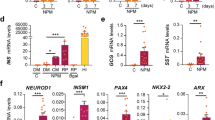
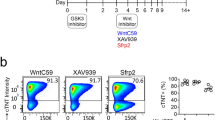
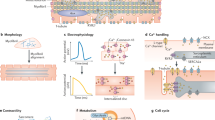
Abstract
Induction of tissue-specific cell types via a conventional transdifferentiation strategy typically uses overexpression of the corresponding lineage-specific transcription factors. Alternatively, somatic cells can be temporarily activated via a common set of reprogramming factors into a transition state, which can then be directed into various cell types via soluble lineage-specific signals, without establishing a pluripotent state. Here, we provide protocols for the generation of cardiomyocytes, neural stem cells and hepatocytes from fibroblasts with such a cell activation (CA) and signaling-directed (SD; CASD) strategy. In these protocols, beating cardiomyocytes can be induced from mouse fibroblasts in 2–5 weeks; expandable neural stem cells and definitive endoderm progenitors can be obtained from human fibroblasts as early as 2.5 weeks; and human definitive endoderm progenitors can be differentiated into functional hepatocytes in 2 weeks. Through further developments, the CASD strategy can serve as a unique avenue for generating diverse functional cell types for biomedical research and therapeutic applications.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
References
Takahashi, K. & Yamanaka, S. Induction of pluripotent stem cells from mouse embryonic and adult fibroblast cultures by defined factors. Cell 126, 663–676 (2006).
Yu, J. et al. Induced pluripotent stem cell lines derived from human somatic cells. Science 318, 1917–1920 (2007).
Takahashi, K. et al. Induction of pluripotent stem cells from adult human fibroblasts by defined factors. Cell 131, 861–872 (2007).
Murry, C.E. & Keller, G. Differentiation of embryonic stem cells to clinically relevant populations: lessons from embryonic development. Cell 132, 661–680 (2008).
Huang, P. et al. Induction of functional hepatocyte-like cells from mouse fibroblasts by defined factors. Nature 475, 386–389 (2011).
Sekiya, S. & Suzuki, A. Direct conversion of mouse fibroblasts to hepatocyte-like cells by defined factors. Nature 475, 390–393 (2011).
Vierbuchen, T. et al. Direct conversion of fibroblasts to functional neurons by defined factors. Nature 463, 1035–1041 (2010).
Ieda, M. et al. Direct reprogramming of fibroblasts into functional cardiomyocytes by defined factors. Cell 142, 375–386 (2010).
Zhou, Q., Brown, J., Kanarek, A., Rajagopal, J. & Melton, D.A. In vivo reprogramming of adult pancreatic exocrine cells to beta-cells. Nature 455, 627–632 (2008).
Qian, L. et al. In vivo reprogramming of murine cardiac fibroblasts into induced cardiomyocytes. Nature 485, 593–598 (2012).
Song, K. et al. Heart repair by reprogramming non-myocytes with cardiac transcription factors. Nature 485, 599–604 (2012).
Efe, J.A. et al. Conversion of mouse fibroblasts into cardiomyocytes using a direct reprogramming strategy. Nat. Cell Biol. 13, 215–222 (2011).
Wang, H. et al. Small molecules enable cardiac reprogramming of mouse fibroblasts with a single factor, Oct4. Cell Rep. 6, 951–960 (2014).
Kurian, L. et al. Conversion of human fibroblasts to angioblast-like progenitor cells. Nat. Methods 10, 77–83 (2013).
Li, J. et al. Conversion of human fibroblasts to functional endothelial cells by defined factors. Arterioscler. Thromb. Vasc. Biol. 33, 1366–1375 (2013).
Margariti, A. et al. Direct reprogramming of fibroblasts into endothelial cells capable of angiogenesis and reendothelialization in tissue-engineered vessels. Proc. Natl. Acad. Sci. USA 109, 13793–13798 (2012).
Kim, J. et al. Direct reprogramming of mouse fibroblasts to neural progenitors. Proc. Natl. Acad. Sci. USA 108, 7838–7843 (2011).
Wang, L. et al. Generation of integration-free neural progenitor cells from cells in human urine. Nat. Methods 10, 84–89 (2013).
Zhu, S. et al. Small molecules enable OCT4-mediated direct reprogramming into expandable human neural stem cells. Cell Res. 24, 126–129 (2014).
Li, K. et al. Small molecules facilitate the reprogramming of mouse fibroblasts into pancreatic lineages. Cell Stem Cell 14, 228–236 (2014).
Zhu, S. et al. Mouse liver repopulation with hepatocytes generated from human fibroblasts. Nature 508, 93–97 (2014).
Warren, L. et al. Highly efficient reprogramming to pluripotency and directed differentiation of human cells with synthetic modified mRNA. Cell Stem Cell 7, 618–630 (2010).
Yuan, X. et al. Brief report: combined chemical treatment enables Oct4-induced reprogramming from mouse embryonic fibroblasts. Stem Cells 29, 549–553 (2011).
Zhu, S. et al. Reprogramming of human primary somatic cells by OCT4 and chemical compounds. Cell Stem Cell 7, 651–655 (2010).
Zhu, S., Wei, W. & Ding, S. Chemical strategies for stem cell biology and regenerative medicine. Annu. Rev. Biomed. Eng. 13, 73–90 (2011).
Nie, B., Wang, H., Laurent, T. & Ding, S. Cellular reprogramming: a small molecule perspective. Curr. Opin. Cell Biol. 24, 784–792 (2012).
Maki, N. et al. Expression of stem cell pluripotency factors during regeneration in newts. Dev. Dyn. 238, 1613–1616 (2009).
Lu, J. et al. Generation of integration-free and region-specific neural progenitors from primate fibroblasts. Cell Rep. 3, 1580–1591 (2013).
Dimmeler, S., Ding, S., Rando, T.A. & Trounson, A. Translational strategies and challenges in regenerative medicine. Nat. Med. 20, 814–821 (2014).
Zhu, S., Ma, T., Li, J. & Ding, S. Recent advances in chemically induced reprogramming. Cell Cycle 10, 871–872 (2011).
Okita, K. et al. A more efficient method to generate integration-free human iPS cells. Nat. Methods 8, 409–412 (2011).
Li, W. et al. Rapid induction and long-term self-renewal of primitive neural precursors from human embryonic stem cells by small molecule inhibitors. Proc. Natl. Acad. Sci. USA 108, 8299–8304 (2011).
Si-Tayeb, K. et al. Highly efficient generation of human hepatocyte-like cells from induced pluripotent stem cells. Hepatology 51, 297–305 (2010).
Song, Z. et al. Efficient generation of hepatocyte-like cells from human induced pluripotent stem cells. Cell Res. 19, 1233–1242 (2009).
Du, Y. et al. Human hepatocytes with drug metabolic function induced from fibroblasts by lineage reprogramming. Cell Stem Cell 14, 394–403 (2014).
Huang, P. et al. Direct reprogramming of human fibroblasts to functional and expandable hepatocytes. Cell Stem Cell 14, 370–384 (2014).
Azuma, H. et al. Robust expansion of human hepatocytes in Fah–/–/Rag2–/–/Il2rg–/– mice. Nat. Biotechnol. 25, 903–910 (2007).
Bhatia, S.N., Underhill, G.H., Zaret, K.S. & Fox, I.J. Cell and tissue engineering for liver disease. Sci. Transl. Med. 6, 245sr2 (2014).
Acknowledgements
We thank all members of Ding laboratory for their valuable suggestion and discussion, particularly X. Wang for her contribution on episomal Nucleofection, and I. Namro Redwan, P. Liu and M. Xie for proofreading of the manuscript. We also thank C. Brennecka for editing the manuscript. S.D. is supported by funding from the California Institute for Regenerative Medicine (CIRM), the US National Institutes of Health (NIH) and the Gladstone Institutes. S.Z. is a CIRM training scholar.
Author information
Authors and Affiliations
Contributions
S.Z., H.W. and S.D. conceived the study and the experimental design and wrote the manuscript. S.Z. performed the NSC and hepatocyte reprogramming experiments. H.W. performed the cardiomyocyte reprogramming experiments. All authors edited and approved the final manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
About this article
Cite this article
Zhu, S., Wang, H. & Ding, S. Reprogramming fibroblasts toward cardiomyocytes, neural stem cells and hepatocytes by cell activation and signaling-directed lineage conversion. Nat Protoc 10, 959–973 (2015). https://doi.org/10.1038/nprot.2015.059
Published:
Issue Date:
DOI: https://doi.org/10.1038/nprot.2015.059
This article is cited by
-
OCT4 protein and gene expression analysis in the differentiation of spermatogonia stem cells into neurons by immunohistochemistry, immunocytochemistry, and bioinformatics analysis
Stem Cell Reviews and Reports (2023)
-
Directly reprogrammed natural killer cells for cancer immunotherapy
Nature Biomedical Engineering (2021)
-
Conversion of human and mouse fibroblasts into lung-like epithelial cells
Scientific Reports (2019)
-
Transdifferentiation: a new promise for neurodegenerative diseases
Cell Death & Disease (2018)
-
The novel tool of cell reprogramming for applications in molecular medicine
Journal of Molecular Medicine (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.