Abstract
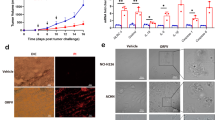
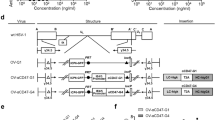
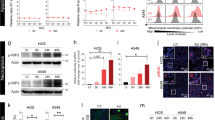
Rodent autonomous parvoviruses (PVs) are endowed with oncotropic properties and represent virotherapeutics with inherent oncolytic features. This work aimed to evaluate the capacity of Minute Virus of Mice (MVMp) to act as an adjuvant stimulating a mouse glioblastoma-specific immune response. MVMp was shown to induce cell death through apoptosis in glioma GL261 cells. Antigen-presenting cells (APCs) provide the initial cue for innate and adaptive immune responses, and thus MVMp-infected GL261 cells were tested for their ability to activate dendritic cells (DCs) and microglia (MG), two distinct cell types that are able to act as APCs. MG and discrete DC subsets were activated after co-culture with MVMp-infected glioma GL261 cells, as evidenced by upregulation of specific activation markers (CD80, CD86) and release of proinflammatory cytokines (tumor necrosis factor-α and interleukin-6). The in vivo analysis of immunodeficient and immunocompetent mice revealed a clear difference in their susceptibility to MVMp-mediated tumor suppression. Immunocompetent mice were fully protected from tumor outgrowth of GL261 cells infected ex vivo with MVMp. In contrast, immunodeficient animals were less competent for MVMp-dependent tumor inhibition, with only 20% of the recipients being protected, arguing for an additional immune component to allow full tumor suppression. In keeping with this conclusion, immunocompetent mice engrafted with MVMp-infected glioma cells developed a level of anti-tumor immunity with isolated splenocytes producing elevated levels of interferon-γ. In rechallenge experiments using uninfected GL261 cells, we could show complete protection against the tumor, arguing for the induction of a T-cell-mediated, tumor-specific, long-term memory response. These findings indicate that the anticancer effect of PVs can be traced back not only for their direct oncolytic effect, but also to their ability to break tumor tolerance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout



Similar content being viewed by others
References
Stupp R, Mason WP, van den Bent MJ, Weller M, Fisher B, Taphoorn MJ et al. Radiotherapy plus concomitant and adjuvant temozolomide for glioblastoma. N Engl J Med 2005; 352: 987–996.
Stummer W, Pichlmeier U, Meinel T, Wiestler OD, Zanella F, Reulen HJ . Fluorescence-guided surgery with 5-aminolevulinic acid for resection of malignant glioma: a randomised controlled multicentre phase III trial. Lancet Oncol 2006; 7: 392–401.
Rommelaere J, Geletneky K, Angelova AL, Daeffler L, Dinsart C, Kiprianova I et al. Oncolytic parvoviruses as cancer therapeutics. Cytokine Growth Factor Rev 2010; 21: 185–195.
Rommelaere J, Cornelis JJ . Antineoplastic activity of parvoviruses. J Virol Methods 1991; 33: 233–251.
Di Piazza M, Mader C, Geletneky K, Herrero YCM, Weber E, Schlehofer J et al. Cytosolic activation of cathepsins mediates parvovirus H-1-induced killing of cisplatin and TRAIL-resistant glioma cells. J Virol 2007; 81: 4186–4198.
Geletneky K, Kiprianova I, Ayache A, Koch R, Herrero YCM, Deleu L et al. Regression of advanced rat and human gliomas by local or systemic treatment with oncolytic parvovirus H-1 in rat models. Neuro Oncol 2010; 12: 804–814.
Moehler MH, Zeidler M, Wilsberg V, Cornelis JJ, Woelfel T, Rommelaere J et al. Parvovirus H-1-induced tumor cell death enhances human immune response in vitro via increased phagocytosis, maturation, and cross-presentation by dendritic cells. Hum Gene Ther 2005; 16: 996–1005.
Raykov Z, Grekova S, Galabov AS, Balboni G, Koch U, Aprahamian M et al. Combined oncolytic and vaccination activities of parvovirus H-1 in a metastatic tumor model. Oncol Rep 2007; 17: 1493–1499.
Giese NA, Raykov Z, DeMartino L, Vecchi A, Sozzani S, Dinsart C et al. Suppression of metastatic hemangiosarcoma by a parvovirus MVMp vector transducing the IP-10 chemokine into immunocompetent mice. Cancer Gene Ther 2002; 9: 432–442.
Szatmari T, Lumniczky K, Desaknai S, Trajcevski S, Hidvegi EJ, Hamada H et al. Detailed characterization of the mouse glioma 261 tumor model for experimental glioblastoma therapy. Cancer Sci 2006; 97: 546–553.
Dranoff G, Jaffee E, Lazenby A, Golumbek P, Levitsky H, Brose K et al. Vaccination with irradiated tumor cells engineered to secrete murine granulocyte-macrophage colony-stimulating factor stimulates potent, specific, and long-lasting anti-tumor immunity. Proc Natl Acad Sci USA 1993; 90: 3539–3543.
Mach N, Gillessen S, Wilson SB, Sheehan C, Mihm M, Dranoff G . Differences in dendritic cells stimulated in vivo by tumors engineered to secrete granulocyte-macrophage colony-stimulating factor or Flt3-ligand. Cancer Res 2000; 60: 3239–3246.
Burudi EM, Riese S, Stahl PD, Regnier-Vigouroux A . Identification and functional characterization of the mannose receptor in astrocytes. Glia 1999; 25: 44–55.
Zolotukhin S, Byrne BJ, Mason E, Zolotukhin I, Potter M, Chesnut K et al. Recombinant adeno-associated virus purification using novel methods improves infectious titer and yield. Gene Therapy 1999; 6: 973–985.
Feinberg AP, Vogelstein B . A technique for radiolabeling DNA restriction endonuclease fragments to high specific activity. Anal Biochem 1983; 132: 6–13.
Cole GA . Interferon-gamma ELISPOT assay for the quantitative measurement of antigen-specific murine CD8+ T-cells. Methods Mol Biol 2005; 302: 191–204.
Ran Z, Rayet B, Rommelaere J, Faisst S . Parvovirus H-1-induced cell death: influence of intracellular NAD consumption on the regulation of necrosis and apoptosis. Virus Res 1999; 65: 161–174.
Abschuetz A, Kehl T, Geibig R, Leuchs B, Rommelaere J, Regnier-Vigouroux A . Oncolytic murine autonomous parvovirus, a candidate vector for glioma gene therapy, is innocuous to normal and immunocompetent mouse glial cells. Cell Tissue Res 2006; 325: 423–436.
Tesniere A, Apetoh L, Ghiringhelli F, Joza N, Panaretakis T, Kepp O et al. Immunogenic cancer cell death: a key-lock paradigm. Curr Opin Immunol 2008; 20: 504–511.
Steinman RM, Banchereau J . Taking dendritic cells into medicine. Nature 2007; 449: 419–426.
Nestle FO, Banchereau J, Hart D . Dendritic cells: on the move from bench to bedside. Nat Med 2001; 7: 761–765.
Mellman I, Steinman RM . Dendritic cells: specialized and regulated antigen processing machines. Cell 2001; 106: 255–258.
Kaur G, Han SJ, Yang I, Crane C . Microglia and central nervous system immunity. Neurosurg Clin N Am 2010; 21: 43–51.
Lumniczky K, Desaknai S, Mangel L, Szende B, Hamada H, Hidvegi EJ et al. Local tumor irradiation augments the antitumor effect of cytokine-producing autologous cancer cell vaccines in a murine glioma model. Cancer Gene Ther 2002; 9: 44–52.
Acknowledgements
We are grateful to B Leuchs and to Dr N Salome for providing virus stock and the 3D9 monoclonal antibody, respectively. SG was supported by a grant from the German Cancer Aid (Deutsche Krebshilfe, Grant No. 10-2183-Mö-1 (Koch/Möhler)).
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on Cancer Gene Therapy website
Supplementary information
Rights and permissions
About this article
Cite this article
Grekova, S., Raykov, Z., Zawatzky, R. et al. Activation of a glioma-specific immune response by oncolytic parvovirus Minute Virus of Mice infection. Cancer Gene Ther 19, 468–475 (2012). https://doi.org/10.1038/cgt.2012.20
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2012.20