Abstract
The anticancer activities of ginsenosides are widely reported. The structure-activity relationship of ginsenosides against cancer is not well elucidated because of the unavailability of these compounds. In this work, we developed a transformation method to rapidly produce rare dehydroxylated ginsenosides by acid treatment. The optimized temperature, time course and concentration of formic acid were 120°C, 4 h and 0.01%, respectively. From 100 mg of Rh1, 8.3 mg of Rk3 and 18.7 mg of Rh4 can be produced by acid transformation. Similarly, from 100 mg of Rg3, 7.4 mg of Rk1 and 15.1 mg of Rg5 can be produced. From 100 mg of Rh2, 8.3 mg of Rk2 and 12.7 mg of Rh3 can be generated. Next, the structure-activity relationships of 23 ginsenosides were investigated by comparing their cytotoxic effects on six human cancer cells, including HCT-116, HepG2, MCF-7, Hela, PANC-1 and A549. The results showed that: (1) the cytotoxic effect of ginsenosides is inversely related to the sugar numbers; (2) sugar linkages rank as C-3 > C-6 > C-20; (3) the protopanaxadiol-type has higher activities; (4) having the double bond at the terminal C20-21 exhibits stronger activity than that at C20-22; and (5) 20(S)-ginsenosides show stronger effects than their 20(R)-stereoisomers.
Similar content being viewed by others
Introduction
Ginseng is one of the best-selling herbal products in the world. It has gained increasing attention in the treatment of cardiovascular diseases, diabetes and central nervous diseases1,2,3. It is generally believed that triterpenoid saponins called ginsenosides are the major active constituents and these are also used as the quality control markers4,5. There is also a popular use of ginsenosides with prominent efforts on anticancer activities associated with cell proliferation6, apoptosis7, angiogenesis8, oxidative stress9, inflammation10 and metastasis of cancer11.
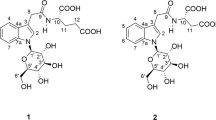
Most ginsenosides share a dammarane triterpenoid structure. Differences in sugar types, numbers and attachment positions provide diversity in structures. By the presence of a hydroxyl moiety at C-6, ginsenosides can be divided into two groups, the protopanaxadiol (PPD) and protopanaxatriol (PPT) group. Ginsenoside Rb1, Rd, Rc and Rb2 are the major compounds of the PPD-type (Fig. 1a) present in ginseng herbs, with sugar moieties attached to the β-OH at C-3 and/or C-20. Ginsenosides Re, Rg1, Rg2, Rf and Rh1 are the main compounds of the PPT-type (Fig. 1b), with sugar moieties linked to the α-OH at C-6 and/or β-OH at C-20.
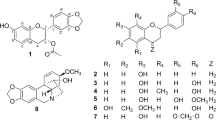
Besides naturally occurring substances, some rare ginsenosides with less sugar moieties and hydroxyl groups continue to be identified. Since most of rare ginsenosides have been commercially unavailable, various methods were developed to generate them, such as chemical treatment12, heating13, enzymatic14 and microbial15,16 treatment. Wang et al. observed that transformation of ginsenosides by artificial gastric juice showed rapid degradation of ginsenosides and could increase cytotoxicity toward cancer cells17. As the main functional component detected in blood or organs after oral administration of ginsenosides, large quantities of compound K (CK) can be produced from cheap monosaccharide via microbial fermentation18. We have previously reported that a steaming process caused extensive conversion of ginsenosides in Panax ginseng to new degraded ones13. The major markers including Rh3, Rk3, Rh4, Rk1, Rk2 and Rg5 (Fig. 1c) were observed in the steamed ginseng, but not detected in raw samples. Rare ginsenosides were reported to show remarkable chemopreventive effects. For example, ginsenoside Rk1 could induce both G1 phase arrest and autophagy at an earlier stage of treatment in HepG2 cells. Rk1 could inhibit telomerase activity and induce apoptosis in HepG2 cells19,20.
To date, efficient methods are limited for producing ginsenosides, especially for the rare types. Correspondingly, because of the unavailability of ginsenosides, the correlation of structures to anticancer activities is still not fully elucidated. In this study, three readily available ginsenosides (Rh1, Rg3 and Rh2) were used as the materials to produce three pairs of C-20(21)/C-20(22) double-bond isomers by chemical transformation. The effects of main factors on transformation of ginsenosides were investigated by ultra-performance liquid chromatography (UPLC) analysis. We explored the anticancer effects of 23 ginsenosides on six human cancer cell lines and uncovered the possible structural-activity relationships based on the number of sugar moieties, position of the sugar moieties, types of aglycone, position of double bond and stereoselectivity. An understanding of these relationships may provide insights into chemical and pharmacological approaches for developing novel cancer chemopreventive compounds.
Results
Transformation of ginsenosides
Effects of temperature on saponin transformation
The effects of temperature on ginsenosides transformation were investigated. Temperatures at 80°C, 100°C, 120°C and 140°C were involved. The results showed that the ginsenoside products increased with increasing temperature and achieved to the highest at 120°C. With a temperature of 140°C, concentrations of products were lower than those at 120°C (Fig. 2a).
Effects of time course on saponin transformation
The effects of heating time on the conversion of ginsenosides were also investigated (Fig. 2b). The contents of original ginsenosides declined gradually at 120°C from 1 to 6 h. Correspondingly, six newly occurring saponins increased gradually during the first 4 h and showed limited increase from 4–6 h. To improve the efficiency, 4 h was selected for ginsenosides transformation.
Effects of acid on saponin transformation
The pH environment is another determinant factor in the transformation process. To investigate the effects of acid concentration on saponin transformation, different concentrations of formic acid (0.01%, 0.1%, 0.5%, 2% and 5%) were employed (Fig. 2c). The results demonstrated that 0.01% of formic acid provided the highest yields.
Yields of rare ginsenosides
Preparation of ginsenoside Rk3/Rh4, Rk1/Rg5 and Rk2/Rh3 were achieved by dehydration at C-20 with 0.01% of formic acid at 120°C for 4 h. The reaction products were separated by UPLC (Fig. 3) and their structures were determined by 13C NMR spectroscopy (Supplementary Fig. S1–S6). The peaks with retention times of 4.7, 6.9, 7.0, 7.4, 8.3, 8.4, 8.7, 10.6 and 10.8 min corresponded to ginsenoside Rh1, Rk3, Rh4, Rg3, Rk1, Rg5, Rh2, Rk2 and Rh3, respectively. As shown in Table 1, from 100 mg of Rh1, 8.3 mg of Rk3 and 18.7 mg of Rh4 can be produced by acid transformation. Similarly, from 100 mg of Rg3, 7.4 mg of Rk1 and 15.1 mg of Rg5 can be produced. From 100 mg of Rh2, 8.3 mg of Rk2 and 12.7 mg of Rh3 can be generated. It seemed the yield of C-20(22) was higher than that of C-20(21) isomer.
Monitoring of ginsenosides transformation by ultra-performance liquid chromatography with UV detection at 203 nm.
(a) Transformation of ginsenoside Rh1 to Rk3 and Rh4; (b) ginsenoside Rg3 to Rk1 and Rg5; (c) ginsenoside Rh2 to Rk2 and Rh3. The chromatographic peaks were identified by comparing with the reference compounds.
Structure-activity relationships of ginsenosides against cancer
A total of 23 ginsenosides were collected, from which the six rare ginsenosides produced by transformation were involved. Six human cancer cell lines were tested to uncover the structure-activity relationships of ginsenosides, including HCT-116, HepG2, MCF-7, Hela, PANC-1 and A549. The results from HepG2 cancer cells were used to elucidate structure-activity relationships of ginsenosides. Similar structure-activity relationships were obtained on other cancer cell lines shown in Supplementary Fig. S7.
Number of sugar moieties
The anticancer activities of ginsenosides with tetrasaccharide glycoside (ginsenosides Rb1, Rb2), trisaccharide glycoside (ginsenosides Re), disaccharide glycoside (ginsenosides Rk1, Rg1), monosaccharide glycoside (ginsenosides Rh2, Rh3, CK) and sapogenin (protopanaxadiol, protopanaxatriol) were compared. As shown in Fig. 4a, ginsenosides Rb1, Rb2, Re and Rd with three or four sugar moieties could almost not inhibit the growth of human cancer cells even at the concentration of 80 μM. In contrast, these ginsenosides promoted the proliferation at low concentrations. Rf and Rg1 having two sugar moieties showed weak cytotoxicity with IC50 values more than 80 μM. Ginsenosides with one sugar moiety could significantly inhibit the cell proliferation. Ginsenoside Rh2 and metabolite CK showed strongest cytotoxic effect among all the ginsenosides at a concentration at 10 μM. PPD and PPT, the aglycones of two dammarane types of ginsenoside, had medium cytotoxic effects with IC50 between 20–40 μM.
Antiproliferative effects of ginsenosides on human cancer HepG2 cells.
(a) Comparison of ginsenosides with different sugar numbers; (b) ginsenosides with different linkage positions of sugars; (c) ginsenosides with different types of aglycones; (d) ginsenosides with different positions of double bonds; (e) ginsenosides with different stereoselectivities of 20(R) and 20(S). The anitproliferative effects were determined by the MTT assay and calculated by comparison with the control after exposure to 5, 10, 20, 40 and 80 μM of ginsenosides in each group for 24 h. The data were expressed as the mean ± SD.
The results demonstrated that the activity of the ginsenosides ranked in a decreasing order as: monosaccharide glycoside > sapogenin > disaccharide glycoside > trisaccharide glycoside > tetrasaccharide glycoside. It implied that the sugar numbers is inversely related to the cytotoxic effects of ginsenosides.
Position of sugar
As shown in Fig. 4b, ginsenosides Rh1, Rh2 and F1 have one glucose but different linkage positions. Results showed that these ginsenosides exhibited differences in cytotoxic activities. For instance, ginsenoside Rh1, with sugar linkage at C-6, exerted stronger antiproliferative effect than ginsenoside F1 with sugar linkage at C-20. Ginsenoside Rh2, with a connection site at C-3, showed the strongest cytotoxic activities. Taken together, the cytotoxic potency of ginsenosides has been demonstrated to be in the order: C-3 > C-6 > C-20.
Different types of aglycone
PPD and PPT are the two major types of dammarane saponins. Compounds protopanaxadiol and protopanaxatriol, the aglycones of PPD-type and PPT-type ginsenosides, inhibited growth of cancer cells in a similar dose-dependent manner, but showed no significant difference. Rh2 and S-Rh1 along with CK and F1, possess the same sugar moiety but different types of aglycone. As shown in Fig. 4c, Rh1 (PPT-type) did not exert significant antiproliferative effects, while Rh2 (PPD-type) could inhibit the proliferation of 80% cells at the concentration of 10 μM. As an important ginsenoside of PPT-type, F1 could not inhibit the growth of cancer cells even at 80 μM, while CK (PPD-type) showed stronger anticancer activity than F1. Our observations suggest that different types of aglycone may make different contributions to cytotoxic activity and it is possible that PPD-type ginsenosides have better anticancer activity.
Position of double bond
Ginsenosides Rk3/Rh4, Rk1/Rg5 and Rk2/Rh3 are the dehydroxylated products of Rh1, Rg3 and Rh2 at C-20, respectively. As shown in Fig. 4d, ginsenosides Rk1 and Rk3 with the double bond at C20-21 showed higher anticancer effects than Rg5 and Rh4 with double bond at C20-22. Ginsenoside Rh3 showed moderate cytotoxic effects from concentration range of 5 to 80 μM; while, Rk2 just showed some antiproliferative effect at a high concentration higher than 80 μM. In summary, ginsenosides with double bond at C20-21 exhibited more effective antitumor activities than those at C20-22.
Stereoselectivity of 20(R) and 20(S)
20(R) and 20(S) is a pair of stereoisomers with different position of C-20 hydroxyl in ginsenosides. 20(S)-OH is geometrically closer to the C-12 hydroxyl than 20(R)-OH. As shown in Fig. 4e, the stereostructure of the C-20 hydroxyl may influence the biological and pharmacological effects of ginsenosides. 20(S)-PPD showed stronger chemopreventive effect than its 20(R)-stereoisomer.
Discussion
C-20 dehydroxylated dammarane-type ginsenosides, a group of rare ginsenosides with remarkable anticancer activities, mainly exist in steamed ginseng21. In this study, we aimed to prepare these rare ginsenosides by chemical transformation. Biotransformation methods were widely used to transfer original ginsenosides to deglycosylated products, such as Rd and CK22,23. Such biotransformation is not suitable to gain the dehydroxylated ones. Since C-20 rare dehydroxylated ginsenosides were present with high concentrations in ginseng after a steaming process, transformation by heating was considered in this study.
Ginsenosides could not be transformed under the neutral condition and more by-products were produced when exposed to strong acids24. It was also known that ginsenosides tend to be transformed by deglycosylation in high acidic environment12. In this work, a low amount of acid was used to obtain dehydroxylated products and also avoid deglycosylation reactions. This chemical transformation method was first applied to prepare rare dehydroxylated ginsenosides. After heating at 120°C for 4 h with 0.01% formic acid, ginsenoside Rh1, Rg3 and Rh2 were ultimately converted to three pairs of raw ginsenosides. The total transformation efficiency was more than 20%.
Due to the low specificity of chemical transformation, by-products produced during the conversion reduced the yield of the target products. To compare the chromatograms after transformation, two unknown by-products with larger polarity were observed. In addition, we found that acid treatments can induce epimerization reactions of ginsenosides and their stereoisomers were further identified in reaction solution. Taken together, the existence of the by-products and the epimerization of the raw materials may result in the incompleteness of this conversion. Reduction of the side-reactions and ways to maximize the target-products, will be our emphases in further researches.
To the best of our knowledge, there is no systematic evaluation of the structure-activity relationship of the rare ginsenosides on the antiproliferative effect of cancer cells. Six human cancer cell lines were used to screen the activity of compounds and to uncover the structure-activity relationship of 23 ginsenosides (Supplementary Fig. S7). Sugar moiety is a key factor in moderating the activity of ginsenosides. The increasing number of sugar moieties reduces the cytotoxic effect of the compound. These observations imply that the anticancer effects of ginsenosides are inversely related to the number of sugar moieties and get the strongest data when only one sugar is linked. As the hydrophobicity influences absorption of compounds, the presence of sugar moieties may reduce the hydrophobic character of the compounds and decrease their permeabilities to cell membranes25. This hydrophobic property is required to interact with specific membrane proteins or to pass into the nucleus and may be the reason why their anticancer activities increase with the decrease of number of sugar moieties for ginsenosides. Besides, the anticancer activities of ginsenosides also depend on their hydrophilicity-lipophilicity balance.
Compared with C-3 linkage, sugar moieties at C-6 or C-20 increases steric hindrance for the entrance of ginsenosides and blocks compounds from extracellular binding to their targets, thus significantly reducing the anticancer activities of ginsenosides26. Previously, it was reported that 25-OH-PPD had stronger chemopreventive effect than 25-OH-PPT27, which is consistent with our observations that PPD-type ginsenosides have better anticancer activity.
Using the above optimal transformation conditions, rare ginsenosides with double bond at C-20 were obtained. We can speculate that the terminal double bond contribute more greatly to the antitumor activity in product ginsenosides in most cases. 20(S)-PPD and 20(R)-PPD, another pair of stereoisomers on the position of the C-20, have been compared for their anticancer activities. Wang et al. observed that 20(S)-PPD showed stronger chemopreventive effect than its 20(R)-stereoisomer28, which is in accordance with our speculation. The reason may be that C-20 hydroxyl group is spatially closer to the C-12 hydroxyl for 20(S) stereoisomer and stereoselective interactions with lipid membranes may affect the antiproliferative activity of 20(R)/(S)-ginsenosides29. In summary, we determined that the ginsenosides (1) with one sugar or just aglycone; (2) with the sugar linkage at C-3; (3) PPD type of aglycone; (4) with double bond at C20-21; (5) stereoselectivity of 20(S) contribute most to their anticancer effects.
With respect to the potency of these ginsenosides, our results indicated that most of the rare ginsenosides have dominant chemotherapeutic activities, such as Rk1, Rk3 and Rh3. Further research in terms of pharmacological and mechanisms of activities are underway to bring clarity to these findings. Based on the uncovered structure-activity relationships, we will not only obtain useful information for modifying ginsenosides structure, but also select ginsenosides with superior anticancer activity for further testing and eventual drug development.
Methods
Materials
High performance liquid chromatography (HPLC) grade acetonitrile and methanol were obtained from Merck (Darmstadt, Germany). Formic acid with a purity of 99% was from ROE (Newark, New Castle, USA). Deionized water (18 MΩ cm−1) was prepared by distilled water through a Milli-Q system (Millipore, Bedford, MA, USA). Other reagents of analytical purity were utilized for sample preparation.
Reference ginsenosides, Rb1, Rb2, Rc, Rd, Re, Rf, 20(R)/20(S)-Rh1, Rh2, Rg1, Rg2, 20(R)/20(S)-Rg3, F1, CK, protopanaxadiol and protopanaxatriol were purchased from Jilin University (Changchun, China). Their structures shown in Fig. 1 were further elucidated by spectroscopic methods (1H, 13C NMR and MS) with a purity of more than 95% for each compound in the authors' laboratory.
Sample preparation
A sealed tube (Beijing Synthware Glass, China) was charged with reference ginsenosides (100 mg) and formic acid in 50 mL of 30% (v/v) methanol. The mixture was stirred at 80°C, 100°C, 120°C and 140°C for 1, 2, 4 and 6 h, respectively. Then the reaction mixture was cooled at room temperature, evaporated to dryness and redissolved with DMSO. The solution was centrifuged at 13,000 rpm for 10 min. Two microliters of the supernatants were subjected to UPLC for analysis.
UPLC analysis
Analysis was performed on an Agilent 1290 Infinity UPLC system (Agilent Technology, Waldbronn, Germany) equipped with a binary pump, a diode-array detector, an auto sampler and a thermostatically controlled column compartment. Samples were separated on an Agilent ZORBAX Extend-C18 column (2.1 mm × 50 mm, 1.8 μm) at 25°C using water (solvent A) and acetonitrile (solvent B). A gradient elution program was used according to the following profile: 20% B at 0–0.5 min, 20–26% B at 0.5–3 min, 26–38% B at 3–6 min, 38–55% B at 6–8 min, 55% B at 8–10 min and 55–100% B at 10–15 min. The flow rate was kept at 0.8 mL/min and the optimum wavelength was set at 203 nm.
HPLC preparation
An Agilent 1100 HPLC system (Agilent Technologies, Santa Clara, CA, USA) was utilized for ginsenosides preparation. The chromatographic separation was achieved on an Agilent ZORBAX SB-C18 semi-preparative column (9.4 mm × 250 mm, 5 μm) with a solvent flow rate of 1 mL/min at the temperature of 25°C. The mobile phase was composed of water (A) and acetonitrile (B). The products of ginsenoside Rh1, Rg3 and Rh2 were carried out with the constant elution of 50% B, 65% B and 85% B, respectively. The collected samples were evaporated under vacuum and lyophilized, then stored at 4°C.
Cell culture
The cancer cell lines used in the study include human colon carcinoma (HCT-116), human liver carcinoma (HepG2), human cervical carcinoma (Hela), human breast adenocarcinoma (MCF-7), human pancreatic cancer (PANC-1) and human lung carcinoma (A549). All cells were purchased from American Type Culture Collection (Manassas, VA, USA) and grown in DMEM (Dulbecco's modified Eagles Medium). To ensure growth and viability of the cells, the medium was supplemented with 10% fetal bovine serum and 1% penicillin/streptomycin (Gibco, USA). The cells were cultured at 37°C in a humid incubator with 5% CO2.
Cell proliferation assay
Assessment of cell proliferation was performed by MTT [3-(4,5-dimethylthiazol-2-yl)-2,5-diphenyl-2H-tetrazolium bromide] assay. Cells were seeded in 96-well plates at a density of 5 × 103 per well and allowed to attach for 24 h. Then they were treated with different concentrations of ginsenosides. Controls were exposed to culture medium with DMSO. After 24 h, 10 μL of MTT (5 mg/mL) was added to culture medium in each well and the plates were incubated for additional 4 h. Then the medium was removed and 200 μL DMSO was added to dissolve the blue formazan crystals converted from MTT. Finally, absorbance values were measured at 490 nm with Synergy 2 Multi-Mode Microplate Reader (BioTek Instruments Inc., USA). Results were expressed as a percentage versus control (vehicle set at 100%).
Statistical analysis
Data were presented as means ± standard deviation (SD) from triplicate experiments performed in a parallel manner. Statistical analysis was conducted using Student's t-test.
References
Lee, M. S., Yang, E. J., Kim, J. I. & Edzard, E. Ginseng for cognitive function in Alzheimer's disease: a systematic review[J]. J Alzheimer's Dis, 18, 339–344 (2009).
Lee, H., Kim, M., Shin, S. S. & Yoon, M. Ginseng treatment reverses obesity and related disorders by inhibiting angiogenesis in female db/db mice. J. Ethnopharmacol. 155, 1342–1352 (2014).
Shishtar, E., Jovanovski, E., Jenkins, A. & Vuksan, V. Effects of Korean White Ginseng (Panax Ginseng C. A. Meyer) on Vascular and Glycemic Health in Type 2 Diabetes: Results of a Randomized, Double Blind, Placebo-controlled, Multiple-crossover, Acute Dose Escalation Trial. Clin. Nutr. Res. 3, 89–97 (2014).
Jia, L., Zhao, Y. & Liang, X. J. Current evaluation of the millennium phytomedicine- ginseng (II): Collected chemical entities, modern pharmacology and clinical applications emanated from traditional Chinese medicine. Curr. Med. Chem. 16, 2924–2942 (2009).
Qi, L. W., Wang, C. Z. & Yuan, C. S. Isolation and analysis of ginseng: advances and challenges. Nat. Prod. Rep. 28, 467–495 (2011).
Chung, K. S. et al. Ginsenoside Rh2 induces cell cycle arrest and differentiation in human leukemia cells by upregulating TGF-beta expression. Carcinogenesis. 34, 331–340 (2013).
Li, J. et al. Ginsenoside Rg1 induces apoptosis through inhibition of the EpoR-mediated JAK2/STAT5 signalling pathway in the TF-1/Epo human leukemia cell line. Asian. Pac. J. Cancer. Prev. 15, 2453–2459 (2014).
Zeng, D., Wang, J., Kong, P., Chang, C. & Li, J. Ginsenoside Rg3 inhibits HIF-1alpha and VEGF expression in patient with acute leukemia via inhibiting the activation of PI3K/Akt and ERK1/2 pathways. Int. J. Clin. Exp. Pathol. 7, 2172–2178 (2014).
Mao, Q., Zhang, P. H., Wang, Q. & Li, S. L. Ginsenoside F(2) induces apoptosis in humor gastric carcinoma cells through reactive oxygen species-mitochondria pathway and modulation of ASK-1/JNK signaling cascade in vitro and in vivo. Phytomedicine. 21, 515–522 (2014).
Shin, Y. M., Jung, H. J., Choi, W. Y. & Lim, C. J. Antioxidative, anti-inflammatory and matrix metalloproteinase inhibitory activities of 20(S)-ginsenoside Rg3 in cultured mammalian cell lines. Mol. Biol. Rep. 40, 269–279 (2013).
Yu, H. et al. Development of liposomal Ginsenoside Rg3: formulation optimization and evaluation of its anticancer effects. Int. J. Pharm. 450, 250–258 (2013).
Kim, M. H. et al. The changes of ginsenoside patterns in red ginseng processed by organic acid impregnation pretreatment. J. Gins. Res. 35, 497–503 (2011).
Qi, L. W. et al. Diagnostic ion filtering to characterize ginseng saponins by rapid liquid chromatography with time-of-flight mass spectrometry. J. Chromatogr. A. 1230, 93–99 (2012).
Kim, B. H. et al. Biotransformation of Korean Panax ginseng by Pectinex. Biol. Pharm. Bull. 29, 2472–2478 (2006).
Choi, J. R. et al. Metabolic activities of ginseng and its constituents, ginsenoside Rb1 and Rg1, by human intestinal microflora. J. Gins. Res. 35, 301–307 (2011).
Chang, K. H., Jo, M. N., Kim, K. T. & Paik, H. D. Evaluation of glucosidases of Aspergillus niger strain comparing with other glucosidases in transformation of ginsenoside Rb1 to ginsenosides Rg3. J. Gins. Res. 38, 47–51 (2014).
Wang, J. R. et al. Transformation of ginsenosides from notoginseng by artificial gastric juice can increase cytotoxicity toward cancer cells. J. Agric. Food Chem. 62, 2558–2573 (2014).
Yan, X. et al. Production of bioactive ginsenoside compound K in metabolically engineered yeast. Cell Res. 24, 770–773 (2014).
Kim, Y. J. et al. Anti-tumor activity of the ginsenoside Rk1 in human hepatocellular carcinoma cells through inhibition of telomerase activity and induction of apoptosis. Biol. Pharm. Bull. 31, 826–830 (2008).
Ko, H., Kim, Y. J., Park, J. S., Park, J. H. & Yang, H. O. Autophagy inhibition enhances apoptosis induced by ginsenoside Rk1 in hepatocellular carcinoma cells. Biosci. Biotechnol. Biochem. 73, 2183–2189 (2009).
Sun, S. et al. Red notoginseng: higher ginsenoside content and stronger anticancer potential than Asian and American ginseng. Food Chem. 125, 1299–1305 (2011).
Zhao, X. et al. Highly selective biotransformation of ginsenoside Rb1 to Rd by the phytopathogenic fungus Cladosporium fulvum (syn. Fulvia fulva). J. Ind. Microbiol. Biot. 36, 721–726 (2009).
Kim, S. H. et al. Enzymatic Transformation of Ginsenoside Rb1 by Lactobacillus pentosus Strain 6105 from Kimchi. J. Gins. Res. 36, 291–297 (2012).
Bae, E. A., Han, M. J., Kim, E. J. & Kim, D. H. Transformation of ginseng saponins to ginsenoside Rh2 by acids and human intestinal bacteria and biological activities of their transformants. Arch. Pharm. Res. 27, 61–67 (2004).
Jia, L. & Zhao, Y. Current evaluation of the millennium phytomedicine--ginseng (I): etymology, pharmacognosy, phytochemistry, market and regulations. Curr. Med. Chem. 16, 2475–2484 (2009).
Li, W. et al. Anti-androgen-independent prostate cancer effects of ginsenoside metabolites in vitro: mechanism and possible structure-activity relationship investigation. Arch. Pharm. Res. 32, 49–57 (2009).
Chen, R. J., Chung, T. Y., Li, F. Y., Lin, N. H. & Tzen, J. T. Effect of sugar positions in ginsenosides and their inhibitory potency on Na+/K+-ATPase activity. Acta. Pharmacol. Sin. 30, 61–69 (2009).
Wang, W. et al. In vitro anti-cancer activity and structure-activity relationships of natural products isolated from fruits of Panax ginseng. Cancer Chemoth. Pharm. 59, 589–601 (2007).
Qi, L. W., Wang, C. Z. & Yuan, C. S. American ginseng: potential structure-function relationship in cancer chemoprevention. Biochem. Pharmacol. 80, 947–954 (2010).
Acknowledgements
This work was financially supported in part by the National Natural Science Foundation of China (Nos.81222052, 81173497 and 81421005) and Jiangsu Province Science Fund for Distinguished Young Scholars (BK20130025).
Author information
Authors and Affiliations
Contributions
L.W.Q. and P.L. designed the research; J.Y.W. and R.Z.G. conducted the chemical preparation; K.Q. and Y.J.Z. did the anticancer screening; K.Q. and Q.L. involved in data analysis and co-wrote the manuscript; L.W.Q. and R.N.A. edited the manuscript. All authors approved the final version of the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Electronic supplementary material
Supplementary Information
supplementary information
Rights and permissions
This work is licensed under a Creative Commons Attribution 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/
About this article
Cite this article
Quan, K., Liu, Q., Wan, JY. et al. Rapid preparation of rare ginsenosides by acid transformation and their structure-activity relationships against cancer cells. Sci Rep 5, 8598 (2015). https://doi.org/10.1038/srep08598
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep08598
This article is cited by
-
Ginsenoside Rg5 as an anticancer drug: a comprehensive review on mechanisms, structure–activity relationship, and prospects for clinical advancement
Pharmacological Reports (2024)
-
Biocatalytic strategies for the production of ginsenosides using glycosidase: current state and perspectives
Applied Microbiology and Biotechnology (2020)
-
Cloning and Characterization of a Ginsenoside-Hydrolyzing α-L-Arabinofuranosidase, CaAraf51, From Cellulosimicrobium aquatile Lyp51
Current Microbiology (2020)
-
Microbial transformation of ginsenoside Rb1, Re and Rg1 and its contribution to the improved anti-inflammatory activity of ginseng
Scientific Reports (2017)
-
Ginseng fermented by mycotoxin non-producing Aspergillus niger: ginsenoside analysis and anti-proliferative effects
Food Science and Biotechnology (2017)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.