Abstract
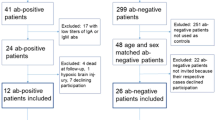
In 1983, reports of antibodies in subjects with major depressive disorder (MDD) to an as-yet uncharacterized infectious agent associated with meningoencephalitis in horses and sheep led to molecular cloning of the genome of a novel, negative-stranded neurotropic virus, Borna disease virus (BDV). This advance has enabled the development of new diagnostic assays, including in situ hybridization, PCR and serology based on recombinant proteins. Since these assays were first implemented in 1990, more than 80 studies have reported an association between BDV and a wide range of human illnesses that include MDD, bipolar disorder (BD), schizophrenia (SZ), anxiety disorder, chronic fatigue syndrome, multiple sclerosis, amyotrophic lateral sclerosis, dementia and glioblastoma multiforme. However, to date there has been no blinded case–control study of the epidemiology of BDV infection. Here, in a United States-based, multi-center, yoked case–control study with standardized methods for clinical assessment and blinded serological and molecular analysis, we report the absence of association of psychiatric illness with antibodies to BDV or with BDV nucleic acids in serially collected serum and white blood cell samples from 396 subjects, a study population comprised of 198 matched pairs of patients and healthy controls (52 SZ/control pairs, 66 BD/control pairs and 80 MDD/control pairs). Our results argue strongly against a role for BDV in the pathogenesis of these psychiatric disorders.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout
Similar content being viewed by others
Accession codes
References
Rott R, Herzog S, Fleischer B, Winokur A, Amsterdam J, Dyson W et al. Detection of serum antibodies to Borna disease virus in patients with psychiatric disorders. Science 1985; 228: 755–756.
Amsterdam J, Winokur A, Dyson W, Herzog S, Gonzalez F, Rott R et al. Borna Disease Virus: a possible etiologic factor in human affective disorders? Arch Gen Psychiatry 1985; 42: 1093–1096.
Hornig M, Briese T, Lipkin WI . Borna disease virus. J Neurovirol 2003; 9: 259–273.
Chalmers RM, Thomas DR, Salmon RL . Borna disease virus and the evidence for human pathogenicity: a systematic review. QJM 2005; 98: 255–274.
Yolken RH, Torrey EF . Viruses, schizophrenia, and bipolar disorder. Clin Microbiol Rev 1995; 8: 131–145.
Lipkin WI, Hornig M . Neurovirology. Microbes and the brain. Lancet 1998; 352 (Suppl 4): SIV21.
Schlitt M, Lakeman FD, Whitly RJ . Psychoses and herpes simplex encephalitis. South Med J 1985; 78: 1347–1350.
Steinberg D, Hirsch SR, Marston SD, Reynolds K, Sutton NP . Influenza infection causing manic psychosis. Br J Psychiatry 1972; 120: 531–535.
Goswami U, Shankar SK, Channabasavanna SM, Chattopadhyay A . Psychiatric presentations in rabies. A clinico-pathologic report from South India with a review of literature. Trop Geogr Med 1984; 36: 77–81.
Menninger KA . Psychoses associated with influenza, I. General data: statistical analysis. JAMA 1919; 72: 235–241.
Mednick SA, Machón RA, Huttunen MO, Bonett D . Adult schizophrenia following prenatal exposure to an influenza epidemic. Arch Gen Psychiatry 1988; 45: 189–192.
O'Callaghan E, Sham P, Takei N, Glover G, Murray RM . Schizophrenia after prenatal exposure to 1957 A2 influenza epidemic. Lancet 1991; 337: 1248–1250.
Lipkin WI, Schneemann A, Solbrig MV . Borna disease virus: implications for neuropsychiatric illness. Trends Microbiol 1995; 3: 64–69.
Ludwig H, Bode L, Gosztonyi G . Borna disease: a persistent disease of the central nervous system. Prog Med Virol 1988; 35: 107–151.
Stitz L, Krey H, Ludwig H . Borna disease in rhesus monkeys as a model for uveo-cerebral symptoms. J Med Virol 1981; 6: 333–340.
Kistler AL, Gancz A, Clubb S, Skewes-Cox P, Fischer K, Sorber K et al. Recovery of divergent avian bornaviruses from cases of proventricular dilatation disease: identification of a candidate etiologic agent. Virol J 2008; 5: 88.
Honkavuori KS, Shivaprasad HL, Williams BL, Quan PL, Hornig M, Street C et al. Novel borna virus in psittacine birds with proventricular dilatation disease. Emerg Infect Dis 2008; 14: 1883–1886.
Horie M, Honda T, Suzuki Y, Kobayashi Y, Daito T, Oshida T et al. Endogenous non-retroviral RNA virus elements in mammalian genomes. Nature 2010; 463: 84–87.
Belyi VA, Levine AJ, Skalka AM . Unexpected inheritance: multiple integrations of ancient bornavirus and ebolavirus/marburgvirus sequences in vertebrate genomes. PLoS Pathog 2010; 6: e1001030.
Bode L, Ferszt R, Czech G . Borna disease virus infection and affective disorders in man. Arch Virol Suppl 1993; 7: 159–167.
Rackova S, Janu L, Kabickova H . Borna disease virus (BDV) circulating immunocomplex positivity in addicted patients in the Czech Republic: a prospective cohort analysis. BMC Psychiatry 2010; 10: 70.
Heinrich A, Adamaszek M . Anti-Borna disease virus antibody responses in psychiatric patients: longterm follow up. Psychiatry Clin Neurosci 2010; 64: 255–261.
Evengård B, Briese T, Lindh G, Lee S, Lipkin WI . Absence of evidence of Borna disease virus infection in Swedish patients with Chronic Fatigue Syndrome. J Neurovirol 1999; 5: 495–499.
Bode L, Reckwald P, Severus WE, Stoyloff R, Ferszt R, Dietrich DE et al. Borna disease virus-specific circulating immune complexes, antigenemia, and free antibodies—the key marker triplet determining infection and prevailing in severe mood disorders. Mol Psychiatry 2001; 6: 481–491.
Wolff T, Heins G, Pauli G, Burger R, Kurth R . Failure to detect Borna disease virus antigen and RNA in human blood [letter]. J Clin Virol 2006; 36: 309–311.
Kishi M, Nakaya T, Nakamura Y, Kakinuma M, Takahashi TA, Sekiguchi S et al. Prevalence of Borna disease virus RNA in peripheral blood mononuclear cells from blood donors. Med Microbiol Immunol 1995; 184: 135–138.
Wolff T, Burger R, Kurth R . Re: Absence of Borna virus in human blood [reply to letter]. J Clin Virol 2006; 36: 314.
Lipkin WI, Hornig M, Briese T . Borna disease virus and neuropsychiatric disease—a reappraisal. Trends Microbiol 2001; 9: 295–298.
American Psychiatric Association. Diagnostic and Statistical Manual of Mental Disorders, 4th edn. Text Revision (DSM-IV-TR). American Psychiatric Press: Washington, DC, USA, 2000.
First MB, Gibbon M, Spitzer RL, Williams JBW . Structured Clinical Interview for DSM-IV Axis I Disorders. Biometrics Research: New York, NY, USA, 1996.
Guy W . ECDEU Assessment Manual for Psychopharmacology. U.S. Department of Health, Education and Welfare: Washington, DC, USA, 1976, pp 218–222.
Hamilton M . A rating scale for depression. Neurol Neurosurg Psychiatry 1960; 23: 56–62.
Williams JBW . A structured interview guide for the Hamilton Depression Rating Scale. Arch Gen Psychiatry 1988; 45: 742–747.
Young RC, Biggs JT, Zeigler VE, Meyer DA . A rating scale for mania: reliability, validity and sensitivity. Br J Psychiatry 1978; 133: 429–433.
Kay SR, Opler LA, Lindenmayer JP . Reliability and validity of the positive and negative syndrome scale for schizophrenics. Psychiatry Res 1988; 23: 99–110.
Amsterdam JD, Hornig-Rohan M . Treatment algorithms in treatment-resistant depression. In: Hornig-Rohan M, Amsterdam JD (eds). Psychiatric Clinics of North America: Treatment-Resistant Depression. W.B. Saunders: Philadelphia, 1996, pp 371–386.
Khot V, DeVane CL, Korpi ER, Venable D, Bigelow LB, Wyatt RJ et al. The assessment and clinical implications of haloperidol acute-dose, steady-state, and withdrawal pharmacokinetics. J Clin Psychopharmacol 1993; 13: 120–127.
Van Putten T, Marder SR, Mintz J, Poland RE . Haloperidol plasma levels and clinical response: a therapeutic window relationship. Am J Psychiatry 1992; 149: 500–505.
Tamura K, Dudley J, Nei M, Kumar S . MEGA4: Molecular Evolutionary Genetics Analysis (MEGA) software version 4.0. Mol Biol Evol 2007; 24: 1596–1599.
Briese T, Hatalski CG, Kliche S, Park YS, Lipkin WI . An enzyme-linked-immunosorbent assay for detecting antibodies to Borna disease virus-specific proteins. J Clin Microbiol 1995; 69: 348–351.
Billich C, Sauder C, Frank R, Herzog S, Bechter K, Takahashi K et al. High-avidity human serum antibodies recognizing linear epitopes of Borna disease virus proteins. Biol Psychiatry 2002; 51: 979–987.
Acknowledgements
This study was supported by NIH award MH57467 (WIL). We thank Meera Bhat for data derived from linkage to the United States Census Bureau database, and Vishal Kapoor for assistance with Western immunoblot analyses.
Author information
Authors and Affiliations
Corresponding authors
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies the paper on the Molecular Psychiatry website
Rights and permissions
About this article
Cite this article
Hornig, M., Briese, T., Licinio, J. et al. Absence of evidence for bornavirus infection in schizophrenia, bipolar disorder and major depressive disorder. Mol Psychiatry 17, 486–493 (2012). https://doi.org/10.1038/mp.2011.179
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2011.179
Keywords
This article is cited by
-
Detection of bornavirus-reactive antibodies and BoDV-1 RNA only in encephalitis patients from virus endemic areas: a comparative serological and molecular sensitivity, specificity, predictive value, and disease duration correlation study
Infection (2024)
-
Antiviral treatment perspective against Borna disease virus 1 infection in major depression: a double-blind placebo-controlled randomized clinical trial
BMC Pharmacology and Toxicology (2020)
-
Bornavirus
Bundesgesundheitsblatt - Gesundheitsforschung - Gesundheitsschutz (2019)
-
Primary psychosis and Borna disease virus infection in Lithuania: a case control study
BMC Psychiatry (2016)
-
A novel intranuclear RNA vector system for long-term stem cell modification
Gene Therapy (2016)