Abstract
Chronic inflammation is the cause of chronic kidney disease (CKD). The nucleotide-binding domain-like receptor protein 3 (NLRP3) inflammasome plays a vital role in the inflammation process and is associated with the regulatory effects of NLRP3 gene polymorphisms. This study evaluated the association between NLRP3 gene polymorphisms and CKD, and further explored whether the association of environmental metals with CKD varied by the NLRP3 genotypes. A total of 218 CKD patients and 427 age- and sex-matched healthy controls were recruited in this clinic-based case–control study. Patients were identified as having CKD if their estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2 and stage 3–5 for at least 3 months. We examined the genotypes of fifteen common ssingle-nucleotide polymorphisms in NLRP3 genes. Concentrations of total urinary arsenic were examined by summing of urinary inorganic arsenic species. Concentrations of selenium, cadmium, and lead were measured from blood samples. Associations between NLRP3 polymorphisms, environmental metals exposure, and CKD were evaluated using multivariable logistic regression while controlling for confounders. We observed that the odds of carrying NLRP3 rs4925650 GA/AA genotypes, NLRP3 rs1539019 CA/AA genotypes, and NLRP3 rs10157379 CT/TT genotypes were significantly higher among CKD cases compared to controls, with the adjusted odds ratio (95% confidence interval) were 1.54 (1.01–2.36), 1.56 (1.04–2.33), and 1.59 (1.05–2.38), respectively. The significant multiplicative interactions were identified between high levels of blood lead and NLRP3 rs4925650 GA/AA genotypes; high levels of blood cadmium or low levels of plasma selenium and the NLRP3 haplotype (rs4925648, rs4925650, rs12048215, and rs10754555) C-A-A-C multiplicatively interacted to increase the risk of CKD. Our results imply that NLRP3 polymorphisms may play an important role in the development of environmental metals exposure related CKD.
Similar content being viewed by others
Introduction
Chronic kidney disease (CKD) affects 8–16% of the world’s population1, resulting in CKD being a common public health problem worldwide2. Using estimated glomerular filtration rate (eGFR) < 60 mL/min/1.73 m2 to define CKD, the prevalence of CKD in Taiwan was 11.9%, and only 3.5% of patients aware their disease3. As the incidence of end-stage renal disease in Taiwan (426/106 in 2012) was the highest globally4, CKD is an extremely important health issue in Taiwan.
Our previous study has reported a significantly increased odds of high total urinary arsenic levels and low plasma lycopene levels among the CKD cases compared to the health controls5. We also found an increased odds of high blood cadmium and lead levels among CKD cases compared to the controls, whereas a decreased odds of high plasma selenium levels were observed among CKD cases compared to the controls6. Recently, a study has shown that plasma concentrations of arsenic and lead were significantly related to the decline of renal function7. In addition, a study reported that increasing concentrations of blood cadmium and lead were associated with an increased risk of proteinuria and related to decreased eGFR8. These studies suggested that low levels of selenium and high levels of arsenic, cadmium, and lead may increase the risk of CKD. However, the mechanism underlying the effect of these metals on CKD remains unclear.
Inflammation is the cause of several kinds of kidney diseases, such as acute kidney injury and CKD9. The nucleotide-binding domain-like receptors 3 (NLRP3) inflammasome is a multi-protein complex that plays an important role in the inflammation process10. A previous study has indicated that NLRP3-induced inflammation may promote kidney inflammation and causes CKD11. Also, a study has reported that the NLRP3 inflammasome may be involved in the pathogenesis of acute kidney injury, CKD, diabetic nephropathy, and crystal-related nephropathy12. Recent evidence has been built to implicate arsenic, cadmium, lead, and selenium on NLRP3 related inflammatory. A study has reported that arsenic activates the NLRP3 inflammasome and induces inflammatory cell death13. However, another study showed that arsenic inhibits the secretion of interleukin (IL)-1β and IL-18, which was caused by activating the NLRP3 inflammasomes in macrophages; thus, indicating that exposure to arsenic may affect inflammasome-mediated inflammation14. These inconsistent findings reveal an unclear relationship between arsenic exposure and NLRP3 inflammasomes. In addition to arsenic exposure, a carp experiment showed that cadmium exposure induced apoptosis of the anterior spleen and splenic lymphocytes by activating NLRP315. Moreover, lead may activate the NLRP3 signaling pathway to cause oxidative stress and inflammation in chicken testicles, thereby reducing testicular function16. In contrast, selenium has a mitigating effect by modifying activation of the NLRP3 signaling pathway in chicken testes caused by lead16.
Variations in the NLRP3 gene may affect mRNA stability and NLRP3 performance17. The NLRP3 gene has nine exons within its 32.9 kb sequence17 and is located on chromosome 1q4418. There are 60 common single nucleotide polymorphisms (SNPs) that have been identified in the NLRP3 gene19. Several studies have suggested that NLRP3 polymorphisms were associated with the risk of cardiovascular diseases. A study has found that the 50-year-old subjects with NLRP3 rs7512998 CC or CT genotypes had a higher blood pressure levels compared to those with TT genotype20. In Chinese Han population, NLRP3 rs10754556 CC genotype was associated with the occurrence of coronary artery disease compared to those with CG or GG genotypes21. The NLRP3 rs4612666 T allele is associated with an increased risk of aorta sclerosis-like ischemic stroke22. To date, few studies have explored the association between NLRP3 gene polymorphisms and CKD. The present study aimed to examine the association between NLRP3 genotypes and CKD, and to further explore whether NLRP3 gene polymorphisms may modify the associations of total urinary arsenic, blood cadmium and lead, and plasma selenium with CKD.
Materials and methods
Study subjects
This study was a clinic-based case–control study. In total, 218 clinically confirmed CKD patients and 427 age- and sex-matched controls who volunteered to participate in this study were recruited previously23. All participants measured serum creatinine concentrations by isotope dilution mass spectrometry (IDMS). The eGFR (mL/min/1.73 m2) was calculated using the equation: 186.3 × (serum creatinine)−1.154 × (age)−0.203 × (0.742 for females)24. CKD patients were clinically diagnosed at the Department of Internal Medicine/Nephrology, Taipei Medical University Hospital and Taipei Municipal Wan Fang Hospital, with an eGFR < 60 mL/min/1.73 m2 for at least 3 months (stages 3–5) and without hemodialysis according to KDIGO 2012 Clinical Practice Guideline for the Evaluation and Management of Chronic Kidney Disease25. Matched controls, with no CKD diagnosis, were recruited from receiving adult and senior citizen health examinations in the Department of Family Medicine. The Research Ethics Committee of Taipei Medical University approved this study (TMU-Joint Institutional Review Board, N201812007) which was conducted in accordance with the Declaration of Helsinki. All participants provided informed consent before the questionnaire interview and specimens collection.
Interview and specimen collection
The questionnaire interview and the urine and blood samples collection were described previously5,23. We measured the arsenic species concentrations in single spot urine samples. EDTA-vacuum syringes were used to collect 5–8 mL peripheral blood samples, and the plasma, red blood cell, and buffy coat were separated. Buffy coat was separated for DNA extraction and to determine NLRP3 polymorphisms. We analyzed the cadmium and lead concentrations from red blood cells, and measured selenium concentrations from plasma.
Environmental metals exposure measurement
Urinary concentrations of inorganic-related arsenic species, including trivalent arsenite (AsIII), pentavalent arsenate (AsV), monomethylarsonic acid (MMAV), and dimethylarsinic acid (DMAV) were measured as described previously26. We assessed total urinary arsenic concentrations by summing the urinary concentrations of AsIII, AsV, MMAV, and DMAV. The measure of arsenic in urine specimens was a direct method of excluding nontoxic organic arsenic that contributed to total arsenic exposure27. Urinary concentrations were adjusted for urinary creatinine concentrations due to variations in the hydration state28. In addition, concentrations of blood cadmium and lead and plasma selenium were determined as described previously6. The validity and reliability of the environmental metals exposure measurement are shown in Supplementary Table S1.
Determination of the genetic polymorphisms
DNA was extracted by digestion with proteinase K followed by phenol and chloroform. We selected 15 common NLRP3 SNPs based on their minor-allele frequencies (≥ 0.2) in the Han Chinese in the Beijing HapMap database. The Agena Bioscience MassARRAY iPLEX system was used according to the manufacturer’s instructions (National Genome Medicine Center, Taipei, Taiwan) to determine the genotypes of 15 SNPs, including rs4925654, rs4925650, rs12239046, rs4925648, rs10925025, rs10925019, rs1539019, rs3806265, rs10925026, rs10157379, rs12143966, rs10754555, rs3806268, rs12048215, and rs12137901. Among them, rs12137901 did not meet the Hardy–Weinberg equilibrium and was excluded from the statistical analysis. Linkage disequilibrium (LD) strength was determined by calculating D′ and r2 of the Lewontin using Haploview 4.1 software29.
Statistical analysis
Differences in the continuous variables were compared between the two groups using the Wilcoxon rank-sum test. The Kruskal–Wallis test was used to compare continuous variables of more than two groups. Multiple logistic regression models were performed estimating odds ratios (ORs) and 95% confidence intervals (CIs) to evaluate the associations between NLRP3 polymorphism, environmental metal exposure, and CKD while adjusting for confounders. The measures of metal concentrations were categorized into three groups based on the tertile distribution of concentrations among controls. We further treated each tertile group as an ordinal variable in the models to conduct the trend test. Additionally, multiple linear regression models were used to assess associations between NLRP3 polymorphisms and eGFR while adjusting for confounders. Confounding was informed by prior knowledge and met the criterion that changed the ORs of exposure variables at least by 10% when adding to models assessing between environmental metal exposure and CKD5. We explored the interaction by estimating the combined effects of environmental metals exposure (median of concentrations among controls as a cutoff point) and NLRP3 genotypes on CKD. The product term was added to the logistic regression models to conduct for the multiplicative interaction between metal and NLRP3 genotypes. The SAS 9.4 (SAS Institute, Cary, NC, USA) was used for statistical analyses. A two-sided p value < 0.05 was considered statistically significant.
Results
The sociodemographic characteristics, eGFR, lifestyle, and disease histories of diabetes and hypertension between CKD cases and controls are shown in Table 1. The mean and standard deviation of age and eGFR in the 218 CKD patients and 427 controls were 65.11 ± 13.52 and 64.21 ± 12.49 years, and 31.54 ± 14.57 and 84.21 ± 15.62 mL/min/1.73 m2 respectively. The CKD cases had a lower level of education. Most cases were less likely to have habits of drinking coffee, tea, and alcohol than controls. The CKD patients had significantly increased odds of regularly used analgesics compared to controls, with OR (95% CI) = 2.94 (1.58–5.44). The odds of having disease histories of diabetes and hypertension were 3–5-fold increase among cases compared to controls.
Table 2 presents the association between 14 NLRP3 gene polymorphisms and CKD. We observed that the odds of carrying NLRP3 rs4925650 GA/AA genotypes were 1.54-fold (95% CI 1.15–3.34) increased among CKD cases compared to controls after adjusting for covariates. In addition, participants with CKD had a 1.56–1.79 fold increased odds of carrying NLRP3 rs12239046 CT/TT, NLRP3 rs10925025 AG/GG, NLRP3 rs1539019 CA/AA, NLRP3 rs10925026 CA/AA, and NLRP3 rs10157379 CT/TT genotypes compared to controls. After additionally adjusting environmental metals exposure, we observed that NLRP3 rs4925650 [GA/AA vs. GG, OR (95% CI) = 1.89 (0.98–3.35)], NLRP3 rs1539019 [CA/AA vs. CC, OR (95% CI) = 1.52 (0.95–2.44)], and NLRP3 rs10157379 [CT/TT vs. CC, OR (95% CI) = 1.50 (0.94–2.41)] were associated with CKD though the confidence interval marginally included the null value. No association was found between other NLRP3 genotypes and CKD after adjusting for covariates and metals concentrations. NLRP3 rs4925650, NLRP3 rs1539019, and NLRP3 rs10157379 genotypes were selected for further analyzed the effect modification of gene and metal on CKD.
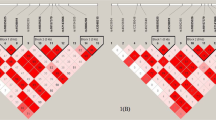
LD and haplotype analyses revealed that the NLRP3 genes exhibited the four haplotype blocks shown in Supplementary Figure S1. D’ of Lewontin of the haplotype NLRP3 block 1 (NLRP3 rs4925648, NLRP3 rs4925650, NLRP3 rs12048215, and NLRP3 rs10754555), NLRP3 block 2 (NLRP3 rs3806265 and NLRP3 rs38062628), NLRP3 block 3 (NLRP3 rs10925019 and NLRP3 rs4925654), and NLRP3 block 4 (NLRP3 rs1539019, NLRP3 rs10925025, NLRP3 rs12143966, NLRP3 rs12239046, NLRP3 rs10925026, and NLRP3 rs10157379) ranged from 0.99 to 1.00. The association between the NLRP3 gene haplotypes and CKD is shown in Table 3. We observed that the odds of C-G-A-C and C-G-A-G haplotypes in the NLRP3 block 1 respectively decreased by 47% and 52% among the CKD cases compared to the controls. In addition, the combined T-G-G-G, C-G-A-C, C-G-A-G, and C-G-G-G haplotypes in the NLRP3 block 1 were significantly inversely associated with CKD compared to the C-A-A-C haplotype. These associations remained statistically significant after further adjusting for metals concentrations in the models. The odds of C-A haplotype in NLRP3 block 3 were decreased by 35% among the CKD cases compared to the controls. No association was observed between NLRP3 blocks 2 and 4 and CKD.
Associations between NLRP3 rs4925650, NLRP3 rs1539019, and NLRP3 rs10157379 genotypes and eGFR are shown in Table 4. Participants who carried the NLRP3 rs4925650 AA genotype decreased 7.62 mL/min/1.73 m2 of eGFR when compared to those carrying the GG genotype. Participants who carried the NLRP3 rs1539019 A allele and the NLRP3 rs10157379 T allele decreased eGFR by 6 mL/min/1.73 m2 when compared with those carrying the NLRP3 rs1539019 C allele and the NLRP3 rs10157379 C allele. These results showed no changes after adjusting for blood cadmium and lead or plasma selenium levels in models.
Environmental metals exposure was found to be associated with CKD in our study (Supplementary Table S2). As the levels of total urinary arsenic and blood cadmium and lead increased, the OR of CKD increased significantly in a dose–response manner. In contrast, as the levels of plasma selenium increased, the OR of CKD decreased significantly in a dose–response relationship. No difference was observed in comparing concentrations of environmental metals exposure by different genotypes of NLRP3 rs4925650, NLRP3 rs1539019, and NLRP3 rs10157379.
Figure 1 shows the combined effect of NLRP3 rs4925650, NLRP3 rs1539019, NLRP3 rs10157379, and levels of environmental metals exposure on the CKD. The trend analysis showed that the OR of CKD increased gradually with exposure to no risk factors, one risk factor, or two risk factors (risk genotypes, high levels of arsenic, cadmium, and lead, or low levels of selenium). We observed that the odds of carrying NLRP3 rs4925650 GA/AA genotypes and high levels of blood lead (> 37.40 μg/L) were 5.03-fold increased (95% CI 2.46–10.27) among CKD cases compared to controls (Fig. 1C). The p-value of the interaction term of NLRP3 rs4925650 and levels of blood lead was 0.0229, which indicated a multiplicative interaction between NLRP3 rs4925650 and blood lead on CKD. In addition, we observed that NLRP3 block 1 (risk haplotype: C-A-A-C) interacted with total urinary arsenic, blood lead and cadmium, and plasma selenium to significantly enhance the OR of CKD, respectively (Fig. 2). High levels of blood cadmium and the NLRP3 block 1 C-A-A-C haplotype, and low levels of plasma selenium and the NLRP3 block 1 C-A-A-C haplotype significantly and multiplicatively interacted to increase the OR of CKD, respectively.
The combined effect of NLRP3 rs4925650, NLRP3rs 1,539,019, NLRP3 rs10157379, and levels of environmental metals exposure on the CKD. (a) Total urinary arsenic; (b) Blood cadmium; (c) Blood lead; (d) Plasma selenium. The estimates of OR were adjusted for age, sex, educational level, alcohol, coffee and tea consumption, analgesic usage, disease histories of diabetes and hypertension, and levels of other metals.
The combined effect of the NLRP3 block 1 haplotypes (rs4925648, rs4925650, rs12048215, and rs10754555) and levels of total urinary arsenic, blood lead and cadmium, and plasma selenium on CKD. The estimates of OR were adjusted for age, sex, educational level, alcohol, coffee and tea consumption, analgesic usage, disease histories of diabetes and hypertension, and levels of other metals.
Discussion
To the best of our knowledge, this study is the first to evaluate the associations between NLRP3 polymorphisms, environmental metals exposure, and CKD. We found that the odds of carrying NLRP3 rs4925650 GA/AA genotypes, NLRP3 rs1539019 CA/AA genotypes, and NLRP3 rs10157379 CT/TT genotypes were significantly higher among CKD cases compared to controls. In addition, certain NLRP3 genotypes were interacting with environmental metals exposure on the risk of CKD. Specifically, high levels of blood lead and NLRP3 rs4925650 GA/AA genotypes; high levels of blood cadmium and NLRP3 block 1 C-A-A-C haplotype, and low levels of plasma selenium and NLRP3 block 1 C-A-A-C haplotype significantly and multiplicatively interacted to increase the risk of CKD.
Persistent inflammation and activation of the innate immune system is a chronic kidney damage phenomenon, which is important for the development of CKD30. The mechanism of inflammation-induced CKD is still unknown. The pattern recognition receptors, such as nucleotide oligomerization domain-like receptors (NLRs), C-type lectin-like receptors, toll-like receptors, and retinoic acid-inducible gene I-like receptors act as sensors of the innate immune system31. The most characteristic inflammasome is NLRP3, which responds to endogenous damage signals caused by entry of pathogen or tissue damage32. The NLRP3 inflammasome is believed to play a key role in the underlying inflammatory response in many chronic diseases, including CKD33.
Genetic variation in the NLRP3 gene may be an important determinant of the degree of the immune inflammatory response, which affects susceptibility to inflammatory diseases34,35. In the present study, NLRP3 rs1539019 CA/AA genotypes significantly increased with the risk of CKD after adjusting for confounders. Various studies have evaluated the association of NLRP3 rs1539019 polymorphism with several health-related outcomes. Patients with chronic hepatitis C virus and the NLRP3 rs1539019 AA genotype do not respond to interferon therapy36, and the NLRP3 rs1539019 TT genotype is related to pneumoconiosis in Chinese coal workers37. In addition, it has been reported that the NLRP3 rs1539019 A allele is related to circulating fibrinogen concentration and therefore to the risk of cardiovascular disease38. The rs1539019 G>T locus is close to a 12-nucleotide sequence identified as a consensus binding site for epidermal growth factor 1 that may influence the vertebrate blood coagulation network39. NLRP3 rs1539019 A allele has been indicated to increase circulating fibrinogen levels38, an indicator of inflammation, which may be associated with blood coagulation, followed by leading to CKD. However, another study found no significant association between the NLRP3rs1539019 polymorphism and the risk of essential hypertension in a Japanese population40. The NLRP3 rs1539019 polymorphism was not associated with primary gouty arthritis in the Chinese Han population41. NLRP3 rs1539019 is an intronic polymorphism and it is unclear how genetic variations in introns affect gene function. However, one study reported that many transcription factors bind to intron sites, and these intron sites may play a role regulating gene expression42.
In addition to NLRP3 rs1539019 polymorphism, we also found that NLRP3 rs4925650 GA/AA genotypes and NLRP3 rs10157379 CT/TT genotypes significantly increased the risk of CKD after adjusting for confounders. Hence, the effect of NLRP3 rs4925650 GA/AA genotypes and NLRP3 rs10157379 CT/TT genotypes on susceptibility to CKD appears to be independent of age, sex, educational level, consumption of tea, alcohol, and coffee, analgesic usage, and disease histories of diabetes and hypertension. The present study found an association between the NLRP3 rs10157379 CT/TT genotypes and CKD in the Taiwanese population, which is in accordance with our previous research showed that the NLRP3 rs10157379 T allele has a borderline association with renal cell carcinoma43. These results imply that subjects with NLRP3 rs10157379 T allele may be associated with renal damage than those with C allele. In addition, a recent study found that the NLRP3 rs10157379 CT genotype was associated with the severity of severe acute respiratory syndrome (SARS)44. NLRP3 may also be related to host immunity and susceptibility to inflammation disorders45 . Further, NLRP3 can interact with thioredoxin-interacting protein (TXNIP), a protein involved in insulin resistance. TXNIP deficiency may impair the activation of the NLRP3 inflammasome and subsequent secretion of interleukin 1β, which was involved in the pathogenesis of diabetes46. As diabetes is a recognized risk factor for CKD, NLRP3 genes may be involved in diabetes-related CKD. Additionally, the haplotype analyses were performed showing that the NLRP3 block 1 C-G-A-C, C-G-A-G, C-G-G-G, or T-G-G-G haplotypes significantly decreased the OR of CKD compared to that of the C-A-A-C haplotype (which includes the NLRP3 rs4925650 A allele). To date, epidemiological studies evaluating the effect of NLRP3 polymorphisms on CKD are limited, further studies are needed to explore the possible mechanism of the associations between these genotypes, the haplotype, and CKD.
Several in vitro and in vivo studies have indicated that metals exposure may affect NLRP3 functional changes. An in vitro study has reported that the insulin resistance induced by NaAsO2 is due to activation of the NLRP3 inflammasome47. Chicken experiments have shown that the NLRP3 signaling pathway is activated by lead-induced oxidative stress after lead administration, which causes testicular damage16. Other animal studies have shown that cadmium chloride induces testicular injury48 or liver injury49 in mice by activating the NLRP3 signaling pathway. A selenium-rich basal diet may inhibit lipopolysaccharide-induced inflammation in chicken liver by suppressing the toll like receptor 4-nuclear factor kB-NLRP3 signaling pathway50. Also, a study has reported that dietary selenium attenuates Staphylococcus aureus mastitis in mice by inhibiting the NLRP3 inflammasome51. Our study found no difference when comparing concentrations of environmental metals exposure by different genotypes of NLRP3 rs4925650, NLRP3 rs1539019, and NLRP3 rs10157379, which suggests that metals exposure and the NLRP3 genes have independent effects on CKD. Additional studies are needed to better understand the effect of environmental metals exposure in NLRP3 function and its mechanism.
In the present study, we observed that high levels of blood lead and NLRP3 rs4925650 GA/AA genotypes significantly interacted to increase the risk of CKD after multivariate adjustment. This may be because lead in the blood can induce alterations of inflammatory marker NLRP3 inflammasome activation16, and reduced eGFR6, leading to an increase in the risk of CKD. We also found that high levels of blood cadmium and the NLRP3 block 1 haplotype C-A-A-C multiplicatively interacted to increase the risk of CKD after adjusting for multiple risk factors. Evidence has shown that cadmium in the blood may inhibit heme oxygenase 1 and nuclear factor erythroid 2-related factor 2, and activate the NLRP3 inflammasome49 or increase reactive oxygen species to activate the NLRP3 inflammasome48, and decreased eGFR6, which may jointly cause CKD pathogenesis52. In addition, we observed that low plasma selenium level and the NLRP3 block 1 haplotype C-A-A-C multiplicatively interacted to increase the risk of CKD after adjusting for multiple risk factors. Studies have found that a low level of plasma selenium may not inhibit the expression of NLRP351, or downregulate the toll-like receptor 4-nuclear factor-kB-NLRP3 signaling pathway50, which may increase kidney inflammation to increase the risk of CKD53. Our study did not precisely measure the levels of serum NLRP3 inflammasome. Therefore, whether the NLRP3 inflammasome was affected by levels of blood cadmium and lead, low levels of plasma selenium or the NLRP3 polymorphisms remain unknown. The underlying mechanism of joint effect of environmental metals exposure and NLRP3 polymorphisms affecting CKD needs further exploration.
The sample size of this study was small with limited statistical power. Further studies with larger sample size are needed to improve the precision of point estimates when assessing the effect modification of NLRP3 gene polymorphisms and environmental metals exposure on CKD. Additionally, unmeasured factors such as blood pressure medication and treatment might have potentially influenced our observed results. Future studies should consider these factors in multiple regression models. The analysis of 15 NLRP3 gene polymorphisms may not represent those of the entire gene functions. Our study did not analyze gene polymorphisms that regulate NLRP3 expression differently according to the disease conditions. Further studies should be carried out to evaluate the function of NLRP3 and its related gene polymorphisms in order to determine their role in CKD development.
Conclusions
In conclusion, the present study found evidence that the NLRP3 rs4925650 GA/AA genotypes or NLRP3 block 1 haplotype C-A-A-C altered the risk of CKD related to high levels of blood lead and cadmium, or low levels of plasma selenium. Future studies are warranted to measure the levels of serum NLRP3 inflammasome, to elucidate the biological mechanism underlying the associations between NLRP3 polymorphisms, environmental metals exposure, and CKD.
Change history
30 December 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41598-022-27056-3
References
Jha, V. et al. Chronic kidney disease: Global dimension and perspectives. Lancet 382, 260–272 (2013).
Couser, W. G., Remuzzi, G., Mendis, S. & Tonelli, M. The contribution of chronic kidney disease to the global burden of major noncommunicable diseases. Kidney Int. 80, 1258–1270 (2011).
Wen, C. P. et al. All-cause mortality attributable to chronic kidney disease: A prospective cohort study based on 462 293 adults in Taiwan. Lancet 371, 2173–2182 (2008).
Lin, Y. C. et al. Incidence and prevalence of ESRD in Taiwan Renal Registry Data System (TWRDS): 2005–2012. Acta Nephrol. 28, 65–66 (2014).
Hsueh, Y. M. et al. Urinary arsenic species and CKD in a Taiwanese population: A case–control study. Am J Kidney Dis 54, 859–870 (2009).
Wu, C. Y. et al. The association between plasma selenium and chronic kidney disease related to lead, cadmium and arsenic exposure in a Taiwanese population. J. Hazard. Mater. 375, 224–232 (2019).
Liu, Y. et al. Associations of plasma metal concentrations with the decline in kidney function: A longitudinal study of Chinese adults. Ecotoxicol. Environ. Saf. 189, 110006 (2020).
Lee, J. et al. Environment-wide association study of CKD. Clin. J. Am. Soc. Nephrol. 15, 766–775 (2020).
Anders, H. J. & Muruve, D. A. The inflammasomes in kidney disease. J. Am. Soc. Nephrol. 22, 1007–1018 (2011).
Zhang, C. et al. Activation of Nod-like receptor protein 3 inflammasomes turns on podocyte injury and glomerular sclerosis in hyperhomocysteinemia. Hypertension 60, 154–162 (2012).
Vilaysane, A. et al. The NLRP3 inflammasome promotes renal inflammation and contributes to CKD. J. Am. Soc. Nephrol. 21, 1732–1744 (2010).
Hutton, H. L., Ooi, J. D., Holdsworth, S. R. & Kitching, A. R. The NLRP3 inflammasome in kidney disease and autoimmunity. Nephrology 21, 736–744 (2016).
Qiu, T. et al. Taurine attenuates arsenic-induced pyroptosis and nonalcoholic steatohepatitis by inhibiting the autophagic-inflammasomal pathway. Cell Death. Dis. 9, 946 (2018).
Ahn, H. et al. Mercury and arsenic attenuate canonical and non-canonical NLRP3 inflammasome activation. Sci. Rep. 8, 13659 (2018).
Zhang, Y., Liu, Q., Yin, H. & Li, S. Cadmium exposure induces pyroptosis of lymphocytes in carp pronephros and spleens by activating NLRP3. Ecotoxicol. Environ. Saf 202, 110903 (2020).
Huang, H. et al. Anti-inflammatory effect of selenium on lead-induced testicular inflammation by inhibiting NLRP3 inflammasome activation in chickens. Theriogenology 155, 139–149 (2020).
Zhang, A. Q. et al. Clinical relevance of single nucleotide polymorphisms within the entire NLRP3 gene in patients with major blunt trauma. Crit. Care 15, R280 (2011).
Zaki, M. H., Lamkanfi, M. & Kanneganti, T. D. The Nlrp3 inflammasome: Contributions to intestinal homeostasis. Trends Immunol. 32, 171–179 (2011).
The International HapMap Project. The international HapMap project. Nature 426, 789–796 (2003).
Kunnas, T., Maatta, K. & Nikkari, S. T. NLR family pyrin domain containing 3 (NLRP3) inflammasome gene polymorphism rs7512998 (C>T) predicts aging-related increase of blood pressure, the TAMRISK study. Immun. Ageing 12, 19 (2015).
Zhou, D. et al. The NLRP3 rs10754558 polymorphism is associated with the occurrence and prognosis of coronary artery disease in the Chinese Han population. Biomed. Res. Int. 2016, 3185397 (2016).
Cheng, L., Yin, R., Yang, S., Pan, X. & Ma, A. Rs4612666 polymorphism of the NLRP3 gene is associated with the occurrence of large artery atherosclerotic ischemic strokes and microembolic signals. Biomed. Res. Int. 2018, 6345805 (2018).
Chen, W. J. et al. Renin–angiotensin–aldosterone system related gene polymorphisms and urinary total arsenic is related to chronic kidney disease. Toxicol. Appl. Pharmacol. 279, 95–102 (2014).
National Kidney Foundation. K/DOQI clinical practice guidelines for chronic kidney disease: Evaluation, classification, and stratification. Am. J. Kidney Dis. 39, S1-266 (2002).
KDIGO. Clinical practice guideline for the evaluation and management of chronic kidney disease. Kidney Int. Suppl. 2013(3), 1–150 (2012).
Hsueh, Y. M. et al. Serum beta-carotene level, arsenic methylation capability, and incidence of skin cancer. Cancer Epidemiol. Biomark. Prev. 6, 589–596 (1997).
National Research Council Arsenic in drinking water (1999).
Barr, D. B. et al. Urinary creatinine concentrations in the U.S. population: Implications for urinary biologic monitoring measurements. Environ. Health Perspect. 113, 192–200 (2005).
Barrett, J. C., Fry, B., Maller, J. & Daly, M. J. Haploview: Analysis and visualization of LD and haplotype maps. Bioinformatics 21, 263–265 (2005).
Granata, S., Dalla, G. A., Bellin, G., Lupo, A. & Zaza, G. Transcriptomics: A step behind the comprehension of the polygenic influence on oxidative stress, immune deregulation, and mitochondrial dysfunction in chronic kidney disease. Biomed. Res. Int. 2016, 9290857 (2016).
Wang, X. & Yi, F. Implication of pattern-recognition receptors in cardiovascular diseases. Antioxid. Redox. Signal. 22, 1130–1145 (2015).
Guo, H., Callaway, J. B. & Ting, J. P. Inflammasomes: Mechanism of action, role in disease, and therapeutics. Nat. Med. 21, 677–687 (2015).
Masood, H., Che, R. & Zhang, A. Inflammasomes in the pathophysiology of kidney diseases. Kidney Dis. 1, 187–193 (2015).
Paramel, G. V., Sirsjo, A. & Fransen, K. Role of genetic alterations in the NLRP3 and CARD8 genes in health and disease. Mediat. Inflamm. 2015, 846782 (2015).
Verma, D. et al. Gene polymorphisms in the NALP3 inflammasome are associated with interleukin-1 production and severe inflammation: Relation to common inflammatory diseases?. Arthritis Rheum. 58, 888–894 (2008).
Estfanous, S. Z. K., Ali, S. A., Seif, S. M., Soror, S. H. A. & Abdelaziz, D. H. A. Inflammasome genes’ polymorphisms in Egyptian chronic hepatitis C patients: Influence on vulnerability to infection and response to treatment. Mediat. Inflamm. 2019, 3273645 (2019).
Ji, X. et al. Polymorphisms in inflammasome genes and risk of coal workers’ pneumoconiosis in a Chinese population. PLoS ONE 7, e47949 (2012).
Dehghan, A. et al. Association of novel genetic Loci with circulating fibrinogen levels: A genome-wide association study in 6 population-based cohorts. Circ. Cardiovasc. Genet. 2, 125–133 (2009).
Davidson, C. J., Tuddenham, E. G. & McVey, J. H. 450 million years of hemostasis. J. Thromb. Haemost. 1, 1487–1494 (2003).
Omi, T. et al. An intronic variable number of tandem repeat polymorphisms of the cold-induced autoinflammatory syndrome 1 (CIAS1) gene modifies gene expression and is associated with essential hypertension. Eur. J. Hum. Genet. 14, 1295–1305 (2006).
Zhang, Q. B., Qing, Y. F., He, Y. L., Xie, W. G. & Zhou, J. G. Association of NLRP3 polymorphisms with susceptibility to primary gouty arthritis in a Chinese Han population. Clin. Rheumatol. 37, 235–244 (2018).
Rose, A. B. Introns as gene regulators: A brick on the accelerator. Front. Genet. 9, 672 (2018).
Chung, C. J. et al. Polymorphism of nucleotide binding domain-like receptor protein 3 (NLRP3) increases susceptibility of total urinary arsenic to renal cell carcinoma. Sci. Rep. 10, 6640 (2020).
Maes, M. et al. In COVID-19, NLRP3 inflammasome genetic variants are associated with critical disease and these effects are partly mediated by the sickness symptom complex: A nomothetic network approach. Mol. Psychiatry https://doi.org/10.1038/s41380-021-01431-4 (2022).
Fritz, J. H., Ferrero, R. L., Philpott, D. J. & Girardin, S. E. Nod-like proteins in immunity, inflammation and disease. Nat. Immunol. 7, 1250–1257 (2006).
Zhou, R., Tardivel, A., Thorens, B., Choi, I. & Tschopp, J. Thioredoxin-interacting protein links oxidative stress to inflammasome activation. Nat. Immunol. 11, 136–140 (2010).
Jia, X. et al. Arsenic induces hepatic insulin resistance via mtROS-NLRP3 inflammasome pathway. J. Hazard. Mater. 399, 123034 (2020).
Han, C. et al. Protective effect of Polygonatum sibiricum against cadmium-induced testicular injury in mice through inhibiting oxidative stress and mitochondria-mediated apoptosis. J. Ethnopharmacol. 261, 113060 (2020).
Liu, C. et al. Cadmium induces acute liver injury by inhibiting Nrf2 and the role of NF-kappaB, NLRP3, and MAPKs signaling pathway. Int. J. Environ. Res. Public Health 17, 138 (2019).
Qu, J., Wang, W., Zhang, Q. & Li, S. Inhibition of lipopolysaccharide-induced inflammation of chicken liver tissue by selenomethionine via TLR4-NF-kappaB-NLRP3 signaling pathway. Biol. Trace Elem. Res. 195, 205–214 (2020).
Bi, C. L. et al. Selenium plays an anti-inflammatory role by regulation NLRP3 inflammasome in Staphylococcus aureus-infected mouse mammary gland. Biol. Trace Elem. Res. 199, 604–610 (2020).
Mulay, S. R. Multifactorial functions of the inflammasome component NLRP3 in pathogenesis of chronic kidney diseases. Kidney Int. 96, 58–66 (2019).
Li, L., Tang, W. & Yi, F. Role of inflammasome in chronic kidney disease. Adv. Exp. Med. Biol. 1165, 407–421 (2019).
Acknowledgements
This study was supported by grants from the Taipei Medical University-Wanfang Hospital Research Project (110TMU-WFH-01) and the Ministry of Science and Technology of Taiwan (MOST 103-2314-B-038-021-MY2 (1-2), MOST 103-2314-B-038-021-MY2 (2-2), MOST 105-2314-B-038-082, MOST 106-2314-B-038-066, MOST 107-2320-B-039-010, MOST 106-2314-B-002-235-MY3, MOST 107-2314-B-038-073, MOST 108-2314-B-038 -089, MOST 109-2314-B-038-081- and MOST 109-2314-B-038-067-).
Author information
Authors and Affiliations
Contributions
Y.L.H., H.S.S., and R.L.H. partly contributed to the conception and design of the work, and H.H.C., Y.F.L., and Y.C.L. recruited the study subjects; W.J.C. has done the experiment; Y.M.H. contributed to the statistical analysis and analyzed the data. Y.M.H. wrote the manuscript; W.J.C. reviewed and editing the manuscript; H.H.C. performed the study design and executed the whole research plan.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
The original online version of this Article was revised: The original version of this Article contained an error in the Materials and methods section, under the subheading ‘Study subjects’, where the TMU-Joint Institutional Review Board assigned number was incorrect. It now reads: “The Research Ethics Committee of Taipei Medical University approved this study (TMU-Joint Institutional Review Board, N201812007) which was conducted in accordance with the Declaration of Helsinki.”
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Hsueh, YM., Chen, WJ., Lin, YC. et al. Combined effects of nucleotide-binding domain-like receptor protein 3 polymorphisms and environmental metals exposure on chronic kidney disease. Sci Rep 12, 6307 (2022). https://doi.org/10.1038/s41598-022-10098-y
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-10098-y
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.