Abstract
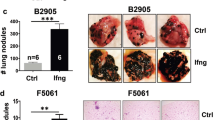
Approximately 90% of melanomas retain wild-type p53, a characteristic that may help shape the development of novel treatment strategies. Here, we employed an adenoviral vector where transgene expression is controlled by p53 to deliver the p19 alternate reading frame (Arf) and interferon-β (IFNβ) complementary DNAs in the B16 mouse model of melanoma. In vitro, cell death was enhanced by combined gene transfer (63.82±15.30% sub-G0 cells); yet introduction of a single gene resulted in significantly fewer hypoploid cells (37.73±7.3% or 36.96±11.58%, p19Arf or IFNβ, respectively, P<0.05). Annexin V staining and caspase-3 cleavage indicate a cell death mechanism consistent with apoptosis. Using reverse transcriptase quantitative PCR, we show that key transcriptional targets of p53 were upregulated in the presence of p19Arf, although treatment with IFNβ did not alter expression of the genes studied. In situ gene therapy revealed significant inhibition of subcutaneous tumors by IFNβ (571±25 mm3) or the combination of p19Arf and IFNβ (489±124 mm3) as compared with the LacZ control (1875±33 mm3, P<0.001), whereas p19Arf yielded an intermediate result (1053±169 mm3, P<0.01 vs control). However, only the combination was associated with increased cell death and prolonged survival (P<0.01). As shown here, the combined transfer of p19Arf and IFNβ using p53-responsive vectors enhanced cell death both in vitro and in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Ravnan MC, Matalka MS . Vemurafenib in patients with BRAF V600E mutation-positive advanced melanoma. Clin Ther 2012; 34: 1474–1486.
Garbe C, Eigentler TK, Keilholz U, Hauschild A, Kirkwood JM . Systematic review of medical treatment in melanoma: current status and future prospects. Oncologist 2011; 16: 5–24.
Giglia-Mari G, Sarasin A . TP53 mutations in human skin cancers. Hum Mutat 2003; 21: 217–228.
Freedberg DE, Rigas SH, Russak J, Gai W, Kaplow M, Osman I et al. Frequent p16-independent inactivation of p14ARF in human melanoma. J Natl Cancer Inst 2008; 100: 784–795.
Bishop DT, Demenais F, Goldstein AM, Bergman W, Bishop JN, Bressac-de Paillerets B et al. Geographical variation in the penetrance of CDKN2A mutations for melanoma. J Natl Cancer Inst 2002; 94: 894–903.
Amos CI, Wang LE, Lee JE, Gershenwald JE, Chen WV, Fang S et al. Genome-wide association study identifies novel loci predisposing to cutaneous melanoma. Hum Mol Genet 2011; 20: 5012–5023.
Tao W, Levine AJ . P19(ARF) stabilizes p53 by blocking nucleo-cytoplasmic shuttling of Mdm2. Proc Natl Acad Sci USA 1999; 96: 6937–6941.
Tao W, Levine AJ . Nucleocytoplasmic shuttling of oncoprotein Hdm2 is required for Hdm2-mediated degradation of p53. Proc Natl Acad Sci USA 1999; 96: 3077–3080.
Pomerantz J, Schreiber-Agus N, Liegeois NJ, Silverman A, Alland L, Chin L et al. The Ink4a tumor suppressor gene product, p19Arf, interacts with MDM2 and neutralizes MDM2's inhibition of p53. Cell 1998; 92: 713–723.
Zhang Y, Xiong Y, Yarbrough WG . ARF promotes MDM2 degradation and stabilizes p53: ARF-INK4a locus deletion impairs both the Rb and p53 tumor suppression pathways. Cell 1998; 92: 725–734.
Sherr CJ . Divorcing ARF and p53: an unsettled case. Nat Rev Cancer 2006; 6: 663–673.
Merkel CA, da Silva Soares RB, de Carvalho AC, Zanatta DB, Bajgelman MC, Fratini P et al. Activation of endogenous p53 by combined p19Arf gene transfer and nutlin-3 drug treatment modalities in the murine cell lines B16 and C6. BMC Cancer 2010; 10: 316.
Qin XQ, Runkel L, Deck C, DeDios C, Barsoum J . Interferon-beta induces S phase accumulation selectively in human transformed cells. J Interferon Cytokine Res 1997; 17: 355–367.
Ekmekcioglu S, Mumm JB, Udtha M, Chada S, Grimm EA . Killing of human melanoma cells induced by activation of class I interferon-regulated signaling pathways via MDA-7/IL-24. Cytokine 2008; 43: 34–44.
Lokshin A, Mayotte JE, Levitt ML . Mechanism of interferon beta-induced squamous differentiation and programmed cell death in human non-small-cell lung cancer cell lines. J Natl Cancer Inst 1995; 87: 206–212.
Biron CA, Nguyen KB, Pien GC, Cousens LP, Salazar-Mather TP . Natural killer cells in antiviral defense: function and regulation by innate cytokines. Annu Rev Immunol 1999; 17: 189–220.
Xie K, Bielenberg D, Huang S, Xu L, Salas T, Juang SH et al. Abrogation of tumorigenicity and metastasis of murine and human tumor cells by transfection with the murine IFN-beta gene: possible role of nitric oxide. Clin Cancer Res 1997; 3 (12 Pt 1): 2283–2294.
Spear GT, Paulnock DM, Jordan RL, Meltzer DM, Merritt JA, Borden EC . Enhancement of monocyte class I and II histocompatibility antigen expression in man by in vivo beta-interferon. Clin Exp Immunol 1987; 69: 107–115.
Fujimura T, Yamasaki K, Hidaka T, Ito Y, Aiba S . A synthetic NOD2 agonist, muramyl dipeptide (MDP)-Lys (L18) and IFN-beta synergistically induce dendritic cell maturation with augmented IL-12 production and suppress melanoma growth. J Dermatol Sci 2011; 62: 107–115.
Sato M, Taniguchi T, Tanaka N . The interferon system and interferon regulatory factor transcription factors -- studies from gene knockout mice. Cytokine Growth Factor Rev 2001; 12: 133–142.
Biron CA . Interferons alpha and beta as immune regulators--a new look. Immunity 2001; 14: 661–664.
Clemens MJ . Interferons and apoptosis. J Interferon Cytokine Res 2003; 23: 277–292.
Sarna GP, Figlin RA, Pertcheck M . Phase II study of betaseron (beta ser17-interferon) as treatment of advanced malignant melanoma. J Biol Response Mod 1987; 6: 375–378.
Kubo H, Ashida A, Matsumoto K, Kageshita T, Yamamoto A, Saida T . Interferon-beta therapy for malignant melanoma: the dose is crucial for inhibition of proliferation and induction of apoptosis of melanoma cells. Arch Dermatol Res 2008; 300: 297–301.
Salmon P, Le Cotonnec JY, Galazka A, Abdul-Ahad A, Darragh A . Pharmacokinetics and pharmacodynamics of recombinant human interferon-beta in healthy male volunteers. J Interferon Cytokine Res 1996; 16: 759–764.
Schiller JH, Storer B, Bittner G, Willson JK, Borden EC . Phase II trial of a combination of interferon-beta ser and interferon-gamma in patients with advanced malignant melanoma. J Interferon Res 1988; 8: 581–589.
Einhorn S, Grander D . Why do so many cancer patients fail to respond to interferon therapy? J Interferon Cytokine Res 1996; 16: 275–281.
Yoshida J, Mizuno M, Fujii M, Kajita Y, Nakahara N, Hatano M et al. Human gene therapy for malignant gliomas (glioblastoma multiforme and anaplastic astrocytoma) by in vivo transduction with human interferon beta gene using cationic liposomes. Hum Gene Ther 2004; 15: 77–86.
Yoshida J, Mizuno M, Wakabayashi T . Interferon-beta gene therapy for cancer: basic research to clinical application. Cancer Sci 2004; 95: 858–865.
Wakabayashi T, Natsume A, Hashizume Y, Fujii M, Mizuno M, Yoshida J . A phase I clinical trial of interferon-beta gene therapy for high-grade glioma: novel findings from gene expression profiling and autopsy. J Gene Med 2008; 10: 329–339.
Lu W, Fidler IJ, Dong Z . Eradication of primary murine fibrosarcomas and induction of systemic immunity by adenovirus-mediated interferon beta gene therapy. Cancer Res 1999; 59: 5202–5208.
Alizadeh H, Howard K, Mellon J, Mayhew E, Rusciano D, Niederkorn JY . Reduction of liver metastasis of intraocular melanoma by interferon-beta gene transfer. Invest Ophthalmol Vis Sci 2003; 44: 3042–3051.
Dickson PV, Hamner JB, Streck CJ, Ng CY, McCarville MB, Calabrese C et al. Continuous delivery of IFN-beta promotes sustained maturation of intratumoral vasculature. Mol Cancer Res 2007; 5: 531–542.
Xiao HB, Zhou WY, Chen XF, Mei J, Lv ZW, Ding FB et al. Interferon-beta efficiently inhibited endothelial progenitor cell-induced tumor angiogenesis. Gene Ther 2012; 19: 1030–1034.
Takaoka A, Hayakawa S, Yanai H, Stoiber D, Negishi H, Kikuchi H et al. Integration of interferon-alpha/beta signalling to p53 responses in tumour suppression and antiviral defence. Nature 2003; 424: 516–523.
Sandoval R, Xue J, Pilkinton M, Salvi D, Kiyokawa H, Colamonici OR . Different requirements for the cytostatic and apoptotic effects of type I interferons. Induction of apoptosis requires ARF but not p53 in osteosarcoma cell lines. J Biol Chem 2004; 279: 32275–32280.
Garcia MA, Collado M, Munoz-Fontela C, Matheu A, Marcos-Villar L, Arroyo J et al. Antiviral action of the tumor suppressor ARF. EMBO J 2006; 25: 4284–4292.
Zhang F, Sriram S . Identification and characterization of the interferon-beta-mediated p53 signal pathway in human peripheral blood mononuclear cells. Immunology 2009; 128 (1 Suppl): e905–e918.
Bajgelman MC, Strauss BE . Development of an adenoviral vector with robust expression driven by p53. Virology 2008; 371: 8–13.
Strauss BE, Bajgelman MC, Costanzi-Strauss E . A novel gene transfer strategy that combines promoter and transgene activities for improved tumor cell inhibition. Cancer Gene Ther 2005; 12: 935–946.
Strauss BE, Costanzi-Strauss E . pCLPG: a p53-driven retroviral system. Virology 2004; 321: 165–172.
Leon RP, Hedlund T, Meech SJ, Li S, Schaack J, Hunger SP et al. Adenoviral-mediated gene transfer in lymphocytes. Proc Natl Acad Sci USA 1998; 95: 13159–13164.
Yamashita M, Ino A, Kawabata K, Sakurai F, Mizuguchi H . Expression of coxsackie and adenovirus receptor reduces the lung metastatic potential of murine tumor cells. Int J Cancer 2007; 121: 1690–1696.
Mizuguchi H, Hayakawa T . Adenovirus vectors containing chimeric type 5 and type 35 fiber proteins exhibit altered and expanded tropism and increase the size limit of foreign genes. Gene 2002; 285: 69–77.
Ghattas IR, Sanes JR, Majors JE . The encephalomyocarditis virus internal ribosome entry site allows efficient coexpression of two genes from a recombinant provirus in cultured cells and in embryos. Mol Cell Biol 1991; 11: 5848–5859.
Zhang F, Lu W, Dong Z . Tumor-infiltrating macrophages are involved in suppressing growth and metastasis of human prostate cancer cells by INF-beta gene therapy in nude mice. Clin Cancer Res 2002; 8: 2942–2951.
Diamond MS, Kinder M, Matsushita H, Mashayekhi M, Dunn GP, Archambault JM et al. Type I interferon is selectively required by dendritic cells for immune rejection of tumors. J Exp Med 2011; 208: 1989–2003.
Wilderman MJ, Sun J, Jassar AS, Kapoor V, Khan M, Vachani A et al. Intrapulmonary IFN-beta gene therapy using an adenoviral vector is highly effective in a murine orthotopic model of bronchogenic adenocarcinoma of the lung. Cancer Res 2005; 65: 8379–8387.
Roos WP, Jost E, Belohlavek C, Nagel G, Fritz G, Kaina B . Intrinsic anticancer drug resistance of malignant melanoma cells is abrogated by IFN-beta and valproic acid. Cancer Res 2011; 71: 4150–4160.
Ambjorn M, Ejlerskov P, Liu Y, Lees M, Jaattela M, Issazadeh-Navikas S . IFNB1/interferon-beta-induced autophagy in MCF-7 breast cancer cells counteracts its proapoptotic function. Autophagy 2012; 9: 3.
Acknowledgements
We thank José Eduardo Krieger and lab for their support and helpful discussions, the lab of Roger Chammas for helpful discussion and for providing use of the FACS, Ana Lucia Garipo for assistance with fluorescence microscopy, Marcio Chaves for facilitating the histological analyses and Paulo Roberto Del Valle for assistance with the RT-qPCR experiment. Funding was provided by FAPESP (Fundação de Amparo à Pesquisa do Estado de São Paulo) in the form of fellowships (06/57823-5, CAM; 2010/03958-2, RFVM) and grants (07/50210-0, 11/50911-0, BES).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Cancer Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Merkel, C., Medrano, R., Barauna, V. et al. Combined p19Arf and interferon-beta gene transfer enhances cell death of B16 melanoma in vitro and in vivo. Cancer Gene Ther 20, 317–325 (2013). https://doi.org/10.1038/cgt.2013.23
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2013.23
Keywords
This article is cited by
-
Potentiation of combined p19Arf and interferon-beta cancer gene therapy through its association with doxorubicin chemotherapy
Scientific Reports (2022)
-
Combination of cabazitaxel and p53 gene therapy abolishes prostate carcinoma tumor growth
Gene Therapy (2020)
-
Response of human melanoma cell lines to interferon-beta gene transfer mediated by a modified adenoviral vector
Scientific Reports (2020)
-
Reestablishment of p53/Arf and interferon-β pathways mediated by a novel adenoviral vector potentiates antiviral response and immunogenic cell death
Cell Death Discovery (2017)
-
Harnessing combined p19Arf and interferon-beta gene transfer as an inducer of immunogenic cell death and mediator of cancer immunotherapy
Cell Death & Disease (2017)