Abstract
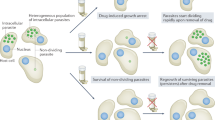
Visceral leishmaniasis causes considerable mortality and morbidity in many parts of the world. There is an urgent need for the development of new, effective treatments for this disease. Here we describe the development of an anti-leishmanial drug-like chemical series based on a pyrazolopyrimidine scaffold. The leading compound from this series (7, DDD853651/GSK3186899) is efficacious in a mouse model of visceral leishmaniasis, has suitable physicochemical, pharmacokinetic and toxicological properties for further development, and has been declared a preclinical candidate. Detailed mode-of-action studies indicate that compounds from this series act principally by inhibiting the parasite cdc-2-related kinase 12 (CRK12), thus defining a druggable target for visceral leishmaniasis.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 51 print issues and online access
$199.00 per year
only $3.90 per issue
Buy this article
- Purchase on SpringerLink
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout





Similar content being viewed by others
Change history
04 July 2023
A Correction to this paper has been published: https://doi.org/10.1038/s41586-023-06364-2
References
Alvar, J. et al. Leishmaniasis worldwide and global estimates of its incidence. PLoS ONE 7, e35671 (2012).
Ritmeijer, K. & Davidson, R. N. Royal Society of Tropical Medicine and Hygiene joint meeting with Médecins Sans Frontières at Manson House, London, 20 March 2003: field research in humanitarian medical programmes. Médecins Sans Frontières interventions against kala-azar in the Sudan, 1989–2003. Trans. R. Soc. Trop. Med. Hyg. 97, 609–613 (2003).
Sundar, S. et al. Efficacy of miltefosine in the treatment of visceral leishmaniasis in India after a decade of use. Clin. Infect. Dis. 55, 543–550 (2012).
den Boer, M. L., Alvar, J., Davidson, R. N., Ritmeijer, K. & Balasegaram, M. Developments in the treatment of visceral leishmaniasis. Expert Opin. Emerg. Drugs 14, 395–410 (2009).
Mueller, M. et al. Unresponsiveness to AmBisome in some Sudanese patients with kala-azar. Trans. R. Soc. Trop. Med. Hyg. 101, 19–24 (2007).
Khare, S. et al. Proteasome inhibition for treatment of leishmaniasis, Chagas disease and sleeping sickness. Nature 537, 229–233 (2016).
Don, R. & Ioset, J.-R. Screening strategies to identify new chemical diversity for drug development to treat kinetoplastid infections. Parasitology 141, 140–146 (2014).
Woodland, A. et al. From on-target to off-target activity: identification and optimisation of Trypanosoma brucei GSK3 inhibitors and their characterisation as anti-Trypanosoma brucei drug discovery lead molecules. ChemMedChem 8, 1127–1137 (2013).
De Rycker, M. et al. Comparison of a high-throughput high-content intracellular Leishmania donovani assay with an axenic amastigote assay. Antimicrob. Agents Chemother. 57, 2913–2922 (2013).
Nühs, A. et al. Development and validation of a novel Leishmania donovani screening cascade for high-throughput screening using a novel axenic assay with high predictivity of leishmanicidal intracellular activity. PLoS Negl. Trop. Dis. 9, e0004094 (2015).
Henderson, C. J., Pass, G. J. & Wolf, C. R. The hepatic cytochrome P450 reductase null mouse as a tool to identify a successful candidate entity. Toxicol. Lett. 162, 111–117 (2006).
Miles, T. J. & Thomas, M. G. Pyrazolo[3,4-d]pyrimidin derivative and its use for the treatment of leishmaniasis. WIPO patent WO/2016/116563 (2016).
Ding, Q., Jiang, N. & Roberts, J. L. Pyrazolo pyrimidines. WIPO patent WO/2005/121107 (2005).
Bantscheff, M. et al. Quantitative chemical proteomics reveals mechanisms of action of clinical ABL kinase inhibitors. Nat. Biotechnol. 25, 1035–1044 (2007).
Terstappen, G. C., Schlüpen, C., Raggiaschi, R. & Gaviraghi, G. Target deconvolution strategies in drug discovery. Nat. Rev. Drug Discov. 6, 891–903 (2007).
Park, J., Koh, M. & Park, S. B. From noncovalent to covalent bonds: a paradigm shift in target protein identification. Mol. Biosyst. 9, 544–550 (2013).
Lee, H. & Lee, J. W. Target identification for biologically active small molecules using chemical biology approaches. Arch. Pharm. Res. 39, 1193–1201 (2016).
Ursu, A. & Waldmann, H. Hide and seek: identification and confirmation of small molecule protein targets. Bioorg. Med. Chem. Lett. 25, 3079–3086 (2015).
Urbaniak, M. D., Guther, M. L. S. & Ferguson, M. A. J. Comparative SILAC proteomic analysis of Trypanosoma brucei bloodstream and procyclic lifecycle stages. PLoS ONE 7, e36619 (2012).
Liu, Y. & Gray, N. S. Rational design of inhibitors that bind to inactive kinase conformations. Nat. Chem. Biol. 2, 358–364 (2006).
Zhang, L. et al. Design, synthesis, and biological evaluation of pyrazolopyrimidine-sulfonamides as potent multiple-mitotic kinase (MMK) inhibitors (part I). Bioorg. Med. Chem. Lett. 21, 5633–5637 (2011).
Freyne, E. J. E. et al. Pyrazolopyrimidines as cell cycle kinase inhibitors. WIPO patent WO/2006/074984 (2006).
Rogers, M. B. et al. Chromosome and gene copy number variation allow major structural change between species and strains of Leishmania. Genome Res. 21, 2129–2142 (2011).
Downing, T. et al. Whole genome sequencing of multiple Leishmania donovani clinical isolates provides insights into population structure and mechanisms of drug resistance. Genome Res. 21, 2143–2156 (2011).
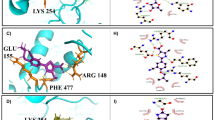
Monnerat, S. et al. Identification and functional characterisation of CRK12:CYC9, a novel cyclin-dependent kinase (CDK)–cyclin complex in Trypanosoma brucei. PLoS ONE 8, e67327 (2013).
Hassan, P., Fergusson, D., Grant, K. M. & Mottram, J. C. The CRK3 protein kinase is essential for cell cycle progression of Leishmania mexicana. Mol. Biochem. Parasitol. 113, 189–198 (2001).
Tu, X. & Wang, C. C. Pairwise knockdowns of cdc2-related kinases (CRKs) in Trypanosoma brucei identified the CRKs for G1/S and G2/M transitions and demonstrated distinctive cytokinetic regulations between two developmental stages of the organism. Eukaryot. Cell 4, 755–764 (2005).
Médard, G. et al. Optimized chemical proteomics assay for kinase inhibitor profiling. J. Proteome Res. 14, 1574–1586 (2015).
Bergamini, G. et al. A selective inhibitor reveals PI3Kγ dependence of TH17 cell differentiation. Nat. Chem. Biol. 8, 576–582 (2012).
Bantscheff, M. et al. Chemoproteomics profiling of HDAC inhibitors reveals selective targeting of HDAC complexes. Nat. Biotechnol. 29, 255–265 (2011).
Bradley, D. J. & Kirkley, J. Regulation of Leishmania populations within the host. I. The variable course of Leishmania donovani infections in mice. Clin. Exp. Immunol. 30, 119–129 (1977).
Croft, S. L., Snowdon, D. & Yardley, V. The activities of four anticancer alkyllysophospholipids against Leishmania donovani, Trypanosoma cruzi and Trypanosoma brucei. J. Antimicrob. Chemother. 38, 1041–1047 (1996).
Seifert, K. & Croft, S. L. In vitro and in vivo interactions between miltefosine and other antileishmanial drugs. Antimicrob. Agents Chemother. 50, 73–79 (2006).
Escobar, P., Yardley, V. & Croft, S. L. Activities of hexadecylphosphocholine (miltefosine), AmBisome, and sodium stibogluconate (Pentostam) against Leishmania donovani in immunodeficient scid mice. Antimicrob. Agents Chemother. 45, 1872–1875 (2001).
Acknowledgements
The authors acknowledge the Wellcome Trust for funding (grants 092340, 105021, 100476, 101842, 079838 and 098051).
Reviewer information
Nature thanks R. Guy, J. Mottram and the other anonymous reviewer(s) for their contribution to the peer review of this work.
Author information
Authors and Affiliations
Contributions
In brief, S.W., M.D.U., T.D.O, H.P., M.Bo. and S.M. carried out the mode of action, genomic and proteomic studies. M.T., S.P. and S.A. carried out the chemistry studies. M.D.R., S.M., L.M.M. and L.Sa. carried out the parasite screening. S.C., L.S., F.R.C.S. and P.C. carried out the drug metabolism and pharmacokinetic studies. F.Z. and N.H. carried out the molecular modelling. R.L. and S.G. carried out the safety studies. S.W., M.T., S.P., M.D.R., R.L., S.G., M.D.U., L.M.M., F.Z., M.Be., G.D., D.W.G., S.G.-D., S.D., J.M.F., P.W.G., M.A.J.F., A.H.F., T.J.M., K.D.R. and I.H.G. designed experiments, managed parts of the project and contributed to the writing. See Supplementary Information for further details.
Corresponding authors
Ethics declarations
Competing interests
These authors have shares in GlaxoSmithKline: P.G.W., S.D., T.J.M., K.D.R., S.C., R.L., S.G., M.Bo., H.P., P.C., G.D., D.G., S.G.-D. and J.M.F. The other authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data figures and tables
Extended Data Fig. 1 Rate-of-kill of L. donovani axenic amastigotes by compound 7.
Chart shows relative luminescence units (RLU) versus time from axenic amastigote rate-of-kill experiment with compound 7 (representative results for one of two independent experiments are shown; data are mean and s.d. of three technical replicates). Concentrations are as follows (µM): 50, open circles; 16.7, closed circles; 5.6, open squares; 1.85, closed squares; 0.62, open triangles; 0.21, closed triangles; 0.069, open inverted triangles; 0.023, closed inverted triangles, 0.0076, open diamonds and 0.0025, closed diamonds.
Extended Data Fig. 2 Linker-containing target molecules synthesized for chemical proteomic experiments and their corresponding EC50 values.
Potencies of the compounds in the cidal axenic and intra-macrophage assays are shown; data are from at least three independent replicates.
Supplementary information
Supplementary Information
This file contains author contributions, methods, characterisation of compounds and ethical statements. It also contains supplementary figures S1-S74 and supplementary tables S1-S9.
Supplementary Data
This file contains Proteomic data from the work at Cellzome.
Source Data
Rights and permissions
About this article
Cite this article
Wyllie, S., Thomas, M., Patterson, S. et al. Cyclin-dependent kinase 12 is a drug target for visceral leishmaniasis. Nature 560, 192–197 (2018). https://doi.org/10.1038/s41586-018-0356-z
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41586-018-0356-z
This article is cited by
-
In vitro evolution and whole genome analysis to study chemotherapy drug resistance in haploid human cells
Scientific Reports (2024)
-
Identification of Novel Antileishmanial Chemotypes By High-Throughput Virtual and In Vitro Screening
Acta Parasitologica (2024)
-
Some novel peptides containing a modified pyrazolopyrimidine moiety: design, synthesis, and in vitro antibacterial screening
Applied Biological Chemistry (2023)
-
Anti-trypanosomatid drug discovery: progress and challenges
Nature Reviews Microbiology (2023)
-
The emerging role of mass spectrometry-based proteomics in drug discovery
Nature Reviews Drug Discovery (2022)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.