Abstract
Extraction of natural resources through mining and logging activities provides revenue and employment across sub-Saharan Africa, a region with the highest burden of malaria globally. The extent to which mining and logging influence malaria transmission in Africa remains poorly understood. Here, we evaluate associations between mining, logging, and malaria in the high transmission setting of the Democratic Republic of the Congo using population-representative malaria survey results and geographic data for environmental features and mining and logging concessions. We find elevated malaria prevalence among individuals in rural areas exposed to mining; however, we also detect significant spatial confounding among locations. Upon correction, effect estimates for mining and logging shifted toward the null and we did not find sufficient evidence to detect an association with malaria. Our findings reveal a complex interplay between mining, logging, space, and malaria prevalence. While mining concessions alone may not drive the high prevalence, unobserved features of mining-exposed areas, such as human migration, changing vector populations, or parasite genetics, may instead be responsible.
Similar content being viewed by others
Introduction
Malaria remains a major cause of illness and mortality across sub-Saharan Africa with an estimated 215 million cases reported in 2019 resulting in over 384,000 deaths1. The high burden of malaria in Africa has been attributed to a complex network of proximal causes, but the region’s environmental suitability for high transmission of malaria is an undeniable root cause2,3. Disturbances to the natural environment through urbanization, deforestation, and agriculture have been shown to significantly alter malaria transmission paradigms, resulting in varied levels of the disease3,4,5,6. Extraction of natural resources through mining or logging also markedly changes natural environments and ecologies, but the effects of these larger scale government allocated operations, referred to as concessions, on malaria remain poorly characterized in sub-Saharan Africa.
Mining and logging industries provide critical streams of revenue and employment opportunities across much of Africa. Exportation of minerals alone accounts for an estimated 70% of the continent’s total exports7 and in Central Africa, nearly 50 million hectares of the Congo Basin are permitted for industrial logging concessions8. Extraction of minerals and timber disrupts the environment in ways that can intensify malaria transmission6,9,10. However, the economic gains from mining and logging activities often contribute to improved housing quality, strengthened infrastructure and access to healthcare, and higher incomes8,11—all of which help reduce the burden of malaria4,12. Additionally, many large mining corporations sponsor malaria control interventions to improve the health of their workforce11,13,14,15. Thus, while mining and logging activities undoubtedly shift local environmental and ecological dynamics, there is also increased potential that malaria transmission may be counterbalanced by the positive impact of these concessions on rural economies and employment stability.
The knowledge base regarding the effects of extractive industries on malaria largely stems from studies of gold mining and deforestation in South America10,16,17,18,19. In Venezuela and Suriname, a large percentage of malaria cases have been attributed to miners20, whose mobility and high risk exposure patterns challenge malaria detection and prevention efforts16,20. Of the limited malaria studies conducted in African mining regions13,15,21,22, only one examined associations between proximal residence to an active mining concession and malaria prevalence22. Knoblauch and colleagues identified a protective effect of close residence to a Zambian copper mine among children; however, their focus on a singular mine limits generalizability of their findings. Studies of deforestation in the Amazon basin have repeatedly linked forest loss to increased malaria10,19; however, associations between forest loss and malaria in Africa have been mixed. In the East African Highlands, decades of forest loss have been associated with increased malaria incidence6,23. A recent multi-national analysis of malaria and remotely sensed forest loss in the Congo Basin found a null effect of deforestation on malaria prevalence in children; however, their use of aggregate survey data may have precluded detection of associations at local scales24. Others have postulated that effects of forest loss on malaria may change over time as vector populations respond to the altered environment25.
Here, we conduct a population-based study to examine associations between exposure to mining and logging concessions and malaria prevalence in the Democratic Republic of the Congo (DRC). The DRC has the second highest prevalence of malaria in the world1, exhibits a booming mining sector7,9, and is home to the largest contiguous area of the Congo Rainforest8. The presence of mining and logging concessions and high occurrence of malaria in the DRC allows for an ideal setting in which to evaluate the effect of these extractive industries on malaria.
Methods
Study design
The primary data source for this study is the cross-sectional 2013–2014 Demographic and Health Surveys for the DRC which is joined with remote-sensed environmental measures and land use data for mining and logging concessions extracted to DHS survey cluster locations. The DHS was administered using a multi-stage cluster survey design to represent the population of the DRC26. Briefly, survey clusters were selected to be representative of all 26 DRC provinces. Within clusters, households were randomly selected proportional to the population size, and within each household, adults ages 15–59 years were consented, interviewed, and asked to provide a dried blood spot (DBS) sample. Only adults who provided a DBS and consented for biospecimen use in future studies were included in this analysis. The outcome of prevalent malaria infections in the DRC was measured through PCR detection of the P. falciparum lactate dehydrogenase gene from DBS samples collected during DHS administration as described previously12.
The main exposures were residence within 15 km of a mining concession and residence within 15 km of a logging concession. Additional covariates included individual-level variables for participant age, sex, use of a long-lasting insecticidal net (LLIN), education, and occupation; household variables for wealth, house roofing material, and the ratio of the number of household members using a bed-net to the total number of household members; and cluster variables for elevation, temperature, precipitation, vegetation, percentage of land cover identified as cropland, grassland, forest, and flooded/swamp land. All individual and household variables were obtained through the DHS. Occupation was recoded such that the manual labor and army category included laborers in mining and logging industries. Cluster variables were extracted from various satellite imagery platforms and other spatial datasets; the methods are described in more detail in the “Appendix”. The main exposures were extracted from geographic data sources as described below.
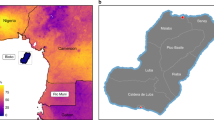
Mining and logging concession data were obtained from the Global Forest Watch online repository27. Mining concessions were subset to only include operations that were active or in remediation spanning the DHS study years (2013–2014); logging concessions only included active operations during 2013. Distance to a mining or logging concession was measured from each cluster location to the boundary of a concession. Clusters were considered exposed to mining or logging if they were located within 15 km of a concession. This distance was chosen to account for the estimated 10 km maximum flight distance of a blood-fed mosquito5, with an additional 5 km to compensate for boundaries and non-residential land near the concessions. This range also accounts for the 5–10 km random spatial offset implemented by the DHS. Locations of mining and logging concessions along with cluster locations were mapped across the DRC. All maps were created in ArcGIS version 10.7.1, shapefiles for administrative boundaries were obtained from GADM.org.
Data analysis
Characteristics of the study population were evaluated across quantiles of P. falciparum cluster prevalence and grouped by individual, household, and cluster level variables. To further examine distributions of malaria interventions and risk factors such as age, sex, LLIN use, occupation, household wealth, and household roof materials by mining and logging exposure, we compared mining exposed and logging exposed clusters with mining and logging doubly unexposed clusters stratified by urban and rural residence.
We then modelled the prevalence odds of malaria across the DRC using hierarchical logistic regression models to account for the nested structure of the DHS data and to allow for inclusion of spatially varying effects. Models were implemented in a Bayesian framework using Integrated Nested Laplace Approximation (INLA) and stochastic partial differential equations for spatial effects28. In all models, we included two separate indicator terms for proximity to a mining concession and to a logging concession; since these areas are non-overlapping, the referent condition for each of these exposures is therefore locations exposed neither to mining nor to logging.
The model fitting process followed two approaches. The first approach evaluated population-level effects of mining and logging on malaria prevalence adjusting for covariates and accounting for cluster-level random effects, which were assumed to vary independently across clusters. The second approach retained covariates and the cluster-level random intercept from the first model and additionally incorporated a spatial field to account for confounding due to space. For the spatial approach, two models were constructed. The first included a spatially varying intercept which borrowed information from neighboring cluster locations assuming a Gaussian random field. The second spatial model explored possible residual confounding due to environmental covariates by allowing spatially varying slopes for temperature, precipitation, vegetation, elevation, and land cover classes while including both independently and spatially varying intercepts across clusters. We introduced spatially varying slopes to account for the unobserved vector population across the DRC. Temperature, precipitation, vegetation, elevation, and various land cover classes have been shown to influence vector composition, survival, and competence for P. falciparum5,23,25, and associations with these covariates may vary due to their effects on the unobserved vector population. Using the spatial modelling approach, we also constructed a smoothed predicted prevalence map of malaria across the DRC, additional details are in the “Appendix”.
For all models, confounding variables were selected based on a directed acyclic graph analysis and retained for adjustment if the 95% uncertainty interval (UI) of the variable excluded the null. Variables were coded as they were presented in the DHS with the exception of collapsing wealth into moderate or higher versus low wealth and recategorization of occupation as: professional, sales, or services; not working; manual labor or army; and agricultural work. All environmental variables were coded as continuous and scaled. Land cover variables were coded in intervals of 10 percentage points. Model comparison was done using Deviance Information Criterion (DIC), with the best fitting model having the smallest DIC29. All models were run using the ‘INLA’ package in R version 4.0.428, additional details are described in the “Appendix”.
Differences in urban and rural residence were considered an important potential source of bias. Urban residence has been associated with lower prevalence of malaria due to many factors including different vector habitats, better access to healthcare, improved housing construction, and overall higher wealth4,12. To address possible bias introduced by urban residence, we stratified all models by urban and rural residence based on the DHS classification of clusters as urban or rural.
A discrete set of confounding variables was identified from fixed effect models for mining and logging in rural and urban areas. The final adjustment set included age, sex, LLIN use, household wealth, temperature, precipitation, vegetation, and elevation. These variables had statistical or substantive significance and were adjusted for in all consecutive analyses.
Ethical approval for this study was obtained from the University of North Carolina Institutional Review Board (UNC IRB# 20-3175) and the Kinshasa School of Public Health. Informed consent was obtained from all participants and all methods were conducted in accordance with guidelines and regulations set forth by the UNC IRB and the Kinshasa School of Public Health.
Results
A total of 16,277 adults across 489 clusters had available P. falciparum PCR results, consented for participation in downstream analyses, and had available cluster location data (Fig. 1). The prevalence of malaria in rural areas was 36.0% (95% UI 35.0, 36.9%) and in urban areas was 28.5% (95% UI 27.4, 29.6%). A predicted surface map of malaria prevalence across the DRC indicated higher prevalence in the north and south-eastern regions and lower prevalence along the eastern border and throughout the Congo River basin (Fig. 2a). The median age of participants was 28 years (IQR: 20–38) and did not differ significantly across quantiles of P. falciparum cluster prevalence (Table 1). Lower quantiles of prevalence correlated with higher individual LLIN use, higher education, more skilled work (professional, sales, and services) and less agricultural work. Households with a higher wealth index, finished roof material, and a higher proportion of household nets per person were more likely to fall into a lower quantile of P. falciparum prevalence. Lower cluster level precipitation, vegetation, and forest coverage and higher population density were also associated with lower cluster P. falciparum prevalence.
Comparisons of malaria risk factors by mining and logging exposure revealed that LLIN use was lowest among individuals residing in mining exposed clusters and highest among individuals in logging exposed clusters (Table 2). Wealth was considerably higher among mining exposed households and in urban areas with 93% of mining exposed urban households reporting moderate or higher wealth. Household wealth was lowest in clusters not exposed to mining or logging in rural areas, but in urban areas, household wealth was lower among those exposed to logging than among doubly unexposed households. This is consistent with findings that mining communities have higher earnings than non-mining communities9,11. Finished roofing materials were consistently more common in mining exposed clusters than either logging exposed clusters or doubly unexposed clusters in both rural and urban areas. In general, finished roofs were more common in urban areas, as was higher household wealth.
Mining and malaria
Mining concessions were prevalent throughout the DRC with 48% of clusters (234/489) located within 15 km of one or more concessions (Fig. 2b). In rural settings, mining was associated with a significant increase in the probability of malaria relative to non-mining and non-logging clusters (POR 1.82, 95% UI 1.35, 2.44) allowing a random intercept for clusters and adjusting for confounding variables. Adding a spatially varying intercept shifted the effect of mining toward the null with a POR of 0.93 (95% UI 0.69, 1.26), adjusting for confounding variables. DIC model fit statistics favored inclusion of the spatially varying intercept (Table 3), suggesting the presence of spatially correlated residual confounding.
In urban settings no significant association with mining was detected when accounting for intra-cluster correlation alone (POR: 0.88, 95% UI 0.57, 1.37), or with a spatially varying intercept included (POR: 0.90, 95% UI 0.54, 1.52). Model fit statistics again favored inclusion of the spatially varying intercept over the random cluster intercept alone (Table 3). Addition of spatially varying slopes for environmental variables in both rural and urban areas failed to improve model fit over the spatially varying intercept model (“Appendix”).
Logging
In the DRC, logging concessions are mostly located in the north-western part of the country, following along the Congo River (Fig. 2c). A total of 48 clusters (9%) were located within 15 km of a logging concession. In rural and urban areas, logging appeared to have a slightly protective association with malaria when controlling for intra-cluster correlation alone; however, the uncertainty intervals for these estimates included the null (Table 3). Inclusion of a spatially varying intercept shifted estimates upward and closer to the null in rural and urban strata with model fit statistics again favoring inclusion of a spatially varying intercept. Adjusting for space, cluster effects, and confounding variables, the prevalence odds ratio of malaria in logging exposed rural areas was 0.97 (0.61, 1.53), and in urban areas was 0.63 (0.28, 1.44).
Discussion
We evaluated associations between exposure to mining and logging concessions and malaria prevalence among Congolese adults. Using population representative malaria survey results and geographic data for mining and logging concessions, our results revealed complex relationships between mining, logging, malaria, and space. Without accounting for space, we found malaria prevalence was higher among individuals in rural areas exposed to mining concessions than among individuals not exposed to mining or logging. When accounting for spatial relationships, exposure to mining and logging operations was unassociated with malaria prevalence. The difference in effect estimates for mining and malaria between the nonspatial and spatial modelling approaches suggests two important conclusions. First, malaria prevalence is high among many individuals in rural areas exposed to mining concessions, a pattern that is also apparent in the predicted malaria prevalence map in Fig. 2a. Areas of elevated prevalence highlight the importance of targeting malaria control interventions in communities proximal to active mining operations. Secondly, the spatial models suggest that the high prevalence of malaria around mining areas was not explicitly associated with exposure to mining concessions themselves. This suggests that mining concessions tend to be located in areas of high malaria prevalence due to factors not measured in these analyses.
The mining sector in the DRC is large and has a complex history of human migration, private and governmental interests, and rapid expansion. In the context of malaria, these features may have led to a mixing of parasite populations30 and different approaches to malaria prevention interventions and vector control9,13—all of which could have contributed to the spatial confounding of mining and malaria that we observed. Spatial variation in parasite genotypes, particularly related to antimalarial resistance, has been shown throughout the DRC, with clusters of drug resistant strains persisting in and around the eastern border and south-central mining regions30. The presence of these genotypes further complicate malaria control and prevention efforts in this high transmission area. Additionally, high rates of human in-migration to mining areas contribute to the mixing of parasite populations and can strain local infrastructure, impacting access to healthcare and administration of malaria control interventions11,13. We did not collect data on parasite genetics or human migration, and the magnitude of bias that each may have contributed to the effects of mining and logging remains unknown and should be studied further.
Another potential source of spatial confounding is the distribution of malaria vector species. In the DRC, Anopheles populations vary widely and have different feeding behaviors, vectorial capacities, and biting rates5,9, all of which influence malaria transmission dynamics and can be difficult to accurately model without extensive entomological survey data. We did not have access to data on vector populations, however we used spatially varying terms for environmental and land cover variables commonly associated with vector dynamics to adjust for unobserved vector populations. Inclusion of these terms had little impact on effect estimates for mining and logging, suggesting that these models may have failed to adequately capture the complexity of the relationships between the environment, Anopheles vector species, and malaria.
This study has several notable strengths including the use of population representative data for malaria results and geographic data to evaluate mining and logging concessions. To our knowledge, this is the first study to evaluate associations between mining, logging, and malaria using a nationally representative sample of adults in Sub-Saharan Africa. This study also had several limitations. First, the DHS was designed to target households and may have missed individuals engaged in mining or logging labor who reside outside of a traditional household living structure. These individuals are likely at the highest risk of any mining or logging associated malaria and are difficult to capture in malaria surveys. Future studies of mining or logging and malaria focused on individual-level health effects might consider constructing surveys that could capture individuals living outside of traditional household structures. Secondly, the ages of participants in this study ranged from 15 to 59 years, thus it is possible that results could be different for children and the elderly who were not included in our study. Thirdly, the DHS does not collect information on environmental variables, land use, or land cover, therefore these measures were all derived from other data sources and linked to malaria results based on cluster location. Measurement error may be present in the derivation of these additional variables, and may also have been introduced when linking to the DHS clusters and averaging throughout the 10 km cluster buffer. Additionally, our mining and logging datasets relied on government reported data of industrial operations. Smaller scale artisanal mining7 and informal timber extraction activities8 are prevalent throughout the DRC and may not have been captured by our data sources. Any effect that local mining and forest clearing activities had on malaria would have been misclassified as unexposed and shifted estimates toward the null. Finally, we were unable to evaluate differences by mining practice or types of extracted minerals. Mining concessions in the DRC often extract multiple types of minerals and close geographic clustering of mining concessions engaged in different practices limited our ability to look for differing effects of mining type or minerals on malaria ecologies given the resolution of our data.
Previously published studies of mining and logging on malaria in Africa have mixed results. In the Brong Ahafo region of Ghana, exposure to an area under development for mining was unassociated with malaria among children15, similar to our results. In a study of a Zambian copper mine, children living in mining-exposed communities experienced a lower odds of malaria than unexposed children22. This finding contrasts with our results and may reflect higher wealth in mining-exposed areas and better healthcare access which may have stronger, more positive impacts on children’s health than adults. Other studies of mining and malaria in sub-Saharan Africa have focused on malaria control interventions implemented within communities around mining concessions13,21. While these studies do not compare malaria infections between communities that are exposed and unexposed to mining, they illustrate successful engagement of the mining sector in malaria control and prevention13,14,21, efforts which should continue to be supported. In the DRC, large mining concessions have historically provided insecticide treated nets and administered indoor residual spraying to reduce malaria transmission11,13. In Fungurume, a town in southern DRC with active copper and cobalt mining operations, entomological surveillance conducted contemporaneously with the DHS found that the local mosquito population had collapsed, likely because of vector control5. We assessed for differences in the relationship between malaria and mining across individual mining concessions in this study, however we did not detect any significant variation. Finally, the results of our logging analysis align with a recent ecological study of deforestation and malaria in sub-Saharan Africa, which found malaria prevalence unassociated with increasing levels of deforestation24. While our results are similar to other null findings in Africa, they contrast with strong relationships between mining and malaria16,17 and deforestation and malaria10,19 in South America, suggesting that the effects of resource extraction on malaria may depend on regional malaria ecologies. Because of this, our results may have limited generalizability outside of the DRC and sub-Saharan Africa.
In our evaluation of mining, logging, and malaria in the DRC we found that mining and logging concessions did not have a detectable effect on malaria prevalence when controlling for spatial confounding. While these results suggest that the mining and logging industries alone may not intensify the burden of malaria in the DRC, it is important to note that malaria prevalence remains high across the country and is further elevated in many areas with active mining operations. Therefore, transmission of malaria should continue to be monitored throughout the DRC as a whole, with particular attention to areas with larger scale resource extraction which should remain a high priority for malaria control interventions especially as demand for natural resources grows and these activities expand.
Data availability
The datasets generated and analyzed during the present study are available from the corresponding author upon request.
References
WHO. World malaria report 2020: 20 years of global progress and challenges. Geneva: World Health Organization; 2020. Licence: CC BY-NC-SA 3.0 IGO.
Paaijmans, K. P., Read, A. F. & Thomas, M. B. Understanding the link between malaria risk and climate. Proc. Natl. Acad. Sci. USA 106(33), 13844–13849 (2009).
Weiss, D. J. et al. Re-examining environmental correlates of Plasmodium falciparum malaria endemicity: A data-intensive variable selection approach. Malar. J. 14(1), 68 (2015).
Tatem, A. J., Gething, P. W., Smith, D. L. & Hay, S. I. Urbanization and the global malaria recession. Malar. J. 12(1), 133 (2013).
Janko, M. M. et al. The links between agriculture, Anopheles mosquitoes, and malaria risk in children younger than 5 years in the Democratic Republic of the Congo: A population-based, cross-sectional, spatial study. Lancet Planet Health 2(2), e74-82 (2018).
Himeidan, Y. E. & Kweka, E. J. Malaria in East African highlands during the past 30 years: Impact of environmental changes. Front. Physiol. 3, 315–315 (2012).
African Natural Resources Center. Catalyzing growth and development through effective natural resources management. Abidjan, Côte d’Ivoire: African Development Bank; 2016. https://www.afdb.org/fileadmin/uploads/afdb/Documents/Publications/anrc/AfDB_ANRC_BROCHURE_en.pdf. Accessed 25 March 2021.
Karsenty, A. & Ferron, C. Recent evolutions of forest concessions status and dynamics in Central Africa. Int. For. Rev. 19(4), 10–26 (2017).
PNLP, KSPH, Swiss KSPH, INRB, INFORM. An epidemiological profile of malaria in the Democratic Republic of Congo. UK (2014).
MacDonald, A. J. & Mordecai, E. A. Amazon deforestation drives malaria transmission, and malaria burden reduces forest clearing. Proc. Natl. Acad. Sci. USA 116, 22212–22218 (2019).
Golder Associates. Environmental and Social Impact Assessment: Executive Summary. Democratic Republic of the Congo; 2007. https://www.eib.org/attachments/pipeline/20070004-nts-en.pdf. Accessed 28 April 2021.
Deutsch-Feldman, M. et al. Spatial and epidemiological drivers of Plasmodium falciparum malaria among adults in the Democratic Republic of the Congo. BMJ Glob. Health. 5(6), e002316 (2020).
Swana, E. K. et al. School-based malaria prevalence: Informative systematic surveillance measure to assess epidemiological impact of malaria control interventions in the Democratic Republic of the Congo. Malar. J. 17, 141 (2018).
Utzinger, J., Tozan, Y. & Singer, B. H. Efficacy and cost-effectiveness of environmental management for malaria control. Trop. Med. Int. Health 6(9), 677–687 (2001).
Asante, K. P. et al. Malaria epidemiology in the Ahafo area of Ghana. Malar. J. 10(1), 211 (2011).
Recht, J. et al. Malaria in Brazil, Colombia, Peru and Venezuela: Current challenges in malaria control and elimination. Malar. J. 16(1), 273 (2017).
PommierdeSanti, V. et al. Malaria in French Guiana linked to illegal gold mining. Emerg. Infect. Dis. 22(2), 344–346 (2016).
Adhin, M. R., Labadie-Bracho, M. & Vreden, S. Gold mining areas in Suriname: Reservoirs of malaria resistance?. Infect. Drug Resist. 7, 111–116 (2014).
Vittor, A. Y. et al. Linking deforestation to malaria in the Amazon: Characterization of the breeding habitat of the principal malaria vector, Anopheles darlingi. Am. J. Trop. Med. Hyg. 81(1), 5–12 (2009).
Pan American Health Organization. Plan of action for malaria elimination 2016–2020. Washington D.C.: Pan American Health Organization/World Health Organization; 2016. https://iris.paho.org/bitstream/handle/10665.2/31440/CD55-13-e.pdf?sequence=1&isAllowed=y. Accessed 25 Mar 2021.
Knoblauch, A. M. et al. The epidemiology of malaria and anaemia in the Bonikro mining area, central Côte d’Ivoire. Malar. J. 13(1), 194 (2014).
Knoblauch, A. M. et al. Community health impacts of the trident copper mine project in Northwestern Zambia: Results from repeated cross-sectional surveys. Int. J. Environ. Res. Public Health 17(10), 3633 (2020).
Lindsay, S. W. & Martens, W. J. M. Malaria in the African highlands: Past, present and future. Bull. World Health Organ. 76, 33–45 (1998).
Bauhoff, S. & Busch, J. Does deforestation increase malaria prevalence? Evidence from satellite data and health surveys. World Dev. 127, 104734 (2020).
Guerra, C. A., Snow, R. W. & Hay, S. I. A global assessment of closed forests, deforestation and malaria risk. Ann. Trop. Med. Parasitol. 100(3), 189–204 (2006).
Ministère du Plan et Suivi de la Mise en œuvre de la Révolution de la Modernité (MPSMRM), Ministère de la Santé Publique (MSP), ICF International. Democratic Republic of Congo Demographic and Health Survey 2013–2014: Key Findings. Rockville, Maryland, USA: MPSMRM, MSP et ICF International; 2014.
Global Forest Watch Open Data Portal. https://data.globalforestwatch.org/. Accessed 9 September 2020.
Lindgren F, Rue H. Bayesian spatial modelling with R—INLA. J. Stat. Softw. 2015; 63(19). http://www.jstatsoft.org/v63/i19/. Accessed 1 Apr 2021.
Spiegelhalter, D. J., Best, N. G., Carlin, B. P. & Linde, A. V. D. Bayesian measures of model complexity and fit. J. R. Stat. Soc. Ser. B Stat. Methodol. 64(4), 583–639 (2002).
Verity, R. et al. The impact of antimalarial resistance on the genetic structure of Plasmodium falciparum in the DRC. Nat. Commun. 11(1), 1–10 (2020).
Acknowledgements
We thank the 2013–2014 DRC DHS study team and participants, without whom this study would not have been possible. We also thank Steve Meshnick for his insight and enthusiasm for this project—his mentorship, keen knowledge of malaria in the DRC, and passion for public health helped shape this study and is deeply missed. Funding support was provided by the National Institutes of Health National Institute for Allergy and Infectious Disease [T32 AI 070114 to CLM]. MMJ is supported by the Bill and Melinda Gates Foundation [OPP1152504]. Funding sources had no role in the study design, analysis, or writing of the manuscript.
Author information
Authors and Affiliations
Contributions
C.L.M. wrote the original draft of the manuscript, M.M.J. help write consecutive drafts. C.L.M., M.M.J., J.K.E., B.W.P., J.J.J., and M.E. conceptualized and designed the study. MKM and AKT provided access to the 2013–2014 DRC Demographic and Health Surveys data. C.L.M. curated and analyzed the data, M.M.J. assisted with data analysis, M.M.J., J.K.E., B.W.P., J.J.J., and M.E. provided analysis tools. C.L.M., M.M.J., J.K.E., B.W.P., M.K.M., J.J.J., and M.E. interpreted the results, and reviewed and edited drafts of the manuscript. All authors approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher's note
Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary Information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons licence, and indicate if changes were made. The images or other third party material in this article are included in the article's Creative Commons licence, unless indicated otherwise in a credit line to the material. If material is not included in the article's Creative Commons licence and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this licence, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Mitchell, C.L., Janko, M.M., Mwandagalirwa, M.K. et al. Impact of extractive industries on malaria prevalence in the Democratic Republic of the Congo: a population-based cross-sectional study. Sci Rep 12, 1737 (2022). https://doi.org/10.1038/s41598-022-05777-9
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-022-05777-9
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.