Key Points
-
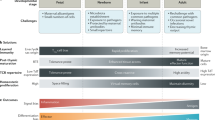
Recent thymic emigrants (RTEs), the youngest peripheral T cells, undergo progressive phenotypic and functional maturation during their first few weeks in the peripheral lymphoid tissue. Here, RTEs and their more mature, although still naive, T cell counterparts occupy largely overlapping niches.
-
RTEs are a functionally distinct subset of the peripheral T cell pool, characterized by reduced cytokine and transcription factor expression compared with mature naive T cells.
-
Post-thymic maturation requires thymic egress and access to secondary lymphoid organs; however, the molecular trigger(s) that drive RTE maturation are still unknown, although the transcriptional repressor NKAP may be required.
-
RTEs are not simply the cellular midpoint between single-positive progenitor thymocytes and mature peripheral T cells, and this suggests that transit through this developmental stage is necessary.
-
Post-thymic maturation may be crucial for ensuring that RTEs are self tolerant or that they are fit to receive homeostatic signals.
Abstract
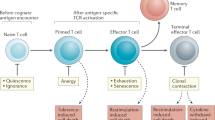
T cell maturation was once thought to occur entirely within the thymus. Now, evidence is mounting that the youngest peripheral T cells in both mice and humans comprise a distinct population from their more mature, yet still naive, counterparts. These cells, termed recent thymic emigrants (RTEs), undergo a process of post-thymic maturation that can be monitored at the levels of cell phenotype and immune function. Understanding this final maturation step in the process of generating useful and safe T cells is of clinical relevance, given that RTEs are over-represented in neonates and in adults recovering from lymphopenia. Post-thymic maturation may function to ensure T cell fitness and self tolerance.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
References
Hale, J. S., Boursalian, T. E., Turk, G. L. & Fink, P. J. Thymic output in aged mice. Proc. Natl Acad. Sci. USA 103, 8447–8452 (2006).
Vrisekoop, N. et al. Sparse production but preferential incorporation of recently produced naive T cells in the human peripheral pool. Proc. Natl Acad. Sci. USA 105, 6115–6120 (2008).
Haines, C. J. et al. Human CD4+ T cell recent thymic emigrants are identified by protein tyrosine kinase 7 and have reduced immune function. J. Exp. Med. 206, 275–285 (2009).
Yager, E. J. et al. Age-associated decline in T cell repertoire diversity leads to holes in the repertoire and impaired immunity to influenza virus. J. Exp. Med. 205, 711–723 (2008).
Mackall, C. L., Hakim, F. T. & Gress, R. E. Restoration of T-cell homeostasis after T-cell depletion. Semin. Immunol. 9, 339–346 (1997).
Elgbratt, K., Kurlberg, G., Hahn-Zohric, M. & Hornquist, E. H. Rapid migration of thymic emigrants to the colonic mucosa in ulcerative colitis patients. Clin. Exp. Immunol. 162, 325–336 (2010).
Li, Y. et al. Decreased level of recent thymic emigrants in CD4+ and CD8+T cells from CML patients. J. Transl. Med. 8, 47 (2010).
Armengol, M. P. et al. Influx of recent thymic emigrants into autoimmune thyroid disease glands in humans. Clin. Exp. Immunol. 153, 338–350 (2008).
Boursalian, T. E., Golub, J., Soper, D. M., Cooper, C. J. & Fink, P. J. Continued maturation of thymic emigrants in the periphery. Nature Immunol. 5, 418–425 (2004). This study provided the first definitive evidence that RTEs undergo progressive phenotypic and functional maturation in secondary lymphoid organs.
Lewis, D. B., Haines, C. & Ross, D. Protein tyrosine kinase 7: a novel surface marker for human recent thymic emigrants with potential clinical utility. J. Perinatol. 31, S72–S81 (2011).
Zaghouani, H., Hoeman, C. M. & Adkins, B. Neonatal immunity: faulty T-helpers and the shortcomings of dendritic cells. Trends Immunol. 30, 585–591 (2009).
PrabhuDas, M. et al. Challenges in infant immunity: implications for responses to infection and vaccines. Nature Immunol. 12, 189–194 (2011).
Di Rosa, F., Ramaswamy, S., Ridge, J. P. & Matzinger, P. On the lifespan of virgin T lymphocytes. J. Immunol. 163, 1253–1257 (1999).
Miller, N. E., Bonczyk, J. R., Nakayama, Y. & Suresh, M. Role of thymic output in regulating CD8 T-cell homeostasis during acute and chronic viral infection. J. Virol. 79, 9419–9429 (2005).
Vezys, V. et al. Continuous recruitment of naive T cells contributes to heterogeneity of antiviral CD8 T cells during persistent infection. J. Exp. Med. 203, 2263–2269 (2006).
Houston, E. G. Jr, Higdon, L. E. & Fink, P. J. Recent thymic emigrants are preferentially incorporated only into the depleted T-cell pool. Proc. Natl Acad. Sci. USA 108, 5366–5371 (2011). This study established that the incorporation of RTEs into the peripheral T cell pool is dependent on existing lymphocyte occupancy in secondary lymphoid organs.
Schnell, F. J. & Kersh, G. J. Control of recent thymic emigrant survival by positive selection signals and early growth response gene 1. J. Immunol. 175, 2270–2277 (2005).
Berzins, S. P., Boyd, R. L. & Miller, J. F. A. P. The role of the thymus and recent thymic migrants in the maintenance of the adult peripheral lymphocyte pool. J. Exp. Med. 187, 1839–1848 (1998).
Li, O., Zheng, P. & Liu, Y. CD24 expression on T cells is required for optimal T cell proliferation in lymphopenic host. J. Exp. Med. 200, 1083–1089 (2004).
Stutman, O. Intrathymic and extrathymic T cell maturation. Immunol. Rev. 42, 138–184 (1978).
Scollay, R. Thymus cell migration: cells migrating from thymus to peripheral lymphoid organs have a “mature” phenotype. J. Immunol. 128, 1566–1570 (1982).
Scollay, R., Chen, W. F. & Shortman, K. The functional capabilities of cells leaving the thymus. J. Immunol. 132, 25–30 (1984).
Kelly, K. A. & Scollay, R. Analysis of recent thymic emigrants with subset- and maturity-related markers. Int. Immunol. 2, 419–425 (1990).
Lee, C. K. et al. Thymic emigrants isolated by a new method possess unique phenotypic and functional properties. Blood 97, 1360–1369 (2001). This study provided evidence that RTEs are functionally distinct from mature naive T cells, and that this characteristic may play a part in peripheral T cell tolerance.
Staton, T. L., Johnston, B., Butcher, E. C. & Campbell, D. J. Murine CD8+ recent thymic emigrants are αE integrin-positive and CC chemokine ligand 25 responsive. J. Immunol. 172, 7282–7288 (2004).
Staton, T. L. et al. CD8+ recent thymic emigrants home to and efficiently repopulate the small intestine epithelium. Nature Immunol. 7, 482–488 (2006). This study provided definitive evidence that, despite their naive phenotype, CD8+ RTEs efficiently traffic to the gut epithelium.
Priyadharshini, B., Welsh, R. M., Greiner, D. L., Gerstein, R. M. & Brehm, M. A. Maturation-dependent licensing of naive T cells for rapid TNF production. PLoS ONE 5, e15038 (2010). This study provided evidence that post-thymic maturation is crucial for rapid and optimal production of TNF by mature naive T cells.
Makaroff, L. E., Hendricks, D. W., Niec, R. E. & Fink, P. J. Postthymic maturation influences the CD8 T cell response to antigen. Proc. Natl Acad. Sci. USA 106, 4799–4804 (2009). This study provided the first observation that T cell maturation status at the time of antigen encounter can influence future cell fate decisions.
Joshi, N. S. & Kaech, S. M. Effector CD8 T cell development: a balancing act between memory cell potential and terminal differentiation. J. Immunol. 180, 1309–1315 (2008).
Chang, J. F., Thomas, C. A. & Kung, J. T. Induction of high level IL-2 production in CD4+8− T helper lymphocytes requires post-thymic development. J. Immunol. 147, 851–859 (1991).
Hendricks, D. W. & Fink, P. J. Recent thymic emigrants are biased against the T-helper type 1 and toward the T-helper type 2 effector lineage. Blood 117, 1239–1249 (2011). This study provided the first observation that post-thymic maturation influences CD4+ T cell lineage commitment and function in RTEs from young adult mice.
Clise-Dwyer, K., Huston, G. E., Buck, A. L., Duso, D. K. & Swain, S. L. Environmental and intrinsic factors lead to antigen unresponsiveness in CD4+ recent thymic emigrants from aged mice. J. Immunol. 178, 1321–1331 (2007).
Opiela, S. J., Koru-Sengul, T. & Adkins, B. Murine neonatal recent thymic emigrants (RTE) are phenotypically and functionally distinct from adult RTE. Blood 113, 5635–5643 (2009).
Rose, S., Lichtenheld, M., Foote, M. R. & Adkins, B. Murine neonatal CD4+ cells are poised for rapid Th2 effector-like function. J. Immunol. 178, 2667–2678 (2007).
Smith-Garvin, J. E., Koretzky, G. A. & Jordan, M. S. T cell activation. Annu. Rev. Immunol. 27, 591–619 (2009).
Wells, A. D. New insights into the molecular basis of T cell anergy: anergy factors, avoidance sensors, and epigenetic imprinting. J. Immunol. 182, 7331–7341 (2009).
Harding, F. A., McArthur, J. G., Gross, J. A., Raulet, D. H. & Allison, J. P. CD28-mediated signaling co-stimulates murine T cells and prevents induction of anergy of T-cell clones. Nature 356, 607–609 (1992).
Macian, F. et al. Transcriptional mechanisms underlying lymphocyte tolerance. Cell 109, 719–731 (2002).
Gavin, M. A. & Bevan, M. J. Increased peptide promiscuity provides a rationale for the lack of N regions in the neonatal T cell repertoire. Immunity 3, 793–800 (1995).
Garcia, A. M., Fadel, S. A., Cao, S. & Sarzotti, M. T cell immunity in neonates. Immunol. Res. 22, 177–190 (2000).
Lukacs-Kornek, V. & Turley, S. J. Self-antigen presentation by dendritic cells and lymphoid stroma and its implications for autoimmunity. Curr. Opin. Immunol. 23, 138–145 (2011).
Gardner, J. M. et al. Deletional tolerance mediated by extrathymic Aire-expressing cells. Science 321, 843–847 (2008).
King, C., Ilic, A., Koelsch, K. & Sarvetnick, N. Homeostatic expansion of T cells during immune insufficiency generates autoimmunity. Cell 117, 265–277 (2004).
Houston, E. G. Jr, Nechanitzky, R. & Fink, P. J. Cutting edge: contact with secondary lymphoid organs drives postthymic T cell maturation. J. Immunol. 181, 5213–5217 (2008).
Surh, C. D. & Sprent, J. Homeostasis of naive and memory T cells. Immunity 29, 848–862 (2008).
Houston, E. G. Jr & Fink, P. J. MHC drives TCR repertoire shaping, but not maturation, in recent thymic emigrants. J. Immunol. 183, 7244–7249 (2009).
Hsu, F. C., Pajerowski, A. G., Nelson-Holte, M., Sundsbak, R. & Shapiro, V. S. NKAP is required for T cell maturation and acquisition of functional competency. J. Exp. Med. 208, 1291–1304 (2011). This study provided evidence that RTE maturation depends on the transcriptional repressor NKAP.
Chen, Y. T., Chen, F. L. & Kung, J. T. Age-associated rapid and Stat6-independent IL-4 production by NK1−CD4+CD8− thymus T lymphocytes. J. Immunol. 163, 4747–4753 (1999).
Makar, K. W. et al. Active recruitment of DNA methyltransferases regulates interleukin 4 in thymocytes and T cells. Nature Immunol. 4, 1183–1190 (2003).
Thangavelu, G. et al. Programmed death-1 is required for systemic self-tolerance in newly generated T cells during the establishment of immune homeostasis. J. Autoimmun. 36, 301–312 (2011).
Jacobs, S. R. et al. Glucose uptake is limiting in T cell activation and requires CD28-mediated Akt-dependent and independent pathways. J. Immunol. 180, 4476–4486 (2008).
Kyewski, B. & Klein, L. A central role for central tolerance. Annu. Rev. Immunol. 24, 571–606 (2006).
Elson, C. O., Cong, Y., Iqbal, N. & Weaver, C. T. Immuno-bacterial homeostasis in the gut: new insights into an old enigma. Semin. Immunol. 13, 187–194 (2001).
Bourgeois, C., Hao, Z., Rajewsky, K., Potocnik, A. J. & Stockinger, B. Ablation of thymic export causes accelerated decay of naive CD4 T cells in the periphery because of activation by environmental antigen. Proc. Natl Acad. Sci. USA 105, 8691–8696 (2008).
Butcher, E. C. & Weissman, I. L. Direct fluorescent labeling of cells with fluorescein or rhodamine isothiocyanate. I. Technical aspects. J. Immunol. Methods 37, 97–108 (1980).
Scollay, R. G., Butcher, E. C. & Weissman, I. L. Thymus cell migration. Quantitative aspects of cellular traffic from the thymus to the periphery in mice. Eur. J. Immunol. 10, 210–218 (1980).
Tough, D. F. & Sprent, J. Turnover of naive- and memory-phenotype T cells. J. Exp. Med. 179, 1127–1135 (1994).
Kong, F. K., Chen, C. L., Six, A., Hockett, R. D. & Cooper, C. J. T cell receptor gene deletion circles identify recent thymic emigrants in the peripheral T cell pool. Proc. Natl Acad. Sci. USA 96, 1536–1540 (1999).
Douek, D. C. et al. Changes in thymic function with age and during the treatment of HIV infection. Nature 396, 690–695 (1998).
Berzins, S. P., Godfrey, D. I., Miller, J. F. A. P. & Boyd, R. L. A central role for thymic emigrants in peripheral T cell homeostasis. Proc. Natl Acad. Sci. USA 96, 9787–9791 (1999).
McCaughtry, T. M., Wilden, M. S. & Hogquist, K. A. Thymic emigration revisited. J. Exp. Med. 204, 2513–2520 (2007).
Kohler, S. & Thiel, A. Life after the thymus: CD31+ and CD31− human naive CD4+ T-cell subsets. Blood 113, 769–774 (2009).
Acknowledgements
This work was supported by US National Institutes of Health grants AI064318 (to P.J.F. with a supplement to D.W.H.) and DK091953 (to P.J.F.). The content is solely the responsibility of the authors and does not necessarily represent the official views of the US National Institute of Allergy and Infectious Diseases or the US National Institutes of Health.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Glossary
- Recent thymic emigrants
-
T cells that have completed thymic development and have recently entered the lymphoid periphery. These young T cells undergo a maturation process that includes changes in function and cell surface phenotype.
- Lymphoreplete
-
A lymphoid periphery that is relatively full of lymphocytes.
- Lymphopenic
-
A lymphoid periphery that is depleted of lymphocytes.
- Homeostasis
-
The controlled turnover of cell populations in which the balance between cell proliferation and cell death maintains constancy in the size of the lymphocyte pool.
- Memory precursor CD8+ T cells
-
A subset of effector CD8+ T cells, defined as IL7RαhiKLRG1low that have an enhanced potential to become long-lived memory CD8+ T cells.
- DNA methylation
-
A repressive epigenetic modification in which methyl groups are present on cytosine bases that are followed by guanine bases (CpGs). CpG-rich areas are typically found in gene promoter and other regulatory regions.
- Anergy
-
A state of immune unresponsiveness. Anergic B and T cells do not respond fully to their cognate antigens.
- Secondary lymphoid organs
-
Organs, including the spleen and lymph nodes, that support lymphocyte homeostasis, maturation and activation-induced differentiation.
- Graft-versus-host disease
-
(GVHD). A disease that results from the immunological attack on target recipient organs or tissues (such as the skin or gut) by donor allogeneic T cells that are transferred along with the allograft (such as bone marrow, liver or gut allografts). GVHD occurs in graft recipients that are unable to eliminate the host-reactive donor T cells owing to immunosuppression, immunological immaturity or tolerance.
Rights and permissions
About this article
Cite this article
Fink, P., Hendricks, D. Post-thymic maturation: young T cells assert their individuality. Nat Rev Immunol 11, 544–549 (2011). https://doi.org/10.1038/nri3028
Published:
Issue Date:
DOI: https://doi.org/10.1038/nri3028
This article is cited by
-
Progressive multifocal leukoencephalopathy in a patient with mediastinal teratoma: a case report
BMC Neurology (2022)
-
Protein abundance of the cytokine receptor γc controls the thymic generation of innate-like T cells
Cellular and Molecular Life Sciences (2022)
-
Selective involution of thymic medulla by cyclosporine A with a decrease of mature thymic epithelia, XCR1+ dendritic cells, and epithelium-free areas containing Foxp3+ thymic regulatory T cells
Histochemistry and Cell Biology (2021)
-
Clonal expansion of innate and adaptive lymphocytes
Nature Reviews Immunology (2020)
-
Genetic T-cell receptor diversity at 1 year following allogeneic hematopoietic stem cell transplantation
Leukemia (2020)