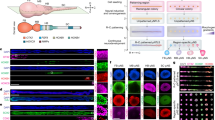
Abstract
Vascular smooth muscle cells (SMCs) arise from diverse developmental origins. Regional distribution of vascular diseases may, in part, be attributed to this inherent heterogeneity in SMC lineage. Therefore, systems for generating human SMC subtypes of distinct embryonic origins would represent useful platforms for studying the influence of SMC lineage on the spatial specificity of vascular disease. Here we describe how human pluripotent stem cells can be differentiated into distinct populations of SMC subtypes under chemically defined conditions. The initial stage (days 0–5 or 0–7) begins with the induction of three intermediate lineages: neuroectoderm, lateral plate mesoderm and paraxial mesoderm. Subsequently, these precursor lineages are differentiated into contractile SMCs (days 5–19+). At key stages, the emergence of lineage-specific markers confirms recapitulation of embryonic developmental pathways and generation of functionally distinct SMC subtypes. The ability to derive an unlimited supply of human SMCs will accelerate applications in regenerative medicine and disease modeling.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Carmeliet, P. Mechanisms of angiogenesis and arteriogenesis. Nat. Med. 6, 389–395 (2000).
Majesky, M.W. Developmental basis of vascular smooth muscle diversity. Arterioscler. Thromb. Vasc. Biol. 27, 1248–1258 (2007).
DeBakey, M.E. & Glaeser, D.H. Patterns of atherosclerosis: effect of risk factors on recurrence and survival-analysis of 11,890 cases with more than 25-year follow-up. Am. J. Cardiol. 85, 1045–1053 (2000).
Ruddy, J.M., Jones, J.A., Spinale, F.G. & Ikonomidis, JS Regional heterogeneity within the aorta: relevance to aneurysm disease. J. Thoracic Cardiovasc. Surg. 136, 1123–1130 (2008).
Owens, G.K., Kumar, M.S. & Wamhoff, B.R. Molecular regulation of vascular smooth muscle cell differentiation in development and disease. Physiol. Rev. 84, 767–801 (2004).
Tang, Z. et al. Differentiation of multipotent vascular stem cells contributes to vascular diseases. Nat. Commun. 3, 875 (2012).
Nguyen, A.T. et al. Smooth muscle cell plasticity: fact or fiction? Circ. Res. 112, 17–22 (2013).
Cheung, C. & Sinha, S. Human embryonic stem cell-derived vascular smooth muscle cells in therapeutic neovascularisation. J. Mol. Cell. Cardiol. 51, 651–664 (2011).
Kattman, S.J., Huber, T.L. & Keller, G.M. Multipotent Flk-1+ cardiovascular progenitor cells give rise to the cardiomyocyte, endothelial, and vascular smooth muscle lineages. Dev. Cell 11, 723–732 (2006).
Yang, L. et al. Human cardiovascular progenitor cells develop from a KDR plus embryonic-stem-cell-derived population. Nature 453, 524–526 (2008).
Cai, C.L. et al. Isl1 identifies a cardiac progenitor population that proliferates prior to differentiation and contributes a majority of cells to the heart. Dev. Cell 5, 877–889 (2003).
Cai, C.L. et al. A myocardial lineage derives from Tbx18 epicardial cells. Nature 454, 104–108 (2008).
Zhou, B. et al. Epicardial progenitors contribute to the cardiomyocyte lineage in the developing heart. Nature 454, 109–113 (2008).
D'Souza, S.L., Elefanty, A.G. & Keller, G. SCL/Tal-1 is essential for hernatopoietic commitment of the hemangioblast but not for its development. Blood 105, 3862–3870 (2005).
Lu, S.J., Ivanova, Y., Feng, Q., Luo, C.M. & Lanza, R. Hemangioblasts from human embryonic stem cells generate multilayered blood vessels with functional smooth muscle cells. Regen. Med. 4, 37–47 (2009).
Pouget, C., Pottin, K. & Jaffredo, T. Sclerotomal origin of vascular smooth muscle cells and pericytes in the embryo. Dev. Biol. 315, 437–447 (2008).
Esner, M. et al. Smooth muscle of the dorsal aorta shares a common clonal origin with skeletal muscle of the myotome. Development 133, 737–749 (2006).
Pouget, C., Gautier, R., Teillet, M.A. & Jaffredo, T. Somite-derived cells replace ventral aortic hemangioblasts and provide aortic smooth muscle cells of the trunk. Development 133, 1013–1022 (2006).
Minasi, M.G. et al. The meso-angioblast: a multipotent, self-renewing cell that originates from the dorsal aorta and differentiates into most mesodermal tissues. Development 129, 2773–2784 (2002).
Jiang, X.B., Rowitch, D.H., Soriano, P., McMahon, A.P. & Sucov, H.M. Fate of the mammalian cardiac neural crest. Development 127, 1607–1616 (2000).
Wang, A. et al. Derivation of smooth muscle cells with neural crest origin from human induced pluripotent stem cells. Cells Tissues Organs 195, 5–14 (2012).
Cheung, C., Bernardo, A.S., Trotter, M.W., Pedersen, R.A. & Sinha, S. Generation of human vascular smooth muscle subtypes provides insight into embryological origin-dependent disease susceptibility. Nat. Biotechnol. 30, 165–173 (2012).
Dosch, R., Gawantka, V., Delius, H., Blumenstock, C. & Niehrs, C. Bmp-4 acts as a morphogen in dorsoventral mesoderm patterning in Xenopus. Development 124, 2325–2334 (1997).
Jain, R.K. Molecular regulation of vessel maturation. Nat. Med. 9, 685–693 (2003).
Vazão, H., das Neves, R.P., Grãos, M. & Ferreira, L. Towards the maturation and characterization of smooth muscle cells derived from human embryonic stem cells. PLoS ONE 6, e17771 (2011).
Raphel, L., Talasila, A., Cheung, C. & Sinha, S. Myocardin overexpression is sufficient for promoting the development of a mature smooth muscle cell-like phenotype from human embryonic stem cells. PLoS ONE 7, e44052 (2012).
Bernardo, A.S. et al. BRACHYURY and CDX2 mediate BMP-induced differentiation of human and mouse pluripotent stem cells into embryonic and extraembryonic lineages. Cell Stem Cell 9, 144–155 (2011).
Zhang, J. et al. A human iPSC model of Hutchinson-Gilford progeria reveals vascular smooth muscle and mesenchymal stem cell defects. Cell Stem Cell 8, 31–45 (2010).
Trigueros-Motos, L. et al. Embryological-origin-dependent differences in homeobox expression in adult aorta: role in regional phenotypic variability and regulation of NF-κB activity. Arterioscler. Thromb. Vasc. Biol. 33, 1248–1256 (2013).
Acknowledgements
We thank L. Vallier for the established work on neuroectoderm differentiation. S.S. is funded by the British Heart Foundation (FS/13/29/30024 and NH/11/1/28922) and the Cambridge National Institute for Health Research Cambridge Biomedical Research Centre. C.C. is supported by an Independent Fellowship from the Institute of Molecular and Cell Biology (Singapore). A.S.B. is supported by the British Heart Foundation (FS/12/37/29516).
Author information
Authors and Affiliations
Contributions
C.C. and S.S. designed the protocol. C.C. performed the differentiation experiments, analyzed the data and wrote and prepared the manuscript. A.S.B. and R.A.P. established the mesoderm induction conditions and coedited the manuscript. S.S. supervised the project and edited the manuscript.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Methods
Immunofluorescence and flow cytometry (PDF 112 kb)
Supplementary Table 1
Approximate yields of SMC subtypes from hPSCs. Values represent (mean ± s.d.). (PDF 113 kb)
Rights and permissions
About this article
Cite this article
Cheung, C., Bernardo, A., Pedersen, R. et al. Directed differentiation of embryonic origin–specific vascular smooth muscle subtypes from human pluripotent stem cells. Nat Protoc 9, 929–938 (2014). https://doi.org/10.1038/nprot.2014.059
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2014.059
This article is cited by
-
Nuclear smooth muscle α-actin participates in vascular smooth muscle cell differentiation
Nature Cardiovascular Research (2023)
-
Spike-based adenovirus vectored COVID-19 vaccine does not aggravate heart damage after ischemic injury in mice
Communications Biology (2022)
-
The CAD risk locus 9p21 increases the risk of vascular calcification in an iPSC-derived VSMC model
Stem Cell Research & Therapy (2021)
-
Induced Pluripotent Stem Cells (iPSCs) in Vascular Research: from Two- to Three-Dimensional Organoids
Stem Cell Reviews and Reports (2021)
-
Human iPS Cell-derived Tissue Engineered Vascular Graft: Recent Advances and Future Directions
Stem Cell Reviews and Reports (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.