206, but that the level of neither protein changed significantly when cells were transfected with an miR-206 inhibitor (Figures 4a and b). qPCR analysis showed that the level of HIF-1α mRNA did not change significantly between groups, whereas the change in the expression levels of Fhl-1 mRNA were consistent with its corresponding protein (Figure 4c).
Effect of microRNA-206 (miR-206) on the hypoxia-inducible factor-1α (HIF-1α)/four-and-a-half LIM (Lin-11, Isl-1 and Mec-3) domain 1 (Fhl-1) pathway. Western blotting (a) and immunofluorescence (b) showed that, compared with the normoxia and pre-miR negative control transfection groups, HIF-1α and Fhl-1 proteins were strongly upregulated in the pre-miR-206 transfection group, but that the level of neither protein changed significantly when transfected in parallel with the miR-206 inhibitor. (c) Quantitative PCR analysis revealed that the level of HIF-1α mRNA did not change significantly between groups, whereas the changes in Fhl-1 mRNA expression levels were consistent with its protein levels. (d) Western blotting confirmed the increased levels of HIF-1α and Fhl-1 proteins after hypoxia, which were further strongly upregulated in cells transfected with pre-miR-206 compared with hypoxia exposure only, but it did not show statistical significance. (e) Immunofluorescence staining for HIF-1α and Fhl-1 (red) and nuclear staining using 4′,6-diamidino-2-phenylindole (blue) showed weaker expression of HIF-1α and Fhl-1 followed by hypoxia exposure compared with pre-miR-206 transfection followed by hypoxia exposure. (f) Quantitative mRNA analysis revealed no significant change in HIF-1 and an upregulation in Fhl-1 mRNAs in pulmonary artery smooth muscle cells transfected with pre-miR-206 and exposed to hypoxia compared with normoxia-treated cells. (*P<0.05; **P<0.01 relative to the normoxia group; n=5 per group). GAPDH, glyceraldehyde 3-phosphate dehydrogenase; NC, negative control.
To observe the effect of elevated miR-206 on the HIF-1α/Fhl-1 pathway in the context of hypoxia, we overexpressed miR-206 and then subjected the PASMCs to 24 h of a hypoxic environment. We found that HIF-1α and Fhl-1 protein are strongly upregulated in hypoxic group compared with the normoxia group, and the signals of both proteins were stronger in the group that was transfected with pre-miR-206 after hypoxia treatment compared with a single-hypoxia exposure group, but no significant difference exists between treatment groups (P>0.05) (Figure 4d), the latter finding was consistent with the finding of IF staining (Figure 4e). However, the levels of HIF-1α mRNA did not change significantly in any cohort, whereas Fhl-1 mRNA was significantly upregulated in the treated groups (Figure 4f). To study whether the changes in Fhl-1 expression affect miR-206 status, an siRNA specifically targeting Fhl-1 was applied. Inhibition of Fhl-1 (Figure 5a) did not significantly change the expression of miR-206 under hypoxic conditions (Figure 5b). In addition, HIF-1α mRNA levels did not change significantly (Figure 4f). These results suggest that miR-206 affects the HIF-1α/Fhl-1 pathway at the post-transcriptional level, which was completely contrary to our expectations.
Changes in the expression of four-and-a-half LIM (Lin-11, Isl-1 and Mec-3) domain 1 (Fhl-1) did not affect the expression of microRNA-206 (miR-206). (a) Inhibition of Fhl-1 protein expression by small interfering (si)RNA transfection. (b) MiR-206 expression was significantly decreased after hypoxia but was not influenced by repression of the Fhl-1 gene. (*P<0.05; **P<0.01, relative to the normoxia group; n=5 per group). GAPDH, glyceraldehyde 3-phosphate dehydrogenase; NC, negative control.
Overexpression of miR-206 Promotes HIF-1α/Fhl-1-Induced PASMC Proliferation
To study the effect of miR-206 on PASMC proliferation in vitro, cultured PASMCs were transfected with pre-miR-206 or an miR-206 inhibitor, and transfected with pre-miR-206 or si-Fhl-1, followed by 24 h of hypoxia, respectively. When compared with control PASMCs, a significant increase in PASMC proliferation (as assessed using Ki-67 staining) and a higher proportion of cells in the S phase of the cell cycle were observed in PASMCs containing higher levels of miR-206 (Figures 6a–c), whereas PASMCs with si-Fhl-1 treatment displayed a lower cell proliferation rate (Figure 6a) and a higher proportion of cells in the G1/G0 phase (Figure 6c).
Effect of microRNA-206 (miR-206) or small interfering-four-and-a-half LIM (Lin-11, Isl-1 and Mec-3) domain 1 (si-Fhl-1) transfection on pulmonary artery smooth muscle cell (PASMC) proliferation. (a) Upregulation of miR-206 promotes PASMC proliferation compared with the miR-206 inhibitor. Compared with the pre-miR-206 transfection group, PASMCs expressing si-Fhl-1 displayed a lower cell proliferation rate after hypoxia exposure. (b and c) Phase distribution of the cell cycle in each cohort of PASMCs, as determined using flow cytometry (*P<0.05; **P<0.01, relative to the normoxia group; n=5 per group). NC, negative control.
miR-206 Directly Targets the 3′-UTR of HIF-1α Through mREs
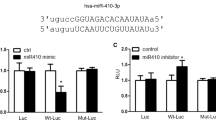
We screened the 3′-UTR region of HIF-1α for mREs (5′-CAUUCC-3′) and identified putative binding sites of miR-206 and miR-1 (Table 5). As shown in Figure 7, miR-206 significantly decreased the relative luciferase reporter activity of the wild-type HIF-1α 3′-UTR, whereas the luciferase reporter activity of the mutant HIF-1α 3′-UTR did not change significantly, which suggests that miR-206 could directly bind to the 3′-UTR of HIF-1α. Taken together, these findings indicate that HIF-1α is a direct downstream target of miR-206 in PASMCs.
MicroRNA-206 (miR-206) directly binds to the 3′-untranslated region (3′-UTR) of hypoxia-inducible factor-1α (HIF-1α). The wild- or mutant-type 3′-UTR of HIF-1α was subcloned downstream of the luciferase (luc) gene. Different effects of miR-206 on the relative luciferase reporter activity of wild/mutant-type HIF-1α 3′-UTR were noted (**P<0.01 relative to control group; n=5 per group). NC, negative control.
DISCUSSION
COPD is a major cause of mortality and morbidity worldwide and is often complicated by the development of PH.30, 31, 32 Cor pulmonale characterized by right ventricular hypertrophy and dilatation is also commonly secondary to PH caused by respiratory disorders.33 It is known that hypoxia, inflammation and other elements have important roles in the development of PH.34 In the process of PH, vascular remodeling contributes to a sustained elevation of pulmonary vascular resistance and pulmonary artery pressure as a result of vascular remodeling characterized largely by vascular smooth muscle cell proliferation and medial hypertrophy.35, 36
The aberrant expression of some miRNAs (such as the downregulation of miR-204 and miR-22 and the upregulation of miR-322) has been implicated recently in PH.37, 38 As one of the muscle-specific miRs,18, 39, 40, 41, 42, 43 miR-206 belongs to the miR-1 family (which, based on the sequence conservation of a 2–8-nucleotide seed region, includes miR-1/206 and miR-133a/b44 that has a critical role in muscle cell biology and pathophysiology. However, no report has linked the miR-1 family to hypoxic PH thus far. For the first time, we confirmed that miR-206 was involved in the mechanism of hypoxia-induced PH. The expression of miR-206 was significantly downregulated in a hypoxia-induced PH rat model, and there was no correlation between the downregulation of miR-206 and the duration of hypoxia exposure, which is in contrast to findings for other miRs (eg, miR-204, as determined from an analysis of 377 different miRNAs) expressed in human PH that have previously been shown to correlate with PH severity,37 suggesting that miR-206 could be involved in the onset stage of hypoxia-induced PH.
Circulating miR-206 in the peripheral blood of the hypoxic PH rat model was also detected. In contrast to other RNA molecules, the circulating miRs are stable and consistent across individuals of the same species,45 and recent reports also indicate that mature miRs are highly stable in circulation.12, 46 Most of the published studies that have examined circulating miRs as biomarkers have focused on cancer,47 but a few reports studied the role of circulating miRs in non-neoplastic diseases. Our research showed that the change in miR-206 expression in the serum in the hypoxic PH rat model was likely to be time-dependent. Our study also showed that miR-206 in the serum did not reflect the molecular response of the lungs. Rather, serum levels may reflect systemic tissue responses and thus may be a nonspecific marker of hypoxemia. The different expression patterns of miR-206 in the serum and lung tissue led to the implication that peripheral cell-free miR-206 may be released directly from microvesicles that are abundant in blood, which reveals a completely different source and mechanism from that derived from the organization-driven miR.48, 49
We believe that circulating miRNAs could be helpful for diagnosis, permitting the detection of early stages of non-neoplastic diseases with samples that are obtained noninvasively. Many miRNAs are already considered biomarkers for cancer; similarly, this should be the case for PH in the coming years. A clinical trial proposes to determine whether miRNA expression in peripheral blood could be used to identify PH development and to aid in clinical diagnosis, and miR-206 could be a triggering factor for the early stage of PH.
Fhl-1 is a member of the LIM family that acts as an early key protein in the mechanism of PH via HIF-1α binding to the hypoxic response element within the Fhl-1 promoter region.4 Previous reports have indicated the presence of a feedback loop between HIF-1α and Fhl-1, and the HIF-1α/Fhl-1 pathway may serve to regulate HIF activity under conditions of prolonged hypoxia.5 Our results showed significantly increased levels of both Fhl-1 mRNA and protein after one day of hypoxic exposure, which were not consistent with previous reports.4 It was puzzling that the mRNA expression level of Fhl-1 was very low at day 30, whereas the protein expression appeared to peak at this time point, which may be due to changed regulatory mechanisms (such as synthesis and degradation rates and post-translational modifications) of the Fhl-1 gene in hypoxic conditions.5 Further research is required to investigate this interesting phenomenon.
It is known that miRs regulate gene expression by interacting with mREs (sequence-guided) and complementary sequences (within a region of the 3′-UTR) of target mRNAs, and that HIF-1α has a key role in hypoxia-related physiologic and pathophysiologic responses.50, 51 A variety of hypoxia-regulated miRs have been identified52, 53, 54, 55, 56, 57 that reveal the link between oxygen-specific stress factors and gene expression control. Our study showed that both HIF-1α and Fhl-1 were upregulated, whereas miR-206 in lungs and PASMCs was significantly downregulated in hypoxia. Bioinformatics analysis and dual-luciferase reporter assays were used to identify and confirm that miR-206 directly targets the mREs of the HIF-1a 3′-UTR. Taken together, we believe there could be a close relationship between miR-206 and the HIF-1α/FHL-1 pathway in hypoxia-induced PH.
Contrary to expectations, compared with those in normoxia group, HIF-1α and Fhl-1 were significantly upregulated at protein level rather than mRNA level in both groups of pre-miR-206 transfection and transfection plus hypoxia, and there was no statistical significance between groups of miR-206 plus hypoxia and hypoxia alone. miRs are generally considered to be negative regulators of target genes on post-transcriptional level, and the change of the target genes normally occurs on protein level rather than on mRNA level. These unexpected results cause confusion on the exact function and regulation mechanism of miR-206 in hypoxia-induced PH. We cannot as yet provide a reasonable explanation for these reproducible phenomena. Possible mechanisms could include the following: other miRs, such as miR-20b58 and 519c,59 triggering reverse effects upon HIF-1α may contribute to the hypoxia response in a complicated network involving the HIF-1α/Fhl-1 pathway; miR-206 may switch its regulating function in a quantity-dependent manner in response to hypoxia; and when miR-206 mimics were applied, competition and saturation effects from endogenous miR-206 could have been involved in the process.60, 61 These effects would be expected to relieve repression of the HIF-1α from other endogenous miRs. The fact that miR-206 is involved in hypoxia-induced PH and that its expression changes frequently in different microenvironments suggests that multiple modes of transcriptional regulation exist for this miR. Other intrinsic or extrinsic mechanisms may exist that control miR-206 targeting of the HIF-1α/Fhl-1 pathway that remains to be defined.
Previous research has shown that a significant inhibition or promotion of PASMC proliferation and migration occurs after silencing or overexpressing Fhl-1, and silencing of Fhl-1 downregulated cyclin D1 expression.4 In our research, cell cycle analysis revealed that cells in the G1 phase were increased after silencing Fhl-1 expression. The low level of cyclin D1 extended the length of the G1 phase, where cells exhibit an increased dependence on growth factors, thereby making it difficult to cross the G1/S transition point for continuous proliferation. Therefore, our experiments further identified that overexpression of miR-206 increased HIF-1α/Fhl-1 upregulation and the percentage of the S-phase cells, suggesting enhanced proliferation of PASMCs, and that silencing of Fhl-1 inhibited cell proliferation. Immunofluorescence staining for Ki-67 confirmed the results from the flow cytometry analysis. These data indicate that the abnormal expression of miR-206 led to a change in the biological response of PASMCs to hypoxia by targeting the HIF-1α/Fhl-1 pathway.
Although we believe that our study provides, for the first time, evidence that miR-206 is involved in hypoxia-induced PH by targeting the HIF-1α/Fhl-1 pathway, we have not elucidated the exact molecular mechanism by which miR-206 impacts the HIF-1α/Fhl-1 pathway. This mechanism remains to be established and will constitute further study.
In summary, our data demonstrate that miR-206 and the HIF-1α/Fhl-1 pathway are involved in a regulatory circuit that modulates PASMC proliferation in hypoxia and likely impacts hypoxia-induced PH. This novel regulatory circuit provides additional insight into how miRs interact with signaling molecules during hypoxia-induced PH and has implications for the treatment of COPD and high-altitude diseases. Circulating miR-206 could be a triggering factor for the early stage of hypoxia-induced PH.
References
Farber HW, Loscalzo J . Pulmonary arterial hypertension. N Engl J Med 2004;351:1655–1665.
Sheikh F, Raskin A, Chu PH et al. An FHL1-containing complex within the cardiomyocyte sarcomere mediates hypertrophic biomechanical stress responses in mice. J Clin Invest 2008;118:3870–3880.
Schessl J, Zou Y, McGrath MJ et al. Proteomic identification of FHL1 as the protein mutated in human reducing body myopathy. J Clin Invest 2008;118:904–912.
Kwapiszewska G, Wygrecka M, Marsh LM et al. Fhl-1, a new key protein in pulmonary hypertension. Circulation 2008;118:1183–1194.
Hubbi ME, Gilkes DM, Baek JH et al. Four-and-a-half LIM domain proteins inhibit transactivation by hypoxia-inducible factor 1. J Biol Chem 2012;287:6139–6149.
Zeng Y, Yi R, Cullen BR . MicroRNAs and small interfering RNAs can inhibit mRNA expression by similar mechanisms. Proc Natl Acad Sci USA 2003;100:9779–9784.
Hutvagner G, Zamore PD . A microRNA in a multiple-turnover RNAi enzyme complex. Science 2002;297:2056–2060.
Bartel DP . MicroRNAs: target recognition and regulatory functions. Cell 2009;136:215–233.
Filipowicz W, Bhattacharyya SN, Sonenberg N . Mechanisms of post-transcriptional regulation by microRNAs: are the answers in sight? Nat Rev Genet 2008;9:102–114.
Ucar A, Vafaizadeh V, Jarry H et al. MiR-212 and miR-132 are required for epithelial stromal interactions necessary for mouse mammary gland development. Nat Genet 2010;42:1101–1108.
Gilad S, Meiri E, Yogev Y et al. Serum microRNAs are promising novel biomarkers. PLoS One 2008;3:e3148.
Mitchell PS, Parkin RK, Kroh EM et al. Circulating microRNAs as stable blood-based markers for cancer detection. Proc Natl Acad Sci USA 2008;105:10513–10518.
Huang Z, Huang D, Ni S et al. Plasma microRNAs are promising novel biomarkers for early detection of colorectal cancer. Int J Cancer 2010;127:118–126.
Jackson DB . Serum-based microRNAs: are we blinded by potential? Proc Natl Acad Sci USA 2009;106:E5.
Gaughwin PM, Ciesla M, Lahiri et al. Hsa-miR-34b is a plasma-stable microRNA that is elevated in pre-manifest Huntington’s disease. Hum Mol Genet 2011;20:2225–2237.
Scholer N, Langer C, Kuchenbauer F . Circulating microRNAs as biomarkers—true blood? Genome Med 2011;3:72.
Cheng H, Zhang L, Cogdell DE et al. Circulating plasma MiR-141 is a novel biomarker for metastatic colon cancer and predicts poor prognosis. PLoS One 2011;6:e17745.
Kloosterman WP, Plasterk RH . The diverse functions of microRNAs in animal development and disease. Dev Cell 2006;11:441–450.
Zhao Y, Ransom JF, Li A et al. Dysregulation of cardiogenesis, cardiac conduction, and cell cycle in mice lacking miRNA-1-2. Cell 2007;129:303–317.
van Rooij E, Sutherland LB, Qi X et al. Control of stress-dependent cardiac growth and gene expression by a microRNA. Science 2007;316:575–579.
Care A, Catalucci D, Felicetti F et al. MicroRNA-133 controls cardiac hypertrophy. Nat Med 2007;13:613–618.
Sempere LF, Freemantle S, Pitha-Rowe I et al. Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol 2004;5:R13.
Zhu P, Huang L, Ge X et al. Transdifferentiation of pulmonary arteriolar endothelial cells into smooth muscle-like cells regulated by myocardin involved in hypoxia-induced pulmonary vascular remodelling. Int J Exp Pathol 2006;87:463–474.
Shimizu RT, Blank RS, Jervis R et al. The smooth muscle alpha-actin gene promoter is differentially regulated in smooth muscle versus non-smooth muscle cells. J Biol Chem 1995;270:7631–7643.
Schmittgen TD, Livak KJ . Analyzing real-time PCR data by the comparative C(T) method. Nat Protoc 2008;3:1101–1108.
Bustin SA, Benes V, Garson JA et al. The MIQE guidelines: minimum information for publication of quantitative real-time PCR experiments. Clin Chem 2009;55:611–622.
Livak KJ, Schmittgen TD . Analysis of relative gene expression data using real-time quantitative PCR and the 2(−Delta Delta C(T)) Method. Methods 2001;25:402–408.
Wang G, Tam LS, Li EK et al. Serum and urinary cell-free MiR-146a and MiR-155 in patients with systemic lupus erythematosus. J Rheumatol 2010;37:2516–2522.
Schermuly RT, Dony E, Ghofrani HA et al. Reversal of experimental pulmonary hypertension by PDGF inhibition. J Clin Invest 2005;115:2811–2821.
Barbera JA, Peinado VI, Santos S . Pulmonary hypertension in chronic obstructive pulmonary disease. Eur Respir J 2003;21:892–905.
Wrobel JP, Thompson BR, Williams TJ . Mechanisms of pulmonary hypertension in chronic obstructive pulmonary disease: a pathophysiologic review. J Heart Lung Transpl 2012;31:557–564.
Simonneau G, Robbins IM, Beghetti M et al. Updated clinical classification of pulmonary hypertension. J Am Coll Cardiol 2009;54 (Suppl):S43–S54.
Lee-Chiong TL Jr, Matthay RA . Pulmonary hypertension and cor pulmonale in COPD. Semin Respir Crit Care Med 2003;24:263–272.
Nakanishi K, Tajima F, Nakata Y et al. Expression of endothelin-1 in rats developing hypobaric hypoxia-induced pulmonary hypertension. Lab Invest 1999;79:1347–1357.
Stenmark KR, McMurtry IF . Vascular remodeling versus vasoconstriction in chronic hypoxic pulmonary hypertension: a time for reappraisal? Circ Res 2005;97:95–98.
Martin KB, Klinger JR, Rounds SI . Pulmonary arterial hypertension: new insights and new hope. Respirology 2006;11:6–17.
Courboulin A, Paulin R, Giguere NJ et al. Role for miR-204 in human pulmonary arterial hypertension. J Exp Med 2011;208:535–548.
Caruso P, MacLean MR, Khanin R et al. Dynamic changes in lung microRNA profiles during the development of pulmonary hypertension due to chronic hypoxia and monocrotaline. Arterioscler Thromb Vasc Biol 2010;30:716–723.
Lee RC, Ambros V . An extensive class of small RNAs in Caenorhabditis elegans. Science 2001;294:862–864.
Lagos-Quintana M, Rauhut R, Lendeckel W et al. Identification of novel genes coding for small expressed RNAs. Science 2001;294:853–858.
Boutz PL, Chawla G, Stoilov P et al. MicroRNAs regulate the expression of the alternative splicing factor nPTB during muscle development. Genes Dev 2007;21:71–84.
Winbanks CE, Wang B, Beyer C et al. TGF-beta regulates miR-206 and miR-29 to control myogenic differentiation through regulation of HDAC4. J Biol Chem 2011;286:13805–13814.
Dey BK, Gagan J, Dutta A . MiR-206 and -486 induce myoblast differentiation by downregulating Pax7. Mol Cell Biol 2011;31:203–214.
Hertel J, Lindemeyer M, Missal K et al. The expansion of the metazoan microRNA repertoire. BMC Genom 2006;7:25.
Chen X, Ba Y, Ma L et al. Characterization of microRNAs in serum: a novel class of biomarkers for diagnosis of cancer and other diseases. Cell Res 2008;18:997–1006.
Mraz M, Malinova K, Mayer J et al. MicroRNA isolation and stability in stored RNA samples. Biochem Biophys Res Commun 2009;390:1–4.
Scholer N, Langer C, Dohner H et al. Serum microRNAs as a novel class of biomarkers: a comprehensive review of the literature. Exp Hematol 2010;38:1126–1130.
Yuan A, Farber EL, Rapoport AL et al. Transfer of microRNAs by embryonic stem cell microvesicles. PLoS One 2009;4:e4722.
Chen TS, Lai RC, Lee MM et al. Mesenchymal stem cell secretes microparticles enriched in pre-microRNAs. Nucleic Acids Res 2010;38:215–224.
Adams JM, Difazio LT, Rolandelli RH et al. HIF-1: a key mediator in hypoxia. Acta Physiol Hung 2009;96:19–28.
Brahimi-Horn MC, Pouyssegur J . HIF at a glance. J Cell Sci 2009;122 (Part 8):1055–1057.
Kulshreshtha R, Davuluri RV, Calin GA . A microRNA component of the hypoxic response. Cell Death Differ 2008;15:667–671.
Kulshreshtha R, Ferracin M, Negrini M et al. Regulation of microRNA expression: the hypoxic component. Cell Cycle 2007;6:1426–1431.
Hebert C, Norris K, Scheper MA et al. High mobility group A2 is a target for miRNA-98 in head and neck squamous cell carcinoma. Mol Cancer 2007;6:5.
Hua Z, Lv Q, Ye W et al. MiRNA-directed regulation of VEGF and other angiogenic factors under hypoxia. PLoS One 2006;1:e116.
Bruning U, Cerone L, Neufeld Z et al. MicroRNA-155 promotes resolution of hypoxia-inducible factor 1alpha activity during prolonged hypoxia. Mol Cell Biol 2011;31:4087–4096.
Wu C, So J, Davis-Dusenbery BN et al. Hypoxia potentiates microRNA-mediated gene silencing through posttranslational modification of argonaute2. Mol Cell Biol 2011;31:4760–4774.
Lei Z, Li B, Yang Z et al. Regulation of HIF-1alpha and VEGF by miR-20b tunes tumor cells to adapt to the alteration of oxygen concentration. PLoS One 2009;4:e7629.
Cha ST, Chen PS, Johansson G et al. MicroRNA-519c suppresses hypoxia-inducible factor-1alpha expression and tumor angiogenesis. Cancer Res 2010;70:2675–2685.
Khan AA, Betel D, Miller ML et al. Transfection of small RNAs globally perturbs gene regulation by endogenous microRNAs. Nat Biotechnol 2009;27:549–555.
Saito T, Saetrom P . Target gene expression levels and competition between transfected and endogenous microRNAs are strong confounding factors in microRNA high-throughput experiments. Silence 2012;3:3.
Acknowledgements
We thank Dr Xiaoqing Chen for rigorous statistical technical assistance and Dr Fang He for critical reading of the manuscript. This research was supported by research grants 30800403 (to PZ) and 81270176 (to GW) from the Natural Science Foundation of China.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
This paper reveals the role of muscle-specific microRNA-206 in a rat model of hypoxia-induced pulmonary hypertension. Hypoxia induces down-regulation of miR-206 by means of the hypoxia-inducible factor-1α/four-and-a-half LIM domain 1 pathway in pulmonary artery smooth muscle cells, suggesting that miR-206 may be an important factor in chronic obstructive pulmonary disease.
Rights and permissions
About this article
Cite this article
Yue, J., Guan, J., Wang, X. et al. MicroRNA-206 is involved in hypoxia-induced pulmonary hypertension through targeting of the HIF-1α/Fhl-1 pathway. Lab Invest 93, 748–759 (2013). https://doi.org/10.1038/labinvest.2013.63
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2013.63
Keywords
This article is cited by
-
Activation of AMPK inhibits TGF-β1-induced airway smooth muscle cells proliferation and its potential mechanisms
Scientific Reports (2018)
-
microRNA-206 is involved in survival of hypoxia preconditioned mesenchymal stem cells through targeting Pim-1 kinase
Stem Cell Research & Therapy (2016)
-
Experimental Study of the Mechanisms of Intracellular Defense in Cardiomyocytes Associated with Stages of Anthracosilicosis Development
Bulletin of Experimental Biology and Medicine (2015)