Abstract
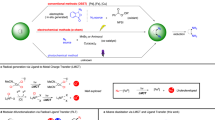
Iron is inexpensive, non-toxic and the most abundant transition metal in the Earth’s crust, rendering iron-catalysed C–H activations attractive yet particularly challenging. Despite major advances, iron-catalysed C–H activations have been linked to high reaction temperatures or the use of reactive Grignard reagents. Here we present iron-catalysed ketimine C–H activations at ambient reaction temperature with the help of blue light in the absence of additives, utilizing easily accessible cis-[Fe(H)2(dppe)2] (where dppe is 1,2-bis(diphenylphosphino)ethane) as a single component precatalyst. Mild reaction conditions, high atom economy and the lack of Grignard reagents are distinguishing features of the iron-catalysed C–H alkenylation manifold. Detailed mechanistic investigations by deuterium labelling, isolation of organometallic intermediates and in operando light-emitting diode nuclear magnetic resonance spectroscopy revealed the role of the light and an oxidative addition to an iron(0) complex as the modus operandi for the C–H activation.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 digital issues and online access to articles
$119.00 per year
only $9.92 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
Data availability
The data supporting the findings of this study are available within the article and its Supplementary Information, or from the authors on reasonable request. Crystal structure data have been deposited at the Cambridge Crystallographic Data Centre (CCDC nos. 2192211–2192227 and 2280102–2280104), and crystallographic data are provided in Supplementary Information. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk. Spectroscopic and kinetic data that support the findings of this study are freely available in Zenodo data repository, with https://doi.org/10.5281/zenodo.10138296.
References
Rogge, T. et al. C–H activation. Nat. Rev. Methods Prim. 1, 1–31 (2021).
Ackermann, L. Carboxylate-assisted transition metal-catalyzed C–H bond functionalizations: mechanism and scope. Chem. Rev. 111, 1315–1345 (2011).
Colby, D. A., Tsai, A. S., Bergman, R. G. & Ellman, J. A. Rhodium-catalyzed chelation-assisted C–H bond functionalization reactions. Acc. Chem. Res. 45, 814–825 (2012).
Arockiam, P. B., Bruneau, C. & Dixneuf, P. H. Ruthenium(II)-catalyzed C–H bond activation and functionalization. Chem. Rev. 112, 5879–5918 (2012).
Davies, H. M. L. & Morton, D. Collective approach to advancing C–H functionalization. ACS Cent. Sci. 3, 936–943 (2017).
Gensch, T., Hopkinson, M. N., Glorius, F. & Wencel-Delord, J. Mild metal-catalyzed C–H activation: examples and concepts. Chem. Soc. Rev. 45, 2900–2936 (2016).
Park, Y., Kim, Y. & Chang, S. Transition metal-catalyzed C–H amination: scope, mechanism, and applications. Chem. Rev. 117, 9247–9301 (2017).
Rej, S., Ano, Y. & Chatani, N. Bidentate directing groups: an efficient tool in C–H bond functionalization chemistry for the expedient construction of C–C bonds. Chem. Rev. 120, 1788–1887 (2020).
Sun, C.-L., Li, B.-J. & Shi, Z.-J. Direct C–H transformation via iron catalysis. Chem. Rev. 111, 1293–1314 (2010).
Satoh, T. & Miura, M. Transition metal-catalyzed regioselective arylation and vinylation of carboxylic acids. Synthesis 2010, 3395–3409 (2010).
Manan, R. S. & Zhao, P. Merging rhodium-catalysed C–H activation and hydroamination in a highly selective [4+2] imine/alkyne annulation. Nat. Commun. 7, 11506 (2016).
Colby, D. A., Bergman, R. G. & Ellman, J. A. Rhodium-catalyzed C–C bond formation via heteroatom-directed C–H bond activation. Chem. Rev. 110, 624–655 (2010).
Gao, K., Lee, P.-S., Fujita, T. & Yoshikai, N. Cobalt-catalyzed hydroarylation of alkynes through chelation-assisted C–H bond activation. J. Am. Chem. Soc. 132, 12249–12251 (2010).
Lee, P.-S., Fujita, T. & Yoshikai, N. Cobalt-catalyzed room-temperature addition of aromatic imines to alkynes via directed C–H bond activation. J. Am. Chem. Soc. 133, 17283–17295 (2011).
Fallon, B. J. et al. C–H activation/functionalization catalyzed by simple, well-defined low-valent cobalt complexes. J. Am. Chem. Soc. 137, 2448–2451 (2015).
Gandeepan, P. et al. 3d transition metals for C–H activation. Chem. Rev. 119, 2192–2452 (2019).
Yoshino, T. & Matsunaga, S. Cobalt-catalyzed C(sp3)–H functionalization reactions. Asian J. Org. Chem. 7, 1193–1205 (2018).
Nakao, Y. Hydroarylation of alkynes catalyzed by nickel. Chem. Rec. 11, 242–251 (2011).
Khake, S. M. & Chatani, N. Nickel-catalyzed C–H functionalization using a non-directed strategy. Chem 6, 1056–1081 (2020).
Liu, Y.-H., Xia, Y.-N. & Shi, B.-F. Ni-catalyzed chelation-assisted direct functionalization of inert C–H bonds. Chin. J. Chem. 38, 635–662 (2020).
Hirano, K. & Miura, M. Recent advances in copper-mediated direct biaryl coupling. Chem. Lett. 44, 868–873 (2015).
Shang, R., Ilies, L. & Nakamura, E. Iron-catalyzed C–H bond activation. Chem. Rev. 117, 9086–9139 (2017).
Cera, G. & Ackermann, L. Iron-catalyzed C–H functionalization processes. Top. Curr. Chem. 374, 57 (2016).
Jia, T., Zhao, C., He, R., Chen, H. & Wang, C. Iron-carbonyl-catalyzed redox-neutral [4+2] annulation of N–H imines and internal alkynes by C–H bond activation. Angew. Chem. Int. Ed. 55, 5268–5271 (2016).
Kimura, N., Kochi, T. & Kakiuchi, F. Iron-catalyzed regioselective anti-Markovnikov addition of C–H bonds in aromatic ketones to alkenes. J. Am. Chem. Soc. 139, 14849–14852 (2017).
Kimura, N., Kochi, T. & Kakiuchi, F. Iron‐catalyzed ortho‐selective C−H alkylation of aromatic ketones with N‐alkenylindoles and partial indolylation via 1,4‐iron migration. Asian J. Org. Chem. 8, 1115–1117 (2019).
Messinis, A. M., Finger, L. H., Hu, L. & Ackermann, L. Allenes for versatile iron-catalyzed C–H activation by weak O-coordination: mechanistic insights by kinetics, intermediate isolation, and computation. J. Am. Chem. Soc. 142, 13102–13111 (2020).
Messinis, A. M., Oliveira, J. C. A., Stückl, A. C. & Ackermann, L. Cyclometallated iron(II) alkoxides in iron-catalyzed C–H activations by weak O-carbonyl chelation. ACS Catal. 12, 4947–4960 (2022).
Kimura, N., Katta, S., Kitazawa, Y., Kochi, T. & Kakiuchi, F. Iron-catalyzed ortho C–H homoallylation of aromatic ketones with methylenecyclopropanes. J. Am. Chem. Soc. 143, 4543–4549 (2021).
Twilton, J. et al. The merger of transition metal and photocatalysis. Nat. Rev. Chem. 1, 0052 (2017).
Wang, C.-S., Dixneuf, P. H. & Soulé, J.-F. Photoredox catalysis for building C–C bonds from C(sp2)–H bonds. Chem. Rev. 118, 7532–7585 (2018).
Guillemard, L. & Wencel-Delord, J. When metal-catalyzed C–H functionalization meets visible-light photocatalysis. Beilstein J. Org. Chem. 16, 1754–1804 (2020).
Mendelsohn, L. N. et al. Visible-light-enhanced cobalt-catalyzed hydrogenation: switchable catalysis enabled by divergence between thermal and photochemical pathways. ACS Catal. 11, 1351–1360 (2021).
Cuthbertson, J. D. & Macmillan, D. W. C. The direct arylation of allylic sp3 C–H bonds via organic and photoredox catalysis. Nature 519, 74–77 (2015).
Kalyani, D., Mcmurtrey, K. B., Neufeldt, S. R. & Sanford, M. S. Room temperature C–H arylation: merger of Pd-catalyzed C–H functionalization and visible-light photocatalysis. J. Am. Chem. Soc. 133, 18566–18569 (2011).
Shaw, M. H., Shurtleff, V. W., Terrett, J. A., Cuthbertson, J. D. & MacMillan, D. W. C. Native functionality in triple catalytic cross-coupling: sp3 C–H bonds as latent nucleophiles. Science 352, 1304–1308 (2016).
Wegeberg, C. & Wenger, O. S. Luminescent first-row transition metal complexes. JACS Au 1, 1860–1876 (2021).
Stephenson, C. R. J., Yoon, T. & MacMillan, D. W. C. Visible Light Photocatalysis in Organic Chemistry (Wiley, 2018).
Dombray, T. et al. Iron-catalyzed C–H borylation of arenes. J. Am. Chem. Soc. 137, 4062–4065 (2015).
Zhou, W. J. et al. Light runs across iron catalysts in organic transformations. Chem. Eur. J. 26, 15052–15064 (2020).
Bautista, M. T., Bynum, L. D. & Schauer, C. K. Synthesis of η2-dihydrogen complex, trans-{Fe(η2-H2)(H)[1,2-bis(diphenylphosphino)-ethane]2}[BF4]: an experiment for an advanced inorganic chemistry laboratory involving synthesis and NMR properties of an η2-H2 complex. J. Chem. Educ. 73, 988 (1996).
Azizian, H. & Morris, R. H. Photochemical synthesis and reactions of FeH(C6H4PPhCH2CH2PPh2)(PPh2PCH2CH2PPh2). Inorg. Chem. 22, 6–9 (1983).
Lehnherr, D. et al. Discovery of a photoinduced dark catalytic cycle using in situ LED-NMR spectroscopy. J. Am. Chem. Soc. 140, 13843–13853 (2018).
Hoye, T. R., Eklov, B. M., Ryba, T. D., Voloshin, M. & Yao, L. J. No-D NMR (no-deuterium proton NMR) spectroscopy: a simple yet powerful method for analyzing reaction and reagent solutions. Org. Lett. 6, 953–956 (2004).
Suslick, B. A. & Tilley, T. D. Mechanistic interrogation of alkyne hydroarylations catalyzed by highly reduced single-component cobalt complexes. J. Am. Chem. Soc. 142, 11203–11218 (2020).
Frohnapfel, D. S. & Templeton, J. L. Transition metal η2-vinyl complexes. Coord. Chem. Rev. 206–207, 199–235 (2000).
Goumans, T. P. M. et al. Photodissociation of the phosphine-substituted transition metal carbonyl complexes Cr(CO)5L and Fe(CO)4L: a theoretical study. J. Am. Chem. Soc. 125, 3558–3567 (2003).
Salassa, L., Garino, C., Salassa, G., Gobetto, R. & Nervi, C. Mechanism of ligand photodissociation in photoactivable [Ru(bpy)2L2]2+ complexes: a density functional theory study. J. Am. Chem. Soc. 130, 9590–9597 (2008).
Casitas, A., Krause, H., Goddard, R. & Fürstner, A. Elementary steps of iron catalysis: exploring the links between iron-alkyl and iron-olefin complexes for their relevance in C–H activation and C–C bond formation. Angew. Chem. Int. Ed. 54, 1521–1526 (2015).
Yu, C., Zhang, W.-X. & Xi, Z. Cyclobutadiene sandwich complexes of nickel and iron from cyclization of 1,3-butadiene dianions: synthesis and structural characterization. Organometallics 37, 4100–4104 (2018).
Aranyos, A. et al. Novel electron-rich bulky phosphine ligands facilitate the palladium-catalyzed preparation of diaryl ethers. J. Am. Chem. Soc. 121, 4369–4378 (1999).
Acknowledgements
Generous support by the DFG (SPP 1807 Gottfried-Wilhelm-Leibniz award to L.A.), the European Union’s Horizon 2020 research and innovation programme (Marie Skłodowska-Curie grant agreement no. 895404 to A.M.M. and ERC advanced grant agreement no. 101021358 to L.A.) and FCI Kekulé Fellowship no. 110091 (T.v.M.) is gratefully acknowledged. We thank C. Golz (Göttingen University) for assistance with the X-ray diffraction analysis and I. Maksso for measuring inductively coupled plasma mass spectrometry solutions.
Author information
Authors and Affiliations
Contributions
A.M.M. unravelled the photo-promoted iron-catalysed C–H activation of imines, conducted the mechanistic studies, explored the substrate scope assisted by T.v.M. and M.S., and wrote the paper with revisions provided by the other authors. M.S. performed the Grignard studies. L.A. conceived and directed the research programme and revised the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Catalysis thanks Brian Patrick and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Methods, Figs. 1–52, Tables 1–6 and References.
Supplementary Data 1
CIF file of the crystal structure of compound 4a.
Supplementary Data 2
CIF file of the crystal structure of compound 4b.
Supplementary Data 3
CIF file of the crystal structure of compound 4c.
Supplementary Data 4
CIF file of the crystal structure of compound 4d.
Supplementary Data 5
CIF file of the crystal structure of compound 4e.
Supplementary Data 6
CIF file of the crystal structure of compound 4f.
Supplementary Data 7
CIF file of the crystal structure of compound 4g.
Supplementary Data 8
CIF file of the crystal structure of compound 5a.
Supplementary Data 9
CIF file of the crystal structure of compound 5b.
Supplementary Data 10
CIF file of the crystal structure of compound 5c.
Supplementary Data 11
CIF file of the crystal structure of compound 6a.
Supplementary Data 12
CIF file of the crystal structure of compound 6b.
Supplementary Data 13
CIF file of the crystal structure of compound 6c.
Supplementary Data 14
CIF file of the crystal structure of compound 8.
Supplementary Data 15
CIF file of the crystal structure of compound 10a.
Supplementary Data 16
CIF file of the crystal structure of compound 10b.
Supplementary Data 17
CIF file of the crystal structure of compound E-3aa.
Supplementary Data 18
CIF file of the crystal structure of compound E-3aa″.
Supplementary Data 19
CIF file of the crystal structure of compound E-3aj.
Supplementary Data 20
CIF file of the crystal structure of compound Z-3ea.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Messinis, A.M., von Münchow, T., Surke, M. et al. Room temperature photo-promoted iron-catalysed arene C–H alkenylation without Grignard reagents. Nat Catal 7, 273–284 (2024). https://doi.org/10.1038/s41929-023-01105-0
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41929-023-01105-0