Abstract
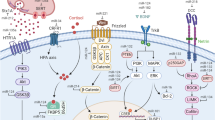
A central question in addiction is how drug-induced changes in synaptic signaling are converted into long-term neuroadaptations. Emerging evidence reveals that microRNAs (miRNAs) have a distinct role in this process through rapid response to cellular signals and dynamic regulation of local mRNA transcripts. Because each miRNA can target hundreds of mRNAs, relative changes in the expression of miRNAs can greatly impact cellular responsiveness, synaptic plasticity and transcriptional events. These diverse consequences of miRNA action occur through coordination with genes implicated in addictions, the most compelling of these being the neurotrophin BDNF, the transcription factor cAMP-responsive element-binding protein (CREB) and the DNA-binding methyl CpG binding protein 2 (MeCP2). In this study, we review the recent progress in the understanding of miRNAs in general mechanisms of plasticity and neuroadaptation and then focus on specific examples of miRNA regulation in the context of addiction. We conclude that miRNA-mediated gene regulation is a conserved means of converting environmental signals into neuronal response, which holds significant implications for addiction and other psychiatric illnesses.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout


Similar content being viewed by others
References
Perkins DO, Jeffries C, Sullivan P . Expanding the ‘central dogma’: the regulatory role of nonprotein coding genes and implications for the genetic liability to schizophrenia. Mol Psychiatry 2005; 10: 69–78.
Mattick JS . Non-coding RNAs: the architects of eukaryotic complexity. Embo Reports 2001; 2: 986–991.
Ambros V . The functions of animal microRNAs. Nature 2004; 431: 350–355.
Kim VN, Han J, Siomi MC . Biogenesis of small RNAs in animals. Nat Rev Mol Cell Biol 2009; 10: 126–139.
Chen CZ, Li L, Lodish HF, Bartel DP . MicroRNAs modulate hematopoietic lineage differentiation. Science 2004; 303: 83–86.
Hwang HW, Mendell JT . MicroRNAs in cell proliferation, cell death, and tumorigenesis. Br J Cancer 2006; 94: 776–780.
Sun JG, Liao RX, Qiu J, Jin JY, Wang XX, Duan YZ et al. Microarray-based analysis of microRNA expression in breast cancer stem cells. J Exp Clin Canc Res 2010; 29: 174.
Friedman RC, Farh KKH, Burge CB, Bartel DP . Most mammalian mRNAs are conserved targets of microRNAs. Genome Res 2009; 19: 92–105.
Hobert O . Gene regulation by transcription factors and microRNAs. Science 2008; 319: 1785–1786.
Ashraf SI, McLoon AL, Sclarsic SM, Kunes S . Synaptic protein synthesis associated with memory is regulated by the RISC pathway in Drosophila. Cell 2006; 124: 191–205.
Martin KC, Zukin RS . RNA trafficking and local protein synthesis in dendrites: an overview. J Neurosci 2006; 26: 7131–7134.
Lugli G, Torvik VI, Larson J, Smalheiser NR . Expression of microRNAs and their precursors in synaptic fractions of adult mouse forebrain. J Neurochem 2008; 106: 650–661.
Sempere LF, Freemantle S, Pitha-Rowe I, Moss E, Dmitrovsky E, Ambros V . Expression profiling of mammalian microRNAs uncovers a subset of brain-expressed microRNAs with possible roles in murine and human neuronal differentiation. Genome Biol 2004; 5: R13.
Abelson JF, Kwan KY, O'Roak BJ, Baek DY, Stillman AA, Morgan TM et al. Sequence variants in SLITRK1 are associated with Tourette's syndrome. Science 2005; 310: 317–320.
Urdinguio RG, Fernandez AF, Lopez-Nieva P, Rossi S, Huertas D, Kulis M et al. Disrupted microRNA expression caused by Mecp2 loss in a mouse model of Rett syndrome. Epigenetics 2010; 5: 656–663.
Kim J, Inoue K, Ishii J, Vanti WB, Voronov SV, Murchison E et al. A microRNA feedback circuit in midbrain dopamine neurons. Science 2007; 317: 1220–1224.
Maes OC, Chertkow HM, Wang E, Schipper HM . MicroRNA: implications for Alzheimer disease and other human CNS disorders. Curr Genomics 2009; 10: 154–168.
Bicker S, Schratt G . microRNAs: tiny regulators of synapse function in development and disease. J Cell Mol Med 2008; 12: 1466–1476.
Sethi P, Lukiw WJ . Micro-RNA abundance and stability in human brain: specific alterations in Alzheimer's disease temporal lobe neocortex. Neurosci Lett 2009; 459: 100–104.
Perkins DO, Jeffries CD, Jarskog LF, Thomson JM, Woods K, Newman MA et al. microRNA expression in the prefrontal cortex of individuals with schizophrenia and schizoaffective disorder. Genome Biol 2007; 8: R27.
Zhu YL, Kalbfleisch T, Brennan MD, Li Y . A microRNA gene is hosted in an intron of a schizophrenia-susceptibility gene. Schizophr Res 2009; 109: 86–89.
Huang WH, Li MD . Nicotine modulates expression of miR-140*, which targets the 3′-untranslated region of dynamin 1 gene (Dnm1). Int J Neuropsychopharmacol 2009; 12: 537–546.
Huang WH, Li MD . Differential allelic expression of dopamine D1 receptor gene (DRD1) is modulated by microRNA miR-504. Biol Psychiatry 2009; 65: 702–705.
Hyman SE, Malenka RC, Nestler EJ . Neural mechanisms of addiction: the role of reward-related learning and memory. Ann Rev Neurosci 2006; 29: 565–598.
Hyman SE, Malenka RC . Addiction and the brain: the neurobiology of compulsion and its persistence. Nat Rev Neurosci 2001; 2: 695–703.
Koob GF . The neurocircuitry of addiction: implications for treatment. Clin Neurosci Res 2005; 5: 89–101.
Kauer JA . Learning mechanisms in addiction: synaptic plasticity in the ventral tegmental area as a result of exposure to drugs of abuse. Annu Rev Physiol 2004; 66: 447–475.
Gerdeman GL, Partridge JG, Lupica CR, Lovinger DM . It could be habit forming: drugs of abuse and striatal synaptic plasticity. Trends Neurosci 2003; 26: 184–192.
Russo SJ, Dietz DM, Dumitriu D, Morrison JH, Malenka RC, Nestler EJ . The addicted synapse: mechanisms of synaptic and structural plasticity in nucleus accumbens. Trends Neurosci 2010; 33: 267–276.
Kalivas PW, Volkow N, Seamans J . Unmanageable motivation in addiction: a pathology in prefrontal-accumbens glutamate transmission. Neuron 2005; 45: 647–650.
Chang LF, Karin M . Mammalian MAP kinase signalling cascades. Nature 2001; 410: 37–40.
Paroo Z, Ye XC, Chen S, Liu QH . Phosphorylation of the human microRNA-generating complex mediates MAPK/Erk signaling. Cell 2009; 139: 112–122.
Pietrzykowski AZ . The role of microRNAs in drug addiction: a big lesson from tiny molecules. Int Rev Neurobiol 2010; 91: 1001–1005.
Smalheiser NR, Lugli G . microRNA regulation of synaptic plasticity. Neuromol Med 2009; 11: 133–140.
Acheson A, Conover JC, Fandl JP, Dechiara TM, Russell M, Thadani A et al. A Bdnf autocrine loop in adult sensory neurons prevents cell death. Nature 1995; 374: 450–453.
Huang EJ, Reichardt LF . Neurotrophins: roles in neuronal development and function. Ann Rev of Neurosci 2001; 24: 677–736.
Remenyi J, Hunter CJ, Cole C, Ando H, Impey S, Monk CE et al. Regulation of the miR-212/132 locus by MSK1 and CREB in response to neurotrophins. Biochem J 2010; 428: 281–291.
Schratt GM, Tuebing F, Nigh EA, Kane CG, Sabatini ME, Kiebler M et al. A brain-specific microRNA regulates dendritic spine development. Nature 2006; 439: 283–289.
Vo N, Klein ME, Varlamova O, Keller DM, Yamamoto T, Goodman RH et al. A cAMP-response element binding protein-induced microRNA regulates neuronal morphogenesis. Proc Natl Acad Sci U S A. 2005; 102: 16426–16431.
Hansen KF, Sakamoto K, Wayman GA, Impey S, Obrietan K . Transgenic miR132 alters neuronal spine density and impairs novel object recognition memory. Plos One 2010; 5: e15497.
Endo M, Ohashi K, Sasaki Y, Goshima Y, Niwa R, Uemura T et al. Control of growth cone motility and morphology by LIM kinase and slingshot via phosphorylation and dephosphorylation of cofilin. J Neurosci 2003; 23: 2527–2537.
Siegel G, Obernosterer G, Fiore R, Oehmen M, Bicker S, Christensen M et al. A functional screen implicates microRNA-138-dependent regulation of the depalmitoylation enzyme APT1 in dendritic spine morphogenesis. Nat Cell Biol 2009; 11: 705–U736.
Edbauer D, Neilson JR, Foster KA, Wang CF, Seeburg DP, Batterton MN et al. Regulation of synaptic structure and function by FMRP-associated microRNAs miR-125b and miR-132. Neuron 2010; 65: 373–384.
Barco A, Alarcon JM, Kandel ER . Expression of constitutively active CREB protein facilitates the late phase of long-term potentiation by enhancing synaptic capture. Cell 2002; 108: 689–703.
Benito E, Barco A . CREB's control of intrinsic and synaptic plasticity: implications for CREB-dependent memory models. Trends Neurosci 2010; 33: 230–240.
Zhou Y, Won J, Karlsson MG, Zhou M, Rogerson T, Balaji J et al. CREB regulates excitability and the allocation of memory to subsets of neurons in the amygdala. Nat Neurosci 2009; 12: 1438–1443.
Dong Y, Green T, Saal D, Marie H, Neve R, Nestler EJ et al. CREB modulates excitability of nucleus accumbens neurons. Nat Neurosci 2006; 9: 475–477.
Marin MT, Berkow A, Golden SA, Koya E, Planeta CS, Hope BT . Context-specific modulation of cocaine-induced locomotor sensitization and ERK and CREB phosphorylation in the rat nucleus accumbens. Eur J Neurosci 2009; 30: 1931–1940.
Moron JA, Gullapalli S, Taylor C, Gupta A, Gomes I, Devi LA . Modulation of opiate-related signaling molecules in morphine-dependent conditioned behavior: conditioned place preference to morphine induces CREB phosphorylation. Neuropsychopharmacology 2010; 35: 955–966.
Wu J, Xie XH . Comparative sequence analysis reveals an intricate network among REST, CREB and miRNA in mediating neuronal gene expression. Genome Biol 2006; 7: R85.
Becskei A, Serrano L . Engineering stability in gene networks by autoregulation. Nature 2000; 405: 590–593.
Lagos-Quintana M, Rauhut R, Yalcin A, Meyer J, Lendeckel W, Tuschl T . Identification of tissue-specific microRNAs from mouse. Current Biol 2002; 12: 735–739.
Visvanathan J, Lee S, Lee B, Lee JW, Lee SK . The microRNA miR-124 antagonizes the anti-neural REST/SCP1 pathway during embryonic CNS development. Genes Dev 2007; 21: 744–749.
Conaco C, Otto S, Han JJ, Mandel G . Reciprocal actions of REST and a microRNA promote neuronal identity. Proc Natl Acad Sci U S A. 2006; 103: 2422–2427.
Rajasethupathy P, Fiumara F, Sheridan R, Betel D, Puthanveettil SV, Russo JJ et al. Characterization of small RNAs in Aplysia reveals a role for miR-124 in constraining synaptic plasticity through CREB. Neuron 2009; 63: 803–817.
Gao J, Wang WY, Mao YW, Graff J, Guan JS, Pan L et al. A novel pathway regulates memory and plasticity via SIRT1 and miR-134. Nature 2010; 466: 1105–1109.
Renthal W, Kumar A, Xiao GH, Wilkinson M, Covington HE, Maze I et al. Genome-wide analysis of chromatin regulation by cocaine reveals a role for sirtuins. Neuron 2009; 62: 335–348.
Alvarez-Saavedra M, Antoun G, Yanagiya A, Oliva-Hernandez R, Cornejo-Palma D, Perez-Iratxeta C et al. miRNA-132 orchestrates chromatin remodeling and translational control of the circadian clock. Hum Mol Genet 2011; 20: 731–751.
Georgel PT, Horowitz-Scherer RA, Adkins N, Woodcock CL, Wade PA, Hansen JC . Chromatin compaction by human MeCP2. Assembly of novel secondary chromatin structures in the absence of DNA methylation. J Biol Chem 2003; 278: 32181–32188.
Jones PL, Veenstra GJ, Wade PA, Vermaak D, Kass SU, Landsberger N et al. Methylated DNA and MeCP2 recruit histone deacetylase to repress transcription. Nat Genet 1998; 19: 187–191.
Chahrour M, Jung SY, Shaw C, Zhou X, Wong ST, Qin J et al. MeCP2, a key contributor to neurological disease, activates and represses transcription. Science 2008; 320: 1224–1229.
Martinowich K, Hattori D, Wu H, Fouse S, He F, Hu Y et al. DNA methylation-related chromatin remodeling in activity-dependent Bdnf gene regulation. Science 2003; 302: 890–893.
Wu H, Tao JF, Chen PJ, Shahab A, Ge WH, Hart RP et al. Genome-wide analysis reveals methyl-CpG-binding protein 2-dependent regulation of microRNAs in a mouse model of Rett syndrome. Proc Natl Acad Sci U S A. 2010; 107: 18161–18166.
Larimore JL, Chapleau CA, Kudo S, Theibert A, Percy AK, Pozzo-Miller L . Bdnf overexpression in hippocampal neurons prevents dendritic atrophy caused by Rett-associated MECP2 mutations. Neurobiol Dis 2009; 34: 199–211.
Klein ME, Lioy DT, Ma L, Impey S, Mandel G, Goodman RH . Homeostatic regulation of MeCP2 expression by a CREB-induced microRNA. Nat Neurosci 2007; 10: 1513–1514.
Lonetti G, Angelucci A, Morando L, Boggio EM, Giustetto M, Pizzorusso T . Early environmental enrichment moderates the behavioral and synaptic phenotype of MeCP2 null mice. Biol Psychiatry 2010; 67: 657–665.
Caccamo A, Maldonado MA, Bokov AF, Majumder S, Oddo S . CBP gene transfer increases BDNF levels and ameliorates learning and memory deficits in a mouse model of Alzheimer's disease. Proc Natl Acad Sci USA 2010; 107: 22687–22692.
Hollander JA, Im HI, Amelio AL, Kocerha J, Bali P, Lu Q et al. Striatal microRNA controls cocaine intake through CREB signaling. Nature 2010; 466: 197–202.
Belin D, Everitt BJ . Cocaine seeking habits depend upon doparnine-dependent serial connectivity linking the ventral with the dorsal striatum. Neuron 2008; 57: 432–441.
Nestler EJ, Aghajanian GK . Molecular and cellular basis of addiction. Science 1997; 278: 58–63.
Carlezon WA, Thome J, Olson VG, Lane-Ladd SB, Brodkin ES, Hiroi N et al. Regulation of cocaine reward by CREB. Science 1998; 282: 2272–2275.
Im HI, Hollander JA, Bali P, Kenny PJ . MeCP2 controls BDNF expression and cocaine intake through homeostatic interactions with microRNA-212. Nat Neurosci 2010; 13: 1120–1127.
Chang Q, Khare G, Dani V, Nelson S, Jaenisch R . The disease progression of Mecp2 mutant mice is affected by the level of BDNF expression. Neuron 2006; 49: 341–348.
Zhou ZL, Hong EJ, Cohen S, Zhao WN, Ho HYH, Schmidt L et al. Brain-specific phosphorylation of MeCP2 regulates activity-dependent Bdnf transcription, dendritic growth, and spine maturation. Neuron 2006; 52: 255–269.
Schoenbaum G, Stalnaker TA, Shaham Y . A role for BDNF in cocaine reward and relapse. Nat Neurosci 2007; 10: 935–936.
Horger BA, Iyasere CA, Berhow MT, Messer CJ, Nestler EJ, Taylor JR . Enhancement of locomotor activity and conditioned reward to cocaine by brain-derived neurotrophic factor. J Neurosci 1999; 19: 4110–4122.
Choi KH, Whisler K, Graham DL, Self DW . Antisense-induced reduction in nucleus accumbens cyclic AMP response element binding protein attenuates cocaine reinforcement. Neuroscience 2006; 137: 373–383.
Le Foll B, Diaz J, Sokoloff P . A single cocaine exposure increases BDNF and D-3 receptor expression: implications for drug-conditioning. Neuroreport 2005; 16: 175–178.
Chandrasekar V, Dreyer JL . microRNAs miR-124, let-7d and miR-181a regulate cocaine-induced plasticity. Mol Cell Neurosci 2009; 42: 350–362.
Chandrasekar V, Dreyer JL . Regulation of MiR-124, Let-7d, and MiR-181a in the accumbens affects the expression, extinction, and reinstatement of cocaine-induced conditioned place preference. Neuropsychopharmacology 2011; 36: 1149–1164.
Luscher C, Malenka RC . Drug-evoked synaptic plasticity in addiction: from molecular changes to circuit remodeling. Neuron 2011; 69: 650–663.
Mellios N, Huang HS, Grigorenko A, Rogaev E, Akbarian S . A set of differentially expressed miRNAs, including miR-30a-5p, act as post-transcriptional inhibitors of BDNF in prefrontal cortex. Hum Mol Genet 2008; 17: 3030–3042.
Kumar A, Choi KH, Renthal W, Tsankova NM, Theobald DEH, Truong HT et al. Chromatin remodeling is a key mechanism underlying cocaine-induced plasticity in striatum. Neuron 2005; 48: 303–314.
Sadri-Vakili G, Kumaresan V, Schmidt HD, Famous KR, Chawla P, Vassoler FM et al. Cocaine-induced chromatin remodeling increases brain-derived neurotrophic factor transcription in the rat medial prefrontal cortex, which alters the reinforcing efficacy of cocaine. J Neurosci 2010; 30: 11735–11744.
Self DW, Genova LM, Hope BT, Barnhart WJ, Spencer JJ, Nestler EJ . Involvement of cAMP-dependent protein kinase in the nucleus accumbens in cocaine self-administration and relapse of cocaine-seeking behavior. J Neurosci 1998; 18: 1848–1859.
Graham DL, Edwards S, Bachtell RK, DiLeone RJ, Rios M, Self DW . Dynamic BDNF activity in nucleus accumbens with cocaine use increases self-administration and relapse. Nat Neurosci 2007; 10: 1029–1037.
Hnasko TS, Sotak BN, Palmiter RD . Morphine reward in dopamine-deficient mice. Nature 2005; 438: 854–857.
Huang W, Ma JZ, Payne TJ, Beuten J, Dupont RT, Li MD . Significant association of DRD1 with nicotine dependence. Hum Genet 2008; 123: 133–140.
Xing B, Kong H, Meng X, Wei SG, Xu M, Li SB . Dopamine D1 but not D3 receptor is critical for spatial learning and related signaling in the hippocampus. Neurosci 2010; 169: 1511–1519.
Brunzell DH, Mineur YS, Neve RL, Picciotto MR . Nucleus accumbens CREB activity is necessary for nicotine conditioned place preference. Neuropsychopharmacology 2009; 34: 1993–2001.
Xu Q, Huang WH, Payne TJ, Ma JZ, Li MD . Detection of genetic association and a functional polymorphism of dynamin 1 gene with nicotine dependence in European and African Americans. Neuropsychopharmacology 2009; 34: 1351–1359.
Koob GF, Nestler EJ . The neurobiology of drug addiction. J Neuropsychiatry Clin Neurosci 1997; 9: 482–497.
Whistler JL, Chuang HH, Chu P, Jan LY, von Zastrow M . Functional dissociation of mu opioid receptor signaling and endocytosis: implications for the biology of opiate tolerance and addiction. Neuron 1999; 23: 737–746.
Artalejo CR, Elhamdani A, Palfrey HC . Sustained stimulation shifts the mechanism of endocytosis from dynamin-1-dependent rapid endocytosis to clathrin- and dynamin-2-mediated slow endocytosis in chromaffin cells (vol 99, pg 6358, 2002). Proc Natl Acad Sci U S A. 2002; 99: 9082.
Beveridge NJ, Tooney PA, Carroll AP, Gardiner E, Bowden N, Scott RJ et al. Dysregulation of miRNA 181b in the temporal cortex in schizophrenia. Hum Mol Genet 2008; 17: 1156–1168.
Volkow ND . Substance use disorders in schizophrenia - clinical implications of comorbidity. Schizophr Bull 2009; 35: 469–472.
Williams JM, Gandhi KK, Lu SE, Kumar S, Shen JW, Foulds J et al. Higher nicotine levels in schizophrenia compared with controls after smoking a single cigarette. Nicotine Tob Res 2010; 12: 855–859.
Lukiw WJ . Micro-RNA speciation in fetal, adult and Alzheimer's disease hippocampus. Neuroreport 2007; 18: 297–300.
Hebert SS, Horre K, Nicolai L, Papadopoulou AS, Mandemakers W, Silahtaroglu AN et al. Loss of microRNA cluster miR-29a/b-1 in sporadic Alzheimer's disease correlates with increased BACE1/beta-secretase expression. Proc Natl Acad Sci U S A 2008; 105: 6415–6420.
Holsinger RMD, McLean CA, Masters CL, Evin G, Beyreuther K . BACE and beta-secretase product CTF beta are increased in sporadic Alzheimer's disease brain. Neurobiol Aging 2002; 23: S177.
Fukumoto H, Cheung B, Hyman B, Irizarry M . beta-site amyloid precursor protein cleaving enzyme (BACE) activity is increased in temporal neocortex of Alzheimer's disease. Neurobiol Aging 2002; 23: S181.
Kihara T, Shimohama S, Sawada H, Kimura J, Kume T, Kochiyama H et al. Nicotinic receptor stimulation protects neurons against beta-amyloid toxicity. Ann Neurol 1997; 42: 159–163.
Boissonneault V, Plante I, Rivest S, Provost P . MicroRNA-298 and MicroRNA-328 regulate expression of mouse beta-amyloid precursor protein-converting enzyme 1. J Biol Chem 2009; 284: 1971–1981.
Dolganiuc A, Petrasek J, Kodys K, Catalano D, Mandrekar P, Velayudham A et al. MicroRNA expression profile in Lieber-DeCarli diet-induced alcoholic and methionine choline deficient diet-induced nonalcoholic steatohepatitis models in mice. Alcohol Clin Exp Res 2009; 33: 1704–1710.
Pietrzykowski AZ, Friesen RM, Martin GE, Puig SI, Nowak CL, Wynne PM et al. Posttranscriptional regulation of BK channel splice variant stability by miR-9 underlies neuroadaptation to alcohol. Neuron 2008; 59: 274–287.
Shipston MJ . Alternative splicing of potassium channels: a dynamic switch of cellular excitability. Trends Cell Biol 2001; 11: 353–358.
Martin G, Puig SI, Pietrzykowski A, Zadek P, Emery P, Treistman S . Restricted cellular localization of a specific BK-channel subtype controls ethanol sensitivity in the nucleus accumbens. Alcohol Clin Exp Res 2004; 28: 61A.
Volkow ND, Wang GJ, Begleiter H, Porjesz B, Fowler JS, Telang F et al. High levels of dopamine D-2 receptors in unaffected members of alcoholic families:possible protective factors. Arch Gen Psychiatry 2006; 63: 999–1008.
Saba LM, Bennett B, Hoffman PL, Barcomb K, Ishii T, Kechris K et al. A systems genetic analysis of alcohol drinking by mice, rats and men: influence of brain GABAergic transmission. Neuropharmacology 2011; 60: 1269–1280.
Liu J, Yang AR, Kelly T, Puche A, Esoga C, June Jr HL et al. Binge alcohol drinking is associated with GABAA alpha2-regulated Toll-like receptor 4 (TLR4) expression in the central amygdala. Proc Natl Acad Sci USA 2011; 108: 4465–4470.
McClung CA, Nestler EJ . Regulation of gene expression and cocaine reward by CREB and Delta FosB. Nat Neurosci 2003; 6: 1208–1215.
Long JY, Wang Y, Wang WJ, Chang BHJ, Danesh FR . Identification of microRNA-93 as a novel regulator of vascular endothelial growth factor in hyperglycemic conditions. J Biol Chem 2010; 285: 23455–23463.
Santarelli DM, Beveridge NJ, Tooney PA, Cairns MJ . Upregulation of dicer and microRNA expression in the dorsolateral prefrontal cortex Brodmann area 46 in schizophrenia. Biol Psychiatry 2011; 69: 180–187.
Acknowledgements
The preparation of this report was in part supported by NIH Grants DA-12844 and DA-13787 to Ming D Li. We thank Dr David L Bronson for his excellent editing of this report.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
PowerPoint slides
Rights and permissions
About this article
Cite this article
Li, M., van der Vaart, A. MicroRNAs in addiction: adaptation's middlemen?. Mol Psychiatry 16, 1159–1168 (2011). https://doi.org/10.1038/mp.2011.58
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/mp.2011.58
Keywords
This article is cited by
-
Plasma levels of CGRP and expression of specific microRNAs in blood cells of episodic and chronic migraine subjects: towards the identification of a panel of peripheral biomarkers of migraine?
The Journal of Headache and Pain (2020)
-
Investigating the genetic profile of dopaminergic neurons in the VTA in response to perinatal nicotine exposure using mRNA-miRNA analyses
Scientific Reports (2018)
-
Decoding the ubiquitous role of microRNAs in neurogenesis
Molecular Neurobiology (2017)
-
MicroRNA-30a-5p in the prefrontal cortex controls the transition from moderate to excessive alcohol consumption
Molecular Psychiatry (2015)
-
Placental miRNA expression profiles are associated with measures of infant neurobehavioral outcomes
Pediatric Research (2013)