- NEWS FEATURE
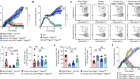
Rogue antibodies could be driving severe COVID-19
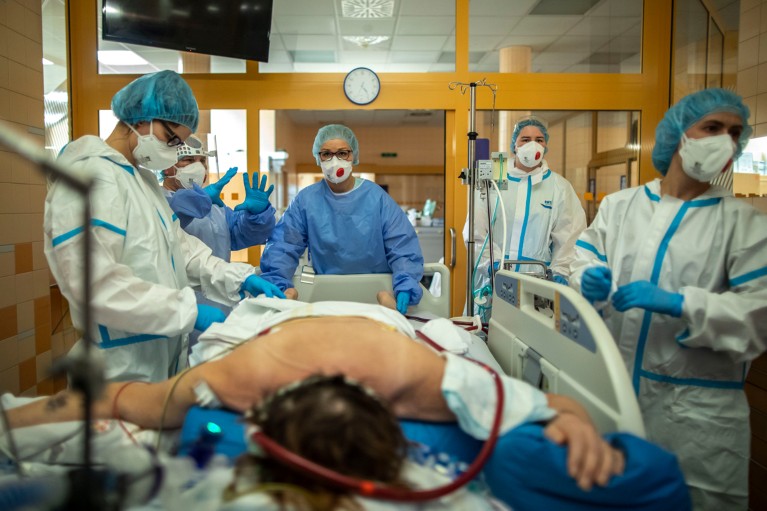
Physicians treat a person with COVID-19 at a hospital’s intensive-care unit in the Czech Republic. Credit: Gabriel Kuchta/Getty
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Rent or buy this article
Prices vary by article type
from$1.95
to$39.95
Prices may be subject to local taxes which are calculated during checkout
Nature 590, 29-31 (2021)
doi: https://doi.org/10.1038/d41586-021-00149-1
References
Bastard, P. et al. Science 370, eabd4585 (2020).
Pozetto, B., Mogensen, K. E., Tovey, M. G. & Gresser, I. J. Infect. Dis. 150, 707–713 (1984).
Wang, E. Y. et al. Preprint at medRxiv https://doi.org/10.1101/2020.12.10.20247205 (2020).
Zuo, Y. et al. Sci. Transl. Med. 12, eabd3876 (2020).
Zuniga, M. et al. Preprint at medRxiv https://doi.org/10.1101/2020.12.28.20248807 (2021).
Ehrenfeld, M. et al. Autoimmun. Rev. 19, 102597 (2020).
Zulfiqar, A.-A., Lorenzo-Villalba, N., Hassler, P. & Andrès, E. N. Engl. J. Med. 382, e43 (2020).
Bhattacharjee, S. & Banerjee, M. SN Compr. Clin. Med. https://doi.org/10.1007/s42399-020-00521-8 (2020).
Monk, P. D. et al. Lancet Respir. Med. https://doi.org/10.1016/S2213-2600(20)30511-7 (2020).
The REMAP-CAP Investigators et al. Preprint at medRxiv https://doi.org/10.1101/2021.01.07.21249390 (2021).

 The lasting misery of coronavirus long-haulers
The lasting misery of coronavirus long-haulers
 Traitorous COVID antibodies and fast-spreading variant
Traitorous COVID antibodies and fast-spreading variant
 Why do COVID death rates seem to be falling?
Why do COVID death rates seem to be falling?
 The latest COVID research updates
The latest COVID research updates