Abstract
Sortilin receptor 1 (SORL1) is involved in cellular trafficking of amyloid precursor protein and plays an essential role in amyloid-beta peptide generation in Alzheimer disease (AD). The major A allele in a SORL1 single nucleotide polymorphism (SNP), rs3824968, is associated with an increased AD risk. However, the role of SORL1 rs3824968 in the normal ageing process has rarely been examined in relation to brain structural morphology. This study investigated the association between SORL1 rs3824968 and grey matter (GM) volume in a nondemented Chinese population of 318 adults within a wide age range (21–92 years). Through voxel-based morphometry, we found that participants carrying SORL1 allele A exhibited significantly smaller GM volumes in the right posterior cingulate, left middle occipital, medial frontal and superior temporal gyri. Considerable interaction between age and SORL1 suggested a detrimental and accelerated ageing effect of allele A on putamen. These findings provide evidence that SORL1 rs3824968 modulates regional GM volume and is associated with brain trajectory during the adult lifespan.
Similar content being viewed by others
Introduction
The sortilin-related receptor gene (SORL1) encodes a mosaic protein belonging to at least two families: the vacuolar protein sorting 10 domain-containing receptor family and the low density lipoprotein receptor family. The encoded protein acts as an apolipoprotein E (APOE) binder and sorting receptor that regulates the intracellular transport and processing of the amyloid precursor protein (APP) in neurons1. In addition to the ε4 allele of APOE, the major genetic risk factor for late-onset Alzheimer disease (AD)2, SORL1 is genetically associated with late-onset and sporadic AD3. SORL1 regulates and traps APP in the Golgi apparatus and reduces the production of the amyloid-beta peptide (Aβ), the main component of senile plaques in AD4. Cell biology experiments have indicated that SORL1 underexperssion leads to overproduction of Aβ3, whereas SORL1 overexpression considerably reduces cellular APP and extracellular Aβ5. Although the underlying mechanisms of SORL1 in AD and neurodegenerative processes remain unclear, a recent comprehensive review on multiple functional domains of SORL1 suggested that SORL1 can assume various characters in AD pathogenesis6.
Among single nucleotide polymorphisms (SNPs) in SORL1, Rogaeva et al.3 observed a substantial association between a synonymous SNP in exon 34 (rs3824968) and AD in a northern European case–control cohort and implied that the SNP alters the physiological role of SORL1 in the processing of APP holoprotein. The association between SORL1 rs3824968, neurodegenerative disease and ageing process has recently been reported6,7,8,9,10,11. Lower levels of CSF Aβ1-42 were observed in carriers of the AD risk allele A in SORL1 rs3824968 compared with noncarriers12 and the decrease in these levels was associated with AD progression13. Two studies in the Chinese population9,14 also reported a similar association between SORL1 rs3824968 A allele and increased AD risk. However, the association between SORL1 rs3824968 and AD was not consistent across different populations. This discrepancy may be owing to ethnic difference, suggesting the existence of population-specific locus. It should be noted the reference allele for the SORL1 rs3824968 is the T over all populations. In the Chinese population, however, the reference allele is the A. The association between SORL1 variants and accelerating cognitive ageing in relation to spatial and episodic memory performance were observed in a follow-up study of a nondemented population15. In the Lothian Birth Cohort comprising 1936 participants, an association between the SORL1 rs3824968 A allele and reduced spatial-span tasking in cognitively healthy participants was noted16. Bralten et al.17 observed associations between SORL1 variants and hippocampal volume, as an intermediate phenotype, in a gene-wide association analysis of healthy young adults.
In addition to the differences in the effects of the SNP alleles within the brain, the expression of SORL1 can vary according to the age grades during the lifespan. A previous study reported that healthy elderly individuals exhibit two-fold higher SORL1 expressions than do younger adults18. Notably, SORL1 risk variants may predict decrease in white matter (WM) integrity and SORL1 mRNA expression, most prominently during the ageing process and they may be associated with high Aβ deposition in the postmortem brain, thus conferring neuropathological risk for SORL1 variants through the amyloidogenic pathway11. Nonetheless, few studies have systemically investigated the interactions of age, SORL1 effect and regional grey matter (GM) volume changes in a healthy population. Thus, deducing the risk factors for accelerated brain ageing by evaluating the interaction of SORL1 variants with age and their association with the regional brain volume changes over the adult lifespan would benefit current ageing-related genetic imaging studies.
Considering previous findings and the role of SORL1 in the normal ageing process, this study examined regional GM volume changes as an intermediate phenotype in nondemented adults during their lifespan to infer the relationship between the genetic effect of SORL1 rs3824968 and brain ageing. By using a relatively large sample size with a broad age range in combination with an unbiased and whole-brain voxel-based morphometry (VBM) approach, we assessed two hypotheses based on previous studies: (1) independent of the age effect, the variants of SORL1 rs3824968 are associated with the difference of regional GM volume in nondemented adults and (2) SORL1 rs3824968 interacts with age and is associated with regional GM volume alterations across lifespan.
Results
Demographics
Twelve participants were excluded because of failure in SORL1 genotyping or extensive motion problems during MRI scanning. Of the 318 participants that completed SORL1 genotyping, MRI acquisitions and cognitive assessments, 42, 146 and 130 carried the SORL1 TT, AT and AA genotypes, respectively. APOE genotypes were also determined to reveal five genotypes: ε2/ε2 (n = 3, 0.9%), ε2/ε3 (n = 44, 13.8%), ε3/ε3 (n = 222, 69.8%), ε2/ε4 (n = 6, 1.9%) and ε3/ε4 (n = 42, 13.2%). The frequencies of the genotypes followed the Hardy–Weinberg equilibrium. The demographics and clinical characteristics of the study group are shown in Table 1. The participants carrying the SORL1 TT, AT and AA genotypes showed no significant differences in age (P = 0.230), sex (P = 0.695), educational level (P = 0.380), APOE allele distribution (P = 0.291), total GM volume (P = 0.304), total WM volume (P = 0.243), total CSF volume (P = 0.849), total intracranial volume (P = 0.425), or neuropsychological characteristics.
Main Effect of Age and SORL1 rs3824968 on Regional GM Volume
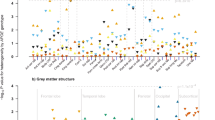
Regarding the main effect of age, DARTEL-based T1 VBM analyses showed widespread age-related GM volume differences in the study participants (Fig. 1), in accordance with previous studies19,20. Regarding the main effect of the gene obtained from F tests (Table 2, Fig. 2A), on comparing the participants carrying the SORL1 TT, AT and AA genotypes, the post hoc analyses revealed positive dosage effect of the T allele on GM volume of the left middle occipital gyrus (Fig. 2B). In addition, participants carrying AA showed lower GM volume in the right cerebellar tonsil than did those carrying AT (Fig. 2C). Moreover, participants carrying AA had lower GM volumes in the left medial frontal and right posterior cingulate gyri than did those carrying the T allele (Fig. 2D,E). The participants carrying the A allele showed significantly lower GM volume in the left superior temporal gyrus than did those carrying TT (Fig. 2F). After controlling APOE genotype as additional nuisance variable, the findings remain consistent with current findings (Supplementary Table 1).
Age effect: GM differences in a nondemented elderly participants.
Regions of significant GM differences from 318 normal participants superimposed on surface space. T-score maps show clusters at a voxel threshold with P of <0.005 as well as extended voxel sizes of 298, all of the clusters remained significant and survived from the criteria of corrected Palpha of <0.05 by Monte Carlo simulation.
Regional GM volume differences among the three SORL1 rs3824968 genotypic groups.
T-score map shows significant smaller GM volume in SORL1 A allele carriers compared with those carrying TT (A). The clusters were set at a voxel threshold with P of <0.005 as well as extended voxel sizes of 298, all of the clusters remained significant and survived from the criteria of corrected Palpha of <0.05 by Monte Carlo simulation. Bottom bar graph shows the GM volume difference between the SORL1 genotypes and the regions with significant gene main effect in the left middle occipital gyrus (B), right cerebellum tonsil (C), left medial frontal gyrus (D), right posterior cingulate gyrus (E) and left superior temporal gyrus (F). *Bonferroni-corrected P < 0.05 (post hoc tests in analysis of covariance).
Age and SORL1 Interaction Effect on Regional GM Volume
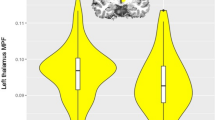
Regarding the voxelwise covariate interaction analysis, we observed considerable interaction effect between age and the three SORL1 genotypes in the right putamen region (Fig. 3) and the downward slope was steeper in the participants carrying AA and AT than in those carrying TT. To verify whether this accelerated reduction in the putaminal volume was related to cognitive performance, we applied partial correlation for each genotypic group (results are presented in Section 3.4).
Interaction between the SORL1 genotype and age on right putamen GM volume.
The scatter plot demonstrates the interaction between the SORL1 genotype and age on right putamen GM volume using voxel-wised covariate analysis with the SORL1 genotypes as the condition and age as the covariate, while controlling for sex and education level as nuisance variables (corrected Palpha of <0.05 by Monte Carlo simulation)).
Correlation Analysis Between Regional GM Volume and Cognitive Function
In the partial correlation analysis between regional GM volume and cognitive performance levels in all participants, none of the regional GM volume changes for the effect of SORL1 showed correlation with MMSE, digit span forward (DSF), or digit span backward (DSB) scores. For the correlation between the cluster of voxelwise interaction and cognitive tests, although the results failed to survive from the criteria of Bonferroni correction, a positive trend between DSB score and putaminal volume was observed in the participants carrying AA (r = 0.189, P = 0.034).
Discussion
We performed VBM analysis to examine the effect of SORL1 rs3824968 on regional GM volume and age-related changes in the brain GM volume of nondemented participants within a wide age range. The findings of this study showed that the SORL1 rs3824968 A allele carriers had lower GM volume in the left superior temporal, left medial frontal, right posterior cingulate and left middle occipital gyri than did those carrying TT. A considerable interaction between age and the SORL1 genotypes was observed, showing an accelerated (downward) slope of GM volume differences in the right putamen of the participants carrying the AD-risk (A) allele compared with those carrying TT. Thus, our findings support the hypothesis that SORL1 rs3824968 is associated with regional GM volume differences in early adulthood and can influence ageing-related GM morphological changes during the nondemented adult lifespan.
The SORL1 rs3824968 A carriers showed significantly decreased GM volume in the left superior temporal, left medial frontal, right posterior cingulate and left middle occipital gyrus, suggesting detrimental effects of the A allele on brain GM volume. A trend towards gene dosage effect of the A allele was also observed in the left middle occipital, medial frontal and right posterior cingulate gyri. SORL1 SNPs have been found to be associated with an increased risk of neurodegenerative disease in Swedish and African American population through meta-analysis21,22. Nonetheless, association studies have further identified that haplotype SNPs including rs3824968 are associated with lower Aβ42 levels in CSF and higher AD risk in Asian population14,23,24. Therefore, the SORL1 risk variants may alter the Aβ expression levels in either healthy adults or patients with neurodegenerative disease12,13,25, thus suggesting that SORL1 rs3824968 variants may influence amyloid pathology during ageing. Greater amyloid burden has been shown to be positively associated with regional GM atrophy26,27,28, particularly in the superior temporal, medial frontal and cingulate gyri in both structural and functional MRI studies29,30. In addition to the hypothesis of amyloid pathology, Cuenco et al.31 proposed another viewpoint that SORL1 increases dementia risk through effects on cerebrovascular abnormalities; SORL1 rs3824968 and its haplotypes were found to be associated with brain volume and WM hyperintensities in their study. This biological and neuroimaging evidence may suggest that SORL1 rs3824968 is associated with brain morphology in terms of A allele-related volumetric reduction caused by high Aβ load, cardiovascular disease, or both. However, additional studies are necessary to explore the potential mechanisms of SORL1 rs3824968 on the cellular and molecular determinants of brain structural differences in both demented and nondemented individuals.
Here, we revealed the first evidence of the influence of SORL1 on age-related changes in the brain cortical morphology of nondemented participants over a wide age range. The right putamen of the participants carrying AA and AT showed a considerable effect of interaction between age and the SORL1 genotype with a steeper downward slope of age-related GM volume reduction compared with that of those carrying TT. This finding supports the hypothesis that SORL1 polymorphism plays a role in the ageing process of the human brain with a partially detrimental effect on age-related differences of regional GM volume, particularly in the putamen of the participants with the A allelic variant. A previous study reported that participants with presenilin-1, presenilin-2, or APP mutations showed higher amyloid load and greater GM atrophy in the putamen32. In other words, if the SORL1 variants contribute to the risk of AD neuropathogenesis through the amyloidogenic pathway, putaminal volume could be a vulnerable subcortical area in the brain ageing process with regard to the SORL1 effect. In addition, such an accelerated regional GM volume reduction effect in the putamen further links to cognitive decline in participants carrying the A allele. A trend of positive correlation between putamen volume and DSB scores was observed in the participants carrying AA. Studies have shown that the basal ganglia play a vital role in working memory and executive function, particularly in the putaminal region33,34. Moreover, increased activity in the putamen during a card sorting test was observed to be related to working memory function, as evident on functional MRI35. In the current study, imaging indicated an association between SORL1 rs3824968 and putaminal volume and a trend of neural correlation of working memory (DSB), which may be a marker of accelerated cognitive impairment at preclinical stage36,37,38.
We observed no age-by-SNP interaction effect in the significant clusters that detected the main effect of genotype: the left superior temporal gyrus, left middle occipital gyrus, left medial frontal gyrus, right cerebellar tonsil and right posterior cingulate gyrus. In addition to a previous study, which proposed a pathway by which SORL1 variants could mediated neural risk of AD beginning from teenage years25, current data support two pathways of SORL1 rs3824968 effect on GM volume during the ageing process: (1) differences of regional cortical volume between the three genotypes occur from early adult life and preferentially stay stable during adulthood and (2) in contrast to the first pathway, variants of SORL1 rs3824968 do not modulate putaminal volume directly but interact with age during the adult lifespan. Further research is necessary to explore underlying mechanisms for various SORL1 genetic effects along brain ageing trajectory.
The necessity for statistically sufficient sample sizes in imaging studies of genetic variation is increasingly being recognised. The relatively large and (by international standards) homogenous cohort of participants with a wide age range recruited in this study increased the credibility of our findings, based on previously proposed recommendations regarding cohort sizes39. However, the characteristic cross-sectional design of this study may be a limitation. We did not measure Aβ load levels in our participants; the genotypic effect of the SORL1 SNP on GM volume might be affected through Aβ deposition or other mechanisms such as inflammatory signalling modulation40. Therefore, the associations between SORL1 variants, biochemical data and brain morphometry should be investigated simultaneously in future studies to explain the effect of SORL1 on GM volume reduction. In addition, rather than having a direct effect on regional GM volume, SORL1 rs3824968 may be in linkage disequilibrium with truly associated variants; in addition, it may be an intermediate phenotype. Since such a linkage could vary among different populations and the association between rs3824968 and AD was not replicated in studies of other populations, these may confound the generalisability of our findings, which were based on a homogenous Chinese cohort. In addition, no significant correlation was found between general cognitive function (MMSE, DSF and DSB) and regional GM volume differences of the SORL1 effect. Although MMSE is a widely used tool for detecting dementia, it may lack sensitivity to and specificity for early signs of subtle cognitive changes. Despite the current samples being from nondemented participants, the ceiling effect of using MMSE, DSF and DSB for cognition assessment may also have yielded false negative results41. Specific cognitive assessments regarding short- and long-term memory should be examined in future to associate cognitive decline with genetic effect and brain structural changes.
The AD-risk SORL1 rs3824968 A allele was associated with decreased GM volume and exhibited a trend towards gene dosage effect in several brain regions during the adult lifespan. Considerable interaction between age and SORL1 suggested a detrimental and accelerated ageing effect of the AD-risk allele on putaminal volume. Although the underlying molecular mechanisms remain unclear, our findings support the hypothesis that SORL1-related genetic factors play a vital role in the process of normal ageing. The regional GM volume alterations associated with the effect of SORL1 rs3824968 might be a potential neuroimaging biomarker for accelerated brain ageing during the adult lifespan.
Methods
Participants and Instruments
This study initially recruited 330 nondemented ethnic Chinese participants from Northern Taiwan (mean age: 55.9 ± 22.1 years; range: 21–92 years; 57.5% males) through an advertisement in local communities and universities. Each participant was administered a diagnostic structured Mini-International Neuropsychiatric Interview (MINI)42, Mini-Mental State Examination (MMSE) Chinese version43 and Clinical Dementia Rating (CDR) scale44. Participants with any of the following conditions were excluded: (1) a Diagnostic and Statistical Manual of Mental Disorders, Fourth Edition (DSM-IV) axis I diagnosis such as mood disorder or psychotic disorder; (2) a neurobiological disorder, such as dementia, head injury, stroke, or Parkinson disease; (3) illiteracy; (4) an MMSE score of ≤24; or (5) ≥65 years of age with a CDR of >0.5 [exclusion criteria (4) and (5) were applied to prevent possible inclusion of dementia patients].
The cognitive functioning of the participants was evaluated using the MMSE for general cognitive status and the Wechsler Digit Span subtest for verbal working memory abilities. All participants exhibited sufficient visual and auditory acuity to undergo cognitive testing. This study was conducted in accordance with the Declaration of Helsinki and approved by the Institutional Review Board of Taipei Veterans General Hospital. Written informed consent was obtained from all participants after they were adequately apprised of the study.
Genotyping
Genomic DNA was extracted from peripheral blood with a commercial kit (Qiagen, Gentra Puregene Blood Kit). SORL1 rs3824968 genotyping was performed using high-throughput matrix-assisted laser desorption/ionisation time-of-flight mass spectrometry. PCR and single-base extension primers were designed using DNA mass arrays (Sequenom, San Diego). The genotyping analysis was performed using an iPLEX Gold Reagent Kit according to the manufacturer’s instructions. The purified extension products were spotted onto a 384 SpectroCHIP II array by using a MassArray Nanodispenser RS1000, followed by an analysis on a MassARRAY Compact Analyzer. The resulting spectra were processed and the alleles were called using a SpectroTYPER. The APOE genotype was determined using the PCR-restriction fragment length polymorphism method, as described by Hong et al.30.
MRI Acquisition
All MRI scans were performed at National Yang-Ming University, Taipei, Taiwan, on a 3.0-T Siemens MRI scanner with a 12-channel head coil (Siemens Magnetom Tim Trio, Erlangen, Germany). High-resolution structural MR images were acquired through sagittal 3D magnetisation-prepared rapid gradient-echo sequencing (TR = 2530 ms, TE = 3.5 ms, TI = 1100 ms, FOV = 256 mm, flip angle = 7°, matrix size = 256 × 256, 192 sagittal slices, voxel size = 1.0 × 1.0 × 1.0 mm, no gap). All images were acquired parallel to the anterior commissure–posterior commissure line. To minimise motion artefacts generated during image acquisition, each participant’s head was immobilised using cushions inside the coil. An experienced radiologist carefully checked each image to ensure the absence of scanner artefacts, motion problems, or gross anatomical abnormalities.
Diffeomorphic Anatomical Registration Through Exponentiated Lie Algebra-Based T1 VBM Analysis
Individual T1-weighted volumetric images were processed using Gaser’s VBM8 toolbox (http://dbm.neuro.uni-jena.de) within Statistical Parametric Mapping (SPM8; Wellcome Institute of Neurology, University College London, UK). VBM processing was performed using the following procedures: (1) The anterior commissure was set as the origin of each T1-weighted image. (2) A segmentation approach in the VBM8 toolbox was applied in the initial native space. (3) To achieve a higher registration accuracy and account for different brain size among subjects, the native space segments of the GM, WM and cerebrospinal fluid (CSF) were initially affine registered to the tissue probability maps in the Montreal Neurological Institute (MNI) standard space. (4) All affine registered GM and WM tissue segments were used to generate group-specific templates through nonlinear warping by using the Diffeomorphic Anatomical Registration Through Exponentiated Lie (DARTEL) algebra toolbox45 implemented in SPM8. (5) Nonlinear deformation parameters obtained in the previous step were used to modulate the GM, WM and CSF tissue maps of the participants’ brains to compare actual volumetric differences among groups. (6) Modulated tissue segments were converted into an isotropic voxel resolution of 1.5 × 1.5 × 1.5 mm. All normalised, segmented and modulated MNI standard space images were then smoothed with an 8-mm Gaussian kernel before voxelwise group comparisons. Segmented tissue volumes ((i.e. (GM, WM and CSF) were estimated in cubic millimetres by counting the voxels representing the native space of the GM, WM and CSF. Total intracranial volume was calculated as the sum of the GM, WM and CSF volumes.
Statistical Analysis
Statistical analysis was performed using the Statistical Package for Social Sciences (SPSS) software package (SPSS 20 for Windows, Chicago, IL, USA). Analysis of variance and chi-squared test were respectively applied to compare the continuous and categorical variables of the demographic data among the three groups (participants with the AA, AT and TT genotypes). Smoothed and modulated GM segments were analysed with SPM8 by using the framework of a general linear model. Analysis of covariance was used by covarying age, sex and educational levels to reveal the random effect of SORL1 rs3824968 on GM volumes. To prevent possible partial volume effects around the margin between the GM and WM, all voxels with a GM probability value of <0.2 (range: 0–1) were eliminated. In this study, to further investigate the main effects of age-by-SNP interaction on regional GM volumes, voxelwise covariate interaction analysis was used with the SORL1 genotype as a condition and age as a covariate, controlling for sex and education level as nuisance variables. In each model, the main effect and interaction were explored using F tests. Post hoc t tests were subsequentially performed to examine the relationships between genotypic groups for any significant main effects and interaction clusters using Bonferroni corrections for multiple comparisons. The problem of multiple comparisons was corrected using a Monte Carlo simulation created in AlphaSim and implemented in the Analysis of Functional NeuroImages software (http://afni.nimh.nih.gov/afni/). Based on the simulation, the statistical threshold of F tests and post hoc tests were set at corrected Palpha of <0.05, with a minimum cluster size of 298 voxels (AlphaSim with the following parameters: single voxel P value of 0.005, 5000 simulations, FWHMx/y/z = 7.5/8.7/8.1 mm with a GM mask) for multiple comparisons. The coordinates of each significant cluster were transformed from MNI coordinates into Talairach coordinates by using the GingerALE toolbox (BrainMap Development Team; http://brainmap.org/ale/index.html). The anatomical structures of the coordinates representing significant clusters were identified using the Talairach and Tournoux atlas46. The volume of each significant cluster was extracted from modulated GM segments in MNI space for each participant and further correlated to cognitive assessments in the entire study group. Partial correlation analyses between regional GM volume (crucial results of genetic effect/age-by-SNP interaction) and cognitive performance were controlled for nuisance variables (age, sex and educational levels) and corrected for multiple testing with Bonferroni correction. Since APOE is the major risk factor for AD, additional statistical analyses were performed using age, sex, educational level and APOE genotype (ε2/ε2, ε2/ε3, ε3/ε3, ε2/ε4 and ε3/ε4) as nuisance variable to account for potential effect by APOE status.
Additional Information
How to cite this article: Huang, C.-C. et al. Effect of Alzheimer’s Disease Risk Variant rs3824968 at SORL1 on Regional Gray Matter Volume and Age-Related Interaction in Adult Lifespan. Sci. Rep. 6, 23362; doi: 10.1038/srep23362 (2016).
References
Andersen, O. M. et al. Neuronal sorting protein-related receptor sorLA/LR11 regulates processing of the amyloid precursor protein. Proc Natl Acad Sci USA 102, 13461–13466, 10.1073/pnas.0503689102 (2005).
Corder, E. H. et al. Gene dose of apolipoprotein E type 4 allele and the risk of Alzheimer's disease in late onset families. Science 261, 921–923 (1993).
Rogaeva, E. et al. The neuronal sortilin-related receptor SORL1 is genetically associated with Alzheimer disease. Nat Genet 39, 168–177, 10.1038/ng1943 (2007).
Andersen, O. M. et al. Molecular dissection of the interaction between amyloid precursor protein and its neuronal trafficking receptor SorLA/LR11. Biochemistry 45, 2618–2628, 10.1021/bi052120v (2006).
Offe, K. et al. The lipoprotein receptor LR11 regulates amyloid beta production and amyloid precursor protein traffic in endosomal compartments. J Neurosci 26, 1596–1603, 10.1523/JNEUROSCI.4946-05.2006 (2006).
Yin, R. H., Yu, J. T. & Tan, L. The Role of SORL1 in Alzheimer's Disease. Mol Neurobiol 51, 909–918, 10.1007/s12035-014-8742-5 (2015).
Lee, J. H. et al. The association between genetic variants in SORL1 and Alzheimer disease in an urban, multiethnic, community-based cohort. Arch Neurol 64, 501–506, 10.1001/archneur.64.4.501 (2007).
Liu, F. et al. A study of the SORL1 gene in Alzheimer's disease and cognitive function. J Alzheimers Dis 18, 51–64, 10.3233/JAD-2009-1137 (2009).
Tan, E. K. et al. SORL1 haplotypes modulate risk of Alzheimer's disease in Chinese. Neurobiol Aging 30, 1048–1051, 10.1016/j.neurobiolaging.2007.10.013 (2009).
Guo, L. H. et al. SORL1 genetic variants and cerebrospinal fluid biomarkers of Alzheimer's disease. Eur Arch Psychiatry Clin Neurosci 262, 529–534, 10.1007/s00406-012-0295-x (2012).
Felsky, D. et al. The SORL1 gene and convergent neural risk for Alzheimer's disease across the human lifespan. Mol Psychiatry 19, 1125–1132, 10.1038/mp.2013.142 (2014).
Alexopoulos, P. et al. Impact of SORL1 single nucleotide polymorphisms on Alzheimer's disease cerebrospinal fluid markers. Dement Geriatr Cogn Disord 32, 164–170, 10.1159/000332017 (2011).
Kolsch, H. et al. Influence of SORL1 gene variants: association with CSF amyloid-beta products in probable Alzheimer's disease. Neurosci Lett 440, 68–71, 10.1016/j.neulet.2008.05.049 (2008).
Ning, M. et al. Amyloid-beta-related genes SORL1 and ACE are genetically associated with risk for late-onset Alzheimer disease in the Chinese population. Alzheimer Dis Assoc Disord 24, 390–396, 10.1097/WAD.0b013e3181e6a575 (2010).
Reynolds, C. A. et al. Sortilin receptor 1 predicts longitudinal cognitive change. Neurobiol Aging 34, 1710 e1711–1718, 10.1016/j.neurobiolaging.2012.12.006 (2013).
Houlihan, L. M. et al. Replication study of candidate genes for cognitive abilities: the Lothian Birth Cohort 1936. Genes Brain Behav 8, 238–247, 10.1111/j.1601-183X.2008.00470.x (2009).
Bralten, J. et al. Association of the Alzheimer's gene SORL1 with hippocampal volume in young, healthy adults. Am J Psychiatry 168, 1083–1089, 10.1176/appi.ajp.2011.10101509 (2011).
Furuya, T. K. et al. SORL1 and SIRT1 mRNA expression and promoter methylation levels in aging and Alzheimer's Disease. Neurochem Int 61, 973–975, 10.1016/j.neuint.2012.07.014 (2012).
Taki, Y. et al. Correlations among brain gray matter volumes, age, gender and hemisphere in healthy individuals. PLoS One 6, e22734, 10.1371/journal.pone.0022734 (2011).
Good, C. D. et al. A voxel-based morphometric study of ageing in 465 normal adult human brains. Neuroimage 14, 21–36, 10.1006/nimg.2001.0786 (2001).
Reynolds, C. A. et al. Sequence variation in SORL1 and dementia risk in Swedes. Neurogenetics 11, 139–142, 10.1007/s10048-009-0210-4 (2010).
Reitz, C. et al. Meta-analysis of the association between variants in SORL1 and Alzheimer disease. Arch Neurol 68, 99–106, 10.1001/archneurol.2010.346 (2011).
Shibata, N. et al. Genetic association between SORL1 polymorphisms and Alzheimer's disease in a Japanese population. Dement Geriatr Cogn Disord 26, 161–164, 10.1159/000149821 (2008).
Kimura, R. et al. SORL1 is genetically associated with Alzheimer disease in a Japanese population. Neurosci Lett 461, 177–180, 10.1016/j.neulet.2009.06.014 (2009).
Felsky, D. et al. The SORL1 gene and convergent neural risk for Alzheimer's disease across the human lifespan. Mol Psychiatry, 10.1038/mp.2013.142 (2013).
Jack, C. R., Jr. et al. 11C PiB and structural MRI provide complementary information in imaging of Alzheimer's disease and amnestic mild cognitive impairment. Brain 131, 665–680, 10.1093/brain/awm336 (2008).
Oh, H., Madison, C., Villeneuve, S., Markley, C. & Jagust, W. J. Association of Gray Matter Atrophy with Age, β-Amyloid and Cognition in Aging. Cerebral Cortex, 10.1093/cercor/bht017 (2013).
Price, J. L. & Morris, J. C. Tangles and plaques in nondemented aging and “preclinical” Alzheimer's disease. Ann Neurol 45, 358–368 (1999).
Chetelat, G. et al. Relationship between atrophy and beta-amyloid deposition in Alzheimer disease. Ann Neurol 67, 317–324, 10.1002/ana.21955 (2010).
Hong, C. J. et al. Association study of apolipoprotein E epsilon4 with clinical phenotype and clozapine response in schizophrenia. Neuropsychobiology 42, 172–174, 26689 (2000).
Cuenco, K. T. et al. Association of distinct variants in SORL1 with cerebrovascular and neurodegenerative changes related to Alzheimer disease. Arch Neurol 65, 1640–1648, 10.1001/archneur.65.12.1640 (2008).
Cash, D. M. et al. The pattern of atrophy in familial Alzheimer disease: volumetric MRI results from the DIAN study. Neurology 81, 1425–1433, 10.1212/WNL.0b013e3182a841c6 (2013).
Ell, S. W., Marchant, N. L. & Ivry, R. B. Focal putamen lesions impair learning in rule-based, but not information-integration categorization tasks. Neuropsychologia 44, 1737–1751, 10.1016/j.neuropsychologia.2006.03.018 (2006).
Ashby, F. G., Alfonso-Reese, L. A., Turken, A. U. & Waldron, E. M. A neuropsychological theory of multiple systems in category learning. Psychol Rev 105, 442–481 (1998).
Monchi, O., Petrides, M., Petre, V., Worsley, K. & Dagher, A. Wisconsin Card Sorting revisited: distinct neural circuits participating in different stages of the task identified by event-related functional magnetic resonance imaging. J Neurosci 21, 7733–7741 (2001).
Jacobson, M. W., Delis, D. C., Bondi, M. W. & Salmon, D. P. Asymmetry in auditory and spatial attention span in normal elderly genetically at risk for Alzheimer's disease. J Clin Exp Neuropsychol 27, 240–253, 10.1080/13803390490515441 (2005).
Baddeley, A. D., Bressi, S., Della Sala, S., Logie, R. & Spinnler, H. The decline of working memory in Alzheimer's disease. A longitudinal study. Brain 114 (Pt 6), 2521–2542 (1991).
Toepper, M., Beblo, T., Thomas, C. & Driessen, M. Early detection of Alzheimer's disease: a new working memory paradigm. Int J Geriatr Psychiatry 23, 272–278, 10.1002/gps.1873 (2008).
Meyer-Lindenberg, A. & Weinberger, D. R. Intermediate phenotypes and genetic mechanisms of psychiatric disorders. Nat Rev Neurosci 7, 818–827, 10.1038/nrn1993 (2006).
Zhao, Y., Cui, J. G. & Lukiw, W. J. Reduction of sortilin-1 in Alzheimer hippocampus and in cytokine-stressed human brain cells. Neuroreport 18, 1187–1191, 10.1097/WNR.0b013e32821c56c4 (2007).
Simard, M. & van Reekum, R. Memory assessment in studies of cognition-enhancing drugs for Alzheimer's disease. Drugs Aging 14, 197–230 (1999).
Sheehan, D. V. et al. The Mini-International Neuropsychiatric Interview (M.I.N.I.): the development and validation of a structured diagnostic psychiatric interview for DSM-IV and ICD-10. J Clin Psychiatry 59 Suppl 20, 22-33; quiz 34–57 (1998).
Folstein, M. F., Folstein, S. E. & McHugh, P. R. “Mini-mental state”. A practical method for grading the cognitive state of patients for the clinician. J Psychiatr Res 12, 189–198 (1975).
Morris, J. C. The Clinical Dementia Rating (CDR): current version and scoring rules. Neurology 43, 2412–2414 (1993).
Ashburner, J. A fast diffeomorphic image registration algorithm. Neuroimage 38, 95–113, 10.1016/j.neuroimage.2007.07.007 (2007).
Talairach, J. & Tournoux, P. Co-planar Stereotaxic Atlas of the Human Brain. 3-dimensional Proportional System: An Approach to Cerebral Imaging. (Thieme Medical Publishers, 1988).
Acknowledgements
This study was supported in part by grants from the Ministry of Science and Technology (MOST) of Taiwan (NSC-101-2314-B-075-040, NSC-102-2321-B-010-023, MOST-104-2633-B-400-002, MOST-104-2314-B-075-078-MY2); National Health Research Institute (NHRI-EX103-10310EI); Taiwan Veterans General Hospital (VGHUST102-G1-2-1, VGHUST103-G1-4-1 and V103C-048, TCVGH-103G213); Brain Research Center, National Yang-Ming University, Taiwan; and the Ministry of Education, Aim for the Top University Plan, Taiwan. The authors also acknowledge the MRI support received from the MRI Core Laboratory of National Yang-Ming University, Taiwan.
Author information
Authors and Affiliations
Contributions
C.C.H., S.J.T. and C.P.L. conceived the experiment, C.C.H., M.E.L., Y.H.W. and T.R.C. conducted the experiment. C.C.H. and K.H.C. analysed the results. C.C.H. wrote the main manuscript and H.W.K. and M.E.L. edited the manuscript. A.C.Y. gave technical support and conceptual advice. All authors discussed the results and implications and commented on the manuscript at all stages.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Huang, CC., Liu, ME., Kao, HW. et al. Effect of Alzheimer's Disease Risk Variant rs3824968 at SORL1 on Regional Gray Matter Volume and Age-Related Interaction in Adult Lifespan. Sci Rep 6, 23362 (2016). https://doi.org/10.1038/srep23362
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep23362
This article is cited by
-
Case report of first-episode psychotic symptoms in a patient with early-onset Alzheimer’s disease
BMC Psychiatry (2020)
-
Risk factor SORL1: from genetic association to functional validation in Alzheimer’s disease
Acta Neuropathologica (2016)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.