Abstract
Brain tumor initiating cells (BTICs) contribute to the genesis and recurrence of gliomas. We examined whether the microglia and macrophages that are abundant in gliomas alter BTIC growth. We found that microglia derived from non-glioma human subjects markedly mitigated the sphere-forming capacity of glioma patient–derived BTICs in culture by inducing the expression of genes that control cell cycle arrest and differentiation. This sphere-reducing effect was mimicked by macrophages, but not by neurons or astrocytes. Using a drug screen, we validated amphotericin B (AmpB) as an activator of monocytoid cells and found that AmpB enhanced the microglial reduction of BTIC spheres. In mice harboring intracranial mouse or patient-derived BTICs, daily systemic treatment with non-toxic doses of AmpB substantially prolonged life. Notably, microglia and monocytes cultured from glioma patients were inefficient at reducing the sphere-forming capacity of autologous BTICs, but this was rectified by AmpB. These results provide new insights into the treatment of gliomas.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Singh, S.K. et al. Identification of human brain tumour initiating cells. Nature 432, 396–401 (2004).
Kelly, J.J. et al. Proliferation of human glioblastoma stem cells occurs independently of exogenous mitogens. Stem Cells 27, 1722–1733 (2009).
Bao, S. et al. Glioma stem cells promote radioresistance by preferential activation of the DNA damage response. Nature 444, 756–760 (2006).
Eramo, A. et al. Chemotherapy resistance of glioblastoma stem cells. Cell Death Differ. 13, 1238–1241 (2006).
Chen, J. et al. A restricted cell population propagates glioblastoma growth after chemotherapy. Nature 488, 522–526 (2012).
Cheng, L., Bao, S. & Rich, J.N. Potential therapeutic implications of cancer stem cells in glioblastoma. Biochem. Pharmacol. 80, 654–665 (2010).
Dietrich, J., Diamond, E.L. & Kesari, S. Glioma stem cell signaling: therapeutic opportunities and challenges. Expert Rev. Anticancer Ther. 10, 709–722 (2010).
Charles, N.A., Holland, E.C., Gilbertson, R., Glass, R. & Kettenmann, H. The brain tumor microenvironment. Glia 60, 502–514 (2012).
Yang, I., Han, S.J., Kaur, G., Crane, C. & Parsa, A.T. The role of microglia in central nervous system immunity and glioma immunology. J. Clin. Neurosci. 17, 6–10 (2010).
Hussain, S.F. et al. The role of human glioma-infiltrating microglia/macrophages in mediating antitumor immune responses. Neuro-oncol. 8, 261–279 (2006).
Kerber, M. et al. Flt-1 signaling in macrophages promotes glioma growth in vivo. Cancer Res. 68, 7342–7351 (2008).
Markovic, D.S. et al. Gliomas induce and exploit microglial MT1-MMP expression for tumor expansion. Proc. Natl. Acad. Sci. USA 106, 12530–12535 (2009).
Bettinger, I., Thanos, S. & Paulus, W. Microglia promote glioma migration. Acta Neuropathol. 103, 351–355 (2002).
Hwang, S.Y. et al. Induction of glioma apoptosis by microglia-secreted molecules: the role of nitric oxide and cathepsin B. Biochim. Biophys. Acta 1793, 1656–1668 (2009).
Mora, R. et al. TNF-alpha– and TRAIL-resistant glioma cells undergo autophagy-dependent cell death induced by activated microglia. Glia 57, 561–581 (2009).
Brantley, E.C. et al. Nitric oxide–mediated tumoricidal activity of murine microglial cells. Transl. Oncol. 3, 380–388 (2010).
Kees, T. et al. Microglia isolated from patients with glioma gain antitumor activities on poly (I:C) stimulation. Neuro-oncol. 14, 64–78 (2012).
Chicoine, M.R. et al. The in vivo antitumoral effects of lipopolysaccharide against glioblastoma multiforme are mediated in part by Toll-like receptor 4. Neurosurgery 60, 372–380 (2007).
Galarneau, H., Villeneuve, J., Gowing, G., Julien, J.P. & Vallieres, L. Increased glioma growth in mice depleted of macrophages. Cancer Res. 67, 8874–8881 (2007).
Wu, A. et al. Glioma cancer stem cells induce immunosuppressive macrophages/microglia. Neuro-oncol. 12, 1113–1125 (2010).
Ye, X.Z. et al. Tumor-associated microglia/macrophages enhance the invasion of glioma stem-like cells via TGF-β1 signaling pathway. J. Immunol. 189, 444–453 (2012).
Williams, K. et al. Biology of adult human microglia in culture: comparisons with peripheral blood monocytes and astrocytes. J. Neuropathol. Exp. Neurol. 51, 538–549 (1992).
Giuliani, F., Hader, W. & Yong, V.W. Minocycline attenuates T cell and microglia activity to impair cytokine production in T cell-microglia interaction. J. Leukoc. Biol. 78, 135–143 (2005).
Brescia, P. et al. CD133 is essential for glioblastoma stem cell maintenance. Stem Cells 31, 857–869 (2013).
Hu, Y. & Smyth, G.K. ELDA: extreme limiting dilution analysis for comparing depleted and enriched populations in stem cell and other assays. J. Immunol. Methods 347, 70–78 (2009).
Zheng, H. et al. p53 and Pten control neural and glioma stem/progenitor cell renewal and differentiation. Nature 455, 1129–1133 (2008).
Lawrence, D.M. et al. Astrocyte differentiation selectively upregulates CCL2/monocyte chemoattractant protein-1 in cultured human brain-derived progenitor cells. Glia 53, 81–91 (2006).
Edman, L.C. et al. Alpha-chemokines regulate proliferation, neurogenesis, and dopaminergic differentiation of ventral midbrain precursors and neurospheres. Stem Cells 26, 1891–1900 (2008).
Samanani, S. et al. Screening for inhibitors of microglia to reduce neuroinflammation. CNS Neurol. Disord. Drug Targets 12, 741–749 (2013).
Tang, X., Mo, C., Wang, Y., Wei, D. & Xiao, H. Anti-tumour strategies aiming to target tumour-associated macrophages. Immunology 138, 93–104 (2013).
Popovich, P.G. et al. Depletion of hematogenous macrophages promotes partial hindlimb recovery and neuroanatomical repair after experimental spinal cord injury. Exp. Neurol. 158, 351–365 (1999).
Polfliet, M.M. et al. A method for the selective depletion of perivascular and meningeal macrophages in the central nervous system. J. Neuroimmunol. 116, 188–195 (2001).
Wu, A. et al. Persistence of CD133+ cells in human and mouse glioma cell lines: detailed characterization of GL261 glioma cells with cancer stem cell-like properties. Stem Cells Dev. 17, 173–184 (2008).
Bertrand, J. et al. Cancer stem cells from human glioma cell line are resistant to Fas-induced apoptosis. Int. J. Oncol. 34, 717–727 (2009).
Rubissow, M.J. Remission of astrocytoma following amphotericin-B treatment. Lancet 1, 565 (1970).
Tokuda, Y. et al. Augmentation of murine tumor necrosis factor production by amphotericin B in vitro and in vivo. Antimicrob. Agents Chemother. 37, 2228–2230 (1993).
Wilson, E., Thorson, L. & Speert, D.P. Enhancement of macrophage superoxide anion production by amphotericin B. Antimicrob. Agents Chemother. 35, 796–800 (1991).
Neumann, H. & Wekerle, H. Brain microglia: watchdogs with predigree. Nat. Neurosci. 16, 253–255 (2013).
Hanisch, U.K. & Kettenmann, H. Microglia: active sensor and versatile effector cells in the normal and pathologic brain. Nat. Neurosci. 10, 1387–1394 (2007).
Allavena, P., Sica, A., Garlanda, C. & Mantovani, A. The Yin-Yang of tumor-associated macrophages in neoplastic progression and immune surveillance. Immunol. Rev. 222, 155–161 (2008).
Mantovani, A., Allavena, P., Sica, A. & Balkwill, F. Cancer-related inflammation. Nature 454, 436–444 (2008).
Qian, B.Z. & Pollard, J.W. Macrophage diversity enhances tumor progression and metastasis. Cell 141, 39–51 (2010).
Galvão, R.P. & Zong, H. Inflammation and gliomagenesis: bi-directional communication at early and late stages of tumor progression. Curr. Pathobiol. Rep. 1, 19–28 (2013).
Vecil, G.G. et al. Interleukin-1 is a key regulator of matrix metalloproteinase-9 expression in human neurons in culture and following mouse brain trauma in vivo. J. Neurosci. Res. 61, 212–224 (2000).
Sarkar, S., Nuttall, R.K., Liu, S., Edwards, D.R. & Yong, V.W. Tenascin-C stimulates glioma cell invasion through matrix metalloproteinase-12. Cancer Res. 66, 11771–11780 (2006).
Besson, A. & Yong, V.W. Involvement of p21(Waf1/Cip1) in protein kinase C alpha–induced cell cycle progression. Mol. Cell Biol. 20, 4580–4590 (2000).
Nguyen, M.D., D'Aigle, T., Gowing, G., Julien, J.P. & Rivest, S. Exacerbation of motor neuron disease by chronic stimulation of innate immunity in a mouse model of amyotrophic lateral sclerosis. J. Neurosci. 24, 1340–1349 (2004).
Lun, X. et al. Efficacy and safety/toxicity study of recombinant vaccinia virus JX-594 in two immunocompetent animal models of glioma. Mol. Ther. 18, 1927–1936 (2010).
Zhou, Y. et al. The chemokine GRO-alpha (CXCL1) confers increased tumorigenicity to glioma cells. Carcinogenesis 26, 2058–2068 (2005).
Acknowledgements
We acknowledge the technical assistance of Y. Fan, X. Zhang, F. Yong, J. Wang, H. Mowbray, B. Verhaeghe, M. Keough, R. Hassam, B. Mckenzie and A. Luchman. Clodronate was a gift from Roche Diagnostics GmbH. We thank Alberta Innovates – Health Solutions/Alberta Cancer Foundation and the Canadian Institutes of Health Research for operating grant support. V.W.Y. is a Canada Research Chair (Tier 1) in Neuroimmunology, for which salary support is gratefully acknowledged.
Author information
Authors and Affiliations
Contributions
S.S. designed all of the experiments, conducted and analyzed the data from the majority of the experiments, supervised the acquisition of all datasets, and wrote and edited the manuscript. A.D. performed the PCR analyses, conducted the liposome treatment of mice and edited the manuscript. F.J.Z. transfected GL261 with luciferase reporter, performed bioluminescence imaging and analyses, and guided aspects of the study. C.S. helped process the cells from tumor specimens and conducted some of the experiments. X.L. implanted mice with intracranial human BTICs, cross-checked the in vivo datasets and edited the manuscript. X.W. performed the microarray experiments and analyses. J.K. was instrumental in the derivation of BTIC lines from resected human specimens and in the analyses of the genetic alterations of the cells, and edited the manuscript. W.H. provided the surgically resected brain materials from epilepsy cases from which non-transformed adult human microglia were derived and edited the manuscript. M.H. provided the resected glioblastoma and blood specimens from which BTICs, tumor-derived microglia and monocytes were obtained and edited the manuscript. P.M. provided the resected glioblastoma and blood specimens from which BTICs, tumor-derived microglia and monocytes were obtained and edited the manuscript. J.F.D. performed the MRI studies, adapted human imaging to evaluation in mice and edited the manuscript. D.K. supervised the measurements of levels of AmpB in mice. N.V.R. supplied the clodronate liposomes and advised on its use. S.R. supervised the microarray analyses and edited the manuscript. P.F. provided expertise with regards to the implantation of BTICs in mice and the treatment procedures of mice with intracranial tumors and edited the manuscript. G.C. supervised the BTIC bank that houses the patient-derived lines used in this study, provided general expertise on brain tumors and edited the manuscript. S.W. co-supervised the BTIC bank, provided expertise on stem cells and edited the manuscript. V.W.Y. supervised all aspects of this work, designed the majority of experiments, obtained the funding for this study and edited the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Integrated supplementary information
Supplementary Figure 1 a) Glioma patient-derived BTICs exhibit stem-like markers in vitro; (b) when implanted into the brain of NOD-SCID mice, the patient-derived BTICs retain stem-like markers.
(a) The 25EF patient-derived BTIC spheres were labeled for stem-like markers (green) and nuclei (NY: nuclear yellow). The majority of cells express these stem-like markers. Similar results were found for the 48EF line (data not shown). (b) The 25EF and 48EF lines were implanted into the striatum. Two months after, many cells were immunoreactive for stem-like markers (brown, as detected by DAB peroxidase staining). The H&E picture is a low magnification image of the brain to depict the large tumor.
Supplementary Figure 2 Consequence of microglia – BTIC co-culture.
(a) Microglia cultured in BTIC medium. (b) BTIC-microglia ratio for co-culture assay. (c-e) Three other patient-derived BTIC lines are reduced in sphere forming capacity when confronted with microglia. (f) Ratio of MCM:normal medium, where 1:10 begins to lose effectiveness in reducing spheres. Well-formed spheres (g) are rapidly differentiated with microglia conditioned medium MCM (h). (i) The 25EF patient-derived BTIC spheres were exposed to MCM for 72h, with 10 μM bromodeoxyurdine (BrdU) added during the last 24h. (j) Propidium iodide (PI) and annexin V flow cytometry show that while calphostin C (CpC, positive control) exposed BTICs undergo apoptosis, death was not altered by MCM compared to BTIC controls. (k) Graphical analysis of GeneSpring-identified top genes expressed in BTICs after 6h of MCM treatment. Genes up regulated by >10 fold were clustered by hierarchical clustering and visualized by Java TreeView. (n=4 for all groups from b-f and I-j; Error bars, s.d.)
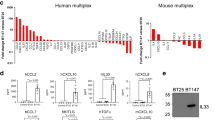
Supplementary Figure 3 Levels of molecules in conditioned media.
The control and AmpB groups refer, respectively, to BTICs alone and BTICs treated with AmpB in the absence of microglia intermediary. MCM refers to the 24h conditioned medium of microglia grown in BTIC medium, while AmpB/MCM represents the 24h conditioned medium of microglia treated with AmpB. Values are mean ± s.d. of 4 wells per condition. p values displayed are of AmpB/MCM compared to MCM alone (1-way ANOVA with Tukey's multiple comparisons, all 4 columns analysed); the statistical analyses for the other groups are not shown.
Supplementary Figure 4 Extreme limiting dilution assay (ELDA) of BTICs in absence or presence of media conditioned by human microglia and monocytes.
Extreme limiting dilution assay (ELDA) was carried out as described previously25 (a,d). Briefly, BTIC cells (25EF and 48EF) were plated in decreasing numbers from 200 cells/well to 1 cell/well in 200μl. Cultures were maintained until day 14, when the number of wells containing spheres for each cell plating density (number of positive cultures) was recorded, calculated and plotted using online ELDA analysis program25 (http://bioinf.wehi.edu.au/software/elda). ELDA analysis for 25EF (b,c) and 48EF lines (e,f). ELDA experiment shows zero sphere formation with MCM or MonoCM in all wells including single cell wells compared to control (a and d), and ELDA indicates significant differences in stem cell frequencies between the groups (c and f). n=4 for all groups; Error bars, s.d.
Supplementary Figure 5 Reduction of growth of BTICs but not their differentiated progenies by monocytoid cells.
The 25EF and 48EF BTIC lines were grown in BTIC medium or were differentiated by culture for 5d in 1% FBS medium. 104 cells/well were then exposed to MCM or AmpB-treated microglia conditioned medium (AmpB/MCM) from 2 separate microglia cultures (MCM1 or MCM2) for 72h, and the total number of cells/well was counted (a); panel b depicts representative images at 72h. Similar experiments were performed for medium conditioned by monocytes (c), or with co-culture with microglia (MG) (d). **p<0.01, ***p<0.001 compared to respective controls (1 way ANOVA) (n of 4 wells/histogram, Error bars, s.d.). The p value at the top of each set of histograms refers to the overall ANOVA value.
Supplementary Figure 6 Cell cycle analyses by flow cytometry confirm the reduction of proliferation of BTICs but not their differentiated cells in response to microglia-derived factors.
The 25EF BTIC line was grown in BTIC medium or was differentiated by culture for 5d in 1% FBS medium. Cells were then exposed to MCM or AmpB-treated microglia conditioned medium (AmpB/MCM) for 72h and subjected to propidium iodide flow cytometry for DNA content. a) Representative panels show 32% of control BTIC cells were in the S-phase of the cell cycle, and this was reduced in BTICs exposed to MCM, and further attenuated in BTICs exposed to AmpB/MCM. In contrast, in cells differentiated by 1%FBS, there was no impact of MCM or AmpB/MCM. These results were reproduced across 3 sets of cultures (b). *p<0.05, ***p<0.001 compared to respective controls (1 way ANOVA). (c) Finally, AmpB, MCM or monoCM from 2 subjects, and those collected from AmpB-treated cultures, were without effects on growth of U251 or U87 differentiated glioma lines. n = 4 for all groups; Error bars, s.d.
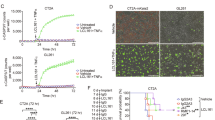
Supplementary Figure 7 Impact of patient-derived BTICs on survival of mice; AmpB treatment reduces tumor growth and prevents the weight loss that occurs in mice with intracranial tumors.
(a) Survival curve of mice implanted intracranially with 100 (e2) to 10,000 (e5) 25EF BTICs, n=5 for all groups, with log-rank analysis. (b) MRI image shows a large intracranial tumor (arrow) 7 weeks post-implantation in a control mouse with 48EF cells which was significantly reduced with AmpB treatment; the quantification of tumor area by MRI is shown in (c) (unpaired t-test, n=4 for all groups). (d) Absolute body weight (left) or weight displayed as change from that at start of experiment (right), n=10 mice each group. Error bars, s.d.
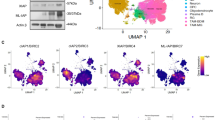
Supplementary Figure 8 Pharmacokinetics and safety profile of AmpB in mice, and CD133 and nestin-positive cells in the tumor core of mice implanted with mouse GL261 BTICs.
Serum (a) and brain (b) level of AmpB in mice, measured using HPLC, following 30 days of AmpB treatment (n=5 for all groups). (c) Differential counts of blood cells, showing elevated level of monocytes, in mice injected with daily AmpB for 30 days; mice were not neutropenic with AmpB treatment, n=5 for all groups, with unpaired t-test. (d,e) In another group of tumor bearing mice injected with daily AmpB for 50 days, normal renal and liver function tests (using Multistix10SG, Bayer Health Care, Elkhart, IN) in urine were demonstrated; these mice had elevation of monocytes in blood consistent with AmpB being a stimulator of monocytoid cells, but normal neutrophil counts. Mean ± s.d. n of 4, with unpaired t-test. (f) Representative images from the brains of C57BL/6 mice that died from the GL261 BTIC implantation of Figure 7. CD133 (red) and nuclear yellow (blue) stain in a region of no tumor (NT) or within the tumor core (TC). Nuclear yellow (blue) stain depicting the tumor mass in the top right hand corner whereby when stained for nestin (green), this stem-like marker was found only in the tumor core (TC). Comparable images were seen in vehicle- or AmpB-treated mice when these were moribund from the tumor and sacrificed.
Supplementary Figure 9 Monocytoid cells from glioblastoma do not inhibit BTIC sphere formation in autologous or heterologous interactions until stimulation with AmpB.
Glioblastoma-derived microglia (MG), their conditioned media (MCM) or conditioned media from monocytes (monoCM) were exposed to autologous (from same subject) or heterologous (25EF or 48EF) BTICs and sphere formation after 72h was enumerated. Control MG refers to microglia from non-glioma subjects with epilepsy. AmpB or LPS was used to treat microglia or monocytes in some experiments. Values are mean ± s.d. of 4 wells per condition, with ANOVA comparison between groups.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–10, Supplementary Table 1 (PDF 13763 kb)
Source data
Rights and permissions
About this article
Cite this article
Sarkar, S., Döring, A., Zemp, F. et al. Therapeutic activation of macrophages and microglia to suppress brain tumor-initiating cells. Nat Neurosci 17, 46–55 (2014). https://doi.org/10.1038/nn.3597
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/nn.3597
This article is cited by
-
Exosome-based nanoimmunotherapy targeting TAMs, a promising strategy for glioma
Cell Death & Disease (2023)
-
Interaction of glioma-associated microglia/macrophages and anti-PD1 immunotherapy
Cancer Immunology, Immunotherapy (2023)
-
Detecting monocyte trafficking in an animal model of glioblastoma using R2* and quantitative susceptibility mapping
Cancer Immunology, Immunotherapy (2023)
-
The expression of B7-H3 isoforms in newly diagnosed glioblastoma and recurrence and their functional role
Acta Neuropathologica Communications (2021)
-
1p/19q co-deletion status is associated with distinct tumor-associated macrophage infiltration in IDH mutated lower-grade gliomas
Cellular Oncology (2021)