Abstract
Lichen sclerosus, high-grade usual vulvar intraepithelial neoplasia (VIN) and differentiated VIN have a different malignant potential. The objective of this study was to quantify the proliferative activity in the basal region of the epithelium of vulvar premalignancies. Furthermore, we investigated whether MIB1 expression in the basal region of vulvar epithelium can be helpful in diagnosing differentiated VIN, which may be hard to discern from normal epithelium. MIB1 was used to immunohistochemically visualise proliferating cells within formalin-fixed, paraffin-embedded, archival tissue sections of different vulvar premalignancies (N=48) and normal vulvar epithelium (N=16). Automatic digital image analysis software was developed to quantify the proliferating fraction in different parts of the epithelium (MIB1 positivity index). MIB1 expression differed among the various vulvar premalignancies; a MIB1-negative basal cell layer was a distinct feature of normal vulvar epithelium. No MIB1-negative basal cell layer was noted in differentiated VIN or other vulvar premalignancies. Owing to this negative cell layer, the MIB1 proliferation index in normal vulvar epithelium was significantly lower than in vulvar premalignancies. In conclusion, MIB1 expression can be a helpful tool in diagnosing a premalignancy and has additional value especially to distinguish differentiated VIN neoplasia from normal vulvar epithelium, but cannot explain the differences in malignant potential.
Similar content being viewed by others
Main
Vulvar cancer is the fourth most common gynaecologic cancer and comprises 5% of all malignancies of the female genital tract. On the basis of clinical and pathological features, vulvar squamous-cell carcinoma can be subdivided into two different types, which seem to develop from their own associated premalignancies.
The most frequent type of vulvar carcinoma occurs mainly as a unifocal lesion in elderly women and is related to lichen sclerosus and/or differentiated vulvar intraepithelial neoplasia (VIN). This type of tumour is probably not associated with human papillomavirus (HPV) infection.1, 2, 3, 4 The less common type, accounting for about one-third of all vulvar squamous-cell carcinomas, is associated with HPV, predominantly HPV types 16 and 18. This type of tumour often occurs as a multifocal lesion in relatively young women and is usually preceded by high-grade usual VIN lesions (also referred to as classic VIN lesions).3, 5, 6 Recently, the International Society for the Study of Vulvovaginal Disease (ISSVD) has proposed a new nomenclature for premalignant vulvar lesions (see Table 1).7
In usual VIN, the epithelium is thickened and accompanied by hyperkeratosis and/or parakeratosis. Although a spectrum of architectural abnormalities may be seen, the lesion is readily recognisable as an intraepithelial neoplasm by the pathologist due to cytological abnormalities of the epithelial cells throughout the whole thickness of the epithelium.8 Differentiated VIN is clinically and pathologically more difficult to recognise. There is little or no atypia above the basal or parabasal layers and it has a high degree of cellular differentiation, which combined with an absence of widespread architectural disarray hinders recognition.2, 3, 9 Differentiated VIN is relatively infrequently diagnosed in its pure form and is often seen adjacent to lichen sclerosus and/or rapidly growing invasive vulvar squamous-cell carcinoma.3, 8, 10 Besides the difficulties in clinical and histological recognition of differentiated VIN, it is suggested that this form of VIN is highly proliferative and might be more likely to progress to an invasive neoplasm than lichen sclerosus and HPV-related VIN lesions.2, 3, 11 Currently, no biomarker or diagnostic tool to predict possible invasive behaviour of premalignant vulvar lesions is available. Because of the risk of malignant progression it is current practice that all patients with vulvar premalignancies undergo regular checkups. However, there is no evidence that this follow-up prevents the development of cancer or results in earlier detection of a malignancy.12
Proliferative activity in tissues can be visualised using a proliferation marker like MIB1, which is a monoclonal antibody against the Ki-67 antigen, a nuclear antigen present in human proliferating cells in all stages of the cell cycle, but not in the G0 phase.13, 14 In many (pre)malignant lesions, qualitative MIB1 expression is used for grading and estimating prognosis.10, 15, 16, 17, 18 Bulten et al15 used a method in which epithelial MIB1 expression patterns in cervical lesions were digitally quantified. The method proved to be an objective, reproducible and reliable method of classification for dysplastic changes in cervical epithelium. Systematic quantitative evaluation of MIB1 expression in the basal region of premalignant vulvar lesions has not yet been performed.10, 19, 20, 21, 22, 23
The primary objective of this study was to quantify the proliferative activity in the epithelial cell layers in differentiated VIN and other vulvar premalignancies with the aim to find an explanation for the differences in malignant progression. Furthermore, we investigated whether MIB1 expression in the basal regions of the vulvar epithelium may be helpful in diagnosing differentiated VIN.
Materials and methods
Specimens
Sixty-four specimens from vulvar biopsies, vulvar excisions or vulvectomies, diagnosed as non- or premalignant vulvar lesions between 1992 and 2002, were retrospectively obtained from the archives of the Department of Pathology at the Radboud University Nijmegen Medical Centre (Nijmegen, the Netherlands). All specimens were routinely fixed (4% buffered formalin) and paraffin-embedded. Standard 4-μm thick haematoxylin and eosin (H&E)-stained sections were used for the classification of the lesions. Two expert pathologists (JB and JMMG), specialised in gynaecological pathology, blindly and independently reexamined the slides and classified them according to current WHO criteria and the recent modification of the ISSVD.7, 24 They agreed on all diagnoses.
The 64 tissue specimens in this study consisted of 22 usual type VIN lesions, 14 differentiated VIN lesions, 12 lesions with lichen sclerosus and 16 normal epithelium samples (10 patients with vulvodynia who were treated with a partial resection of the vulvar vestibule and six normal epithelium samples from free tumour margins in vulvectomy specimens of patients treated for vulvar cancer).
Immunohistochemistry
Four-micrometer-thick paraffin sections were mounted onto polylysine-coated slides and dried overnight at 58°C. The sections were dewaxed in xylene and endogenous peroxidase was blocked using H2O2 in methanol for 15 min and the slides were rinsed three times in phosphate-buffered saline (PBS; pH 7.4) for 5 min. The slides were placed in a citrate buffer (0.01 M; pH 6.0) and heated in a household microwave oven (3 min at 850 W until boiling; followed by 10 min at 180 W). The sections were allowed to cool down to room temperature (RT) and were briefly washed in PBS (10 min). Subsequently, the slides were preincubated with 20% normal goat serum and incubated with the primary antibody Ki-67 (clone MIB1, Dakocytomation, Denmark) 1:100 in PBS with 1% BSA (60 min, RT). Subsequently, the slides were rinsed in PBS (10 min) and post-antibody blocking was done for 15 min (powervision plus). This was followed by incubation with polymeric horseradish peroxidase goat anti-mouse/rabbit/rat IgG (30 min, RT). The slides were developed with diaminobenzidine (mixed with H2O2), counterstained with Mayer's haematoxylin, dehydrated in ethanol and xylene and finally mounted. In each run, a buffer only and a vulvar squamous-cell carcinoma served as negative and positive controls.
Quantification of Immunohistochemical Staining/Image Analysis
Quantitative analysis of MIB1 staining was achieved using digital image analysis of microscopic images. Image acquisition was performed using a 3CCD colour video camera (Sony DXC-950P, Sony Corp., Japan) mounted on a conventional light microscope (Axioskop 2 plus, Carl Zeiss AG, Germany) and attached to a personal computer with frame grabber card (Matrox Meteor-II Multichannel, Matrox Imaging, Dorval, Canada). Images were acquired using a × 20 objective (Plan Neofluar, NA=0.5, resulting specimen level pixel size 0.39 μm2). Before analysis of the immunohistochemical staining, an image of an empty microscopic field was acquired, which was used for correction of unequal illumination. In each tissue section, we aimed to measure eight microscopic fields, representative of the lesion. Image acquisition and analysis were performed using a custom written macro in KS400 image analysis software (Carl Zeiss AG, Germany). For each digitised red green blue (RGB) image, the following procedure was performed. The operator interactively defined the location of the basement membrane by drawing a line with a mouse cursor. From this line, 20 strata of each 5 μm thickness were automatically determined, covering the basal region of the epithelium. Within these areas, pixels with a ratio between the red and green RGB component of >1.03 and a red intensity under 180 were labelled as belonging to MIB1-positive nuclei. In the same way, pixels with a ratio between blue and green intensity over 1.07 and red camera channel under 210 were labelled as belonging to haematoxylin-stained, MIB1-negative nuclei. See Figure 1a and b for the different steps in the process. Thresholds were determined from a set of training slides and were found adequate for almost all slides analysed in this study. When the initial thresholds led to unrealistic patterns, adjustment was performed by the operator (data not shown).
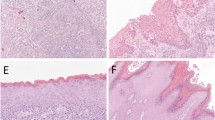
(a and b) Photos of MIB1 staining and the digital image analysis process in normal vulvar epithelium. (a) A red line under the basement membrane, drawn by the operator. (b) An 100 μm wide area covering part of the epithelium was identified. Twenty strata of each 5 μm width, in which MIB1-negative (green) and MIB1-positive (blue) nuclei are shown. Note the negative MIB1 cell layer above the basement membrane. (d–h) H&E and MIB1 staining of 4 μm paraffin sections of a usual VIN lesion, differentiated VIN and lichen sclerosus. The peroxidase-labelled immunohistochemical complexes were visualised with diaminobenzodine (DAB) (brown-black). (c) H&E-stained section of a usual VIN lesion, showing atypical cells throughout the whole thickness of the epithelium. (d) In usual VIN, MIB1-positive cells can be seen in all layers of the epithelium. (e1) An abortive pearl that can be found in differentiated VIN. (e2) The atypia in differentiated VIN is confined to the basal layers of the epithelium. (f) In differentiated VIN, the MIB1-positive cells are confined to the basal layers. (g) In lichen sclerosus little atypia can be seen. (h) In lichen sclerosus, the MIB1-positive cells are confined to the basal layers. Original magnifications: (a and b) × 20; (c, d, e1, f, g, h) × 10; (e2) × 5.
For each measured field, the total area of positive nuclei per stratum of 5 μm and the total area of all nuclei per stratum was automatically recorded. For each stratum, the ratio between the MIB1-positive nuclear area and total nuclear area was calculated (single layer). This ratio, × 100%, was used as a measure of MIB1 positivity: the MIB1 positivity index. In addition, the cumulative MIB1 positivity indices were calculated by dividing the sum of the MIB1-positive nuclear area in multiple strata by the sum of the total nuclear area in the same strata ( × 100%).
In normal vulvar epithelium, the presence or absence of inflammatory cells directly underlying the basement membrane was recorded for each measured field.
Statistical Analysis
All measured fields were averaged to calculate mean values per patient. These values were used to calculate mean values per type of lesion.
All analyses were performed using SPSS 12.0.1 software (SPSS Inc., Chicago, IL, USA). To test whether the positivity index in the lowest 5 μm of normal vulvar epithelium differed between the fields with and without inflammatory cells underneath the basal membrane, the nonparametric Mann–Whitney-U-test for unpaired observations was used. The Kruskal–Wallis one-way analysis of variance by ranks was used to test whether the different positivity indices were different for at least one of the four diagnostic groups. When a significant difference was found, distribution-free all-treatments multiple comparisons based on pairwise rankings with correction for tied observations were used to disclose which of the diagnostic groups differed significantly.25
Results
Pathology
In Figure 1c, e and g, H&E-stained sections of differentiated VIN, usual VIN and lichen sclerosus are shown.
The epithelial cells of usual VIN (Figure 1c) have a high nuclear: cytoplasmic ratio and lack cytoplasmic maturation above the basal and parabasal layers. Mitotic activity is present above the basal layer with mitotic figures that are often abnormal in appearance and reach the upper cell layers. Multinucleation and dyskeratosis, including formation of intraepithelial squamous pearls, may be seen. Nuclear pleomorphism and hyperchromasia are present; however, nucleoli are uncommon.
The atypia in differentiated VIN lesions (Figure 1e) is strictly confined to the basal and parabasal layers of the epithelium, where the cells have abundant cytoplasm and form abortive pearls. The nuclei are relatively uniform in size and contain coarse chromatin and prominent nucleoli leading to paradoxical maturation abutting on the epithelial–stromal junction. The superficial layers of the epithelium have a normal maturation, exhibit hyperkeratosis and do not show koilocytosis.24 Furthermore, in differentiated VIN, the epithelium exhibits elongation of rete pegs.
In lichen sclerosus (Figure 1g), the loss of rete ridges is clearly visible. There is little or no cellular or nuclear atypia. A zone of homogeneous collagenised subepithelial oedema of variable thickness is present as is a band of lymphocytic infiltration beneath this zone. The epithelium is flattened and/or thinned, and mild hyperkeratosis is present.
MIB1 Expression Pattern
In all lesions, MIB1 staining was present in the basal and/or parabasal regions of the epithelium and the basal membrane was always clearly identifiable. As can be seen in Figure 1d, f and g, MIB1 expression varied in the different vulvar premalignancies. In usual VIN all cell layers were positive for MIB1 (see Figure 1d). In differentiated VIN (Figure 1f), the majority of the epithelium was MIB1-negative with a thin layer of MIB1-positive cells parabasally. MIB1 positivity was restricted to the lower one-third of the epithelium in lichen sclerosus (Figure 1h). Furthermore, normal epithelium had a distinct MIB1 expression pattern. Like in lichen sclerosus, the lower one-third of the normal vulvar epithelium was MIB1-positive, but a MIB1-negative cell layer directly above the basement membrane was visible (Figure 1a and b). A simplified schematic representation of the different expression patterns is depicted in Figure 2.
MIB1 Positivity Index
In Figure 3, an overview of the single (Figure 3a) and cumulative (Figure 3b) MIB1 positivity indices in different areas of the epithelium are shown. The thick line represents the median positivity index, boxes represent quartiles and the lines indicate extreme values. The MIB1 positivity indices per patient were based on a median number of 6 (range 4–10) measurements. In normal epithelium, there was no statistically significant difference in MIB1 positivity index in the lowest 5 μm of the epithelium between the measured fields with or without inflammatory cells underneath the basal membrane (data not shown, Mann–Whitney-U-test, P>0.05). The MIB1 positivity indices in normal epithelium from vulvar vestibulitis patients and from patients with a carcinoma did not differ from one other (data not shown, Mann–Whitney-U-test, all P-values>0.05).
In Table 2, an overview of P-values (distribution-free all-treatments multiple comparisons based on pairwise rankings with correction for tied observations) can be found, comparing cumulative and single layer MIB1 positivity indices between the four diagnostic groups.
In normal vulvar epithelium, the lowest 10 μm of the epithelium was almost negative for MIB1. The cumulative MIB1 positivity indices at 5 and 10 μm in normal epithelium were significantly lower than the corresponding areas in any of the other lesion types (all P-values <0.001). In the higher strata, single layer MIB1 positivity indices in normal vulvar epithelium were comparable to the single layer MIB1 positivity indices in differentiated VIN. Owing to the big difference in the lowest two layers, the difference between the cumulative MIB1 positivity indices remained statistically significant. Comparing the MIB1 positivity indices between lichen sclerosus and normal vulvar tissue revealed differences in only the layers up to 15 μm.
In usual VIN lesions, a MIB1 positivity index of almost 40% or more was observed in all layers of the epithelium, causing the cumulative MIB1 positivity indices to be around the same values. In none of the other epithelia, single layer MIB1 positivity indices (at 55 μm or higher) were as high as those in usual VIN (all P-values<0.001). Comparing the cumulative MIB1 positivity indices of usual VIN with the other lesions gave the same results (all P-values <0.05). The differences with normal epithelium were most striking, as all cumulative and single layer MIB1 positivity indices were significantly higher in usual VIN.
The cumulative MIB1 positivity indices up to 10 μm in lichen sclerosus were significantly lower than these strata in differentiated VIN. The single and cumulative MIB1 positivity indices higher in the epithelium were not significantly different and the pattern was comparable (see Figures 2 and 3).
Discussion
In general, it is suggested that differentiated VIN has a higher malignant potential than other vulvar premalignancies.8, 26 This is not explained by our quantitative analysis of proliferative activity. The MIB1 positivity indices in the epithelium of differentiated VIN were not statistically different from the MIB1 positivity indices in well-established other precursors of vulvar carcinoma, that is usual VIN. The proliferative activity in the lower strata was comparable, whereas the MIB1 positivity indices covering a higher part of the epithelium were significantly higher in usual VIN, which is thought to be of lesser malignant potential. Several other investigators calculated a MIB1 positivity or labelling index, measuring the entire thickness of the epithelium or only the basal cell layer, but none of the groups used digital quantitative image analysis and mostly no distinction was made between differentiated and usual VIN, or only usual VIN was analysed.27 Only scoring the MIB1 pattern or using a semiquantitative scoring method is less accurate.28 Owing to deep rete pegs the measurement of the entire epithelium, as previously published by Bulten et al15 for cervical lesions, is technically not possible in differentiated VIN lesions. We calculated the MIB1 positivity index by dividing the measured MIB1-positive nuclear area by the total nuclear area. In this manner, the thickness of the epithelium that can be highly variable among different vulvar premalignancies and among different patients was no confounder in the measurements. When it would be possible to measure the entire epithelium, the differences in cumulative MIB1 positivity indices between differentiated VIN and usual VIN would probably be more prominent.
We noticed a remarkable difference in the MIB1 staining pattern of vulvar premalignancies vs normal vulvar epithelium. In normal vulvar epithelium, a MIB1-negative cell layer above the basal membrane was observed, which was absent in differentiated VIN. There was no MIB1-negative cell layer in other vulvar premalignancies and therefore the presence of a negative MIB1 layer seems to be a characteristic of normal vulvar epithelium and the absence of such a layer above the basal membrane, indicates the presence of a premalignancy. Scurry et al21 describe similar results in their qualitative analysis of Ki-67 of lichen sclerosus and normal epithelium. Although they did not digitally quantify their measurements and did not specifically mention a MIB1-negative basal cell layer, they describe that the MIB1 staining in normal epithelium was localised more parabasally and in lichen sclerosus more basally. The lack of effect of inflammatory cells beneath normal vulvar epithelium on the presence of a MIB1-negative basal cell layer and the absence of a MIB1-negative basal cell layer in vulvar premalignancies can be seen as disturbance of cell growth in vulvar premalignancies.
Lichen sclerosus and usual VIN lesions are readily recognisable as they have distinct characteristics at histopathological examination. Differentiated VIN is often very difficult to recognise and sometimes hard to discern from normal epithelium.2, 3, 9 Using MIB1 expression to detect a proliferating basal cell layer in combination with the characteristic MIB1 staining pattern with only positive cells in the rete pegs and little positivity in the upper layers of the epithelium, might improve the early recognition of differentiated VIN. A simplified schematic representation of the different expression patterns is depicted in Figure 2. Further investigations have to provide evidence whether this feature of MIB1 is applicable in daily practice.
Despite its atrophic clinical aspect, lichen sclerosus has a relatively high proliferative activity, which was significantly higher than in normal vulvar tissue, which might be responsible for its malignant potential. This is in contrast with Tan et al,29 who found a lower MIB1 positivity index in vulvar lichen sclerosus compared to normal controls. However, they did not use digital imaging techniques to quantify MIB1 positivity and only counted the basal and suprabasal cell layers. Lichen sclerosus and differentiated VIN showed comparable patterns of MIB1 positivity indices, both single and cumulative. This attributes to the assumption that lichen sclerosus and differentiated VIN belong to the same entity that might proceed to HPV-negative vulvar carcinoma.1, 2, 3, 30 Differentiated VIN might develop from lichen sclerosus and/or squamous hyperplasia, through loss of apoptosis regulation, like the development of squamous-cell carcinoma in oral lichen planus lesions.31 The role of the regulator of apoptosis, p53, and its mutations, in differentiated VIN and HPV-negative vulvar carcinoma remains to be elucidated.8, 32, 33, 34
In conclusion, the use of MIB1 in the diagnosis of premalignant vulvar lesions might prove helpful, especially to discern differentiated VIN from normal vulvar epithelium. In all vulvar premalignancies, the relationship between basal and parabasal cell layers seems to be disturbed. However, the MIB1 positivity index was not higher in differentiated VIN lesions, which are presumed to be more likely to become invasive, than in usual VIN lesions.
References
Carlson JA, Ambros R, Malfetano J, et al. Vulvar lichen sclerosus and squamous cell carcinoma: a cohort, case control, and investigational study with historical perspective; implications for chronic inflammation and sclerosis in the development of neoplasia. Hum Pathol 1998;29:932–948.
Fox H, Wells M . Recent advances in the pathology of the vulva. Histopathology 2003;42:209–216.
Hart WR . Vulvar intraepithelial neoplasia: historical aspects and current status. Int J Gynecol Pathol 2001;20:16–30.
van der Avoort IAM, Shirango H, Hoevenaars BM, et al. Vulvar squamous cell carcinoma is a multifactorial disease following two separate and independent pathways. Int J Gynecol Pathol 2006;25:22–29.
Hildesheim A, Han CL, Brinton LA, et al. Human papillomavirus type 16 and risk of preinvasive and invasive vulvar cancer: results from a seroepidemiological case–control study. Obstet Gynecol 1997;90:748–754.
Iwasawa A, Nieminen P, Lehtinen M, et al. Human papillomavirus in squamous cell carcinoma of the vulva by polymerase chain reaction. Obstet Gynecol 1997;89:81–84.
Sideri M, Jones RW, Wilkinson EJ, et al. Squamous vulvar intraepithelial neoplasia: 2004 modified terminology, ISSVD Vulvar Oncology Subcommittee. J Reprod Med 2005;50:807–810.
Yang B, Hart WR . Vulvar intraepithelial neoplasia of the simplex (differentiated) type: a clinicopathologic study including analysis of HPV and p53 expression. Am J Surg Pathol 2000;24:429–441.
van Beurden M, ten Kate FJ, Smits HL, et al. Multifocal vulvar intraepithelial neoplasia grade III and multicentric lower genital tract neoplasia is associated with transcriptionally active human papillomavirus. Cancer 1995;75:2879–2884.
Modesitt SC, Groben PA, Walton LA, et al. Expression of Ki-67 in vulvar carcinoma and vulvar intraepithelial neoplasia III: correlation with clinical prognostic factors. Gynecol Oncol 2000;76:51–55.
Hsieh MY, Kuo HW . The simplex (differentiated) variant of vulvar intraepithelial neoplasia. Dermatol Surg 2004;30:948–951.
Maclean AB . Vulval cancer: prevention and screening. Best Pract Res Clin Obstet Gynaecol 2006;20:379–395.
Cattoretti G, Becker MH, Key G, et al. Monoclonal antibodies against recombinant parts of the Ki-67 antigen (MIB 1 and MIB 3) detect proliferating cells in microwave-processed formalin-fixed paraffin sections. J Pathol 1992;168:357–363.
Key G, Becker MH, Baron B, et al. New Ki-67-equivalent murine monoclonal antibodies (MIB 1-3) generated against bacterially expressed parts of the Ki-67 cDNA containing three 62 base pair repetitive elements encoding for the Ki-67 epitope. Lab Invest 1993;68:629–636.
Bulten J, van der Laak JA, Gemmink JH, et al. MIB1, a promising marker for the classification of cervical intraepithelial neoplasia. J Pathol 1996;178:268–273.
Garzetti GG, Ciavattini A, De Nictolis M, et al. MIB 1 immunostaining in cervical intraepithelial neoplasia: prognostic significance in mild and moderate lesions. Gynecol Obstet Invest 1996;42:261–266.
Kruse AJ, Baak JP, de Bruin PC, et al. Ki-67 immunoquantitation in cervical intraepithelial neoplasia (CIN): a sensitive marker for grading. J Pathol 2001;193:48–54.
Salvesen HB, Iversen OE, Akslen LA . Identification of high-risk patients by assessment of nuclear Ki-67 expression in a prospective study of endometrial carcinomas. Clin Cancer Res 1998;4:2779–2785.
Hendricks JB, Wilkinson EJ, Kubilis P, et al. Ki-67 expression in vulvar carcinoma. Int J Gynecol Pathol 1994;13:205–210.
Marchetti M, Salmaso R, Polonio S, et al. Ki-67 expression in vulvar carcinoma. Preliminary results. Eur J Gynaecol Oncol 1996;17:361–364.
Scurry J, Beshay V, Cohen C, et al. Ki67 expression in lichen sclerosus of vulva in patients with and without associated squamous cell carcinoma. Histopathology 1998;32:399–404.
Van Hoeven KH, Kovatich AJ . Immunohistochemical staining for proliferating cell nuclear antigen, BCL2, and Ki-67 in vulvar tissues. Int J Gynecol Pathol 1996;15:10–16.
van Beurden M, de Craen AJ, de Vet HC, et al. The contribution of MIB 1 in the accurate grading of vulvar intraepithelial neoplasia. J Clin Pathol 1999;52:820–824.
Wilkinson EJ . Premalignant and malignant tumors of the vulva. In: Kurman RJ (ed). Blaustein's Pathology of the Female Genital Tract. 5th edn. Springer-Verlag: New York, 2002, pp 99–149.
Hollander M, Wolfe DA . Nonparametric Statistical Methods. 2nd edn. John Wiley & Sons Inc.: New York, 1999.
Preti M, van Seters M, Sideri M, van Beurden M . Squamous vulvar intraepithelial neoplasia. Clin Obstet Gynecol 2005;48:845–861.
Brustmann H, Naude S . Expression of topoisomerase IIalpha, Ki-67, proliferating cell nuclear antigen, p53, and argyrophilic nucleolar organizer regions in vulvar squamous lesions. Gynecol Oncol 2002;86:192–199.
Rolfe KJ, Eva LJ, Maclean AB, et al. Cell cycle proteins as molecular markers of malignant change in vulvar lichen sclerosus. Int J Gynecol Cancer 2001;11:113–118.
Tan SH, Derrick E, McKee PH, et al. Altered p53 expression and epidermal cell proliferation is seen in vulval lichen sclerosus. J Cutan Pathol 1994;21:316–323.
Jones RW, Baranyai J, Stables S . Trends in squamous cell carcinoma of the vulva: the influence of vulvar intraepithelial neoplasia. Obstet Gynecol 1997;90:448–452.
Bascones-Ilundain C, Gonzalez-Moles MA, Esparza-Gomez G, et al. Importance of apoptotic mechanisms in inflammatory infiltrate of oral lichen planus lesions. Anticancer Res 2006;26:357–362.
Pinto AP, Lin MC, Sheets EE, et al. Allelic imbalance in lichen sclerosus, hyperplasia, and intraepithelial neoplasia of the vulva. Gynecol Oncol 2000;77:171–176.
Rosenthal AN, Hopster D, Ryan A, et al. Immunohistochemical analysis of p53 in vulval intraepithelial neoplasia and vulval squamous cell carcinoma. Br J Cancer 2003;88:251–256.
van Seters M, ten Kate FJ, van Beurden M, et al. In the absence of (early) invasive carcinoma vulvar intraepithelial neoplasia associated with lichen sclerosus is mainly of undifferentiated type: new insights in histology and aetiology. J Clin Pathol 2007 (in press).
Acknowledgements
We thank Professor Dr PJ Slootweg, Department of Pathology, Radboud University Nijmegen Medical Centre, for his useful comments and ideas regarding this paper.
Author information
Authors and Affiliations
Corresponding author
Additional information
Disclosure/conflict of interest
The authors state that concerning this manuscript, neither conflict of interest nor funding exists.
Rights and permissions
About this article
Cite this article
van der Avoort, I., van der Laak, J., Paffen, A. et al. MIB1 expression in basal cell layer: a diagnostic tool to identify premalignancies of the vulva. Mod Pathol 20, 770–778 (2007). https://doi.org/10.1038/modpathol.3800796
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/modpathol.3800796
Keywords
This article is cited by
-
Sec62/Ki67 and p16/Ki67 dual-staining immunocytochemistry in vulvar cytology for the identification of vulvar intraepithelial neoplasia and vulvar cancer: a pilot study
Archives of Gynecology and Obstetrics (2019)
-
Präinvasive Veränderungen von Vulva oder Vagina
Der Onkologe (2014)
-
Phosphorylated S6 as an immunohistochemical biomarker of vulvar intraepithelial neoplasia
Modern Pathology (2013)
-
Interobserver variability and the effect of education in the histopathological diagnosis of differentiated vulvar intraepithelial neoplasia
Modern Pathology (2013)
-
Hypermutation in mantle cell lymphoma does not indicate a clinical or biological subentity
Modern Pathology (2009)