- TECHNOLOGY FEATURE
Closing in on a complete human genome

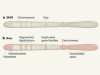
‘Nanopore’ sequencers, such as these GridION units, can decode tens of thousands of bases of DNA without interruption. Credit: Oxford Nanopore Technologies
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 590, 679-681 (2021)
doi: https://doi.org/10.1038/d41586-021-00462-9
References
International Human Genome Sequencing Consortium. Nature 409, 860–921 (2001).
Jain, M. et al. Nature Biotechnol. 36, 338–345 (2018).
Jain, M. et al. Nature Biotechnol. 36, 321–323 (2018).
Miga, K. H. et al. Nature 585, 79–84 (2020).
Logsdon, G. A. et al. Preprint at bioRxiv https://doi.org/10.1101/2020.09.08.285395 (2020).
Wenger, A. M. et al. Nature Biotechnol. 37, 1155–1162 (2019).
Suzuki, Y., Myers, E. W. & Morishita, S. Sci. Adv. 6, eabd9230 (2020).

 A wealth of discovery built on the Human Genome Project — by the numbers
A wealth of discovery built on the Human Genome Project — by the numbers
 The broken promise that undermines human genome research
The broken promise that undermines human genome research
 The next 20 years of human genomics must be more equitable and more open
The next 20 years of human genomics must be more equitable and more open
 Sequence three million genomes across Africa
Sequence three million genomes across Africa
 Breaking through the unknowns of the human reference genome
Breaking through the unknowns of the human reference genome
 How the human genome transformed study of rare diseases
How the human genome transformed study of rare diseases
 From one human genome to a complex tapestry of ancestry
From one human genome to a complex tapestry of ancestry
 Milestones in genomic sequencing
Milestones in genomic sequencing
 An ace in the hole for DNA sequencing
An ace in the hole for DNA sequencing
 NatureTech hub
NatureTech hub






