Abstract
Help seeking (HS) is a core coping strategy that is directed towards obtaining support, advice, or assistance as means of managing stress. Women have been found to use more HS than men. Neural correlates of sex differences have also been reported in prefrontal-limbic system (PLS) regions that are linked to stress and coping, yet structural differences between men and women relating to HS in the PLS are still unknown. Thus, the association between gray matter volume (GMV) and HS was investigated using voxel-based morphometry (VBM) in a large healthy sample (126 men and 156 women). Results indicated women reported more HS than men did. VBM results showed that the relation between HS scores and GMV differed between men and women in regions of the bilateral orbitofrontal cortex extending to the subgenual anterior cingulate cortex(OFC/sgACC). Among women, higher HS scores were associated with smaller GMV in these areas while a positive correlation between GMV and HS scores was observed among men. These results remained significant after controlling for general intelligence, stress, anxiety and depression. Thus, this study suggested that structural differences between men and women are correlated to characteristic brain regions known to be involved in the PLS which is considered critical in stress regulation.
Similar content being viewed by others
Introduction
Help-seeking (HS) is defined as “any communication about a problem or troublesome event that is directed towards obtaining support, advice, or assistance in times of distress”1. Psychologists consider HS to be a core coping strategy frequently adopted to deal with stressful situations in everyday living2. Lazarus and Folkman3 defined coping as constantly changing cognitive and behavioral efforts to manage specific external or internal demands that have been appraised as exceeding the resources of a person. Prior study suggested HS is an effective strategy for coping stress1 and is negatively correlated with depression, anxiety and interpersonal sensitivity4,5,6.
Previous imaging studies indicate that the prefrontal-limbic system (PLS), including the prefrontal cortex (PFC), anterior cingulate cortex (ACC) and hippocampus, are comprised of brain regions that can be affected by environmental stressors, as well as aspects of coping7,8,9. The PLS has an important role in determining which events are experienced as threatening and potentially stressful as well as regulating behavioral, cognitive and physiological response to a given stressor7,8.
Functional magnetic resonance imaging (MRI) studies have directly implicated the orbitofrontal cortex (OFC) extending to the anterior cingulate cortex (ACC) in regulation of the human stress response during the Montreal imaging stress task9 and serial subtraction stress task10, respectively. Structural imaging research has also revealed greater cumulative adversity and more recent stressful life events are related to smaller gray matter volume in regions of the OFC, medial PFC and other areas11.
Furthermore, the hippocampus is a medial temporal lobe structure that has been linked in regulation of the stress response12. For example, recent research on resilience, the capacity to cope effectively in stressful situations13, implicated the hippocampus in the development of neural circuits that control adequate stress adaptations14. Structural imaging studies have also found decreased hippocampal volume in patients with major depression15 and posttraumatic stress disorder (PTSD)16,17 compared to healthy controls. Moreover, Gianaros and colleagues18 found that chronic stress levels predicted decreases in hippocampal volume of a healthy sample 20 years later.
Although findings on sex difference in coping have been mixed, findings related to the reported use of HS have been more consistent19,20,21. For example, in managing stress, females tend to rely more on coping strategies that concern seeking emotional help and positive self-talk while males may be more likely to adopt problem-focused strategies19,22. Sex differences in coping have also been observed in clinical samples having chronic pain23,24, wherein women with chronic pain report using social support more than their male counterparts do. Sex differences in emotion regulation25 indicate men suppress negative emotions more than women do. In addition, there is considerable evidence linking women's relatively more frequent use of interpersonal coping strategies that reflect “tending and befriending” to sex differences in affiliative neurocircuitry, particularly release of the stress hormone, oxytocin, which is enhanced by estrogen and related to increased interpersonal responsiveness26,27,28,29.
Together these findings consistently implicate engagement of the PLS to coping. However, regional differences in PLS brain regions, i.e., gray matter volume (GMV) related to individual variability in HS, have not been investigated in healthy samples. In addition, although functional MRI studies on sex difference in emotion regulation have implicated involvement of the PLS30,31, structural bases for differences between men and women in HS are still unknown. In light of sex differences in behavioral and hormonal bases of HS, we speculate that relations between GMV and the reported use of HS also differs between the sexes.
In summary, the goals of this research were to explore structural differences between men and women in the reported use of HS as a stress management strategy using voxel-based morphometry (VBM). Based on findings from behavioral, imaging and neurobiological studies, we hypothesized that sex differences in the use of HS among healthy young adults would be associated with GMV differences in PLS regions, particularly the PFC, ACC and hippocampus.
Methods
Participants and procedures
Participants were 273 young adults (123 men, 150 women; mean age = 19.98 years, SD = 1.21, age range: 18–25 years) from Southwest University (SWU), Chongqing, China who volunteered as part of an ongoing project examining associations between brain imaging, creativity and mental health. All participants were right-handed and screened to confirm healthy development by a self-report questionnaire before the scan. Thus, a reported history of psychiatric or neurological disorders, mental health treatment or use of psychiatric medications were exclusion criterion. The study was approved by the SWU Brain Imaging Center Institutional Review Board.
In accordance with the Declaration of Helsinki (2008), written informed consent was obtained from all participants prior to engaging in the research tasks. First, participants underwent an MRI scan. During the scan, participants were instructed to keep their heads still and to remain awake. The scan was comprised of anatomical imaging (5 minutes), resting state imaging (8 minutes) and diffusion tensor imaging (17 minutes), but only anatomical imaging data was used in this study. Subsequently, participants completed the Chinese version of the HS scale32, Combined Raven's Test (CRT)33, Adolescent Self-Rating Life Events Checklist (ASLEC)34, Trait Anxiety Inventory (STAT-T)35 and Beck Depression Inventory (BDI)36. Participants were compensated 40 RMB for the MRI scan and 15 RMB per hour for completion of self-report measures.
Measures
Help-seeking scale
The use of help-seeking in managing stress was assessed with the 10-item Help-Seeking (HS) subscale of the Chinese Ways of Coping Scale32. The HS scale is widely used for measuring HS tendencies (e.g., asking someone to help me to get over a difficulty; I tried to keep my unpleasant feelings to myself) used to manage real-life stressors. Participants confirmed whether they had used specific HS strategies in past two years by responding “Yes” or “No” to each item. The HS scale is considered to be a valid measure of the tendency to use HS after experiences of stress4. Previous studies have shown that the HS subscale has acceptable 1-week test-retest reliability (r = 0.69)32. In this sample, the Cronbach's alpha was acceptable, α = 0.65.
Assessment of general intelligence
To control for individual differences in intellectual ability in analyses of relations between coping and GMV37, participants completed the Combined Raven's Test (CRT), a recognized intelligence test with a high degree of reliability and validity38. The CRT, which includes the Raven's standard progressive matrices (C, D, E sets) and Raven's colored progressive matrices (A, B, AB sets), consists of 72 items revised for use in Chinese samples by Sun et al33. Number of correct answers given in 40 minutes was used as a psychometric index of individual intelligence in line with standard practice39.
Assessment of negative life events
The ASLEC is a 26 item self-report questionnaire measuring the impact of stressful life events experienced during the past year on five-point scales (1–5); higher scores indicate more severe influence of stressful events34. Events scored as 0 suggesting no occurrence in the past year. Total scores were based on the sum of all items and provided an index of negative life events. The ASLEC has been reported to have satisfactory 2-week test–retest reliability (r = 0.70)34 and is considered to be a valid measure for assessing the influence of negative life events40. In this sample, the scale had a Cronbach's alpha of α = 0.89.
Assessment of anxiety and depression
Trait anxiety and depressive symptoms were assessed with the STAI-T35 and BDI36. Higher scores reflected higher levels of trait anxiety and greater severity of depressive symptoms. In this sample, alphas for the STAI-T and BDI were α = 0.83 and α = 0.83 respectively.
MRI Data Acquisition
MR images were acquired on a 3.0-T Siemens Trio MRI scanner with an 8-channel head coil (Siemens Medical, Erlangen, Germany). High-resolution T1-weighted anatomical images were acquired using a magnetization-prepared rapid gradient echo (MPRAGE) sequence (TR/TE/TI = 1900 ms/2.52 ms/900 ms; Flip angle = 9°; Slices = 176; Slice thickness = 1.0 mm; Resolution matrix = 256 × 256; Voxel size = 1 × 1 × 1 mm3).
Voxel-Based Morphometry
Images were processed using the SPM8 (Wellcome Department of Cognitive Neurology, London, UK) implemented in Matlab 7.8 (MathWorks Inc., Natick, MA, USA). Each MR image was first displayed in SPM8 to screen for artifacts or gross anatomical abnormalities. For better registration, the re-orientation of images was manually set to the anterior commissure. Segmentation of T1 weighted anatomical images into GM and white matter (WM) was done using new segmentation in SPM8. Subsequently, we performed Diffeomorphic Anatomical Registration through Exponentiated Lie algebra (DARTEL) in SPM8 for registration, normalization (resolution 1.5 × 1.5 × 1.5 mm) and modulation using default parameters41. To ensure regional differences in the absolute amount of GM or WM were conserved, the image intensity of each voxel was modulated by Jacobian determinants. Then, registered images were transformed to Montreal Neurological Institute (MNI) space. Finally, normalized modulated images (GM and WM images) were smoothed with a 10-mm full-width at half-maximum Gaussian kernel (FWHM) to increase signal to noise ratio.
Statistical analysis
Statistical analyses of GMV data were performed using SPM8. In whole-brain analyses, we used a voxel-wise analysis of covariance (ANCOVA) in SPM8 to illuminate whether relations between regional GMV and HS tendencies differed between men and women (i.e., whether the interaction between sex and HS scores was related to GMV). Sex was the group factor (using the one-way ANOVA option of SPM8)42,43 and HS scores, age, CRT score, total GM volume and BDI, STAI-T and ASLEC were covariates. All covariates, except total GM volume, were modeled so that their unique relations with GMV could be observed within each sex using the interactions option in SPM8. Main effects for each covariate and interaction effects of sex on remaining covariates were assessed using t-contrasts. Where significant effects were found, interactions were decomposed via ‘REST’44 to extract regional mean GMV values of each participant from the cluster of voxels showing a significant interaction effect. These effects were analyzed using SPSS 16.0 and the pattern revealed in the interaction was graphed for visualization purposes.
To avoid edge effects around the borders between GM and WM, an absolute threshold masking of 0.2 was used; that is, voxels with gray matter or white matter values lower than 0.2 were excluded from analyses45. For all analyses, the cluster-level statistical threshold was set at P < 0.05 and corrected at the non-stationary cluster correction46 with an underlying uncorrected voxel level of p < 0.001.
Results
Behavioral data
Table 1 presents descriptive statistics and comparisons between men and women on different measures. As shown there, a significant sex difference was observed on HS with women endorsing significantly more use than men did (t = 3.55, p < 0.01, two-tailed t test). Table 2 presents correlations between HS scores and other behavioral variables.
Sex Differences in Associations between HS and GMV
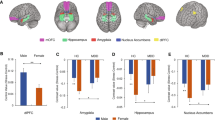
To assess structural differences between men and women in HS, an ANCOVA was used, controlling age, CRT, total GM volume, BDI, STAI-T and ASLEC scores. A sex x HS interaction effect was found on GMV in regions of the bilateral OFC extending to the subgenual ACC (OFC/sgACC; MNI coordinate: 5, 24, −19; t = 4.51, p < 0.05; Cluster size = 4114 mm3; Fig. 1). Specifically, HS scores were negatively related to OFC/sgACC among women but had a positive correlation to OFC/sgACC among men. No other significant effects were found.
Interaction effect between sex and Help-Seeking (HS) scores on regional GMV.
The left panel shows that HS was negatively associated with GMV in regions of the bilateral OFC extending to the subgenual ACC in females, whereas this correlation was positive for males at the peak voxel (x, y, z = 5, 24, −19). The right panel shows the corresponding partial correlation scatterplots of the interaction effects of sex and HS on OFC/sgACC adjusted for age, CRT, total volume of gray matter, negative life events, anxiety and depression for illustration purpose only. The x-axis of each scatterplot represents standardized residuals of HS scores and the y-axis represents standardized residuals of brain regions' GMV. For standardized residual measures, age, CRT and total volume of gray matter, negative life events, anxiety and depression were regressed out. Results were shown with P < 0.05, corrected for multiple comparisons at the cluster-level with an underlying voxel-level of P < 0.001, uncorrected. HS, help-seeking; GMV, gray matter volume; OFC/sgACC, orbitofrontal cortex extending to subgenual anterior cortex; CRT, Combined Raven's Test.
Discussion
To the best of our knowledge, this study is the first to use VBM to assess anatomical differences between men and women in the reported use of HS as a stress management strategy. Consistent with hypotheses and in line with numerous prior behavior studies19,47, women reported more HS than men. More notably, however, differences were found between men and women in the association between HS and GMV in bilateral OFC/sgACC regions. Specifically, among women, HS was negatively correlated to GMV in this region while, among men, this association was positive.
Previous studies have demonstrated that, in PFC regions, reduced GMV (or increased cortical thinning) is associated with increased cortical development reflecting increased functioning of these regions in young adults43,48,49. This finding might suggest sex differences in social maturity that could influence the use of HS in managing stressors. Structural imaging research has also found that gray matter levels peak earlier among women than men50. The OFC region is involved in reward and emotional regulation and more development in this region may contribute to more use of HS. In addition, the OFC combined with the sgACC are core regions involved in stress regulation, aside from the hippocampus51. For examples, functional MRI research using the Montreal imaging stress task9 revealed a profound deactivation of OFC/sgACC among people who reacted to the stressor with a significant increase in cortisol, hence suggesting an inhibitory role of the OFC/sgACC in regulating the stress response. Using a serial subtraction stress paradigm, Wang10 also found an association between OFC/ACC and stress regulation. Moreover, the OFC is involved in adaptive, cognitively-guided coping while OFC dysfunction is linked to less adaptive and emotionally-guided coping52. Previous studies also found that OFC damage dramatically alters the capacity to cope with stressful situations and use HS behavior53.
In addition, the cingulate cortex is considered critical in cognitive and emotional control. Unlike the dorsal ACC which functions in conflict monitoring, error processing and other cognitive manipulations, the sgACC is involved mainly in emotional regulation processing and the prevention of psychiatric disorders such as depression and PTSD54,55. In a functional imaging study of sex difference in emotion regulation, Mak et al.31 found that Women show enhanced activation in regions of the OFC compared to men, when regulating negative emotions. In another study exploring the interaction between cognitive control and emotion, women displayed significantly more OFC activation than men did when regulating the interaction between negative emotion and working memory56. A recent structural imaging also found that larger sgACC volume may related to more negative emotions in teenagers, whereas adult depressive patients show a complementary pattern42. Perhaps, reduced GMV in regions of OFC/sgACC may indicate earlier neuromaturation among young women compared to young men. In turn, this difference may contribute to women's relatively increased use of HS in managing stressors. In conjunction with sex differences in affiliative neurocircuitry28, the more developed OFC/sgACC may equip women with comparatively stronger propensities toward HS behavior.
Despite prior research linking hippocampus involvement with stress responding and regulation9, the current study did not find structural relations between HS and hippocampal volume. Morphological changes of the hippocampus have been observed in patients of stress-related psychiatric disorder such as major depression and PTSD57,58. However, the nature of relations between hippocampal reduction and psychological trauma, PTSD and coping are still controversial59. Because other research found no association between exposure to accumulative stress and reduced hippocampal volume in a non-psychiatric sample11. Given that only healthy young adults participated in the current study, future research is necessary to clarify associations between hippocampal GMV and HS in clinical samples.
Notwithstanding these findings, the main limitations of this study should be mentioned. First, it is not clear whether findings generalize to other age groups or general populations in which HS scores and GMV are more normally-distributed. Second, concerns that automated preprocessing procedures of VBM may result in more preprocessing errors raised in relation to other published studies60 may apply to this research as well. Finally, findings should be considered as provisional. While other GMV differences corresponding to reported use of HS were not revealed or did not survive corrections for multiple comparisons, replications in other large samples are needed to demonstrate the reliability of findings across groups.
In sum, this is the first VBM study to investigate associations between the prefrontal-limbic system and HS as a stress management strategy among women versus men. Associations between GMV in prefrontal-limbic system (OFC/sgACC) regions and HS were found to differ between women and men. In concert with evidence that OFC damage dramatically alters the capacity to cope with stressful events and reduce the use of HS53 and sex differences in OFC activation during the regulation of negative emotion31, this study highlights the importance of the prefrontal-limbic system in stress and emotion regulation. One interesting future topic is how gender differences in HS are associated with the regions of the prefrontal-limbic system when under stress.
References
Gourash, N. Help-seeking: A review of the literature. Am. J. Community. Psychol. 6, 413–423 (1978).
Skinner, E., Edge, K., Altman, J. & Sherwood, H. Searching for the structure of coping: A review and critique of category systems for classifying ways of coping. Psychol. Bull. 129, 216–269 (2003).
Lazarus, R. S. & Folkman, S. Stress, appraisal and coping (Springer, New York, 1984).
Chen, L. et al. Mental health, duration of unemployment and coping strategy: a cross-sectional study of unemployed migrant workers in eastern china during the economic crisis. BMC. Public. Health. 12, 597 (2012).
Li, Y. & Zhang, J. The relationship between personality traits, subjective stress and coping styles in adolescence. Acta. Psychol. Sinica 36, 71–77 (2004).
Zhang, X., Wang, H., Xia, Y., Liu, X. & Jung, E. Stress, coping and suicide ideation in Chinese college students. J. Adolesc. 35, 683–690 (2012).
Compas, B. E. Psychobiological Processes of Stress and Coping. Ann. N. Y. Acad. Sci. 1094, 226–234 (2006).
McEwen, B. S. & Gianaros, P. J. Central role of the brain in stress and adaptation: Links to socioeconomic status, health and disease. Ann. N. Y. Acad. Sci. 1186, 190–222 (2010).
Pruessner, J. C. et al. Deactivation of the Limbic System During Acute Psychosocial Stress: Evidence from Positron Emission Tomography and Functional Magnetic Resonance Imaging Studies. Biol. Psychiatry. 63, 234–240 (2008).
Wang, J. et al. Perfusion functional MRI reveals cerebral blood flow pattern under psychological stress. Proc. Natl. Acad. Sci. U. S. A. 102, 17804–17809 (2005).
Ansell, E. B., Rando, K., Tuit, K., Guarnaccia, J. & Sinha, R. Cumulative Adversity and Smaller Gray Matter Volume in Medial Prefrontal, Anterior Cingulate and Insula Regions. Biol. Psychiatry. 72, 57–64 (2012).
Kim, J. J. & Diamond, D. M. The stressed hippocampus, synaptic plasticity and lost memories. Nat. Rev. Neurosci. 3, 453–462 (2002).
Reynaud, E. et al. Relationship between emotional experience and resilience: An fMRI study in fire-fighters. Neuropsychologia. 51, 845–849 (2013).
Taliaz, D. et al. Resilience to chronic stress is mediated by hippocampal brain-derived neurotrophic factor. J. Neurosci. 31, 4475–4483 (2011).
Frodl, T., Reinhold, E., Koutsouleris, N., Reiser, M. & Meisenzahl, E. M. Interaction of childhood stress with hippocampus and prefrontal cortex volume reduction in major depression. J. Psychiatr. Res. 44, 799–807 (2010).
Gilbertson, M. W. et al. Smaller hippocampal volume predicts pathologic vulnerability to psychological trauma. Nat. Neurosci. 5, 1242–1247 (2002).
Karl, A. et al. A meta-analysis of structural brain abnormalities in PTSD. Neurosci. Biobehav. Rev. 30, 1004–1031 (2006).
Gianaros, P. J. et al. Prospective reports of chronic life stress predict decreased grey matter volume in the hippocampus. Neuroimage. 35, 795–803 (2007).
Al-Bahrani, M., Aldhafri, S., Alkharusi, H., Kazem, A. & Alzubiadi, A. Age and gender differences in coping style across various problems: Omani adolescents' perspective. J. Adolesc. 36, 303–309 (2013).
Matud, M. P. Gender differences in stress and coping styles. Pers. Indiv. Differ. 37, 1401–1415 (2004).
Wang, J. et al. Gender difference in neural response to psychological stress. Soc. Cogn. Affect. Neurosci. 2, 227–239 (2007).
Tamres, L. K., Janicki, D. & Helgeson, V. S. Sex Differences in Coping Behavior: A Meta-Analytic Review and an Examination of Relative Coping. Pers. Soc. Psychol. Rev. 6, 2–30 (2002).
Greenspan, J. D. et al. Studying sex and gender differences in pain and analgesia: a consensus report. Pain 132, S26–S45 (2007).
Keogh, E. & Eccleston, C. Sex differences in adolescent chronic pain and pain-related coping. Pain 123, 275–84 (2006).
Gross, J. J. & John, O. P. Individual differences in two emotion regulation processes: Implications for affect, relationships and well-being. J. Pers. Soc. Psychol. 85, 348–362 (2003).
Depue, R. A. & Morrone-Strupinsky, J. V. A neurobehavioral model of affiliative bonding: Implications for conceptualizing a human trait of affiliation. Behav. Brain Sci. 28, 313–349 (2005).
Kosfeld, M., Heinrichs, M., Zak, P. J., Fischbacher, U. & Fehr, E. Oxytocin increases trust in humans. Nature. 435, 673–676 (2005).
Taylor, S. E. et al. Biobehavioral responses to stress in females: Tend-and-befriend, not fight-or-flight. Psychol. Rev. 107, 411–429 (2000).
Taylor, S. E. [Affiliation and Stress]. The Oxford Handbook of Stress, Health and Coping [Folkman, S. (ed.)] [86–100] (Oxford University Press, New York, 2011).
Domes, G. et al. The neural correlates of sex differences in emotional reactivity and emotion regulation. Hum. Brain. Mapp. 31, 758–769 (2010).
Mak, A. K. Y. et al. Sex-related differences in neural activity during emotion regulation. Neuropsychologia. 47, 2900–2908 (2009).
Xiao, J. & Xu, X. Validity andreliabilityof the Ways of Coping(in Chinese). J. Chinese. Mental. Health. 10, 164–168 (1996).
Sun, C., Wu, Z., Wu, Z. & Xu, S. Age Differences in RAVEN Test and the Relation Between the Differences and Memory Training of “Method of Loci”. Acta Psychol. Sinica. 26, 59–63 (1994).
Liu, X. C., Liu, Q. L., Yang, J. & Zhao, G. F. Reliability and validity of the adolescents self-rating life events checklist. Chinese. J. Clin. Psychol. 5, 34–36 (1997).
Spielberger, C. D., Gorsuch, R. L., Lushene, R., Vagg, P. R. & Jacobs, G. A. Manual for the State-Trait Anxiety Inventory. (Consulting Psychologists Press, California, 1983).
Beck, A. T., Steer, R. A. & Brown, G. Manual for the Beck Depression Inventory-II. (Psychological Corporation, Texas, 1996).
Jung, R. E. & Haier, R. J. The Parieto-Frontal Integration Theory of intelligence: Converging neuroimaging evidence. Behav. Brain Sci. 30, 135–154 (2007).
Tang, C. et al. Effects of lead pollution in SY River on children's intelligence. Life Sci. J. 9, 458–464 (2012).
Li, H. et al. (2014). Examining brain structures associated with perceived stress in a large sample of young adults via voxel-based morphometry. Neuroimage 92, 1–7.
Liu, X. C. & Tein, J. Y. Life events, psychopathology and suicidal behavior in Chinese adolescents. J. Affect. Disord. 86, 195–203 (2005).
Ashburner, J. A fast diffeomorphic image registration algorithm. NeuroImage 38, 95–113 (2007).
Blankstein, U. et al. The complex minds of teenagers: Neuroanatomy of personality differs between sexes. Neuropsychologia. 47, 599–603 (2009).
Takeuchi, H. et al. Regional gray matter density is associated with achievement motivation: evidence from voxel-based morphometry. Brain Struct. Funct. 219, 1–13 (2012).
Song, X. W. et al. REST: a toolkit for resting-state functional magnetic resonance imaging data processing. PloS. One. 6, e25031 (2011).
Mühlau, M. et al. Structural brain changes in tinnitus. Cereb. Cortex. 16, 1283–1288 (2006).
Hayasaka, S., Phan, K. L., Liberzon, I., Worsley, K. J. & Nichols, T. E. Nonstationary cluster-size inference with random field and permutation methods. NeuroImage. 22, 676–687 (2004).
Addis, M. & Mahalik, J. Men, masculinity and the contexts of help seeking. Am. Psychol. 58, 5–14 (2003).
Banissy, M. J., Kanai, R., Walsh, V. & Rees, G. Inter-individual differences in empathy are reflected in human brain structure. Neuroimage. 62, 2034–2039 (2012).
Takeuchi, H. et al. Brain structures associated with executive functions during everyday events in a non-clinical sample. Brain. Struct. Funct. 218, 1017–1032 (2013).
Blakemore, S. J. The social brain in adolescence. Nat. Rev. Neurosci. 9, 267–277 (2008).
McEwen, B. S. Physiology and Neurobiology of Stress and Adaptation: Central Role of the Brain. Physiol. Rev. 87, 873–904 (2007).
Northoff, G., Bermpohl, F., Schoeneich, F. & Boeker, H. How Does Our Brain Constitute Defense Mechanisms? First-Person Neuroscience and Psychoanalysis. Psychother. Psychosom. 76, 141–153 (2007).
Lagopoulos, J., Hermens, D. F., Naismith, S. L., Scott, E. M. & Hickie, I. B. Frontal lobe changes occur early in the course of affective disorders in young people. BMC. Psychiatry. 12, 4 (2012).
Bush, G., Luu, P. & Posner, M. I. Cognitive and emotional influences in anterior cingulate cortex. TICS. 4, 215–222 (2000).
Sekiguchi, A. et al. Brain structural changes as vulnerability factors and acquired signs of post-earthquake stress. Mol. Psychiatry. 18, 618–623 (2012).
Koch, K. et al. Gender differences in the cognitive control of emotion: An fMRI study. Neuropsychologia. 45, 2744–2754 (2007).
Campbell, S., Marriott, M., Nahmias, C. & MacQueen, G. M. Lower hippocampal volume in patients suffering from depression: a meta-analysis. Am. J. Psychiatry. 161, 598–607 (2004).
Videbech, P. & Ravnkilde, B. Hippocampal volume and depression: a meta-analysis of MRI studies. Am. J. Psychiatry. 161, 1957–1966 (2004).
Lupien, S. J., McEwen, B. S., Gunnar, M. R. & Heim, C. Effects of stress throughout the lifespan on the brain, behaviour and cognition. Nat. Rev. Neurosci. 10, 434–445 (2009).
Kennedy, K. M. et al. Age-related differences in regional brain volumes: A comparison of optimized voxel-based morphometry to manual volumetry. Neurobiol. Aging. 30, 1657–1676 (2009).
Acknowledgements
We thank Xin Hao, Lei Zhang, Dan-Dan Tong and Jun-Yi Yang for the assistance in data collection. This work was supported by the National Natural Science Foundation of China (31170983; 31271087), China and Chongqing Postdoctoral Science Foundation funded project (2012M510098; XM2012006), the Key Discipline Fund of National 211 Project (NSKD11007), the Program for New Century Excellent Talents in University (2011) by the Ministry of Education and Program for Talents in colleges and universities in Chongqing (2011).
Author information
Authors and Affiliations
Contributions
H.J.L., J.Z.S., Q.L.Z. and J.Q. planned, collected, analyzed, interpreted the data and wrote the paper. D.T.W. and W.F.L. helped analyze and write the paper. T.J. G.H. reviewed and revised the manuscript.
Ethics declarations
Competing interests
The authors declare no competing financial interests.
Rights and permissions
This work is licensed under a Creative Commons Attribution-NonCommercial-ShareAlike 4.0 International License. The images or other third party material in this article are included in the article's Creative Commons license, unless indicated otherwise in the credit line; if the material is not included under the Creative Commons license, users will need to obtain permission from the license holder in order to reproduce the material. To view a copy of this license, visit http://creativecommons.org/licenses/by-nc-sa/4.0/
About this article
Cite this article
Li, HJ., Sun, JZ., Zhang, QL. et al. Neuroanatomical Differences between Men and Women in Help-Seeking Coping Strategy. Sci Rep 4, 5700 (2014). https://doi.org/10.1038/srep05700
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/srep05700
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.