Abstract
General control nonderepresible 2 (GCN2) is a highly conserved cytosolic kinase that modulates a complex response for coping with the stress owing to lack of amino acids. GCN2 has been recently shown to be involved in the regulation of metabolic balance and lipid degradation rate in the liver. We hypothesized that GCN2 could have a role in in hepatic fibrogenesis and in the response to acute or chronic liver injury. Activation of GCN2 in primary or immortalized human hepatic stellate cells by incubation with medium lacking the essential amino acid histidine correlated with decreased levels of collagen type I protein and mRNA, suggesting an antifibrogenic effect of GCN2. In vivo studies with Gcn2 knock-out mice (Gcn2−/−) showed increased susceptibility to both acute or chronic liver damage induced by CCl4, as shown by higher alanine aminotransferase and aspartate aminotransferase activities, increased necrosis and higher inflammatory infiltrates compared with wild-type mice (WT). Chronic CCl4 treatment increased deposition of interstitial collagen type I more in Gcn2−/− mice than in WT mice. Col1a1 and col1a2 mRNA levels also increased in CCl4-treated Gcn2−/− mice compared with WT mice. These results suggest that GCN2 is a key regulator of the fibrogenic response to liver injury.
Similar content being viewed by others
Main
GCN2 (general control nonderepressible 2), is a cytosolic kinase present in all cell types that becomes activated by the accumulation of uncharged tRNAs,1, 2 triggering a highly conserved and well-orchestrated pathway leading to changes in protein expression and helping to cope with the stress caused by nutrient deficiency.3, 4 The effects of GCN2 activation were initially described to modulate processes related to protein metabolism or amino acid transport. However, recent studies have demonstrated the role played by GCN2 in other cellular responses, such as sensitivity to RNA viral infection5 or T-cell differentiation.6 Although GCN2 is typically activated by amino acid deprivation, UV exposure7, 8 or high levels of urea9 can also lead to GCN2 activation. These data suggest that GCN2 could act as a key regulator to the cellular response under stress conditions.
Previous work indicates that in the liver GCN2 contributes to the control of protein balance during deprivation of essential amino acids10 and has also been shown to regulate hepatic fatty-acid metabolism11 and hepatic insulin sensitivity.12 Gcn2−/− mice are more sensitive to oxidative stress induced in the liver by an imbalanced diet and present decreased levels of glutathione peroxidase in the liver.13 Most of the studies have focused on stress caused by amino acid deprivation, yet the role of GCN2 in liver injury and fibrogenesis has not been studied. Work from our group suggested that GCN2 could be a regulator of the biology of hepatic stellate cells (HCS), the cell type mainly responsible for collagen deposition in the liver. Both in primary and in immortalized HSC, activation of GCN2 induced by either by amino acid restriction or by proteasome inhibition caused a fast and sustained decrease in intracellular hydrogen peroxide levels.14 This result pointed to GCN2 as a potential regulator of fibrogenesis, as hydrogen peroxide has been shown to induce collagen production and to mediate the profibrogenic action of TGF-β on HSC.15, 16, 17
In the present paper we explored the role of GCN2 on the regulation of collagen type I expression in HSC and in the response to acute and chronic liver damage. Using in vitro and in vivo approaches, we demonstrate that GCN2 has a key role in collagen type I production by HSC and protects from liver injury and fibrosis in mice.
MATERIALS AND METHODS
Cell Culture and Treatments
Cell culture experiments were carried out using primary human HSC and the human HSC line LX-2. This non-tumoral cell line was obtained after immortalization in low-serum media culture of HSC isolated from normal primary human HSC18 and was kindly provided by Dr SL Friedman, Mount Sinai School of Medicine, New York. Cells were cultured in Dulbeccos’s modified Eagle’s medium supplemented with 1% fetal bovine serum, penicillin/streptomycin and non-essential amino acids for 36 h. The medium was replaced with serum-free Dulbeccos’s modified Eagle’s medium for 12 h, after which treatments were carried out.
Primary human HSC were kindly provided by Dr F Hong, Mount Sinai School of Medicine, New York. Cells were cultured in minimun essential medium (MEM) supplemented with 10% fetal bovine serum and penicillin/streptomycin for 36 h. The medium was replaced with serum-free MEM for 12 h, after which treatments were added.
Cells were treated by replacing the media with MEM without histidine for 4, 8 and 24 h. Control cells were incubated with complete MEM, containing histidine 270 μM. Cell culture reagents were obtained from Gibco BRL (Grand Island, NY, USA).
Western Blot
The antibodies used on western blot analysis were: total GCN2, phospho-specific GCN2, LC3I and II from Cell Signaling Technology (Beverly, MA, USA), collagen type I from Millipore; β-actin and p62 from Sigma (St Louis, MO, USA). Following incubation with primary antibodies, membranes were washed and incubated with a secondary polyclonal (Bio-Rad, Hercules, CA, USA) or monoclonal (Amersham Life Science, Arlington Heights, IL, USA) antibodies, conjugated to horseradish peroxidase. Immunoreactive proteins were detected by enhanced chemiluminescence system (Roche Molecular Biochemicals, Lewes, UK). Figures are representative of at least three independent experiments.
Cell Viability
To evaluate cell viability in vitro, trypan blue staining was used for determination of dead cells by dye exclusion. For these assays 1 × 106 LX-2 cells were seeded in 100 mm culture dishes and treated during 24 h either with histidine-free medium or complete medium. The cells were trypsinized, incubated with trypan blue (0.2% in phosphate-buffered saline) and counted within 3 min under light microscopy using a hemocytometer. Triplicate samples form two independent experiments were examined for each data point. When indicated, cells were pretreated for 30 min with either chloroquine or 3-methyl adenine, obtained from SIGMA Chemical Company.
Quantitative Real-Time RT-PCR
Sybrgreen quantitative real-time RT-PCR was performed in a Roche Lightcycler 480 using the following PCR primers: human COL1A1 forward 5′-ACGCTAACCCCCTCCCCAGC-3′ and human COL1A1 reverse 5′-CGGTGGCCGCTAAGAGGAGC-3′; human COL1A2 forward 5′-CGGCAGCAGGAGGTTTCGGC-3′ and human COL1A2 reverse 5′-GTCGCAGAGCCCCTGGGTCA-3′; mouse COL1A1 forward 5′-GCGGTAACGATGGTGCTGTT-3′ and mouse COL1A1 reverse 5′-CTTCACCCTTAGCACCAAC-3′; mouse COL1A2 forward 5′-TTGTCGCCAGTGAG-3′ and mouse COL1A2 reverse 5′-CTGGTCCTGCTGGT-3′. All values were normalized to glyceraldehyde-3-phosphate dehydrogenase levels. Results were expressed as relative fold change.
Animal Study Design
Homozygous mice for the GCN2.KO4 mutant locus and their WT littermates were used (The Jackson Laboratory, Bar Harbor, ME, USA). Mice received humane care in compliance with the guidelines of the National Institutes of Health and Animal Care Committee at the Mount Sinai School of Medicine. Acute liver injury was induced by injecting intraperitoneally 0.05 ml of CCl4/kg of body weight (25% v/v in mineral oil). Chronic liver injury was induced by injecting intraperitoneally 0.05 ml of CCl4/kg of body weight (25% v/v in mineral oil) twice a week for 4 weeks. Control groups were injected with mineral oil. Mice were maintained at 22 °C with 12 h light/dark cycle, were fed standard chow diet, had free access to water, and were killed under ketamine/xylazine anesthesia 48 h after the last dose of CCl4.
Serum Transaminases
Blood was collected from the retro-orbital vein under anesthesia, centrifuged at 3000 r.p.m. for 3 min, and serum was separated to assay for alanine aminotransferase (ALT) and aspartate aminotransferase (AST) using kits from Pointe Scientific (Canton, MI, USA).
Liver Histology and Immunohistochemistry
Liver samples were fixed in 10% buffered formalin and embedded in paraffin. Five micrometer sections were dehydrated and stained with hematoxylin and eosin (H&E) and were evaluated by a liver pathologist. The sirius red/fast green staining was carried out as according to López-De León and Rojkind.19 Collagen type I immunostaining was carried out using a monoclonal antibody (Millipore, Darmstadt, Germany) and ImmunoCruz-staining system.
RESULTS
Activation of GCN2 Decreases Collagen Type I Expression in HSC
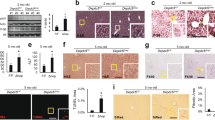
The first experiments were aimed to establish the effect of GCN2 activation on collagen type I expression by HSC. GCN2 was activated by incubation with histidine-free medium, and phosphorylated GCN2 (the active form of the enzyme) and protein levels of collagen type I were analyzed by western blot. As shown in Figure 1a, incubation of LX-2 cells with histidine-free medium increased phosphorylated GCN2 time-dependently up to 4 h. Both in LX-2 cells and in primary human HSC, collagen-I protein expression decreased after incubation with histidine-free medium up to 24 h (Figure 1b).
Time course analysis of GCN2 (general control nonderepresible 2) phosphorylation and collagen type I protein levels in hepatic stellate cells (HSC) treated with histidine-free medium. (a) The cell line LX-2 was treated from 30, 60, 120 and 240 min with medium without histidine and total and phospho-active GCN2 levels were determined by western blot. (b) LX-2 and primary human HSC were treated from 4 to 24 h with medium without histidine and collagen type I production was determined by western blot. Upper panels, representative immunoblots. Lower panels, densitometry values referred to the loading control. Each bar represents the mean±s.d. of protein fold change compared with control of four independent experiments (*P<0.05; **P<0.01, a vs 4 h control; b vs its own control; C control; Hisw/o, histidine-free medium).
To determine whether changes in collagen type I protein expression in response to GCN2 activation were due to transcriptional inhibition, the COL1A1 and COL1A2 genes mRNA levels were analyzed. As shown in Figure 2a, in control LX-2 cells, which were incubated in complete medium, COL1A1 and COL1A2 mRNAs increased after 24 h. In contrast, both mRNAs remained unchanged in cells cultured with histidine-free medium. The same effects were observed in primary human HSC (Figure 2b).
Time course analysis of collagen type I mRNA levels in hepatic stellate cells (HSC) treated with histidine-free medium. Analysis of COL1A1 and COL1A2 mRNA levels by quantitative PCR were carried out in the cell line LX-2 (a) or in primary human HSC (b) treated from 4 to 24 h with histidine-free medium. Values are referred to basal levels of glyceraldehyde-3-phosphate dehydrogenase (GAPDH) mRNA. Control represents non-treated cells and was given the value of one. Each bar represents the mean±s.d. of mRNA fold change compared with control of at least triplicate data from four independent experiments (*P<0.05; **P<0.01, a vs 4 h control; b vs its own control; Hisw/o, histidine-free medium).
Autophagy is Not Induced By GCN2 Activation in HSC
GCN2 has been described in different cell types to mediate autophagy induced by amino acid starvation or by tRNA accumulation, suggesting a possible role of autophagy in the response of HSC to GCN2 activation. We first evaluated the effect of autophagy inhibitors chloroquine and 3-methyl adenine on the viability of HSC in response to GCN2 activation. As shown in Supplementary Figure 1, both autophagy inhibitors caused in LX-2 a decrease on cell viability, which was more significant for chloroquine than for 3-methyl adenine. GCN2 activation by histidine deprivation enhanced LX-2 viability both in control cells and in cells pretreated with autophagy inhibitors.
To analyze whether GCN2 activation does in fact induce autophagy in HSC, western blot analysis of LC3I, II and of p62 was carried out in LX-2 incubated with histidine deprived medium. GCN2 activation did not alter p62 levels and failed to enhance LC3I and II levels (Supplementary Figure 2).
Acute CCl4 Injection Causes More Liver Injury To Gcn2−/− than to WT Mice
We had previously demonstrated that GCN2 activation causes an increase in HSC viability and a decrease in intracellular hydrogen peroxide levels.13 These results as well as the antifibrogenic action described above, suggested that GCN2 could have a role in regulating the response to liver damage. To evaluate this possibility we studied the effects of acute and chronic liver injury in Gcn2−/− mice.
Acute CCl4 treatment caused increased ALT and AST activities by 4.1 and 3.9-fold, respectively, in Gcn2−/− mice compared with WT mice (Figure 3a).
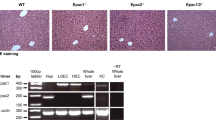
Analysis of the effects of acute carbon tetrachloride (CCl4) treatment on Gcn2−/− mice. Wild-type (WT) or Gcn2−/− mice were injected with CCl4 (0.05 ml/kg of body weight) or with mineral oil (MO) for the controls, and killed 48 h later. (a) Alanine aminotransferase (ALT) and aspartate aminotransferase (AST) activities in blood samples collected from retro-orbital vein 24 h after treatment. Each bar represents the mean±s.d. of IU/l compared with control of at least triplicate data from four independent experiments (*P<0.05; **P<0.01). (b) Liver sections of WT and Gcn2−/− mice treated with either MO or CCl4 and stained with hemotoxylin and eosin. Green arrows point to necrosis ( × 400).
Hematoxylin and eosin (H&E) staining showed more necrotic areas in CCl4-treated Gcn2−/− mice compared with WT mice, although there was more inflammation in acute CCl4-treated WT than in Gcn2−/− mice (Figure 3b). Pathological scores based on the Brunt classification validated these findings. The necrosis score was 2.5±0.26 and 1.5±0.34 for CCl4-treated Gcn2−/− mice and WT mice, respectively. The inflammation score was 1.1±0.22 and 2.0±0.35 for CCl4-treated Gcn2−/− and WT mice, respectively.
Chronic CCl4 Injection Causes More Liver Injury to Gcn2−/− than to WT Mice
Chronic CCl4 treatment increased ALT and AST activities 3.3 and 3.2-fold, respectively, in Gcn2−/− compared with WT mice (Figure 4a).
Analysis of the effects of chronic carbon tetrachloride (CCl4) treatment on Gcn2−/− mice. Wild-type (WT) or Gcn2−/− mice were injected with either mineral oil (MO) for the controls or 0.05 ml/kg of body weight of CCl4 twice a week for 1 month. Mice were killed 48 h after the last injection. Blood was collected from retro-orbital vein. (a) Serum analysis of alanine aminotransferase (ALT) and aspartate aminotransferase (AST) of WT and Gcn2−/− mice. Each bar represents the mean±s.d. of IU/l compared with control of at least triplicate data from four independent experiments (*P<0.05; **P<0.01). (b) Liver sections of WT and Gcn2−/− mice treated with either MO or CCl4 stained with hematoxylin and eosin. Green arrows point to necrosis areas and yellow arrows point to inflammation areas, characterized by lymphocyte infiltration ( × 400).
H&E staining showed more necrosis and inflammation in Gcn2−/− compared with WT mice (Figure 4b). The necrosis scores were 2.9±0.1 and 2.4±0.4 for CCl4-treated Gcn2−/− and WT mice, respectively; the inflammation scores for CCl4-treated Gcn2−/− and WT mice were 2.1±0.7 and 1.7±0.2, respectively.
To evaluate whether the higher sensitivity of Gcn2−/− mice to liver injury was owing to different levels of CYP2E2, protein levels of this enzyme were determined by western blot in liver samples of WT and Gcn2−/− mice. No difference was found (data not shown).
Chronic CCl4 Treatment Increases Collagen-I in Gcn2−/− Compared with WT Mice
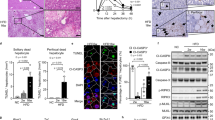
As shown in Figure 5a, Gcn2−/− control mice had increased basal collagenous proteins compared with WT mice. Liver sections from CCl4-treated Gcn2−/− mice showed more collagenous proteins deposition compared with WT mice.
Collagen content on liver sections of chronic carbon tetrachloride (CCl4)-treated Gcn2−/− mice. Wild-type (WT) or Gcn2−/− mice were injected with either mineral oil (MO) for the controls or 0.05 ml/kg of body weight of CCl4 twice a week during one month. Forty-eight hours after the last injection mice were killed. (a) Liver sections stained with fast green/sirius red that specifically stains collagen fibers in red. (b) Collagen-I immunostaining carried out using a monoclonal antibody.
Collagen type I deposition was greater in CCl4-treated Gcn2−/− mice compared with WT mice (Figure 5b). Finally, mRNA levels for Col1a1 and Col1a2 were analyzed. Both col1a1 and col1a2 mRNAs showed a significant increase in CCl4-treated Gcn2−/− mice compared with WT mice (Figure 6).
Collagen type I mRNA levels in chronic carbon tetrachloride (CCl4)-treated Gcn2−/− mice. Wild-type (WT) or Gcn2−/− mice were injected with either mineral oil (MO) for the controls or 0.05 ml/kg of body weight of CCl4 twice a week during 1 month. Mice were killed 48 h after the last injection. Analysis of Col1a1 and Col1a2 mRNA levels was carried out by quantitative PCR in liver homogenates. Values were referred to basal levels of Gadph mRNA. Controls represent non-treated mice and were given the value of one. Each bar represents the mean±s.d. of mRNA fold change compared with controls (**P<0.01).
DISCUSSION
Liver injury triggers a pathological response characterized by tissue damage and inflammatory infiltration leading to hepatic fibrosis. Collagen type I production by activated HSC contributes to the development of liver fibrosis. TGF-β,15 reactive oxygen species16, 17 and acetaldehyde17 regulate collagen type I production by HSC via transcriptional and translational mechanisms. Amino acid levels also exert important regulatory effects in HSC. Studies from our group demonstrated that increased levels of the essential amino acid leucine had a profibrogenic effect on HSC, stimulating selective translation of col1a1 via ROS production and eIF4E phosphorylation.20, 21 Previous results obtained in fibroblasts indicated that amino acid deprivation could be antifibrogenic,22, 23 and our own data in HSC suggested this possibility, as histidine deprivation decreased hydrogen peroxide,14 a key activator of collagen type I production.
In the present study we analyzed the effects of GCN2 activation on collagen type I production by HSC. On the basis of data from other groups and our own, GCN2 activation was induced by incubation of HSC in the absence of histidine, an essential amino acid. There was a decrease in both collagen type I protein and mRNA levels of COL1A1 and COL1A2 mRNAs in response to GCN2 activation by histidine deprivation. As changes in metalloproteinase (MMP) activity or in the levels of its inhibitor TIMP1 could be involved in diminishing collagen-I deposition, protein levels of MMP1, MMP2, MMP9 and MMP13 and TIMP1 were analyzed in primary human HSC treated with histidine-free medium. None of these proteins changed in response to GCN2 activation (data not shown).
GCN2 activation induced by amino acid restriction24 or tRNA accumulation25 has been shown to cause autophagy in different cell types. The role of autophagy in liver injury is complex. On the one hand, autophagy has been shown to protect against liver injury from alcohol and oxidative stress.26, 27 On the other, autophagy contributes to activation of HSC, a key process leading to hepatic fibrosis in HSC.28 In our system, activation of GCN by histidine deprivation failed to induce autophagy in HSC and had a protective effect, counteracting the loss of viability caused by autophagy inhibitors. These results suggest that GCN2 exerts in HSC antifibrogenic and protective effects, which are unrelated to autophagy.
Although further research should establish in greater detail the molecular mechanisms involved in the downregulation of collagen type I by GCN2 activation, there are evidence from the literature pointing to the possibility of collagen-I being transcriptionally regulated. Amino acid deficiency typically activates ATF-4 and other members of the leucine-zipper family such as CHOP and C/EBP-β via GCN2.3, 4 Some members of this family of transcription factors have been shown to participate either in the upregulation15, 29 or in the downregulation30, 31 of the col1a1 and col1a2 genes in rat HSC, and other cells.32 Therefore, downregulation of col1a1 and col1a2 genes could be part of the modification of gene expression mediated by GCN2 to cope with stress situations.
The hepatoprotective role of GCN2 was further established when the effects caused by acute and chronic injury in mice lacking GCN2 were evaluated. Although Gcn2−/− mice have been characterized in other experimental approaches such as their response to amino acid deficiency,10, 11, 12 to our knowledge this is the first study evaluating the ability of these mice to react to stress not directly related to nutritional conditions. After acute and chronic CCl4-treatment, Gcn2−/− mice exhibited increased ALT and AST activities, more necrotic areas and, in the case of chronic treatment, enhanced inflammatory cell infiltration and collagen type I production. Control Gcn2−/− mice showed greater collagen type I deposition compared with control WT animals, suggesting that lack of GNC2 induces by itself a fibrogenic effect that could be further enhanced by the response to chronic liver injury. Thus, GCN2 could be involved in the regulation of basal collagen type I production in the absence of liver injury.
The reason(s) why Gcn2−/− mice are more sensitive to liver damage remain to be established. Recent studies have shown a regulation of hepatic cytochrome P450 (CYP) 3A because of activation of GCN2 by proteasome inhibition,33 pointing to a possible alteration on members of the CYP family on Gcn2−/− animals. However, analysis on WT and Gcn2−/− mice of hepatic levels of CYP2E2, the main responsible for CCl4 metabolization, did not show any difference. Data form our own group and others indicate that oxidative stress could be involved in the higher susceptibility of Gcn2−/− mice to liver injury. Gcn2−/− mice present decreased levels of glutathione peroxidase in the liver13 and we found GCN2 activation by histidine deprivation to have antioxidant effects on hepatocytes and HSC, causing a decrease in intracellular hydrogen peroxide levels.14 Therefore, GCN2-deficient mice would lack a protective pathway than can be triggered by different stimuli and this could contribute to their being more sensitive to liver damage.
In summary, our results indicate that GCN2 can be an important regulator of the liver injury and fibrosis in the acute and chronic setting. Thus, GCN2 may be a potential target for antifibrotic or hepatoprotective strategies.
References
Wek SA, Zhu S, Wek RC . The histidyl-tRNA synthetase-related sequence in the eIF-2 alpha protein kinase GCN2 interacts with tRNA and is required for activation in response to starvation for different amino acids. Mol Cell Biol 1995;15:4497–4506.
Dong J, Qiu H, Garcia-Barrio M et al. Uncharged tRNA activates GCN2 by displacing the protein kinase moiety from a bipartite tRNA-binding domain. Mol Cell 2000;6:269–279.
Kilberg MS, Shan J, Su N . ATF4-dependent transcription mediates signaling of amino acid limitation. Trends Endocrinol Metab 2009;20:436–443.
Kimball SR, Jefferson LS . Role of amino acids in the translational control of protein synthesis in mammals. Semin Cell Dev Biol 2005;16:21–27.
Berlanga JJ, Ventoso I, Harding HP et al. Antiviral effect of the mammalian translation initiation factor 2alpha kinase GCN2 against RNA viruses. EMBO J 2006;25:1730–1740.
Sundrud MS, Koralov SB, Feuerer M et al. Halofuginone inhibits TH17 cell differentiation by activating the amino acid starvation response. Science 2009;324:1334–1338.
Deng J, Harding HP, Raught B et al. Activation of GCN2 in UV-irradiated cells inhibits translation. Curr Biol 2002;12:1279–1286.
Parker SH, Parker TA, George KS et al. The roles of translation initiation regulation in ultraviolet light-induced apoptosis. Mol Cell Biochem 2006;293:173–181.
Cai Q, Brooks HL . Phosphorylation of eIF2α via the general control kinase, GCN2, modulates the ability of renal medullary cells to survive high urea stress. Am J Physiol Renal Physiol 2011;301:F1202–F1207.
Anthony TG, McDaniel BJ, Byerley RL et al. Preservation of liver protein synthesis during dietary leucine deprivation occurs at the expense of skeletal muscle mass in mice deleted for eIF2 kinase GCN2. J Biol Chem 2004;279:36553–36561.
Guo F, Cavener DR . The GCN2 eIF2alpha kinase regulates fatty-acid homeostasis in the liver during deprivation of an essential amino acid. Cell Metab 2007;5:103–114.
Xiao F, Huang Z, Li H et al. Leucine deprivation increases hepatic insulin sensitivity via GCN2/mTOR/S6K1 and AMPK pathways. Diabetes 2011;60:746–756.
Chaveroux C, Lambert-Langlais S, Parry L et al. Identification of GCN2 as new redox regulator for oxidative stress prevention in vivo. Biochem Biophys Res Commun 2011;415:120–124.
Arriazu E, Pérez de Obanos MP, López-Zabalza MJ et al. Amino acid deprivation decreases intracellular levels of reactive oxygen species in hepatic stellate cells. Cell Physiol Biochem 2010;26:281–290.
García-Trevijano ER, Iraburu MJ, Fontana L et al. Transforming growth factor beta1 induces the expression of alpha1(I) procollagen mRNA by a hydrogen peroxide-C/EBPbeta-dependent mechanism in rat hepatic stellate cells. Hepatology 1999;29:960–970.
Nieto N, Friedman SL, Cederbaum AI . Cytochrome P450 2E1-derived reactive oxygen species mediate paracrine stimulation of collagen I protein synthesis by hepatic stellate cells. J Biol. Chem 2002;277:9853–9864.
Greenwel P, Domínguez-Rosales JA, Mavi G et al. Hydrogen peroxide: a link between acetaldehyde-elicited alpha1(I) collagen gene up-regulation and oxidative stress in mouse hepatic stellate cells. Hepatology 2000;31:109–116.
Xu L, Hui AY, Albanis E et al. Human hepatic stellate cell lines, LX-1 and LX-2: new tools for analysis of hepatic fibrosis. Gut 2005;54:142–151.
López-De León A, Rojkind M . A simple micromethod for collagen and total protein determination in formalin-fixed paraffin-embedded sections. J Histochem Cytochem 1985;33:737–743.
Pérez de Obanos MP, López-Zabalza MJ, Prieto J et al. Leucine stimulates procollagen alpha1(I) translation on hepatic stellate cells through ERK and PI3K/Akt/mTOR activation. J Cell Physiol 2006;209:580–586.
Pérez de Obanos MP, López-Zabalza MJ, Arriazu E et al. Reactive oxygen species (ROS) mediate the effects of leucine on translation regulation and type I collagen production in hepatic stellate cells. Biochem Biophys Acta 2007;1773:1681–1688.
Rishikof DC, Ricupero DA, Poliks CF et al. Amino acid availability regulates type I procollagen accumulation in human lung fibroblasts. J Cell Biochem 1999;75:130–137.
Krupsky M, Kuang PP, Goldstein RH . Regulation of type I collagen mRNA by amino acid deprivation in human lung fibroblasts. J Biol Chem 1997;272:13864–13868.
Tallóczy Z, Jiang W, Virgin HW et al. Regulation of starvation- and virus-induced autophagy by the eIF2alpha kinase signaling pathway. Proc Natl Acad Sci USA 2002;99:190–195.
Huynh LN, Thangavel M, Chen T et al. Linking tRNA localization with activation of nutritional stress responses. Cell Cycle 2010;9:3112–3118.
Ding WX, Li M, Chen X et al. Autophagy reduces acute ethanol-induced hepatotoxicity and steatosis in mice. Gastroenterology 2010;139:1740–1752.
Czaja MJ . Functions of autophagy in hepatic and pancreatic physiology and disease. Gastroenterology 2011;140:1895–1908.
Hernández–Gea V, Ghiassi–Nejad Z, Rozenfeld R et al. Autophagy releases lipid that promotes fibrogenesis by activated hepatic stellate cells in mice and in human tissues. Gastroenterology 2012;142:938–946.
Attard FA, Wang L, Potter JJ et al. CCAAT/enhancer binding protein beta mediates the activation of the murine alpha1(I) collagen promoter by acetaldehyde. Arch Biochem Biophys 2000;378:57–64.
Greenwel P, Tanaka S, Penkov D et al. Tumor necrosis factor alpha inhibits type I collagen synthesis through repressive CCAAT/enhancer-binding proteins. Mol Cell Biol 2000;20:912–918.
Iraburu MJ, Domínguez-Rosales JA, Fontana L et al. Tumor necrosis factor alpha down-regulates expression of the alpha1(I) collagen gene in rat hepatic stellate cells through a p20C/EBPbeta- and C/EBPdelta-dependent mechanism. Hepatology 2000;31:1086–1093.
Ghosh AK, Bhattacharyya S, Mori Y et al. Inhibition of collagen gene expression by interferon-gamma: novel role of the CCAAT/enhancer binding protein beta (C/EBPbeta). J Cell Physiol 2006;207:251–260.
Acharya P, Engel JC, Correia MA . Hepatic CYP3A suppression by high concentrations of proteasomal inhibitors: a consequence of endoplasmic reticulum (ER) stress induction, activation of RNA-dependent protein kinase-like ER-bound eukaryotic initiation factor 2alpha (eIF2alpha)-kinase (PERK) and general control nonderepressible-2 eIF2alpha kinase (GCN2), and global translational shutoff. Mol Pharmacol 2009;76:503–515.
Acknowledgements
This work was supported by Fondo de Investigaciones Sanitarias (FIS) grant 00/0143 (to MI, EA, MRG and MJLZ). US Public Health Service Grants 5ROI AA 017733, 5ROI AA017733-0151, 5P20 AA017067, SP20 AA017067-0151 and AA017067-0351 from the National Institute on Alcohol Abuse and Alcoholism (to NN). EA was supported by fellowships from Gobierno de Navarra and Bancaja.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Laboratory Investigation website
This paper examines the role of GCN2, a cytosolic kinase that modulates the cellular response to amino acid starvation, in the hepatic response to injury. (Gcn2-/-) mice show increased susceptibility to liver damage and increased deposition of interstitial collagen type I, which suggests that GCN2 is a key regulator of the fibrogenic response in liver.
Supplementary information
Rights and permissions
About this article
Cite this article
Arriazu, E., Ruiz de Galarreta, M., López-Zabalza, M. et al. GCN2 kinase is a key regulator of fibrogenesis and acute and chronic liver injury induced by carbon tetrachloride in mice. Lab Invest 93, 303–310 (2013). https://doi.org/10.1038/labinvest.2012.173
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.2012.173