Abstract
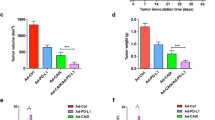
Although Renal Cell Cancer (RCC) is known to be immunogenic, clinical efficacy of various immunotherapeutic approaches remains unsatisfactory. Novel targeted therapies showing cytostatic rather than cytotoxic activity are unable to cure RCC patients. In our studies, we evaluated the therapeutic efficacy of whole-cell vaccine based on irradiated murine RENCA cells genetically modified to secrete designer cytokine—Hyper-IL6 (H6)—comprising IL-6 and soluble IL-6 receptor. An orthotopic RCC model based on a subcapsular implantation of RENCA cells into kidneys of Balb/C mice was employed. The efficacy of RENCA-H6 vaccine was compared with control vaccine (RENCA-wt) in relation to naive (non-immunized) animals. Three sets of vaccination experiments were carried out in a (i) protective, (ii) palliative and (iii) adjuvant (following nephrectomy) setting. The influence of vaccination on survival of RCC-bearing animals was analyzed. Specificity of vaccine-induced immune response was studied using model antigen—GFP. RCC-bearing animals immunized with RENCA-H6 vaccine showed prolonged survival compared with other groups. In palliative and adjuvant settings the survival RENCA-H6-immunized animals exceeded 75%. Administration of RENCA-H6 inhibited formation and recruitment of Treg cells (CD4+CD25+Foxp3+) and increased maturation of DCs. RENCA tumors in RENCA-H6- vaccinated animals contained large populations of NK cells and activated CD4+, CD8+ T cells. In addition, in mice vaccinated with RENCA-H6 cells large population of CD4+ and CD8+ memory cells (CD62Llow) were detected. In the orthotopic RCC model, RENCA-H6 vaccine showed high therapeutic potential, which resulted from modulation of numerous immunological mechanisms.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Hudes G, Carducci M, Tomczak P, Dutcher J, Figlin R, Kapoor A et al. Temsirolimus, interferon alfa, or both for advanced renal-cell carcinoma. N Engl J Med 2007; 356: 2271–2281.
Motzer RJ, Hutson TE, Tomczak P, Michaelson MD, Bukowski RM, Rixe O et al. Sunitinib versus interferon alfa in metastatic renal-cell carcinoma. N Engl J Med 2007; 356: 115–124.
Escudier B, Eisen T, Stadler WM, Szczylik C, Oudard S, Siebels M et al. Sorafenib in advanced clear-cell renal-cell carcinoma. N Engl J Med 2007; 356: 125–134.
Wysocki PJ, Zolnierek J, Szczylik C, Mackiewicz A . Targeted therapy of renal cell cancer. Curr Opin Investig Drugs 2008; 9: 570–575.
Motzer RJ, Rini BI, Bukowski RM, Curti BD, George DJ, Hudes GR et al. Sunitinib in patients with metastatic renal cell carcinoma. JAMA 2006; 295: 2516–2524.
Mach N, Dranoff G . Cytokine-secreting tumor cell vaccines. Curr Opin Immunol 2000; 12: 571–575.
Ward S, Casey D, Labarthe MC, Whelan M, Dalgleish A, Pandha H et al. Immunotherapeutic potential of whole tumour cells. Cancer Immunol Immunother 2002; 51: 351–357.
Scheffer SR, Nave H, Korangy F, Schlote K, Pabst R, Jaffee EM et al. Apoptotic, but not necrotic, tumor cell vaccines induce a potent immune response in vivo. Int J Cancer 2003; 103: 205–211.
Porgador A, Tzehoval E, Katz A, Vadai E, Revel M, Feldman M et al. Interleukin 6 gene transfection into Lewis lung carcinoma tumor cells suppresses the malignant phenotype and confers immunotherapeutic competence against parental metastatic cells. Cancer Res 1992; 52: 3679–3686.
Ledda MF, Adris S, Bravo AI, Bover L, Carbone C, Paleolog E et al. Tumor cells engineered to express interleukin-6 exhibit a reduced tumorigenicity depending on the tumor cell model. Cell Mol Biol (Noisy-le-grand) 1996; 42: 769–778.
Wysocki PJ, Kowalczyk DW, Izycki D, Grabarczyk P, Kwias Z, Mackiewicz A . IL-6 and GM-CSF in tumor rejection model of renal cell cancer. Adv Exp Med Biol 2001; 495: 379–383.
Taga T, Kishimoto T . Gp130 and the interleukin-6 family of cytokines. Annu Rev Immunol 1997; 15: 797–819.
Mackiewicz A, Schooltink H, Heinrich PC, Rose-John S . Complex of soluble human IL-6-receptor/IL-6 up-regulates expression of acute-phase proteins. J Immunol 1992; 149: 2021–2027.
Peters M, Blinn G, Solem F, Fischer M, Meyer zum Buschenfelde KH, Rose-John S . In vivo and in vitro activities of the gp130-stimulating designer cytokine Hyper-IL-6. J Immunol 1998; 161: 3575–3581.
Kowalczyk DW, Wlazlo AP, Shane S, Ertl HC . Vaccine regimen for prevention of sexually transmitted infections with human papillomavirus type 16. Vaccine 2001; 19: 3583–3590.
Weiss J, Zimmermann F . Tribromoethanol (Avertin) as an anaesthetic in mice. Lab Anim 1999; 33: 192–193.
Ahn KS, Jung YS, Kim J, Lee H, Yoon SS . Behavior of murine renal carcinoma cells grown in ectopic or orthotopic sites in syngeneic mice. Tumour Biol 2001; 22: 146–153.
Wysocki PJ, Kwiatkowska EP, Kazimierczak U, Suchorska W, Kowalczyk DW, Mackiewicz A . Captopril, an angiotensin-converting enzyme inhibitor, promotes growth of immunogenic tumors in mice. Clin Cancer Res 2006; 12: 4095–4102.
Vieweg J, Heston WD, Gilboa E, Fair WR . An experimental model simulating local recurrence and pelvic lymph node metastasis following orthotopic induction of prostate cancer. Prostate 1994; 24: 291–298.
Tan MH, Holyoke ED, Goldrosen MH . Murine colon adenocarcinoma: syngeneic orthotopic transplantation and subsequent hepatic metastases. J Natl Cancer Inst 1977; 59: 1537–1544.
Ali SA, McLean CS, Boursnell ME, Martin G, Holmes CL, Reeder S et al. Preclinical evaluation of “whole” cell vaccines for prophylaxis and therapy using a disabled infectious single cycle-herpes simplex virus vector to transduce cytokine genes. Cancer Res 2000; 60: 1663–1670.
Wagner JR, Walther MM, Linehan WM, White DE, Rosenberg SA, Yang JC . Interleukin-2 based immunotherapy for metastatic renal cell carcinoma with the kidney in place. J Urol 1999; 162: 43–45.
Sella A, Swanson DA, Ro JY, Putnam Jr JB, Amato RJ, Markowitz AB et al. Surgery following response to interferon-alpha-based therapy for residual renal cell carcinoma. J Urol 1993; 149: 19–21; discussion 21–12.
Hara I, Nagai H, Miyake H, Yamanaka K, Hara S, Micallef MJ et al. Effectiveness of cancer vaccine therapy using cells transduced with the interleukin-12 gene combined with systemic interleukin-18 administration. Cancer Gene Ther 2000; 7: 83–90.
Kausch I, Jiang H, Brocks C, Bruderek K, Kruger S, Sczakiel G et al. Ki-67-directed antisense therapy in an orthotopic renal cell carcinoma model. Eur Urol 2004; 46: 118–124; discussion 124-125.
Chagnon F, Thompson-Snipes L, Elhilali M, Tanguay S . Murine renal cell carcinoma: evaluation of a dendritic-cell tumour vaccine. BJU Int 2001; 88: 418–424.
Chagnon F, Tanguay S, Ozdal OL, Guan M, Ozen ZZ, Ripeau JS et al. Potentiation of a dendritic cell vaccine for murine renal cell carcinoma by CpG oligonucleotides. Clin Cancer Res 2005; 11: 1302–1311.
Wysocki PJ, Grabarczyk P, Mackiewicz-Wysocka M, Kowalczyk DW, Mackiewicz A . Genetically modified dendritic cells—a new, promising cancer treatment strategy? Expert Opin Biol Ther 2002; 2: 835–845.
Wysocki PJ, Karczewska-Dzionk A, Mackiewicz-Wysocka M, Mackiewicz A . Human cancer gene therapy with cytokine gene-modified cells. Expert Opin Biol Ther 2004; 4: 1595–1607.
Wysocki PJ, Zolnierek J, Szczylik C, Mackiewicz A . Recent developments in renal cell cancer immunotherapy. Expert Opin Biol Ther 2007; 7: 727–737.
Shibata J, Murakami K, Abe M, Hashimoto A, Utsugi T, Fukushima M et al. Life prolonging effect of antitumor agents on postoperative adjuvant therapy in the lung spontaneous metastasis model in mice. Anticancer Res 1998; 18: 1203–1209.
Medical Research Council Renal Cancer Collaborators. Interferon-alpha and survival in metastatic renal carcinoma: early results of a randomised controlled trial. Lancet 1999; 353: 14–17.
Levy DA, Slaton JW, Swanson DA, Dinney CP . Stage specific guidelines for surveillance after radical nephrectomy for local renal cell carcinoma. J Urol 1998; 159: 1163–1167.
Linehan WM, Shipley WU, Parkinson DR . Cancer of the kidney and urether. In: DeVita VT, Hellman S, Rosenberg SA (eds). Cancer: Principles and Practice of Oncology, 5th edn, Lippincott-Raven: Philadelphia, 1997, pp 858–911.
Ljungberg B, Alamdari FI, Rasmuson T, Roos G . Follow-up guidelines for nonmetastatic renal cell carcinoma based on the occurrence of metastases after radical nephrectomy. BJU Int 1999; 84: 405–411.
Salup RR, Back TC, Wiltrout RH . Successful treatment of advanced murine renal cell cancer by bicompartmental adoptive chemoimmunotherapy. J Immunol 1987; 138: 641–647.
Banchereau J, Steinman RM . Dendritic cells and the control of immunity. Nature 1998; 392: 245–252.
Ozbek S, Peters M, Breuhahn K, Mann A, Blessing M, Fischer M et al. The designer cytokine hyper-IL-6 mediates growth inhibition and GM-CSF-dependent rejection of B16 melanoma cells. Oncogene 2001; 20: 972–979.
Drakesmith H, O’Neil D, Schneider SC, Binks M, Medd P, Sercarz E et al. In vivo priming of T cells against cryptic determinants by dendritic cells exposed to interleukin 6 and native antigen. Proc Natl Acad Sci USA 1998; 95: 14903–14908.
Shevach EM . CD4+ CD25+ suppressor T cells: more questions than answers. Nat Rev Immunol 2002; 2: 389–400.
Chen W, Jin W, Hardegen N, Lei KJ, Li L, Marinos N et al. Conversion of peripheral CD4+CD25− naive T cells to CD4+CD25+ regulatory T cells by TGF-beta induction of transcription factor Foxp3. J Exp Med 2003; 198: 1875–1886.
Pyzik M, Piccirillo CA . TGF-beta1 modulates Foxp3 expression and regulatory activity in distinct CD4+ T cell subsets. J Leukoc Biol 2007; 82: 335–346.
Zhang Q, Rubenstein JN, Liu VC, Park I, Jang T, Lee C . Restoration of expression of transforming growth factor-beta type II receptor in murine renal cell carcinoma (renca) cells by 5-Aza-2′-deoxycytidine. Life Sci 2005; 76: 1159–1166.
Sonouchi K, Hamilton TA, Tannenbaum CS, Tubbs RR, Bukowski R, Finke JH . Chemokine gene expression in the murine renal cell carcinoma, RENCA, following treatment in vivo with interferon-alpha and interleukin-2. Am J Pathol 1994; 144: 747–755.
Chen X, Das R, Komorowski R, Beres A, Hessner MJ, Mihara M et al. Blockade of interleukin-6 signaling augments regulatory T-cell reconstitution and attenuates the severity of graft-versus-host disease. Blood 2009; 114: 891–900.
Dominitzki S, Fantini MC, Neufert C, Nikolaev A, Galle PR, Scheller J et al. Cutting edge: trans-signaling via the soluble IL-6R abrogates the induction of FoxP3 in naive CD4+CD25 T cells. J Immunol 2007; 179: 2041–2045.
Mendiratta SK, Quezada A, Matar M, Thull NM, Bishop JS, Nordstrom JL et al. Combination of interleukin 12 and interferon alpha gene therapy induces a synergistic antitumor response against colon and renal cell carcinoma. Hum Gene Ther 2000; 11: 1851–1862.
Izycki D, Nawrocki S, Laciak M, Gryska K, Baksalary-Izycka K, Mackiewicz A . Effect of cyclophosphamide on tumorogenesis of the IL-6 and Hyper-IL-6 gene modified murine melanoma cells. Wspolczesna Onkologia 2004; 8: 124–131.
Hillman GG, Younes E, Visscher D, Hamzavi F, Kim S, Lam JS et al. Inhibition of murine renal carcinoma pulmonary metastases by systemic administration of interferon gamma: mechanism of action and potential for combination with interleukin 4. Clin Cancer Res 1997; 3: 1799–1806.
Qin Z, Blankenstein T . CD4+ T cell—mediated tumor rejection involves inhibition of angiogenesis that is dependent on IFN gamma receptor expression by nonhematopoietic cells. Immunity 2000; 12: 677–686.
Qin Z, Schwartzkopff J, Pradera F, Kammertoens T, Seliger B, Pircher H et al. A critical requirement of interferon gamma-mediated angiostasis for tumor rejection by CD8+ T cells. Cancer Res 2003; 63: 4095–4100.
Lefrancois L, Marzo AL . The descent of memory T-cell subsets. Nat Rev Immunol 2006; 6: 618–623.
Seder RA, Ahmed R . Similarities and differences in CD4+ and CD8+ effector and memory T cell generation. Nat Immunol 2003; 4: 835–842.
Wherry EJ, Teichgraber V, Becker TC, Masopust D, Kaech SM, Antia R et al. Lineage relationship and protective immunity of memory CD8 T cell subsets. Nat Immunol 2003; 4: 225–234.
Acknowledgements
This work was supported by a grant from the Ministry of Science and Higher Education—Grant no. PBZ-KBN 091/P05/54/2003 and BioContract Sp. z o.o.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Rights and permissions
About this article
Cite this article
Wysocki, P., Kazimierczak, U., Suchorska, W. et al. Gene-modified tumor vaccine secreting a designer cytokine Hyper-Interleukin-6 is an effective therapy in mice bearing orthotopic renal cell cancer. Cancer Gene Ther 17, 465–475 (2010). https://doi.org/10.1038/cgt.2010.2
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/cgt.2010.2
Keywords
This article is cited by
-
Current status of vaccine immunotherapy for gastrointestinal cancers
Surgery Today (2023)
-
Re-induction using whole cell melanoma vaccine genetically modified to melanoma stem cells-like beyond recurrence extends long term survival of high risk resected patients - updated results
Journal for ImmunoTherapy of Cancer (2018)
-
Safety of targeting tumor endothelial cell antigens
Journal of Translational Medicine (2016)
-
Mutations of the von Hippel–Lindau gene confer increased susceptibility to natural killer cells of clear-cell renal cell carcinoma
Oncogene (2011)