Abstract
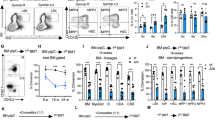
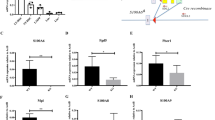
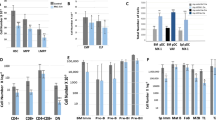
Although hematopoietic stem cells (HSCs) are the most thoroughly characterized type of adult stem cell, the intricate molecular machinery that regulates their self-renewal properties remains elusive. Here we showed that the E3 ubiquitin ligase Itch negatively regulated the development and function of HSCs. Itch−/− mice had HSCs with enhanced frequency, competence and long-term repopulating activity. Itch-deficient HSCs showed accelerated proliferation rates and sustained progenitor properties, as well as more signaling by the transcription factor Notch1, due to more accumulation of activated Notch1. Knockdown of Notch1 in Itch-mutant HSCs resulted in reversion of the phenotype. Thus, we identify Itch as a previously unknown negative regulator of HSC homeostasis and function.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout







Similar content being viewed by others
References
Morrison, S.J., Uchida, N. & Weissman, I.L. The biology of hematopoietic stem cells. Annu. Rev. Cell Dev. Biol. 11, 35–71 (1995).
Orford, K.W. & Scadden, D.T. Deconstructing stem cell self-renewal: genetic insights into cell-cycle regulation. Nat. Rev. Genet. 9, 115–128 (2008).
Orkin, S.H. & Zon, L.I. Hematopoiesis: an evolving paradigm for stem cell biology. Cell 132, 631–644 (2008).
He, S., Nakada, D. & Morrison, S.J. Mechanisms of stem cell self-renewal. Annu. Rev. Cell Dev. Biol. 25, 377–406 (2009).
Blank, U., Karlsson, G. & Karlsson, S. Signaling pathways governing stem-cell fate. Blood 111, 492–503 (2008).
Wilson, A., Laurenti, E. & Trumpp, A. Balancing dormant and self-renewing hematopoietic stem cells. Curr. Opin. Genet. Dev. 19, 461–468 (2009).
Rathinam, C., Thien, C.B., Langdon, W.Y., Gu, H. & Flavell, R.A. The E3 ubiquitin ligase c-Cbl restricts development and functions of hematopoietic stem cells. Genes Dev. 22, 992–997 (2008).
Yokomizo, T. & Dzierzak, E. Fine-tuning of hematopoietic stem cell homeostasis: novel role for ubiquitin ligase. Genes Dev. 22, 960–963 (2008).
Conaway, R.C., Brower, C.S. & Conaway, J.W. Emerging roles of ubiquitin in transcription regulation. Science 296, 1254–1258 (2002).
Hershko, A. & Ciechanover, A. The ubiquitin system. Annu. Rev. Biochem. 67, 425–479 (1998).
Haglund, K., Di Fiore, P.P. & Dikic, I. Distinct monoubiquitin signals in receptor endocytosis. Trends Biochem. Sci. 28, 598–603 (2003).
Hicke, L. & Dunn, R. Regulation of membrane protein transport by ubiquitin and ubiquitin-binding proteins. Annu. Rev. Cell Dev. Biol. 19, 141–172 (2003).
Kodadek, T., Sikder, D. & Nalley, K. Keeping transcriptional activators under control. Cell 127, 261–264 (2006).
Pickart, C.M. Mechanisms underlying ubiquitination. Annu. Rev. Biochem. 70, 503–533 (2001).
Bernassola, F., Karin, M., Ciechanover, A. & Melino, G. The HECT family of E3 ubiquitin ligases: multiple players in cancer development. Cancer Cell 14, 10–21 (2008).
Perry, W.L. et al. The itchy locus encodes a novel ubiquitin protein ligase that is disrupted in a18H mice. Nat. Genet. 18, 143–146 (1998).
Matesic, L.E., Haines, D.C., Copeland, N.G. & Jenkins, N.A. Itch genetically interacts with Notch1 in a mouse autoimmune disease model. Hum. Mol. Genet. 15, 3485–3497 (2006).
Fang, D. et al. Dysregulation of T lymphocyte function in itchy mice: a role for Itch in TH2 differentiation. Nat. Immunol. 3, 281–287 (2002).
Venuprasad, K. et al. The E3 ubiquitin ligase Itch regulates expression of transcription factor Foxp3 and airway inflammation by enhancing the function of transcription factor TIEG1. Nat. Immunol. 9, 245–253 (2008).
Parravicini, V., Field, A.C., Tomlinson, P.D., Basson, M.A. & Zamoyska, R. Itch−/− alphabeta and gammadelta T cells independently contribute to autoimmunity in Itchy mice. Blood 111, 4273–4282 (2008).
Kiel, M.J., Yilmaz, O.H., Iwashita, T., Terhorst, C. & Morrison, S.J. SLAM family receptors distinguish hematopoietic stem and progenitor cells and reveal endothelial niches for stem cells. Cell 121, 1109–1121 (2005).
Yilmaz, O.H., Kiel, M.J. & Morrison, S.J. SLAM family markers are conserved among hematopoietic stem cells from old and reconstituted mice and markedly increase their purity. Blood 107, 924–930 (2006).
Wilson, A. et al. Hematopoietic stem cells reversibly switch from dormancy to self-renewal during homeostasis and repair. Cell 135, 1118–1129 (2008).
Dzierzak, E. & Speck, N.A. Of lineage and legacy: the development of mammalian hematopoietic stem cells. Nat. Immunol. 9, 129–136 (2008).
Huang, X., Cho, S. & Spangrude, G.J. Hematopoietic stem cells: generation and self-renewal. Cell Death Differ. 14, 1851–1859 (2007).
Johnson, G.R. & Moore, M.A. Role of stem cell migration in initiation of mouse foetal liver haemopoiesis. Nature 258, 726–728 (1975).
Spangrude, G.J., Brooks, D.M. & Tumas, D.B. Long-term repopulation of irradiated mice with limiting numbers of purified hematopoietic stem cells: in vivo expansion of stem cell phenotype but not function. Blood 85, 1006–1016 (1995).
Gothot, A., van der Loo, J.C., Clapp, D.W. & Srour, E.F. Cell cycle-related changes in repopulating capacity of human mobilized peripheral blood CD34+ cells in non-obese diabetic/severe combined immune-deficient mice. Blood 92, 2641–2649 (1998).
Lerner, C. & Harrison, D.E. 5-Fluorouracil spares hemopoietic stem cells responsible for long-term repopulation. Exp. Hematol. 18, 114–118 (1990).
Rathinam, C. et al. Generation and characterization of a novel hematopoietic progenitor cell line with DC differentiation potential. Leukemia 20, 870–876 (2006).
Varnum-Finney, B. et al. Pluripotent, cytokine-dependent, hematopoietic stem cells are immortalized by constitutive Notch1 signaling. Nat. Med. 6, 1278–1281 (2000).
Chastagner, P., Israel, A. & Brou, C. AIP4/Itch regulates Notch receptor degradation in the absence of ligand. PLoS ONE 3, e2735 (2008).
Qiu, L. et al. Recognition and ubiquitination of Notch by Itch, a hect-type E3 ubiquitin ligase. J. Biol. Chem. 275, 35734–35737 (2000).
Duncan, A.W. et al. Integration of Notch and Wnt signaling in hematopoietic stem cell maintenance. Nat. Immunol. 6, 314–322 (2005).
Allman, D., Aster, J.C. & Pear, W.S. Notch signaling in hematopoiesis and early lymphocyte development. Immunol. Rev. 187, 75–86 (2002).
Moore, K.A. & Lemischka, I.R. Stem cells and their niches. Science 311, 1880–1885 (2006).
Rathinam, C. & Flavell, R.A. The hematopoiesis paradigm: clarity or ambiguity? Blood 112, 3534–3535 (2008).
Garrison, B.S. & Rossi, D.J. Controlling stem cell fate one substrate at a time. Nat. Immunol. 11, 193–194 (2010).
Trumpp, A., Essers, M. & Wilson, A. Awakening dormant haematopoietic stem cells. Nat. Rev. Immunol. 10, 201–209 (2010).
Min, I.M. et al. The transcription factor EGR1 controls both the proliferation and localization of hematopoietic stem cells. Cell Stem Cell 2, 380–391 (2008).
Kopan, R. & Ilagan, M.X. The canonical Notch signaling pathway: unfolding the activation mechanism. Cell 137, 216–233 (2009).
Maillard, I. et al. Canonical notch signaling is dispensable for the maintenance of adult hematopoietic stem cells. Cell Stem Cell 2, 356–366 (2008).
Radtke, F., Fasnacht, N. & Macdonald, H.R. Notch signaling in the immune system. Immunity 32, 14–27 (2010).
Maillard, I., Adler, S.H. & Pear, W.S. Notch and the immune system. Immunity 19, 781–791 (2003).
Maeda, T. et al. Regulation of B versus T lymphoid lineage fate decision by the proto-oncogene LRF. Science 316, 860–866 (2007).
Radtke, F., Wilson, A. & MacDonald, H.R. Notch signaling in T- and B-cell development. Curr. Opin. Immunol. 16, 174–179 (2004).
Pui, J.C. et al. Notch1 expression in early lymphopoiesis influences B versus T lineage determination. Immunity 11, 299–308 (1999).
Lohr, N.J. et al. Human ITCH E3 ubiquitin ligase deficiency causes syndromic multisystem autoimmune disease. Am. J. Hum. Genet. 86, 447–453 (2010).
Acknowledgements
We thank F. Manzo for help with manuscript submission; and the Yale Cell Sorter Facility for support. Supported by the Howard Hughes Medical Institute (R.A.F.) and the administrative core of the National Institutes of Health Center for Biomedical Research Excellence at Roger Williams Medical Center (P20RR018757).
Author information
Authors and Affiliations
Contributions
C.R. conceived of, designed and did the study, analyzed and interpreted all data, and wrote the manuscript; L.E.M. provided the Itch−/− mice and corrected the manuscript; and R.A.F. provided advice and corrected the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Supplementary information
Supplementary Text and Figures
Supplementary Figures 1–11 (PDF 1265 kb)
Rights and permissions
About this article
Cite this article
Rathinam, C., Matesic, L. & Flavell, R. The E3 ligase Itch is a negative regulator of the homeostasis and function of hematopoietic stem cells. Nat Immunol 12, 399–407 (2011). https://doi.org/10.1038/ni.2021
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/ni.2021
This article is cited by
-
E3 ubiquitin ligase on the biological properties of hematopoietic stem cell
Journal of Molecular Medicine (2023)
-
A20 deficiency in hematopoietic stem cells causes lymphopenia and myeloproliferation due to elevated Interferon-γ signals
Scientific Reports (2019)
-
Notch signaling facilitates hepatitis B virus covalently closed circular DNA transcription via cAMP response element-binding protein with E3 ubiquitin ligase-modulation
Scientific Reports (2019)
-
Notch Signaling in the Regulation of Hematopoietic Stem Cell
Current Stem Cell Reports (2017)
-
G protein-coupled receptor 183 facilitates endothelial-to-hematopoietic transition via Notch1 inhibition
Cell Research (2015)