Abstract
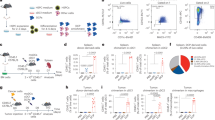
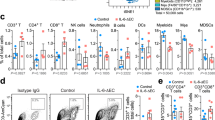
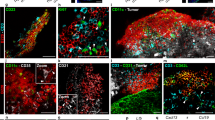
Myeloid-derived suppressor cells (MDSCs) accumulate in the glioma microenvironment during tumor progression and promote immunosuppression. Interleukin-12 (IL-12) immunogene therapy can alter MDSCs toward an antigen-presenting cell phenotype and these mature cells can have a central role in antigen presentation. It remains unclear, however, how MDSC depletion can affect glioma immunotherapy. In this study, we generated a replication-deficient adenoviral vector, Ad.5/3.cRGD-mIL12p70, that transduces the GL261-based murine glioma cell line, resulting in the induction of biologically active, murine IL12p70 expression. Ex vivo, IL-12 expressed by GL261 cells induced interferon-γ synthesis in CD8+ T cells (P<0.001), CD4+ T cells (P=0.009) and natural killer cells (P=0.036). When injected 1 week after tumor implantation, Ad.5/3.cRGD-mIL12p70 successfully prolonged the survival of glioma-bearing mice. Sixty percent of animals treated with IL-12 immunotherapy were long-term survivors over 175 days, whereas all the control group animals expired by 40 days after tumor implantation (P=0.026). Mice receiving Ad.5/3.cRGD-mIL12p70 also accumulated 50% less MDSCs in the brain than the control group (P=0.007). Moreover, in the IL-12 group, MDSCs significantly overexpressed CD80 and major histocompatibility complex class II molecules (P=0.041). Depletion of MDSCs with Gr1+ antibody had no survival benefit induced by IL-12-mediated immunotherapy. Of note, IL-12 therapy increased the presence of myeloid dendritic cells (mDCs) in the glioma microenvironment (P=0.0069). Ultimately, the data show that in the context of IL-12 immunogene therapy, MDSCs are dispensable and mDCs may provide the majority of antigen presentation in the brain.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Malmstrom A, Gronberg BH, Marosi C, Stupp R, Frappaz D, Schultz H et al. Temozolomide versus standard 6-week radiotherapy versus hypofractionated radiotherapy in patients older than 60 years with glioblastoma: the Nordic randomised, phase 3 trial. Lancet Oncol 2012; 13: 916–926.
Becker KP, Yu J . Status quo—standard-of-care medical and radiation therapy for glioblastoma. Cancer J 2012; 18: 12–19.
Stupp R, Hegi ME, Mason WP, van den Bent MJ, Taphoorn MJ, Janzer RC et al. Effects of radiotherapy with concomitant and adjuvant temozolomide versus radiotherapy alone on survival in glioblastoma in a randomised phase III study: 5-year analysis of the EORTC-NCIC trial. Lancet Oncol 2009; 10: 459–466.
Wainwright DA, Nigam P, Thaci B, Dey M, Lesniak MS . Recent developments on immunotherapy for brain cancer. Expert Opin Emerg Drugs 2012; 17: 181–202.
Sampson JH, Heimberger AB, Archer GE, Aldape KD, Friedman AH, Friedman HS et al. Immunologic escape after prolonged progression-free survival with epidermal growth factor receptor variant III peptide vaccination in patients with newly diagnosed glioblastoma. J Clin Oncol 2010; 28: 4722–4729.
Okada H, Kalinski P, Ueda R, Hoji A, Kohanbash G, Donegan TE et al. Induction of CD8+ T-cell responses against novel glioma-associated antigen peptides and clinical activity by vaccinations with {alpha}-type 1 polarized dendritic cells and polyinosinic–polycytidylic acid stabilized by lysine and carboxymethylcellulose in patients with recurrent malignant glioma. J Clin Oncol 2011; 29: 330–336.
Szatmari T, Lumniczky K, Desaknai S, Trajcevski S, Hidvegi EJ, Hamada H et al. Detailed characterization of the mouse glioma 261 tumor model for experimental glioblastoma therapy. Cancer Sci 2006; 97: 546–553.
Colombo MP, Piconese S . Regulatory-T-cell inhibition versus depletion: the right choice in cancer immunotherapy. Nat Rev 2007; 7: 880–887.
Gabrilovich DI, Nagaraj S . Myeloid-derived suppressor cells as regulators of the immune system. Nat Rev Immunol 2009; 9: 162–174.
El Andaloussi A, Han Y, Lesniak MS . Prolongation of survival following depletion of CD4+CD25+ regulatory T cells in mice with experimental brain tumors. J Neurosurg 2006; 105: 430–437.
Fujita M, Kohanbash G, Fellows-Mayle W, Hamilton RL, Komohara Y, Decker SA et al. COX-2 blockade suppresses gliomagenesis by inhibiting myeloid-derived suppressor cells. Cancer Res 2011; 71: 2664–2674.
Zhu X, Fujita M, Snyder LA, Okada H . Systemic delivery of neutralizing antibody targeting CCL2 for glioma therapy. J Neuro-Oncol 2011; 104: 83–92.
Medina-Echeverz J, Fioravanti J, Zabala M, Ardaiz N, Prieto J, Berraondo P . Successful colon cancer eradication after chemoimmunotherapy is associated with profound phenotypic change of intratumoral myeloid cells. J Immunol 2011; 186: 807–815.
Kerkar SP, Goldszmid RS, Muranski P, Chinnasamy D, Yu Z, Reger RN et al. IL-12 triggers a programmatic change in dysfunctional myeloid-derived cells within mouse tumors. J Clin Invest 2011; 121: 4746–4757.
Robertson MJ, Ritz J . Interleukin 12: basic biology and potential applications in cancer treatment. Oncologist 1996; 1: 88–97.
Xu M, Mizoguchi I, Morishima N, Chiba Y, Mizuguchi J, Yoshimoto T . Regulation of antitumor immune responses by the IL-12 family cytokines, IL-12, IL-23, and IL-27. Clin Dev Immunol 2010; 2010 pii 832454.
Soleimani M, Nadri S . A protocol for isolation and culture of mesenchymal stem cells from mouse bone marrow. Nat Protocols 2009; 4: 102–106.
Tyler MA, Ulasov IV, Borovjagin A, Sonabend AM, Khramtsov A, Han Y et al. Enhanced transduction of malignant glioma with a double targeted Ad5/3-RGD fiber-modified adenovirus. Mol Cancer Therap 2006; 5: 2408–2416.
Daley JM, Thomay AA, Connolly MD, Reichner JS, Albina JE . Use of Ly6G-specific monoclonal antibody to deplete neutrophils in mice. J Leukocyte Biol 2008; 83: 64–70.
Talmadge JE, Hood KC, Zobel LC, Shafer LR, Coles M, Toth B . Chemoprevention by cyclooxygenase-2 inhibition reduces immature myeloid suppressor cell expansion. Int Immunopharmacol 2007; 7: 140–151.
Davis FB, Tang HY, Shih A, Keating T, Lansing L, Hercbergs A et al. Acting via a cell surface receptor, thyroid hormone is a growth factor for glioma cells. Cancer Res 2006; 66: 7270–7275.
Liu Y, Ehtesham M, Samoto K, Wheeler CJ, Thompson RC, Villarreal LP et al. In situ adenoviral interleukin 12 gene transfer confers potent and long-lasting cytotoxic immunity in glioma. Cancer Gene Ther 2002; 9: 9–15.
Ehtesham M, Kabos P, Kabosova A, Neuman T, Black KL, Yu JS . The use of interleukin 12-secreting neural stem cells for the treatment of intracranial glioma. Cancer Res 2002; 62: 5657–5663.
Ryu CH, Park SH, Park SA, Kim SM, Lim JY, Jeong CH et al. Gene therapy of intracranial glioma using interleukin 12-secreting human umbilical cord blood-derived mesenchymal stem cells. Hum Gene Ther 2011; 22: 733–743.
Lang FF, Bruner JM, Fuller GN, Aldape K, Prados MD, Chang S et al. Phase I trial of adenovirus-mediated p53 gene therapy for recurrent glioma: biological and clinical results. J Clin Oncol 2003; 21: 2508–2518.
Ali S, King GD, Curtin JF, Candolfi M, Xiong W, Liu C et al. Combined immunostimulation and conditional cytotoxic gene therapy provide long-term survival in a large glioma model. Cancer Res 2005; 65: 7194–7204.
Steding CE, Wu ST, Zhang Y, Jeng MH, Elzey BD, Kao C . The role of interleukin-12 on modulating myeloid-derived suppressor cells, increasing overall survival and reducing metastasis. Immunology 2011; 133: 221–238.
Kerkar SP, Restifo NP . Cellular constituents of immune escape within the tumor microenvironment. Cancer Res 2012; 72: 3125–3130.
Hussain SF, Yang D, Suki D, Aldape K, Grimm E, Heimberger AB . The role of human glioma-infiltrating microglia/macrophages in mediating antitumor immune responses. Neuro-oncology 2006; 8: 261–279.
Chiu TL, Wang MJ, Su CC . The treatment of glioblastoma multiforme through activation of microglia and TRAIL induced by rAAV2-mediated IL-12 in a syngeneic rat model. J Biomed Sci 2012; 19: 45.
Srivastava MK, Zhu L, Harris-White M, Kar UK, Huang M, Johnson MF et al. Myeloid suppressor cell depletion augments antitumor activity in lung cancer. PLoS One 7: e40677.
Acknowledgements
This work was supported by the NCI (R01CA122930, R01CA138587) and the National Institute of Neurological Disorders and Stroke (U01NS069997, F32NS073366, K99NS082381).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Cancer Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Thaci, B., Ahmed, A., Ulasov, I. et al. Depletion of myeloid-derived suppressor cells during interleukin-12 immunogene therapy does not confer a survival advantage in experimental malignant glioma. Cancer Gene Ther 21, 38–44 (2014). https://doi.org/10.1038/cgt.2013.81
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2013.81
Keywords
This article is cited by
-
Phenotypic plasticity of myeloid cells in glioblastoma development, progression, and therapeutics
Oncogene (2021)
-
Intratumoral IL-12 delivery empowers CAR-T cell immunotherapy in a pre-clinical model of glioblastoma
Nature Communications (2021)
-
In vitro exploration of a myeloid-derived suppressor cell line as vehicle for cancer gene therapy
Cancer Gene Therapy (2017)
-
Myeloid cells expressing high level of CD45 are associated with a distinct activated phenotype in glioma
Immunologic Research (2017)
-
Detection of inflammatory cell function using 13C magnetic resonance spectroscopy of hyperpolarized [6-13C]-arginine
Scientific Reports (2016)