Abstract
Auxin response factors (ARFs) play central roles in conferring auxin-mediated responses through selection of target genes in plants. Despite their physiological importance, systematic analysis of ARF genes in potato have not been investigated yet. Our genome-wide analysis identified 20 StARF (Solanum tuberosum ARF) genes from potato and found that they are unevenly distributed in all the potato chromosomes except chromosome X. Sequence alignment and conserved motif analysis suggested the presence of all typical domains in all but StARF18c that lacks B3 DNA-binding domain. Phylogenetic analysis indicated that potato ARF could be clustered into 3 distinct subgroups, a result supported by exon-intron structure, consensus motifs, and domain architecture. In silico expression analysis and quantitative real-time PCR experiments revealed that several StARFs were expressed in tissue-specific, biotic/abiotic stress-responsive or hormone-inducible manners, which reflected their potential roles in plant growth, development or under various stress adaptions. Strikingly, most StARFs were identified as highly abiotic stress responsive, indicating that auxin signaling might be implicated in mediating environmental stress-adaptation responses. Taken together, this analysis provides molecular insights into StARF gene family, which paves the way to functional analysis of StARF members and will facilitate potato breeding programs.
Similar content being viewed by others
Introduction
The phytohormone auxin provoke almost every aspect of plant growth, development, stress responses, including apical dominance, defense responses against pathogens, leaf morphology, flower/fruit development, root elongation, vascular differentiation and embryogenesis1,2,3. Some phytopathogens, such as Agrobacterium, Pseudomonas, Streptomyces, and so on, can produce indole-3-acetic acid (IAA) and/or its analogs to modulate or manipulate plant responsive behaviors through auxin-induced signaling cascades and gene expressions4,5.
Considerable progress has been made over recent years in understanding how the cellular auxin response machinery was triggered in plants6. Genetic studies have revealed that, in the auxin signaling pathway, the core components are the F-box-containing TRANSPORT INHIBITOR RESPONSE 1 (TIR1) and its homologous AUXIN-SIGNALING F-BOX PROTEINS (AFBs) proteins, the transcriptional co-repressors AUXIN/INDOLE-3-ACETIC ACID (Aux/IAA), and the transcription factors AUXIN RESPONSE FACTOR (ARFs)7,8,9.In the absence of auxin, interaction between Aux/IAA and ARF proteins inhibited the transcription of auxin-responsive genes; upon the perception of auxin signals, TIR1/AFB protein forms SCFTIR1/AFBs complex with AtCUL1, AtRbx1 and ASK1/2, and recruits Aux/IAA proteins, leading to their subsequent degradation via ubiquitin-proteasome-dependent pathway. The removal of Aux/IAA conduces to ARF-ARF dimerization or interaction with other transcriptional regulators, which determines the transcription of auxin-responsive genes10. As the last step in auxin signaling hierarchy prior to gene regulation, ARFs play central roles in conferring specificity to auxin response through selection of target genes.
The ARF proteins are a set of plant-specific transcription factors, whose typical architecture consists of a conserved amino-terminal DNA binding domain (DBD), a highly conserved carboxyl-terminal domain (CTD) and a variable middle region (MR)11. The DBD is composed of plant-specific B3-type motif, responsible for specifically binding to cis elements like AuxREs (TGTCTC) or its variant (TGTCCC or TGTCAC) in ARF-regulated genes, and an ancillary motif (Auxin_Resp) of unknown function12. The C-terminal Phox and Bem1 (PB1) domain, which were originally described as motif III and IV in Aux/IAA family of proteins, facilitates homo- and hetero-dimerization with Aux/IAA proteins as well as between ARF proteins13. The non-conserved MR is enriched by biased amino acid like glycine (Q), leucine (L), serine (S), and/or proline (P) residues, and the amino acid composition of MR sequence determines the transcriptional ability to activate or repress auxin-responsive genes14,15. To date, ARF gene family has been investigated in several plant species16, including Arabidopsis thanliana, Oryza sativa, Solanum lycopersicum, Zea mays, Glycine max and Gossypium raimondii17,18,19,20,21. The genome of model plant Arabidopsis contains 23 ARF genes, suggesting that ARFs are encoded by a moderate-sized gene family. Hereafter, 25, 51, 36, 21 ARF members were identified in rice, soybean, corn and tomato, respectively17,19,20,21,22. Given that multiple members in ARF family, there might be significant functional diversification between individual members.
Current knowledge of the biological roles of individual ARFs is largely from the characterization of model plants, Arabidopsis and rice. In Arabidopsis, ARF3 identified as a mediator of crosstalk between self-incompatibility (SI) signaling and pistil development, acts non-cell-autonomously to enhance the SI response and simultaneously attenuate auxin responses in stigma epidermal cells, probably by regulating a mobile signal derived from the stylar vasculature23; ARF5/MONOPTEROS directly binds to promoter region of ATHB8, a homeodomain-leucine zipper transcription factor gene involved in procambial cell fate acquisition, and is also required for the expression of the auxin efflux facilitator PIN1, further mediating organ and vascular tissue formation24,25; ARF6, redundantly with ARF8, dynamically regulates the flower maturation including stamen filament elongation, anther dehiscence, stigmatic papillae elongation, gynoecium maturation and flower bud opening26,27. In rice, down-regulation of OsARF1 leads to growth retardation, short curled leaves and sterile phenotype in transgenic plants, suggesting its essential role in vegetative organs and seed development28; OsARF12 is regarded as one of major player in phosphate-induced auxin responses, indicating that ARFs might be involved in phosphate homeostasis in crops29.
Potato (Solanum tuberosum), the third most important global food crop, is regarded as the most widely grown non-cereal crop, and over 1 billion people have potato as daily diet30. Solanum tuberosum Group phureja DM1-3 516 R44 (hereafter referred to as DM) is a homozygous doubled-monopoloid potato, ~727 Mb genome of which was annotated at Potato Genome Sequencing Consortium (PGSC)31. Considering that ARFs are implicated in plant growth, development and stress adaptions, information on potato ARF gene family is needed for better understanding molecular mechanism between auxin signaling and physiological processes in this crop plants. Yet potato ARF gene family, to our knowledge, still remains unexplored. In this study, taking advantage of the DM potato reference genome, we conducted a genome-wide, comprehensive analysis of ARF family genes in potato. A total of 20 StARF (Solanum tuberosum ARF) genes was identified, and the physical and chemical characteristics, genomic structures, chromosomal locations, evolutionary relationship/scenario, expression profiles and protein-protein interaction network of StARF family were investigated in detail. These analyses will contribute to the better understanding the role of ARF genes in auxin-involved responses in potato and will also provide a robust database for the potato research community.
Results
Genome-wide identification of ARF genes from Solanum tuberosum
In order to identify ARF genes in potato, we first used combination of B3 DNA binding (Pfam 02362), Auxin_Resp (Pfam 06507) and PB1 (Pfam 02309) domains as the query to perform local BLAST searches against the potato genome database in Phytozome as well as Potato Genomics Resource. After removing the non-representative splicing forms of same gene locus, sequences of non-redundant ARF candidates were further verified by a Hidden Markov Model (HMM)-based for the presence of both B3 DNA binding and Auxin_Resp motifs, and a total of 20 ARF members were identified in the genome of Solanum tuberosum. The nomenclature for StARF members in the present study was given according to the homologies against Arabidopsis ARFs (Supplementary Fig. S1). Information on the gene name, loci number chromosomal location, predicted characteristics of putative proteins of individual StARF genes are listed (Table 1). Of these, the molecular weight of putative StARF proteins ranged from 40.1 to 130.1 kDa. It is noteworthy that the majority of StARFs encoded weakly acidic proteins as supported by the predicted isoelectric points. The results reinforced the assumption that these StARFs might participate diverse biochemical processes under disparate in vivo environments.
Chromosomal distribution of StARF genes
The chromosomal location and direction of transcription for each ARF gene were established in DM potato. As shown in Fig. 1, 20 StARF genes were positioned on all chromosomes except the chromosome X, however the number of StARF genes were unevenly distributed to potato chromosomes. Three each StARFs were present in chromosome VII and XI; two each were on chromosome I, III, V, VIII and XII; one each located on chromosome II, IV, VI and IX.
Genomic distribution of StARF genes on DM1-3 chromosomes. The chromosome numbers and size are indicated at the top and bottom of each bar, respectively. The arrows next to gene names show the transcription directions. The number on the right side of the bars designated the approximate physical position of the first exon of corresponding StARF genes on potato genome.
Gene duplication is one of the most important mechanisms for acquiring new genes and creating genetic novelty in organisms. Although the member of ARF genes is similar between potato and Arabidopsis, five possible duplication events were observed as sequence alignment suggested that some StARF genes exhibited extreme-high sequence similarity. Among them, StARF2a-2b, StARF6a-6b and StARF10a-10b form twin pairs, StARF18a/b/c and StARF19a/b/c from triplet pairs. Surprisingly, no tandem duplication events were identified because most duplicated StARF genes were positioned to different chromosomes, or they were separated by at least several megabases.
Conserved motif and domain analysis of StARFs
It is reported that most ARF proteins carry a conserved N-terminal DNA-binding domain composed of a plant specific B3-type and Auxin_Resp motifs, and a highly conserved C-termianl PB1 domain corresponding to motif III and IV of the Aux/IAA proteins32,33. To better understand the structural similarity of these ARFs, a multiple sequence alignment was conducted with the deduced amino acid sequences of StARF genes (Supplementary Fig. S2). The results revealed that all StARF proteins contained highly conserved and intact DBD domains, except that StARF18c had a truncated DBD domain (Fig. 2). Although the partial sequence of Auxin_Resp motif was retained, it is plausible that the DNA-binding ability of StARF18c could be impaired due to the lack of DNA-binding domain.
Analysis of conserved domains in StARF porteins. (A) Schematic organization of conserved domains in StARF proteins. The B3 DNA-binding domain, Aux_Resp domain, and PB1 domain are shown in blue, red and green, respectively. (B) Amino acid composition of MR domains in StARF proteins. Bars represent the percentage of different amino acid residues in MR domains of StARFs, and each color represents one kind of amino acid.
Unlike conserved DBD and CTD regions, the protein sequence of the Middle Regions (MRs) are high variable. However, the amino acid composition of the MRs exhibits some non-conserved, unique features between ARF proteins32. It has been proposed that ARF proteins whose middle regions are rich in S, P, L and G might act as transcriptional repressors, whereas those rich in Q, Sand L in MR might be activators34,35. Composition analysis of StARF proteins suggested that the MRs of StARF6a/b, StARF8, StARF19a/b/c are abundant in Q residues, suggesting they might be transcriptional activators (Fig. 2B). Surprisingly, it seems that the MR of StARF17 was completely missing, which made it impossible to speculate its transcriptional activity (Fig. 2).
Structural analysis of StARF genes
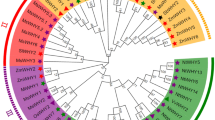
In addition to conserved protein domains, the pattern of intron positions between ARF subgroups can also provide some clues on evolutionary relationships. To identify the introns-exons structure of individual StARF member, a comparison of the full-length cDNA sequences with the corresponding genomic DNA sequences was performed. Accordingly, we constructed a neighbor-joining phylogenetic tree using the amino acids sequences of 20 StARF members, resulting in 3 major ARF clades with very high bootstrap confidence values (Fig. 3A). In the members of StARF5/6/8/19 clade, there are 10–13 introns, and members of StARF1/2/3/4/18 clade have 9–13 introns except that truncated StARF18c contains 5 introns (Fig. 3B). Consistent with previous findings from Arabidopsis and rice, members of StARF10/16/17 clade have relatively lower number of introns (Fig. 3B). Strikingly, the exon-intron structure is conserved within subclades including StARF6a/b, StARF19a/b/c, StARF10a/b, StARF2a/b and StARF18a/b, although the sizes of introns vary greatly for different members; however, the exon-intron structure varies between subclades, which was also supported by the phylogenetic analysis of StARF proteins.
Classification of S. tuberosome ARF proteins. (A) Neighbor-joining tree were generated using MEGA 7.0 to determine the phylogenetic relationship between S. tuberosome ARF proteins (left). According to classification proposed by Finet et al. (2013), StARFs were divided into three subgroups: (A–C) Shadow colors were used to distinguish different StARF subgroups. (B) The intron-exon organization of StARF genes was plotted using Gene Structure Display Server (Version 2.0). Blue boxes represent exons, and grey lines represent introns.
Phylogenetic analysis of StARFs
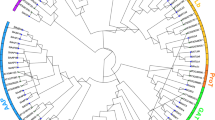
To gain the insight into the evolutionary scenario of StARFs and their homologs in other plant species, we expanded the neighbor-joining analysis to include ARF sequences from other taxa: 4 ARF sequences from the liverwort Marchantia polymorpha, 15 sequences from the moss Physcomitrella patens, 24 sequences from tomato, 23 sequences from rice, and 23 from Arabidopsis (Fig. 4). A B3 domain-containing sequence from green algae Chlamydomonas reinhardtii was used as outgroup. In this analysis, 111 of ARF sequences were joined in a moderately well-supported phylogenetic tree. Cre13.g562400 from C. reinhardtii is the most divergent gene in the overall organization as it showed sequence similarity to ARF genes in B3 DNA-binding region and lacks other motifs. Consistent with analyses by Mutte et al. (2018) and Finet et al. (2013), the ARFs can be classified into 3 major clades on their phylogenetic relationship. Clade A had 49 members, which were orthologs of AtARF5, AtARF7, AtARF6/8 and AtARF13; Clade B included 33 members, and they were homologous to AtARF1, AtARF2, AtARF3 and AtARF4; the remaining ARFs belonged to Clade C, which consisted of AtARF17 and other 20 homologs.
Phylogenetic analysis of ARF proteins in liverwort, the moss, and flowering plants. Neighbor-joining tree was constructed based on the alignment of ARF protein sequences from Marchantia polymorpha, Physcomitrella patens, Oryza sativa, S. lycoperiscum, S. tubersomem and Arabidopsis thaliana. The position of the root was determined from an outgroup consisting of an ARF-like sequence from green algae Chlamydomonas reinhardtii. The percent bootstrap support for 500 replicates is shown on each branch with >50% support. Some nodes were designated with letters in bold and italic. Sequences used in phylogenetic analysis were provided in Supplementary Data.
We further analyzed the phylogeny of the ARF family in more detail (Fig. 4). Clade A contained 3 subclades, which were represented by nodes a, b and c in Fig. 3, and the relationships of subclades in clade A to one another were well supported by bootstrap percentage. The node a (ARF6/8 subclade) subclade was placed as twin-sister to the node b (ARF5/7 subclade), while node c subclade, containing class A-ARFs of bryophytes, was basal to other two subclades, suggesting they might represent precursors of class A-ARFs of land plants. Clade B could be further divided into 5 subclades, represented by node d (ARF3/4 subclade), e (ARF2 subclade), f (ARF1 subclade), g (ARF11/18 subclade) and h (ARF13 subclade). Strikingly, clade B consisted of ARF members only from land plant species, indicating that duplication and diversification class B-ARFs might occur in euphyllophytes. Clade C was composed of node i (ARF17 subclade), j (ARF10/16 subclade) and k (bryophyte C-ARFs). As observed in clade A, we found that node k subclade was only represented by members from bryophytes, implying they might be a group of proto-C-ARFs.
Expression profiles of StARFs among various tissues and developmental stages
To understand the tissue-specific expression profile of StARF genes, FPKM (Fragments per Kilobase of transcript per Million mapped reads) values generated by PGSC were used to assess their expression levels across different organs and developmental stages. The 13 RNA-seq database could be divided into 4 major groups: floral (carpel, petals, sepals, stamens and mature flower), leaf (whole leaf, leaflet and petiole), tuber (tuber, stolon), and others (shoot, root and callus)36. As shown in Fig. 5, of these 20 StARF genes, StARF1/2a/6 were ubiquitously abundant in most tissues, suggesting that they might execute some universal roles in auxin-regulated plant growth and development processes. On the contrary, we also found that expression of several StARFs, including StARF3/10b/16/17/18a, were relatively lower.
Expression profiles of StARF genes with hierarchical clustering in different tissues. The Illumina RNA-Seq data were obtained from PGSC database, and the FPKM value of representative transcripts of StARF genes were log2 transformed for further analysis. The normalized expression data was used to generate heatmap with hierarchical clustering based on the Manhattan correlation with average linkage using MeV software package. Color scale below heatmap shows the expression level; red indicates high transcript abundance while green indicates low abundance.
With respect to floral tissues, transcripts of StARF2/5/6/8/19b were abundant in carpels, StARF1/2a/5/6/18b/19b were preferentially expressed in stamens, StARF6b/19a were stamen-specific ARFs, and StARF2a/6/8 is highly expressed in sepal tissues. The different expression domains of StARFs implied that they might coordinate the expression of target genes involved in the flower development. In addition to StARF1/2a, StARF4/6/8 were highly expressed in leaf or petiole tissues, whereas StARF2b/13 were abundant in root tissues, implying they might play roles in auxin-involved organ development.
Potato tubers develop from underground stolons, and in an in vitro tuberization system, exogenously applied auxin can stimulate the tuberization process. Therefore, ARFs could play a role in tuber growth. We found that a relative high level of expression of StARF1/2a/6 was observed in stolons, which might contribute to the swelling of stolons. However, StARF1/2a/18b expression was much higher than other StARFs, suggesting they might function in root growth.
Differential expression of StARFs during biotic and abiotic stresses
Though auxin was generally regarded as one of major growth-promoting hormones, accumulated evidence highlighted its role in mediating stress responses through cross-talking with other signaling pathways37,38. ARF transcription factors might be one of potential targets in face of abiotic/biotic stresses and hormones. Therefore, the expression patterns of StARF genes under various stress, hormone (and their analog) treatments were investigated by analyzing the log2 fold change between treatments and controls. All 20 StARFs, with the exception of StARF19a, showed strong up-regulation in response to salt stress and mannitol treatment, whereas only half were highly expressed upon heat stress (Fig. 6A). We also found that expression of StARF5/10a/10b/13/18a/18c was decreased in response to thermal conditions (Fig. 6A). Thus these results implied that responsive behavior of StARF genes were associated with plant adaption to diverse environmental stimuli.
Heatmap representation and hierarchical clustering of StARF genes under abiotic stresses (A), biotic stresses (B), and phytohormone treatments (C). The Illumina RNA-Seq data were obtained from PGSC database, and the relative expression of StARF genes was calculated with respect to control samples using FPKM values of representative transcripts corresponding to StARF genes. Fold changes of StARF expression were log2 transformed, and the normalized expression data was used to generate heatmap with MeV software package using the same parameters in Fig. 4. Color scale below heatmap shows the expression level; red indicates high transcript abundance while green indicates low abundance.
The oomycetes P. infestans infection, as well as BABA treatment, resulted in the inhibition of most StARFs (Fig. 6B), clearly suggesting that a majority of ARF-regulated genes were suppressed, and thus auxin could promote disease susceptibility. However, when plants treated with benzothiadiazole S-methyl ester (BTH), an SA analog, only StARF6/17/19a showed strong up-regulation, while StARF5/18b went in the opposite way. Likewise, expression of other StARF members was slightly changed, implying that BTH could induce disease resistance via a molecular mechanism that differs from BABA.
Similarly, we also examined the expression responses of StARFs under various phytohormone treatments (Fig. 6C). Surprisingly, only StARF17/18b were induced by auxin treatment, whereas expression level of other StARF members was slightly decreased or even unchanged. Despite that StARF17/2a expression was induced by GA3, most StARF members slightly decrease their expression in response to gibberellin. In presence of the 6-Benzylaminopurine (BAP), expression of StARF1/4/6a/18a/18b/19a/19c was attenuated, while expression of StARF10b/17 was induced by cytokinin applications. Intriguingly, abscisic acid (ABA), instead of auxin, could induce the expression of nearly half of StARF members like StARF3/13/17, while the induction was not observed in other members. Therefore, these results indicated that some StARF transcription factors, possibly through regulating their target genes, participated diverse abiotic signaling pathways, and were major players in environmental stresses.
Validation of StARFs expression patterns
Although Illumina RNA-seq data provides preliminary information on the expression behavior of StARF genes, in some instances, there are some discrepancies between in silico analysis and experimental data. Thus, we carried out quantitative real-time RT-PCR analysis of StARF genes in several tissues or under several representative conditions, and potato cultivar Shepody was used for the validation experiments. The qRT-PCR results of StARF genes in root, stem and leaf tissues were largely in good agreement with RNA-seq data (Fig. 7A). However, we also found that some members of StARF gene family showed different expression patterns. For example, expression level of StARF4 was similar between root and leaf tissues, which was contradicted the RNA-seq analysis. The inconsistency was due to the differences in collecting potato tissue samples, or probably was the consequence of genotype-dependent expressions.
qRT-PCR analysis of StARF genes in different tissues (A) and phytohormones (B) StARF transcript levels measured by real-time qRT-PCR from the various tissues or under phytohormone treatments at indicated time points. Data are means of three biological replicates (8 pooled plants each), and error bars denote SE. Potato elongation factor StEF-1α gene was used as an internal control. Stars above the error bars indicate significant differences between treatments and controls (according to student’s t-test). qRT-PCR primers for StARF and StEF-1α genes were provided in Supplementary Table S1.
In another experiment, qRT-PCR experiments were conducted on selected conditions to validate the expression patterns of StARFs in response to IAA and ABA treatments (Fig. 7B). By and large, expression patterns of StARFs under selected conditions were similar to RNA-seq results. For example, we observed that StARF17 was strongly induced upon IAA and ABA treatments, and expression of StARF3 was induced by ABA, but kept unchanged to auxin treatment, which is consistent with expression patterns revealed by RNA-seq analysis. Furthermore, we also investigated the expression patterns of StARFs under ethylene (ETH) treatment (Fig. 7B), as it is implicated in regulating plant growth, defense response and senescence. A noteworthy observation in this analysis most members of StARF gene family exhibited up-regulation in response to ETH treatment; contrary to this, we also found several StARF members, including StARF4, StARF10b, StARF16, and StARF18a, showed decreased expression or were unable to respond. The varying responses of StARF genes to ETH treatment could thus be attributed to their diverse roles in complex regulatory cross-talk between auxin and ethylene signaling.
Interaction network of StARFs
Both ARF and AUX/IAA proteins contain PB1 domain, which contributes to the interaction of ARF-ARF, ARF-IAA, and IAA-IAA. To better understand gene functions, the STRING database was employed to generate the protein-protein network between ARF and AUX/IAA protein families in potato39. As shown in Fig. 8, large protein-protein interaction pairs were predicted between StARFs and StIAAs, indicating that members from the two protein families could homo- or hetero-dimerize to mediate ARF-regulated gene expression. Moreover, several StARFs were found to be core nodes in the whole network, indicating that they might be involved in different physiological functions by working with other PB1-domain containing proteins. For example, StARF4 was speculated to be associated with StIAA1/2/9/13/19/20/22/23 and StARF1/6/8/10a/16. On the other hand, a few StARFs could only interact with specific StIAAs, like StARF3-StIAA1, StARF2a-StIAA20/24, and StARF2b-StIAA20/24 pairs. Thus, these results illustrated how members of StARF and StIAA protein families might form functional transcription factor complexes, mediating the expression of most auxin-responsive genes in potato.
Predicted protein-protein interaction network of StARFs and StIAAs. Each node represents either one StARF protein (blue label) or StIAA (black label) protein. The disconnected nodes in the network were hidden due to the lack of supporting information. Edges between nodes represent protein-protein associations predicted by experimentally determined (pink line) or from curated databases (blue line).
Discussion
Auxin response factors are key players in auxin signaling cascade, which controls many physiological and development process in plants2,40. ARFs exert their roles by mediating the expression of downstream genes, thus regulating the cellular responses to plant hormone IAA or its analogs41. Previously, the ARF gene family has been intensively studied in model plant Arabidopsis18, and also to varying degree in a number of other plant species, such as rice, tomato, and soybean17,21,22. In our analysis, total 20 StARFs were identified from potato genome. Given that the genome size of DM1-3 potato was 740 Mb, nearly 5 times larger than Arabidopsis genome31, it is surprising that the number of StARF genes was much less than that of Arabidopsis. Apparently, our observations on ARF gene family contradicted with genome complexity between Arabidopsis and potato. With respect each clade A- and C-ARFs, there were 7 and 4 members of in potato respectively, whereas only 5 and 3 Arabidopsis counterparts were identified. Clade B-ARFs contained 9 members in potato, and 14 members in Arabidopsis, which leads to the big difference in the overall number of ARF genes between potato and Arabidopsis. One possible explanation for the paradox is that several independent, small-scale, segmental duplication events and chromosome rearrangements occurred at ARF13 loci resulted in multiple members of AtARF13 subclade, finally leading to the expansion of ARF gene family in Arabidopsis.
To assess the evolution trajectory of ARF genes, we also searched the available genome data of other plant lineages including green algae, bryophytes (liverwort and moss) and flowering plants, and nearly one hundred of ARF candidate sequences were obtained with similar blast method previously mentioned. Despite the occurrence of several genes encoding B3, III or IV motifs, Auxin_Resp domain was unable to be identified in algae genome. Nevertheless, Auxin_Resp and other three domains (B3, III and IV) in a single protein could be recognized from liverwort and moss genomes, thus reinforcing the idea that canonical ARF proteins were probably established during the evolutionary transition from algae to bryophyte plants.
Furthermore, we conducted a comprehensive phylogenetic approach using the conserved domains on our 111-ARF sequences from algae to flowering plants. Consistent with previous reports, ARF genes were classified into three main clades: clade A (ARF5/6/7/8), clade B (ARF 1/2/3/4/9) and clade C (ARF10/16/17). Among the three main ARF clades, we found that only clade B consisted of ARF sequences only from angiosperms, strongly suggesting the clade might be evolved from flowering plant species. Conversely, bryophyte ARF sequences were present in clade A and C, indicating that the ARFs had diverged into at least two group of transcriptional factors in the common ancestor of bryophyte, and this ancient event possibly predated the divergence of liverwort and moss. Strikingly, we detected that 5 and 4 ARF protein sequences from bryophyte, in our phylogenetic analysis, were loosely associated clade A and C, respectively, and these ARF sequences formed 3 independent clusters on basal of each subclade. As determined by morphology data, fossil evidence and molecular phylogenetic analyses, bryophytes diverged earlier that other land plants42,43,44. With these observations, it is likely that these bryophyte ARFs might resemble the counterparts of ancestral ARF genes in land plants.
Accumulating evidence suggested a link between plant growth hormone auxin and disease resistance45,46, while the behavior of auxin in pathogenesis is complicated. Most studies indicated that auxin promote disease symptoms in many plants. Exogenous IAA applications aggravated the disease progression during the infection of rice with Xanthomona oryzae47. Similarly, IAA application attenuated oligogalacturonide-induced resistance to Botrytis cinerea in Arabidopsis48. However, there were also some findings that auxin played positive roles of in disease resistance. For example, a metabolic profiling approach revealed that endogenous auxin level substantially increased during pathogen infection of Arabidopsis with Pseudomonas syringae DC3000 in comparison with mock plants49. In silico analysis also suggested that, in tomato plants, most ARF genes were differentially expressed in response to various pathogen infections of bacteria and virus50. Consistent with its complicated behavior of auxin, our findings revealed that expression patterns of StARF genes were varying when potato challenged by pathogen P. infestans or treated with disease resistance-priming agent BABA and BTH. Thus, we conclude that the different expression patterns of StARFs might reflect their complicated roles in plant immune response.
In our analysis, we found that expression of most StARF genes was closely associated with heat, salt and osmotic stresses, and expression of StARF3/13/17 were also substantially induced by abscisic acid known as the abiotic stress hormone. Similarly, a whole genome microarray indicated that, among the 186 transcription factor genes, nine OsARFs were identified as drought- or salinity-responsive genes51. In soybean (Glycine max), expression of GmARF33 and GmARF50 was induced by water deficit. In sorghum (Sorghum bicolor) roots, SbARF10, SbARF16 and SbARF21 were induced by salinity, whereas expression of others was repressed29. Interestingly, SbARF genes were strongly induced by salt stress in sorghum leaf tissues, which was corroborated by the evidence that most StARF genes were induced upon abiotic stresses29. Additionally, Arabidopsis ARF19 was proved to be an important player linking hormonal crosstalk of auxin and ethylene-mediated responses52. Our qRT-PCR experiments on StARF genes also revealed that in addition to StARF19b, there were a few StARF genes whose expressions were activated by ethylene treatment, and thus it is likely ARFs might be interaction nodes between auxin and ethylene signaling. Therefore, these observations strongly suggested that ARFs could be a class of potential key players, regulating spatio-temporal expression of stress-responsive genes to mitigate the negative effects of environmental or biotic stresses.
Auxin response factors is implicated in a plethora of developmental processes and stress-adaption responses. In this study, genome mining revealed the existence of 20 StARF genes from potato. Chromosomal distribution, conserved domains/motifs, and structural analysis phylogenetic studies have provided valuable insights into the evolution aspects of StARF gene family. Expression profiling of StARFs in different tissues gave us a cue on how these ARF transcriptional regulators participate auxin-signaling cascade in plant development. Moreover, the diverse responsive behaviors of StARFs in response to stresses, phytohormones and chemical inducers suggested that they were actively involved in the regulation of stress responses to biotic and abiotic stimulus. Protein-protein interaction analysis illustrated the translational regulation relationships of StARFs and its interacting proteins. In summary, our results provide new insights of StARF gene family, which will facilitate future selection and breeding for stress-tolerant potato cultivar.
Materials and Methods
Identification of ARF genes in Solanum tuberosum Group phureja
Phytozome (https://phytozome.jgi.doe.gov/), Potato Genome Sequencing Consortium (PGSC, http://solanaceae.plantbiology.msu.edu/), and NCBI (http://blast.ncbi.nlm.nih.gov/) online resources were searched to identify the entire family of ARF in Solanum tuberosum Group phureja DM1-3. The hidden Markov model (HMM) profiles of the ARF gene family which includes B3 DNA-binding domain (Pfam 02362), Auxin_Resp (Pfam 06507) and AUX/IAA family (Pfam 02309) were used to identify ARF candidates from potato genome. The potato ARF protein sequences were further confirmed using SMART (http://smart.embl-heidelberg.de/smart/batch.pl) and Pfam (http://pfam.xfam.org/search) to confirm the presence of conserved motifs mentioned above. In order to obtain non-redundancy ARF sequences, potato ARF sequences were further used as queries to blast against Phytozome database, and any redundancy was manually removed.
Analysis of gene structure and conserved domains
Based on the genome annotation of DM assembly available in Phytozome, the intron-exon structure of individual StARF genes was predicated, and its genomic organization was visualized using Gene Structure Display Server 2.0 (GSDS, http://gsds.cbi.pku.edu.cn/)53.
Conserved domains in protein sequences were verified using ScanProsite (http://pro-site.expasy.org/scanprosite/) which provides information about positions of different domains in the protein sequence. This information was used to draw visual representation of distribution of domains in the deduced amino acid sequences of proteins using Illustrator for Biological Sequences (Version 1.0)54.
Sequence alignment and phylogenetic construction
Phylogenetic analysis of ARF protein sequences used only the conserved N-terminal DNA-binding domain and the conserved C-terminal region corresponding to the Aux/IAA motif III-IV region. Neighbor-joining analyses of the Arabidopsis Aux/IAA and ARF sequences were conducted in PHYLIP 3.5 (http://evolution.genetics.washington.edu/phy-lip.html) using the PAM matrix of Dayhoff, with 500 bootstrap replicates and randomized sequence input order. One B3 domain-containing sequence from Chlamydomonas reinhardtii was used as outgroup.
Expression profiling of ARF genes in different tissues or under various stresses
Transcriptome analyses were performed using the RNA-seq sequence information generated by the PGSC36. For developmental stages, the corresponding FPKM (fragments per kilobase per million reads) values for StARF genes were obtained for 13 tissues representing major organs and developmental stages, including floral (carpel, petals, sepals, stamens and mature flower), leaf (whole leaf, leaflet and petiole), tuber (tuber, stolon), and other organs (shoot, root and callus).
Abiotic stress-treated tissues included 24-h in vitro grown whole plants exposed to heat (35°C), NaCl (150 mM) and Mannitol (260 mM). Hormone treatments of in vitro grown whole plants consisted of abscisic acid (ABA, 50 mM), indole-3-acetid acid, (IAA, 10 mM), gibberellic acid (GA3, 50 mM), and 6-benzylaminopurine (BAP, 10 mM). Biotic stress were represented by detached leaves challenged by Phytophthora infestans, BABA (DL-β-amino-n-butyric acid), BTH (6-benzylaminopurine), and samples were pooled at 24, 48 and 72 h. Control plants were grown in parallel with each stress or hormone treatment. Similarly, FPKM values for abiotic or biotic stress-treated potato plants were analyzed by calculating the fold change of expression levels between treatments and the corresponding controls.
The normalized expression data was used to generate heatmap using the Institute for Genomic Research MeV software package (http://mev.tm4.org), and hierarchical clustering analysis (HCA) was built on the basis of the Manhattan correlation with average linkage method.
RNA extraction and quantitative real-time RT-PCR
Solanum tuberosum (cultivar “Shepody”) plants were in vitro micropropagated on Murashige and Skoog (MS) medium plus 30 gL−1 sucrose and 0.8% agar (Sigma-Aldrich), with pH adjusted to 5.8. Potato seedlings were routinely subcultured as two-node segments every 3–4 weeks and incubated at 23°C with 16 h photoperiod under cool with fluorescent lamps (~70 μmol m−2 s−1 photon flux density). 3-week old plants were subjected to IAA (50 μM), ABA(100 μM) or ethylene (1 mM) treatments. The plant tissues were collected at designated points and immediately frozen in liquid nitrogen. Sample collections were performed on separate days for the replicates. Total RNA was extracted with Trizol (Invitrogen Inc., USA) as described by the manufacturer. RNA quantity and quality were assessed using a NanoDrop8000 (Thermo Scientific™, USA). Total RNA isolation and reverse transcription with oligo(dT)18 (18418-012; Invitrogen, USA) were performed as described previously26. The amounts of individual genes were measured with gene-specific primers by real-time PCR analysis with a cycler IQ real-time PCR instrument CFX96 (Bio-Rad, USA) and SYBR Green mixture (Bio-Rad, USA). The relative expression of specific genes was quantitated with the 2−ΔΔCt calculation method55, where ΔΔCt is the difference in the threshold cycles and the reference gene, which was potato eIF-1α for expression analyses. The sequences of specific primers are shown in Supplementary Table S1.
Construction of interaction network of StARFs
Protein–protein interaction (PPI) analyses of StARF family were performed on the STRING website (http://www.string-db.org) to predict protein interactions39. Briefly, StIAAs previously identified56 were included as potential interacting-partners of StARFs. Amino acid sequences of the two families were used as query sequences with Arabidopsis as the model. In confidence mode, the online program was run with parameters as follows: the box “active interaction sources” was chosen to “Experiments”; the box “minimum required interaction score” was set to 0.9 (highest confidence); other boxes used default settings.
References
Teale, W. D., Paponov, I. A. & Palme, K. Auxin in action: signalling, transport and the control of plant growth and development. Nat Rev Mol Cell Biol 7, 847–59 (2006).
Weijers, D. & Wagner, D. Transcriptional Responses to the Auxin Hormone. Annu Rev Plant Biol 67, 539–74 (2016).
Zhao, Y. Auxin biosynthesis and its role in plant development. Annu Rev Plant Biol 61, 49–64 (2010).
Fu, J. & Wang, S. Insights into auxin signaling in plant-pathogen interactions. Front Plant Sci 2, 74 (2011).
Kazan, K. & Manners, J. M. Linking development to defense: auxin in plant-pathogen interactions. Trends Plant Sci 14, 373–82 (2009).
Weijers, D. & Friml, J. SnapShot: Auxin signaling and transport. Cell 136(1172), 1172 e1 (2009).
Kong, X., Zhang, L. & Ding, Z. 26S Proteasome: Hunter and Prey in Auxin Signaling. Trends Plant Sci 21, 546–548 (2016).
Vanneste, S. & Friml, J. Plant signaling: Deconstructing auxin sensing. Nat Chem Biol 8, 415–6 (2012).
Wright, R. C. & Nemhauser, J. L. New tangles in the auxin signaling web. F1000Prime Rep 7, 19 (2015).
Rademacher, E. H. et al. A cellular expression map of the Arabidopsis AUXIN RESPONSE FACTOR gene family. Plant J 68, 597–606 (2011).
Nanao, M.H. et al. Structural basis for oligomerization of auxin transcriptional regulators. Nat Commun 5, 3617 (2014).
Boer, D. R. et al. Structural basis for DNA binding specificity by the auxin-dependent ARF transcription factors. Cell 156, 577–89 (2014).
Korasick, D. A. et al. Molecular basis for AUXIN RESPONSE FACTOR protein interaction and the control of auxin response repression. Proc Natl Acad Sci USA 111, 5427–32 (2014).
Tiwari, S. B., Hagen, G. & Guilfoyle, T. The roles of auxin response factor domains in auxin-responsive transcription. Plant Cell 15, 533–543 (2003).
Ulmasov, T., Hagen, G. & Guilfoyle, T. J. Activation and repression of transcription by auxin-response factors. Proc Natl Acad Sci USA 96, 5844–9 (1999).
Li, S. B., Xie, Z. Z., Hu, C. G. & Zhang, J. Z. A Review of Auxin Response Factors (ARFs) in Plants. Front Plant Sci 7 (2016).
Ha, C. V. et al. The auxin response factor transcription factor family in soybean: genome-wide identification and expression analyses during development and water stress. DNA Res 20, 511–24 (2013).
Hagen, G. & Guilfoyle, T. Auxin-responsive gene expression: genes, promoters and regulatory factors. Plant Mol Biol 49, 373–85 (2002).
Liu, Y. et al. Genome-wide analysis of the auxin response factor (ARF) gene family in maize (Zea mays). Plant Growth Regul 63, 225–234 (2011).
Sun, R. R. et al. Genome-wide identification of auxin response factor (ARF) genes and its tissue-specific prominent expression in Gossypium raimondii. Funct Integr Genomics 15, 481–493 (2015).
Wang, D. K. et al. Genome-wide analysis of the auxin response factors (ARF) gene family in rice (Oryza sativa). Gene 394, 13–24 (2007).
Wu, J. et al. Identification, isolation and expression analysis of auxin response factor (ARF) genes in Solanum lycopersicum. Plant Cell Rep 30, 2059–2073 (2011).
Tantikanjana, T. & Nasrallah, J. B. Non-cell-autonomous regulation of crucifer self-incompatibility by Auxin Response Factor ARF3. Proc Natl Acad Sci USA 109, 19468–73 (2012).
Donner, T. J., Sherr, I. & Scarpella, E. Regulation of preprocambial cell state acquisition by auxin signaling in Arabidopsis leaves. Development 136, 3235–46 (2009).
Krogan, N. T., Ckurshumova, W., Marcos, D., Caragea, A. E. & Berleth, T. Deletion of MP/ARF5 domains III and IV reveals a requirement for Aux/IAA regulation in Arabidopsis leaf vascular patterning. New Phytol 194, 391–401 (2012).
Liu, N. et al. Down-regulation of AUXIN RESPONSE FACTORS 6 and 8 by microRNA 167 leads to floral development defects and female sterility in tomato. J Exp Bot 65, 2507–20 (2014).
Nagpal, P. et al. Auxin response factors ARF6 and ARF8 promote jasmonic acid production and flower maturation. Development 132, 4107–4118 (2005).
Attia, K. A. et al. Antisense phenotypes reveal a functional expression of OsARF1, an auxin response factor, in transgenic rice. Curr Issues Mol Biol 11(Suppl 1), i29–34 (2009).
Wang, S. et al. Auxin-related gene families in abiotic stress response in Sorghum bicolor. Funct Integr Genomics 10, 533–46 (2010).
Barrell, P. J., Meiyalaghan, S., Jacobs, J. M. & Conner, A. J. Applications of biotechnology and genomics in potato improvement. Plant Biotechnol J 11, 907–20 (2013).
Potato Genome Sequencing, C. et al. Genome sequence and analysis of the tuber crop potato. Nature 475, 189–95 (2011).
Chandler, J. W. Auxin response factors. Plant Cell Environ 39, 1014–28 (2016).
Guilfoyle, T. J. & Hagen, G. Auxin response factors. Curr Opin Plant Biol 10, 453–60 (2007).
Tiwari, S. B., Hagen, G. & Guilfoyle, T. J. Aux/IAA proteins contain a potent transcriptional repression domain. Plant Cell 16, 533–43 (2004).
Tiwari, S. B., Wang, X. J., Hagen, G. & Guilfoyle, T. J. AUX/IAA proteins are active repressors, and their stability and activity are modulated by auxin. Plant Cell 13, 2809–22 (2001).
Massa, A. N. et al. The transcriptome of the reference potato genome Solanum tuberosum Group Phureja clone DM1-3 516R44. PLoS One 6, e26801 (2011).
Quint, M. & Gray, W. M. Auxin signaling. Curr Opin Plant Biol 9, 448–53 (2006).
Remans, R., Spaepen, S. & Vanderleyden, J. Auxin signaling in plant defense. Science 313, 171 (2006).
Szklarczyk, D. et al. The STRING database in 2017: quality-controlled protein–protein association networks, made broadly accessible. Nucleic Acids Res 45, D362–D368 (2017).
Ma, Q., Grones, P. & Robert, S. Auxin signaling: a big question to be addressed by small molecules. J Exp Bot 69, 313–328 (2018).
Woodward, A. W. & Bartel, B. Auxin: regulation, action, and interaction. Ann Bot 95, 707–35 (2005).
Gandolfo, M., Nixon, K. & Crepet, W. A new fossil flower from the Turonian of New Jersey: Dressiantha bicarpellata gen. et sp. nov. (Capparales). Am J Bot 85, 964 (1998).
Ku, H. M., Vision, T., Liu, J. & Tanksley, S. D. Comparing sequenced segments of the tomato and Arabidopsis genomes: large-scale duplication followed by selective gene loss creates a network of synteny. Proc Natl Acad Sci USA 97, 9121–6 (2000).
Yang, Y. W., Lai, K. N., Tai, P. Y. & Li, W. H. Rates of nucleotide substitution in angiosperm mitochondrial DNA sequences and dates of divergence between Brassica and other angiosperm lineages. J Mol Evol 48, 597–604 (1999).
Sanan-Mishra, N., Varanasi, S. P. & Mukherjee, S. K. Micro-regulators of auxin action. Plant Cell Rep 32, 733–40 (2013).
Yamada, T. The role of auxin in plant-disease development. Annu Rev Phytopathol 31, 253–73 (1993).
Ding, X. et al. Activation of the Indole-3-Acetic Acid–Amido Synthetase GH3-8 Suppresses Expansin Expression and Promotes Salicylate- and Jasmonate-Independent Basal Immunity in Rice. Plant Cell 20, 228 (2008).
Savatin, D. V., Ferrari, S., Sicilia, F. & De Lorenzo, G. Oligogalacturonide-Auxin Antagonism Does Not Require Posttranscriptional Gene Silencing or Stabilization of Auxin Response Repressors in Arabidopsis. Plant Physiol 157, 1163 (2011).
Schmelz, E. A., Engelberth, J., Tumlinson, J. H., Block, A. & Alborn, H. T. The use of vapor phase extraction in metabolic profiling of phytohormones and other metabolites. Plant J 39, 790–808 (2004).
Bouzroud, S. et al. Auxin Response Factors (ARFs) are potential mediators of auxin action in tomato response to biotic and abiotic stress (Solanum lycopersicum). Plos One 13 (2018).
Jain, M. & Khurana, J. P. Transcript profiling reveals diverse roles of auxin-responsive genes during reproductive development and abiotic stress in rice. FEBS J 276, 3148–62 (2009).
Li, J. S., Dai, X. H. & Zhao, Y. D. A role for auxin response factor 19 in auxin and ethylene signaling in Arabidopsis. Plant Physiol 140, 899–908 (2006).
Hu, B. et al. GSDS 2.0: an upgraded gene feature visualization server. Bioinformatics 31, 1296–7 (2015).
Liu, W. et al. IBS: an illustrator for the presentation and visualization of biological sequences. Bioinformatics 31, 3359–61 (2015).
Livak, K. J. & Schmittgen, T. D. Analysis of Relative Gene Expression Data Using Real-Time Quantitative PCR and the 2−ΔΔCT Method. Methods 25, 402–408 (2001).
Gao, J. et al. Genome-wide survey of Aux/IAA gene family members in potato (Solanum tuberosum): Identification, expression analysis, and evaluation of their roles in tuber development. Biochem Biophys Res Commun 471, 320–7 (2016).
Acknowledgements
We thank the Prof. Guixian Xia (Institute of Microbiology, Chinese Academy of Sciences) for fruitful discussions on this manuscript. We are also grateful to Prof. Wanmin Sheng (Institute of Biotechnology, Heilongjiang Academy of Agricultural Sciences) for kindly provding potato cultivar “Shepody”. This project was supported by Science Transfer Services of Chinese Academy of Sciences (KFJ-FP-23 and KFJ-STS-ZDTP-027) to N.L., Key R&D Program of Ningxia Hui Autonmous Region (2018BBF02021) to N.Z., and National Natural Science Foundation of China (31601622) to Z.P.
Author information
Authors and Affiliations
Contributions
N.L. conceived and designed experiments; S.S., L.H., Y.X., Z.P. and N.L. conducted experiments; N.L. preformed data analysis and wrote the manuscript with inputs from N.Z. and H.Z. All authors read and approved the manuscript.
Corresponding authors
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Song, S., Hao, L., Zhao, P. et al. Genome-wide Identification, Expression Profiling and Evolutionary Analysis of Auxin Response Factor Gene Family in Potato (Solanum tuberosum Group Phureja). Sci Rep 9, 1755 (2019). https://doi.org/10.1038/s41598-018-37923-7
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-37923-7
This article is cited by
-
Comparative analysis of SIMILAR to RCD ONE (SRO) family from tetraploid cotton species and their diploid progenitors depict their significance in cotton growth and development
Journal of Cotton Research (2024)
-
Genomic survey and expression analysis of LcARFs reveal multiple functions to somatic embryogenesis in Liriodendron
BMC Plant Biology (2024)
-
Identification of ARF genes in Cucurbita pepo L and analysis of expression patterns, and functional analysis of CpARF22 under drought, salt stress
BMC Genomics (2024)
-
Identification and expression analyses of B3 genes reveal lineage-specific evolution and potential roles of REM genes in pepper
BMC Plant Biology (2024)
-
Transcriptome Analysis Reveals Molecular Signatures Associated with Apical Rooted Cutting (ARC) Technology in Seed Potato Production
Journal of Plant Growth Regulation (2024)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.