Abstract
To evaluate the potential roles of Interleukin-3 (IL-3) and c-Kit, the tyrosine kinase receptor for stem cell factor (SCF), in eosinophil responses in vivo, we examined eosinophil numbers in uninfected or nematode-infected wild-type mice, IL-3−/− mice, and IL-3−/− mice that also have a marked reduction in SCF/c-Kit signaling (ie, KitW/KitW−v, IL-3−/− mice). We found no significant differences in the numbers of eosinophils in the blood, bone marrow or various tissues of IL-3−/− vs IL-3+/+ mice, either at baseline or after the induction of bone marrow, blood or tissue eosinophilia in response to infection with Strongyloides venezuelensis (S.v.) or Nippostrongylus brasiliensis (N.b.). However, in mice with markedly impaired SCF/c-Kit signaling, IL-3 contributed significantly to the increased numbers of eosinophils that were observed in multiple tissues during S.v. infection, but not during infection with N.b.
Similar content being viewed by others
Main
Interleukin-3 (IL-3) can promote the development of various hematopoietic cells in vitro, through effects on both multipotential and more mature, lineage-committed progenitor cells.1 While many studies have shown that IL-3 can enhance the development, survival and functional activation of mouse or human eosinophils in vitro,2, 3, 4, 5, 6 the importance of IL-3 in eosinophil development in vivo has been difficult to assess. In part, this reflects the fact that three cytokines, IL-3, IL-5 and GM-CSF, whose genes are closely linked and which all can be expressed by activated T cells or mast cells, express overlapping functions.7, 8, 9, 10 For example, all three cytokines can promote eosinophil development in vitro.5, 6 Moreover, all three of these cytokines bind to cell surface receptors which are heterodimers consisting of the same ‘common’ β chain (βc);3 this βc forms high affinity receptors for IL-3, IL-5 or GM-CSF when paired with the appropriate specific ligand-binding α chain.11, 12 Mice, but not humans, also express an IL-3-specific β chain (βIL−3).13, 14, 15 The mouse βIL−3 is 91% identical to the mouse βc at the amino-acid level, but it does not form functional high-affinity heterodimers with the mouse IL-5 or GM-CSF-specific α chains.11, 12
Work with mice which lack either of the two β receptor genes7, 8, 9 as well as studies in IL-5 −/− or IL-5R α chain −/− mice16, 17 indicate that IL-3 cannot overcome the marked defect in eosinophil production that is associated with a lack of signaling via the receptor for IL-5. Nevertheless, four considerations prompted us to reassess the potential role of IL-3 in mouse eosinophil responses in vivo. First, because βc −/− mice and βc −/− × IL-3 −/− mice lack IL-5 signaling, and therefore have markedly impaired eosinophil production, such animals do not represent an optimal setting in which to search for potentially important effects of IL-3, for example, enhancement of IL-5-dependent eosinophil development.4, 18 Second, we recently reported that eosinophil recruitment after i.p. allergen challenge in ragweed allergen-immunized mice was greater in IL-3 −/− mice than in IL-3 +/+ mice.19 That IL-3 might, under certain circumstances, suppress eosinophil development and/or migration is supported by studies showing that mice deficient in both IL-3 and GM-CSF, but not single knockouts, exhibit increased numbers of circulating eosinophils.20 Third, we reported that while IL-3 −/− mice were normal in many aspects of hematopoiesis in vitro or in vivo21 as would have been predicted based on the phenotype of βc −/− × IL-3 −/− mice,10 IL-3 −/− mice showed marked impairment of two important features of their Th2-associated hematopoietic response to infection with the nematode Strongyloides venezuelensis (S.v.): bone marrow basophil production and tissue mast cell hyperplasia.22 However, the eosinophil responses of these mice were not assessed. Fourth, stem cell factor (SCF), the ligand for the tyrosine kinase receptor c-Kit, has also been reported to enhance eosinophil development in vitro, but only when used in combination with other cytokines such as IL-3.23, 24 SCF also can stimulate VLA-4-mediated adhesion of human eosinophils to fibronectin and VCAM-1 in vitro.25 SCF is required for physiological mast cell development in mice, as mast cells are essentially absent under baseline conditions in the tissues of KitW/KitW−v mice that have markedly reducing c-Kit signaling.26, 27 However, by using KitW/KitW−v, IL-3 −/− mice, we showed that the modest numbers of mast cells that can appear in the intestines or spleen of KitW/KitW−v mice infected with S.v. are dependent on IL-3.22 This work indicated that KitW/KitW−v, IL-3 −/− mice can be useful for revealing biological responses that may depend on both IL-3 and c-Kit/SCF signaling for their full expression.
We therefore used IL-3 −/− and wild-type mice, as well as KitW/KitW−v, IL-3 −/− mice and the corresponding KitW/KitW−v, IL-3 +/+ mice, to assess the extent to which IL-3 might act alone or in concert with c-Kit/SCF to promote eosinophil production and tissue accumulation at baseline or during infection with nematodes.
Materials and methods
Mice
Unless otherwise specified, IL-3 −/− mice were fourth backcross generation on the C57BL/6 or BALB/c background.21, 22 KitW/KitW−v, IL-3 −/− mice were produced as previously described.22 Briefly, WBB6F1-KitW/+ and KitW−v/+ mice were bred with C57BL/6-IL-3 −/− mice and the appropriate F2 and F3 offspring were identified and bred selectively to generate double-mutant mice; WBB6F1-Kit +/+ mice (Jackson Laboratory) were used as controls. All mouse experiments were conducted according to guidelines of the AAALAC-accredited BIDMC IACUC and NIH.
Infection with Parasites
S. venezuelensis (S.v.) or Nippostrongylus brasiliensis (N.b.) were maintained by serial passage in male Wistar rats or female BALB/c mice, respectively. Third-stage infective larvae (L3) of S.v. were obtained by fecal culture on filter paper,28 whereas L3 of N.b. were obtained by the charcoal-vermiculite fecal culture method.29 Mice were infected by subcutaneous (s.c.) inoculation with 400, 2000 or 10 000 L3 of S.v., or were infected by s.c. inoculation with 600 L3 of N.b.
Counting of Blood Leukocytes
Total leukocytes were counted in samples of retro-orbital blood using the UNOPETTE Test Kit (Becton Dickinson and Company, Franklin, NJ, USA). Smears were prepared of retro-orbital blood and were stained with May-Grünwald-Giemsa. A 300-cell leukocyte differential count was performed and the number of eosinophils was calculated with reference to the total leukocyte count for that animal. Bone marrow cells were obtained by washing the cells from a single femur from each mouse and cytospin preparations of these cells were stained with May-Grünwald-Giemsa. The percentage of eosinophils in 500 bone marrow cells was determined for each sample. Bone marrow basophils were identified and quantified by flow cytometry.30
Counting of Eosinophils and Mast Cells in Jejunum, Ileum, Spleen and Lung
Mice were killed by CO2 inhalation before parasite infection (‘baseline’) or on various days after parasite infection and segments of jejunum and ileum were taken from an area 8–10 cm distal to the pylorus or from an area 4–6 cm proximal to the ileocecal junction, respectively. Segments of intestine, lung (taken near the main bronchus) and spleen were fixed in either Carnoy's fluid or in 10% neutral buffered formalin, pH 7.4. The fixed tissues were embedded in paraffin and 4 μm tissue sections were deparaffinized and dehydrated. To demonstrate mast cells, the Carnoy's fixed sections were stained with 1% alcian blue, pH 1.0 (spleen) or 0.5% alcian blue, pH 0.3 (jejunum and ileum) at 37°C for 1 h.22 They were rinsed briefly in distilled water and counterstained with eosin-Y for 30 s and then dehydrated and mounted. To demonstrate eosinophils, the formalin-fixed sections were stained with hematoxylin-2 (Richard-Allan Scientific, Kalamazoo, MI, USA) for 5 s then rinsed briefly in distilled water and immersed in alkaline sodium chloride solution (1.0% sodium hydroxide and saturated sodium chloride in 80% ethanol) for 20 min and stained in 0.5% Congo red solution (0.5% Congo red in 50% ethanol) for 20 min and dehydrated and mounted.31 Mast cells and eosinophils were counted by light microscopy by a single observer who was unaware of the identity (mouse genotype) of the individual sections. Mast cells and eosinophils in the jejunum and ileum were counted in at least 10 consecutive villus crypt units and the results were expressed as the mean (±s.e.m.) cells per villus crypt unit. The spleen and lung mast cells or eosinophils were counted in 4–6 randomly selected sections and the results were expressed as the mean (±s.e.m.) cells per mm2.
Statistics
The data were analyzed for statistical significance (defined as P<0.05) using the two-tailed unpaired Student's t-test; unless otherwise specified, all data are expressed as mean±s.e.m.
Results
IL-3 −/− Mice Exhibit Normal Eosinophil Levels at Baseline and Unimpaired Eosinophil Responses to Infection with the Nematodes S.v. or N.b.
S.v. is a naturally occurring parasite of murine rodents that is rejected by a T-cell-dependent immune response which is associated with extensive mast cell hyperplasia and eosinophil infiltration in the affected intestinal mucosa.32, 33, 34 We reported that IL-3 −/− mice infected with 2000 S.v. L3, in comparison with the corresponding wild-type mice, exhibited significantly prolonged production of the parasites' eggs, as well as two marked abnormalities in their hematopoietic effector cell response to the infection: a virtual lack of parasite-induced hyperplasia of bone marrow basophils and a significant reduction in parasite-induced mast cell hyperplasia in the intestines and spleen.22
When tissues derived from some of the mice used in the study of Lantz et al22 were processed into Congo red-stained sections, we found that BALB/c IL-3 −/− and IL-3 +/+ mice that were infected with 2000 S.v. L3 exhibited nearly identical levels of tissue eosinophilia in the jejunum, (∼2-fold the corresponding levels in uninfected control mice), as well as similar levels of blood eosinophilia (Table 1). Similar results were obtained in a different experiment with C57BL/6 IL-3 −/− and IL-3 +/+ mice (Table 1).
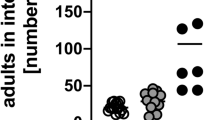
To address the possibility that eosinophil responses in IL-3 −/− mice might differ from those of IL-3 +/+ mice when the animals were infected with numbers of parasites that are different from that (ie, 2000 L3) used in our standard experiments, we infected groups of C57BL/6 IL-3 −/− or IL-3 +/+ mice with 400 or 10 000 S.v. L3. No parasite egg production was observed in any of the mice infected with 400 S.v. L3, indicating that this number of S.v. failed to establish a detectable infection. However, as shown in Figure 1a, substantial parasite egg production occurred in IL-3 −/− or IL-3 +/+ mice that were infected with 10 000 S.v. L3. In accord with our findings in mice infected with 2000 S.v. L3,22 the IL-3 −/− mice exhibited a significant delay in their ability to clear the infection, with parasite egg counts dropping to undetectable levels on day 15 of infection in the IL-3 −/− mice vs day 12 for the wild-type mice.
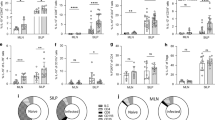
(a) Daily fecal egg production in female 8 to 10-week-old C57BL/6 IL-3 +/+ vs IL-3 −/− mice after infection with 10 000 L3 of S.v. All data are mean±s.e.m. (n=3–6 per group). *P<0.05, †P<0.001 vs corresponding values for IL-3 +/+ mice. All mice were killed on the day of clearance of infection. (b–g) Numbers of mast cells (b), eosinophils per villus crypt unit in jejunum and ileum (c), eosinophils per mm2 in sections of spleen and lung (d), % of bone marrow (BM) eosinophils (e), and numbers of eosinophils (f), and WBC (white blood cells) (g) per mm3 of blood, at baseline (−) or after infection with 400 (+) or 10 000 (++) L3 of S.v. in 8 to 10-week-old female C57BL/6 IL-3 +/+ vs IL-3 −/− mice. All data are mean±s.e.m. (n=3–6 per group), except that only mean values are given for very low values. *P<0.05, †P<0.001, ‡P<0.0001 vs corresponding baseline values for mice of the same genotype or (as indicated by brackets) vs corresponding values for mice of the other genotype. Data for mice infected with 10 000 L3 are from the same mice shown in (a). ND means not done.
However, on days 7 and 8 of the infection with 10 000 S.v. L3, parasite egg production in the IL-3 +/+ mice was ∼3-fold that in the IL-3 −/− mice. This finding, that was not observed in IL-3 −/− and IL-3 +/+ mice which had been infected with 2000 S.v. L3,22 indicates that the effects of IL-3 during primary infection with S.v. may be quite complex: IL-3 can contribute to the rapid resolution of the infection, but, under certain circumstances, IL-3 may also enhance egg production at early stages of the infection.
In accord with results obtained in mice infected with 2000 S.v. L3,22 IL-3 +/+ mice infected with 10 000 S.v. L3 exhibited a significant increase in numbers of bone marrow basophils by the time parasite egg expulsion was complete, whereas the identically challenged IL-3 −/− mice did not (2.4±0.2 vs 0.86±0.05% basophils, P<0.001). By contrast, infection with 400 S.v. L3 produced no significant changes in bone marrow basophil levels (0.64±0.06 or 0.65±0.02% in infected IL-3 +/+ or IL-3 −/− mice, vs corresponding baseline values in uninfected IL-3 +/+ or IL-3 −/− mice of 0.60±0.04 or 0.55±0.08%). Infection with 10 000 S.v. L3 also induced a substantial mast cell hyperplasia in the jejunum and ileum of the IL-3 +/+ mice, and resulted in greatly increased numbers of mast cells in the spleen, whereas the changes in mast cell numbers at these sites in the IL-3 −/− mice were minimal (Figure 1b).
In contrast to the dramatic differences exhibited by IL-3 −/− and IL-3+/+ mice in changes in bone marrow basophil or tissue mast cell levels during S.v. infection, the eosinophil responses to infection in mice of either genotype (and the baseline eosinophil levels in uninfected mice) were statistically indistinguishable (Figure 1c–f). Blood eosinophil levels were slightly lower after S.v. infection in IL-3−/− mice than in IL-3+/+ mice, but this difference was not statistically significant (Figure 1f); eosinophil levels in all of the other tested sites were virtually identical in the IL-3−/− and IL-3+/+ mice (Figure 1c–e). Note that mice infected with only 400 S.v. L3 also developed a significant blood eosinophilia (albeit to levels substantially lower than those in mice infected with 10 000 L3), and the levels in the IL-3−/− mice were slightly, but not significantly lower than those in the IL-3+/+ mice (Figure 1f). Thus, of the three hematopoietic lineages examined (eosinophils, basophils, and mast cells), the eosinophil was the most sensitive indicator of the presence of parasites.
We also examined eosinophil responses in mice infected with the nematode N.b., which, like S.v., induces a Th2-type immune response that is associated with blood and tissue eosinophilia.32, 35 However, even though mice infected with N.b. develop an intestinal mast cell hyperplasia, several lines of evidence indicate that mast cells have little or no role in the resolution of N.b. infection.34, 35, 36 We found that BALB/c IL-3−/− or IL-3+/+ mice infected with 600 N.b. L3 exhibited a virtually identical pattern of parasite egg production, with complete clearance of the infection in mice of either genotype by day 11 (data not shown). However, as in mice infected with S.v., N.b.-infected IL-3+/+ mice developed increased numbers of bone marrow basophils (∼100% increase) and increased numbers of mast cells in the jejunum, ileum and spleen, whereas the N.b.-infected IL-3−/− mice failed to develop increased numbers of bone marrow basophils and exhibited greatly impaired expansion of intestinal and splenic mast cell populations (data not shown). By contrast, both IL-3−/− and IL-3+/+ mice developed substantial, and virtually identical, increases in levels of bone marrow, blood, jejunal, ileal and splenic eosinophils in response to N.b. infection (Figure 2).
Numbers of eosinophils per villus crypt unit in jejunum and ileum (a), eosinophils per mm2 in sections of spleen (b), % of bone marrow (BM) eosinophils (c), and numbers of eosinophils (d) and WBC (white blood cells) (e) per mm3 of blood, at baseline (−) or at day 11 after infection with 600 L3 of N.b. in 3 to 4-month-old male or female BALB/c IL-3 +/+ vs IL-3 −/− mice. All data are mean±s.e.m. (n=3–5 per group). *P<0.05, †P<0.001, ‡P<0.0001 vs the corresponding baseline values for mice of the same genotype.
These results strongly support the conclusion that, despite its well-documented ability to promote eosinophil development in vitro, IL-3 can be dispensable for eosinophil production and tissue accumulation in mice in vivo. In fact, we have shown using a mouse model of ragweed-induced allergic inflammation known to be associated with a selective tissue accumulation of eosinophils that, under some circumstances, IL-3 can actually have a suppressive effect on eosinophil development and/or migration.19 While the potential role of IL-3 in regulating eosinophil production or tissue accumulation of eosinophils in the context of S.v. infection had not previously been investigated, Madden et al32 examined the effects of the administration of polyclonal antibodies to mouse IL-3 on the blood eosinophilia, and enhanced levels of tissue mast cells, which developed in mice that were infected with N.b. They found that treatment with anti-IL-3 antibodies reduced N.b.-induced increases in tissue mast cells by ∼40%, but had no detectable effect on blood eosinophilia on days 11 or l3 of the infection.32
The results of this study, and those reported by Lantz et al,22 clearly show that the requirements for IL-3 for expansion of basophil, intestinal mast cell and eosinophil populations during Th2-type responses to intestinal helminths in mice are distinct; IL-3 is required for the basophil response and importantly contributes to the mast cell response, but makes little or no detectable contribution to the eosinophil response. This conclusion is supported by our findings derived from mice which had been subjected to infection with a wide range of S.v. third stage larvae: 400, 2000 or 10 000, as well as by our results and those of Madden et al32 in N.b.-infected mice.
Infection with S.v., but not N.b., Induces Substantially Less Tissue Accumulation of Eosinophils in KitW/KitW−v, IL-3−/− Mice Than in KitW/KitW−v, IL-3+/+ Mice
Our findings in IL-3−/− and +/+ mice do not exclude the possibility that IL-3 might contribute to eosinophil responses in settings different than those we tested. For example, we reported that mast cell development and parasite immunity are impaired even more severely in S.v.-infected IL-3−/− mice that have markedly reducing c-Kit/SCF signaling (ie KitW/KitW−v, IL-3 −/− mice) than in similarly infected IL-3−/− or KitW/KitW−v, IL-3+/+ mice.22 We found that baseline numbers of blood, bone marrow and tissue eosinophils were essentially identical in KitW/KitW−v, IL-3−/− and KitW/KitW−v, IL-3+/+ mice (Figure 3). By contrast, S.v. infection induced a significant increase in tissue eosinophil levels in KitW/KitW−v, IL-3+/+ mice but, with the exception of the bone marrow, not in the KitW/KitW−v, IL-3−/− mice (Figure 3). Although blood eosinophil levels were comparable in uninfected and S.v.-infected KitW/KitW−v, IL-3+/+ mice, blood eosinophil levels were significantly lower in S.v.-infected KitW/KitW−v, IL-3−/− mice as compared to uninfected KitW/KitW−v, IL-3−/− mice (Figure 3d). It is important to note that all mice were killed when KitW/KitW−v, IL-3+/+ mice cleared their infection (days 18 or 19), a point in the infection in which KitW/KitW−v, IL-3−/− mice still retained most of their parasites.22 Thus, KitW/KitW−v, IL-3−/− mice exhibited a significantly impaired tissue eosinophil response despite the persistent stimulus of a heavy parasite burden.
Numbers of eosinophils per villus crypt unit in jejunum and ileum (a), eosinophils per mm2 in sections of spleen and lung (b), % of bone marrow (BM) eosinophils (c), and numbers of eosinophils (d) and WBC (white blood cells) (e) per mm3 of blood, at baseline (−) or at days 18 or 19 after infection with 2000 L3 of S.v. in 3 to 4-month-old male or female KitW/KitW−v, IL-3 +/+ vs KitW/KitW−v, IL-3 −/− mice. The data for S.v.-infected mice are from the same groups of mice depicted in Figure 3a in Lantz et al.22 All data are mean±s.e.m. (n=3–9 per group). *P<0.05, ‡P<0.0001 vs the corresponding baseline values for mice of the same genotype or (as indicated by brackets) vs corresponding values for mice of the other genotype.
We also examined eosinophil production in KitW/KitW−v, IL-3−/− mice infected with the nematode N.b. In contrast to our previous observations using S.v.,22 KitW/KitW−v, IL-3−/− or KitW/KitW−v, IL-3+/+ mice infected with 600 N.b. L3 exhibited no significant differences in parasite egg production as compared with Kit +/+, IL-3+/+ mice, with clearance of the infection ranging from days 11 to 14 (data not shown). These results are in agreement with other reports indicating that the primary host response to N.b. infection is indistinguishable or only slightly delayed in KitW/KitW−v mice as compared with wild-type mice.36 On the other hand, as in mice infected with S.v.,22 we found that N.b. infection induced an enhancement of bone marrow basophil production that was primarily depended on IL-3 (Figure 4a) and striking increases in mast cells in the intestines and spleen that were primarily dependent on c-Kit (Figure 4b). However, the eosinophil responses induced by infection of KitW/KitW−v mice with N.b. or S.v. differed in their requirement for IL-3. Thus, in contrast to our results with S.v. (see Figure 3), the levels of bone marrow, jejunal, ileal, lung and splenic eosinophils that developed in N.b.-infected KitW/KitW−v, IL-3−/− and KitW/KitW−v, IL-3+/+ mice were virtually identical (Figure 4c–e).
(a–g) Percentage of bone marrow basophils (a), numbers of mast cells (b), and numbers of eosinophils per villus crypt unit in jejunum and ileum (c), eosinophils per mm2 in sections of spleen and lung (d), % of bone marrow (BM) eosinophils (e), and numbers of eosinophils (f) and WBC (white blood cells) (g) per mm3 of blood, at baseline (−) or at days 11–14 after infection with 600 L3 of N.b. in 3 to 4-month-old male or female Kit +/+, IL-3 +/+ vs KitW/KitW−v, IL-3 +/+ vs KitW/KitW−v, IL-3 −/− mice. All data are mean±s.e.m. (n=3–10 per group), except that only mean values are given for very low values (b). *P<0.05, †P<0.001, ‡P<0.0001 vs the corresponding baseline values for mice of the same genotype or (as indicated by brackets) vs corresponding values for mice of another genotype. Baseline data for uninfected mice are from the same groups of mice depicted in Figure 3d in Lantz et al22 (a and b) or from Figure 3(c–g).
Although our results indicate that IL-3 makes little or no contribution to eosinophil responses to N.b. infection in either wild-type (Figure 2c–f) or KitW/KitW−v (Figure 4c–f) mice, they do suggest that c-Kit/SCF significantly contributes to eosinophil responses in this setting. Thus, N.b. infection of KitW/KitW−v, IL-3+/+ or −/− mice resulted in significantly lower levels of eosinophils in the blood and in all tissues (except the bone marrow) than did N.b. infection of wild-type (Kit +/+, IL-3+/+) mice (Figure 4c-f). The extent to which c-Kit/SCF may contribute to eosinophil responses to other nematodes remains to be determined. Wild-type mice were not infected with S.v. during the experiment shown in Figure 3, however, the eosinophil responses the S.v.-infected KitW/KitW−v, IL-3+/+ mice depicted in Figure 3a–d were similar to those observed in the S.v.-infected wild-type (IL-3+/+) mice shown in Table 1 and Figure 1c-f. Taken together, these results indicate that the eosinophil responses to N.b. infection in mice may be more dependent on c-Kit than are those observed during infection with S.v.
Table 2 summarizes some of our key findings regarding the roles of IL-3 and c-Kit signaling in influencing the numbers of blood and tissue eosinophils in mice subjected to the two different models of parasite infection analyzed in this study.
Discussion
IL-3 appeared to make no detectable contribution to eosinophil numbers in normal mice, in that we found no significant differences in the numbers of eosinophils in the blood, bone marrow or various tissues of IL-3−/− vs IL-3+/+ mice, either at baseline or after the induction of bone marrow, blood or tissue eosinophilia in response to infection with S.v. or N.b. However, in KitW/KitW−v mice, which have markedly impaired SCF/c-Kit signaling, our results indicate that IL-3 contributed significantly to the increased numbers of eosinophils that were observed in multiple tissues during S.v. infection, but not during infection with N.b.
A number of mechanisms may contribute to the inability of KitW/KitW−v, IL-3−/− mice to develop a robust eosinophil response to S.v. IL-3 can expand numbers of eosinophils generated from hematopoietic stem cells in vitro2, an effect which can be enhanced by SCF.23, 24 Therefore, both IL-3 and SCF may be required for increased commitment of progenitor cells to the eosinophilic lineage during a primary infection with S.v. Alternatively, or in addition, IL-3 and SCF may be required to promote eosinophil accumulation during S.v. infection by influencing eosinophil migration. Indeed, Yuan et al25 have provided evidence that SCF is an agonist for eosinophil adhesion to endothelial cells. Other studies suggest that eosinophil migration is in part regulated by the ability of resident mast cells to secrete chemoattractants for eosinophils, such as eotaxin and leukotrienes.37, 38, 39, 40 Modest numbers of mast cells do develop in the intestinal mucosal tissue or spleen of KitW/KitW−v, IL-3+/+ mice during infection with S.v.,22, 33 however, mast cell are virtually absent from the corresponding tissues of S.v.-infected KitW/KitW−v, IL-3−/− mice.22 Thus, the presence of low levels of eosinophils in S.v.-infected KitW/KitW−v, IL-3−/− mice may in part reflect the virtual absence of mast cells in these animals.
Our findings show that IL-3 can cooperate with c-Kit/SCF to increase numbers of eosinophils in response to S.v. infection. This is clearly shown in Figure 3, which demonstrates that the ability of S.v. infection to enhance eosinophil numbers in the tissues is almost undetectable in KitW/KitW−v, IL-3−/− mice. Although the precise mechanism(s) by which IL-3 and c-Kit/SCF regulate the eosinophil responses during S.v. infection remain(s) to be determined, one important implication of our findings is that the impaired immunity to S.v. expressed by KitW/KitW−v, IL-3−/− mice22 may in part reflect the combined contributions of mast cells, basophils and eosinophils.
We also noted that, on days 7 and 8 of the infection with 10 000 S.v. L3, parasite egg production in IL-3+/+ mice was ∼3-fold that in the IL-3−/− mice. Thus, in addition to contributing to the rapid resolution of the infection, under certain circumstances, IL-3 may also have effects that can enhance egg production at early stages of the infection. While the explanation for this observation remains to be determined, two studies have shown that egg production during nematode infections can be significantly higher in rats which have normal numbers of mast cells than in rats which are mast cell-deficient either because of loss-of-function mutations in c-kit41 or because of mast cell depletion as a result of the administration of an anti-SCF antibody.42 In the context of these findings in the rat, and in light of evidence that IL-3 can both promote increased numbers of mast cells and basophils22 and also can enhance the function of these cells,43, 44 as well as have effects on eosinophils, we can speculate about at least one mechanism by which IL-3 might promote increased nematode fecundity. Perhaps, in certain stages of such infections, the net effect of IL-3-dependent enhancement of mast cell, basophil and/or eosinophil numbers and/or function is to promote parasite fecundity. This might occur, for example, if IL-3 promoted the increased local release by mast cells, basophils and/or eosinophils of mediators that can enhance intestinal vascular or epithelial cell permeability, thereby favoring parasite nutrition, health and egg production.
References
Ihle JN . Interleukin-3 and hematopoiesis. Chem Immunol 1992;51:65–106.
Sanderson CJ, Warren DJ, Strath M . Identification of a lymphokine that stimulates eosinophil differentiation in vitro. Its relationship to interleukin 3, and functional properties of eosinophils produced in cultures. J Exp Med 1985;162:60–74.
Rothenberg ME, Owen Jr WF, Silberstein DS, et al. Human eosinophils have prolonged survival, enhanced functional properties, and become hypodense when exposed to human interleukin 3. J Clin Invest 1988;81:1986–1992.
Warren DJ, Moore MA . Synergism among interleukin 1, interleukin 3, and interleukin 5 in the production of eosinophils from primitive hemopoietic stem cells. J Immunol 1988;140:94–99.
Wardlaw AJ, Moqbel R, Kay AB . Eosinophils: biology and role in disease. Adv Immunol 1995;60:151–266.
Rothenberg ME . Eosinophilia. N Engl J Med 1998;338:1592–1600.
Robb L, Drinkwater CC, Metcalf D, et al. Hematopoietic and lung abnormalities in mice with a null mutation of the common beta subunit of the receptors for granulocyte-macrophage colony-stimulating factor and interleukins 3 and 5. Proc Natl Acad Sci USA 1995;92:9565–9569.
Nishinakamura R, Nakayama N, Hirabayashi Y, et al. Mice deficient for the IL-3/GM-CSF/IL-5 beta c receptor exhibit lung pathology and impaired immune response, while beta IL3 receptor-deficient mice are normal. Immunity 1995;2:211–222.
Nicola NA, Robb L, Metcalf D, et al. Functional inactivation in mice of the gene for the interleukin-3 (IL-3)-specific receptor beta-chain: implications for IL-3 function and the mechanism of receptor transmodulation in hematopoietic cells. Blood 1996;87:2665–2674.
Nishinakamura R, Miyajima A, Mee PJ, et al. Hematopoiesis in mice lacking the entire granulocyte-macrophage colony-stimulating factor/interleukin-3/interleukin-5 functions. Blood 1996;88:2458–2464.
Hayashida K, Kitamura T, Gorman DM, et al. Molecular cloning of a second subunit of the receptor for human granulocyte-macrophage colony-stimulating factor (GM-CSF): reconstitution of a high-affinity GM-CSF receptor. Proc Natl Acad Sci USA 1990;87:9655–9659.
Kitamura T, Sato N, Arai K, et al. Expression cloning of the human IL-3 receptor cDNA reveals a shared beta subunit for the human IL-3 and GM-CSF receptors. Cell 1991;66:1165–1174.
Gorman DM, Itoh N, Kitamura T, et al. Cloning and expression of a gene encoding an interleukin 3 receptor-like protein: identification of another member of the cytokine receptor gene family. Proc Natl Acad Sci USA 1990;87:5459–5463.
Itoh N, Yonehara S, Schreurs J, et al. Cloning of an interleukin-3 receptor gene: a member of a distinct receptor gene family. Science 1990;247:324–327.
Hara T, Miyajima A . Two distinct functional high affinity receptors for mouse interleukin-3 (IL-3). EMBO J 1992;11:1875–1884.
Kopf M, Brombacher F, Hodgkin PD, et al. IL-5-deficient mice have a developmental defect in CD5+ B-1 cells and lack eosinophilia but have normal antibody and cytotoxic T cell responses. Immunity 1996;4:15–24.
Yoshida T, Ikuta K, Sugaya H, et al. Defective B-1 cell development and impaired immunity against Angiostrongylus cantonensis in IL-5R alpha-deficient mice. Immunity 1996;4:483–494.
Takamoto M, Sugane K . Synergism of IL-3, IL-5, and GM-CSF on eosinophil differentiation and its application for an assay of murine IL-5 as an eosinophil differentiation factor. Immunol Lett 1995;45:43–46.
Neel NF, Creasy BM, Rankin JN, et al. Absence of interleukin-3 does not affect the severity of local and systemic anaphylaxis but does enhance eosinophil infiltration in a mouse model of allergic peritonitis. Immunol Lett 2004;95:37–44.
Gillessen S, Mach N, Small C, et al. Overlapping roles for granulocyte-macrophage colony-stimulating factor and interleukin-3 in eosinophil homeostasis and contact hypersensitivity. Blood 2001;97:922–928.
Mach N, Lantz CS, Galli SJ, et al. Involvement of interleukin-3 in delayed-type hypersensitivity. Blood 1998;91:778–783.
Lantz CS, Boesiger J, Song CH, et al. Role for interleukin-3 in mast-cell and basophil development and in immunity to parasites. Nature 1998;392:90–93.
Kobayashi H . Effect of c-kit ligand (stem cell factor) in combination with interleukin-5, granulocyte-macrophage colony-stimulating factor, and interleukin-3, on eosinophil lineage. Int J Hematol 1993;58:21–26.
Metcalf D, Mifsud S, Di Rago L . Stem cell factor can stimulate the formation of eosinophils by two types of murine eosinophil progenitor cells. Stem Cells 2002;20:460–469.
Yuan Q, Austen KF, Friend DS, et al. Human peripheral blood eosinophils express a functional c-kit receptor for stem cell factor that stimulates very late antigen 4 (VLA-4)-mediated cell adhesion to fibronectin and vascular cell adhesion molecule 1 (VCAM-1). J Exp Med 1997;186:313–323.
Kitamura Y, Go S, Hatanaka K . Decrease of mast cells in W/Wv mice and their increase by bone marrow transplantation. Blood 1978;52:447–452.
Tsai M, Lantz CS, Galli SJ . Regulation of mast cell and basophil development by stem cell factor and interleukin-3. In: Marone G, Lichtenstein LM, Galli SJ (eds). Mast Cells and Basophils. Academic Press: London, 2000, pp 3–20.
Sato Y, Toma H . Strongyloides venezuelensis infections in mice. Int J Parasitol 1990;20:57–62.
Huff TF, Lantz CS . Culture of murine mast cells. In: Lefkovits I (ed). The Immunology Methods Manual, Vol. 3. Academic Press: London, 1997, pp 1393–1407.
Lantz CS, Yamaguchi M, Oettgen HC, et al. IgE regulates mouse basophil Fc epsilon RI expression in vivo. J Immunol 1997;158:2517–2521.
Grouls V, Helpap B . Selective staining of eosinophils and their immature precursors in tissue sections and autoradiographs with Congo red. Stain Technol 1981;56:323–325.
Madden KB, Urban Jr JF, Ziltener HJ, et al. Antibodies to IL-3 and IL-4 suppress helminth-induced intestinal mastocytosis. J Immunol 1991;147:1387–1391.
Khan AI, Horii Y, Tiuria R, et al. Mucosal mast cells and the expulsive mechanisms of mice against Strongyloides venezuelensis. Int J Parasitol 1993;23:551–555.
Nawa Y, Ishikawa N, Tsuchiya K, et al. Selective effector mechanisms for the expulsion of intestinal helminths. Parasite Immunol 1994;16:333–338.
Gause WC, Urban Jr JF, Stadecker MJ . The immune response to parasitic helminths: insights from murine models. Trends Immunol 2003;24:269–277.
Finkelman FD, Shea-Donohue T, Goldhill J, et al. Cytokine regulation of host defense against parasitic gastrointestinal nematodes: lessons from studies with rodent models. Annu Rev Immunol 1997;15:505–533.
Harris RR, Komater VA, Marett RA, et al. Effect of mast cell deficiency and leukotriene inhibition on the influx of eosinophils induced by eotaxin. J Leukoc Biol 1997;62:688–691.
Das AM, Flower RJ, Perretti M . Resident mast cells are important for eotaxin-induced eosinophil accumulation in vivo. J Leukoc Biol 1998;64:156–162.
Hogaboam C, Kunkel SL, Strieter RM, et al. Novel role of transmembrane SCF for mast cell activation and eotaxin production in mast cell-fibroblast interactions. J Immunol 1998;160:6166–6171.
Klein A, Talvani A, Silva PM, et al. Stem cell factor-induced leukotriene B4 production cooperates with eotaxin to mediate the recruitment of eosinophils during allergic pleurisy in mice. J Immunol 2001;167:524–531.
Arizono N, Kasugai T, Yamada M, et al. Infection of Nippostrongylus brasiliensis induces development of mucosal-type but not connective tissue-type mast cells in genetically mast cell-deficient Ws/Ws rats. Blood 1993;81:2572–2578.
Newlands GF, Miller HR, MacKellar A, et al. Stem cell factor contributes to intestinal mucosal mast cell hyperplasia in rats infected with Nippostrongylus brasiliensis or Trichinella spiralis, but anti-stem cell factor treatment decreases parasite egg production during N brasiliensis infection. Blood 1995;86:1968–1976.
Lantz CS, Galli SJ . The role of mast cells, basophils and interleukin-3 (IL-3) in immune responses to parasites: Studies with mast cell- and IL-3-deficient mice. In: Marone G, Lichtenstein LM, Galli SJ (eds). Mast Cells and Basophils. Academic Press: London, 2000, pp 439–452.
Vilarino N, Miura K, MacGlashan Jr DW . Acute IL-3 priming up-regulates the stimulus-induced Raf-1-Mek-Erk cascade independently of IL-3-induced activation of Erk. J Immunol 2005;175:3006–3014.
Acknowledgements
This work was supported by N.I.H. Grants AI-23990 and CA-72074 (SJG), and AI-49932 (CSL). We thank Martin Metz, MD and Caroline Tudor for assistance in producing the figures.
Author information
Authors and Affiliations
Corresponding author
Rights and permissions
About this article
Cite this article
Kimura, K., Song, C., Rastogi, A. et al. Interleukin-3 and c-Kit/stem cell factor are required for normal eosinophil responses in mice infected with Strongyloides venezuelensis. Lab Invest 86, 987–996 (2006). https://doi.org/10.1038/labinvest.3700458
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/labinvest.3700458
Keywords
This article is cited by
-
IgE and mast cells in host defense against parasites and venoms
Seminars in Immunopathology (2016)