Abstract
Innate immune cells quickly infiltrate the site of pathogen entry and not only stave off infection but also initiate antigen presentation and promote adaptive immunity. The recruitment of innate leukocytes has been well studied in the context of extracellular bacterial and fungal infection but less during viral infections. We have recently shown that the understudied cytokine Interleukin (IL)-17D can mediate neutrophil, natural killer (NK) cell and monocyte infiltration in sterile inflammation and cancer. Herein, we show that early immune cell accumulation at the peritoneal site of infection by mouse cytomegalovirus (MCMV) is mediated by IL-17D. Mice deficient in IL-17D or the transcription factor Nuclear factor (erythroid-derived 2)-like 2 (Nrf2), an inducer of IL-17D, featured an early decreased number of innate immune cells at the point of viral entry and were more susceptible to MCMV infection. Interestingly, we were able to artificially induce innate leukocyte infiltration by applying the Nrf2 activator tert-butylhydroquinone (tBHQ), which rendered mice less susceptible to MCMV infection. Our results implicate the Nrf2/IL-17D axis as a sensor of viral infection and suggest therapeutic benefit in boosting this pathway to promote innate antiviral responses.
Similar content being viewed by others
Introduction
Cytomegaloviruses (CMVs) are herpesviruses that can cause fatal disease in their respective hosts. Infections of immune- suppressed patients with human cytomegalovirus (HCMV) still cause significant complications after transplantation, and congenital infection is the leading infectious cause of brain damage and sensorineural deafness in newborns1. Due to its structural and biological similarity to HCMV and its host specificity, mouse cytomegalovirus (MCMV) can be safely studied in mice to gain valuable in vivo insights about the mechanisms of CMV pathogenesis.
Immune responses to MCMV are well described and involve both early innate as well as later adaptive immunity. Indeed, roles for natural killer (NK) cells2, CD8+ T cells3, CD4+ T cells4, dendritic cells (DCs)5, monocytes/macrophages6 and neutrophils7 have been described for the resolution of MCMV infection (reviewed in8). A major role for controlling MCMV infection is attributed to a subtype of NK cells expressing the activating receptor Ly49H in C57BL/6 but not BALB/C mice9.
Although some of the anti-pathogenic functions of different immune subsets during MCMV infection are well described, less is known about their recruitment. It is established that infiltration of leukocytes to local sites of pathogen entry involves cytokine and chemokine production by resident or early-recruited cells. Chemokines shown to be induced after MCMV infection include the neutrophil-attractant macrophage inflammatory protein (MIP)-1α10, the T cell-attractants CXCL911 and CXCL1011,12 and the monocyte-, memory T cell-, neutrophil- and NK cell-attractant CCL213,14. CCL2 has been established as a central mediator for recruiting macrophages and NK cells to MCMV-infected sites14.
Our group has recently established a role for the cytokine Interleukin (IL)- 17D during cancer progression and sterile inflammation15,16. IL-17D is an understudied member of the IL-17 family of cytokines, which has known functions in antipathogenic responses and leukocyte infiltration (reviewed in17). Interestingly, we found that IL-17D induced the chemokine CCL2, leading to the recruitment of NK cells16. We further showed that IL-17D expression was regulated by the transcription factor nuclear factor (erythroid-derived 2)-like 2 (Nrf2), a known sensor of oxidative stress. Notably, activating Nrf2 with the agonist tert-butylhydroquinone (tBHQ) induced IL-17D in vitro and in vivo and led to NK cell-mediated tumor rejection in vivo15.
Immune responses to cancer and viral infections feature several similarities, as both involve NK cells, induction of similar cytokines and chemokines, and local recruitment of immune effector cells. We have previously shown that IL-17D-deficient mice displayed sub-optimal responses to infection by MCMV and local vaccinia virus (VV) scarification15. By using MCMV as a model system, we now analyzed the function of IL-17D and its regulator Nrf2 with specific regard to the immune response at the peritoneal site of virus entry.
Results and Discussion
IL-17D deficient mice are more susceptible to MCMV infection
We previously showed that mice lacking IL-17D are mildly more susceptible to MCMV infection15, as measured by weight loss. Corroborating these findings, Il17d−/− mice also featured a slightly worsened survival rate (Fig. 1a, p = 0.3) and a higher viral burden (Fig. 1b). We assessed viral burden using three different methods: 1) qPCR of the viral transcript Glycoprotein B (gB) of cDNA converted from total mRNA (Fig. 1b left, expressed as fold change compared to WT mice); 2) qPCR of the viral gene Immediate early 1 (IE1) performed on total cellular DNA with viral copy number calculated from a standard curve as read-out (Fig. 1b, middle, expressed as fold change compared to WT mice) and 3) viral plaque assays (Fig. 1b right, expressed as absolute plaque forming units (pfu)/mg organ). Both qPCR methods have been shown to be directly and quantitatively correlated to viral plaque assay measurements18,19, and this was reproduced in our hands. Because we found the data from qPCR of converted mRNA most reproducible, least error-prone and least susceptible to bias and because we were most interested in finding relative differences between transcribed virus gene in WT and Il17d−/− mice, we used this method for all subsequent analyses of viral burden. Corroborating our findings that Il-17d−/− mice feature a mildly more severe phenotype than WT after MCMV infection, viral burdens were significantly increased in some but not all tested organs. For all experiments shown, we used mice on a C57BL/6 background.
Il17d−/− mice are more susceptible to MCMV infection and feature reduced immune cell recruitment into infected peritoneum. (a) Survival of mock- and MCMV-infected WT and Il17D−/− mice. (b) Viral burden 5 days after infection was determined by qPCR of transcript of the viral gene gB (left), qPCR of DNA of the viral gene IE1 (middle) and viral plaque assays (right). gB gene expression is expressed as fold change relative to expression in MCMV-infected WT mice for each organ. The amount of viral copies is expressed as fold change compared to MCMV-infected WT mice for each organ. Viral plaques are expressed as plaque-forming units (pfu)/mg organ. (c), (d) Expression of Il17d and Ccl2 determined by qPCR 24 h after MCMV infection of peritoneal cells in vitro (c) or in vivo (d). Gene expression is expressed as fold change relative to gene expression in mock-infected cells (c) or mice (d). (e) Total numbers of NK cells (7AAD−/CD45+/CD3−/NK1.1+), neutrophils (7AAD−/CD45+/CD11b+/Ly6C+/Ly6Ghigh) and monocytes/macrophage precursors (7AAD−/CD45+/CD11b+/Ly6C+/Ly6Glow) in the peritoneal lavage of mock, heat- inactivated (HI) or live MCMV i.p. infected mice after 24 h. (a,b), left) Representative of three independent experiments with n = 5 or 6 mice per group. (b), middle) Combination of two independent experiments with n = 5 mice per group. (b), right) Representative of two independent experiments with n = 4 or 5 mice per group. (c) Combination of three independent experiments with n = 3-5 replicates per group. (d,e) Representative of three independent experiments with n = 5 mice in each group. Data are represented as mean ± SEM.
MCMV infection induces Il17d and Ccl2 expression within 24 hours at the site of infection
We previously found that MCMV infection induces Il17d expression in primary murine adult fibroblasts15 and therefore wanted to show in our i.p. infection model if peritoneal cells could express Il17d in response to MCMV infection. We first lavaged peritoneal cells from uninfected mice and exposed them to MCMV in vitro. We found that Il17d was significantly upregulated in MCMV-infected cells after 24 h of in vitro infection, compared to mock-infected cells (Fig. 1c). This upregulation correlated with the induction of Ccl2, a gene we showed is a target for IL-17D in tumor models16. To investigate whether Il17d is locally induced at the point of MCMV entry, we peritoneally infected mice with MCMV in vivo and analyzed the lavage after 24 h. Expression of Il17d and Ccl2 transcript (Fig. 1d) as well as CCL2 protein (Suppl Fig. S1b) was locally increased in the peritoneal lavage from MCMV-injected compared to mock-injected mice. IL-17D protein in the peritoneal lavage and supernatant from infected cells was below the detection limit of the ELISA (not shown). There was no difference between mock-infected or heat- inactivated (HI) MCMV-infected mice, suggesting that the observed induction was indeed live virus-induced. The MCMV-mediated expression of Ccl2 might depend on signals additional to IL-17D because Ccl2 was still upregulated in Il17d−/− animals, although not significantly. Because viral stocks of MCMV derived from infected salivary glands (SG- derived) might also contain cytokines that could, in turn, induce virus- independent cytokine activation, we also infected mice with MCMV produced and purified from cultured fibroblasts (tissue culture (TC)- derived). TC-derived MCMV also induced Il17d and Ccl2 transcript and CCL2 protein (Suppl Fig. S1a), indicating that the observed inductions were directly virus-mediated.
It is not obvious which cells express IL-17D in our chosen model. We and others have shown that it is expressed in non-immune cells under baseline and cancer-bearing conditions16,20. Since the major resident cell populations in the peritoneum are macrophages and B cells, comprising up to 90% of the cavity21 (Suppl Figs S3, S6), it is likely that these are the cells that induce IL-17D in this compartment. Macrophages and B cells are immune cells but might have homeostatic, and not effector, functions in the peritoneum due to their resident character. IL-17D could also be expressed in other resident or even newly-recruited cell types.
MCMV infection recruits innate immune cells into the peritoneum dependent on IL-17D
We next performed a detailed analysis of the major immune cell subtypes early in the peritoneum and later in the distal organs after MCMV i.p. infection in WT and Il17d−/− animals (gating strategy in Suppl Fig. S2). We found that early after infection (24 h), the only effector immune cells significantly differing between mock- and MCMV-infected mice were the innate subtypes (NK cells, neutrophils, monocytes; Fig. 1e), while the numbers of T cells (CD4+ and CD8+ T cells) were similar (not shown). Immune cell numbers did not differ between mock-infected and HI MCMV-infected mice, and similar numbers of innate immune cells were again also recruited by TC-derived virus (Suppl Fig. S1c). Interestingly, the numbers of NK cells, neutrophils and monocytes were decreased in Il17d−/− mice infected with MCMV. The results for monocytes are not significant, in line with our published data that recombinant IL-17D only moderately recruits monocytes16. We have previously immune-phenotyped Il17d−/− mice and did not observe baseline differences in immune cell populations compared to WT mice in bone marrow, lymph nodes and spleen15. We extended our analysis to peritoneum, spleen and lung and did not see differences in innate or adaptive immune cell numbers or maturation status of NK cells (Suppl Fig. S3). Thus, the differences in recruited cell numbers after MCMV infection are not due to baseline differences in immune cell composition or NK cell maturation of Il17d−/− mice.
It has been shown that the clearance of MCMV in C57BL/6 mice depends on a specific subtype of NK cells expressing the surface activating receptor Ly49H9. Thus, we analyzed whether MCMV i.p. infection preferably recruited this NK cell subtype early in WT mice, which might explain the increased susceptibility of Il17d−/− mice to MCMV infection. We did not find a difference in Ly49H+ NK cell numbers or activation status of all NK cells (reflected by the expression of CD69) between mock- and MCMV-infected or between WT and Il17d−/− mice at 24 h (Suppl Fig. S4a). We conclude that at this early timepoint, IL-17D does not preferentially recruit an activated subset of NK cells, but rather an increased number of NK and other innate immune cells.
Infiltration of immune cells into distal organs is not dependent on IL-17D during MCMV infection
Since MCMV i.p. infection also leads to systemic disease, we next tested the role of IL-17D in inflammatory responses to MCMV in peripheral organs. In contrast to the induction of Il17d in the peritoneum at 24 h, we did not observe an upregulation in the tested organs spleen, lung, heart, liver and kidney after 36 hours (not shown), three days (not shown) or five days (Suppl Fig. S5a) of MCMV infection. Based on these results, we considered it unlikely that there is an IL-17D-mediated difference in immune cell recruitment, but still analyzed it. We chose the organs spleen and lung because these are highly infected in the MCMV model22 and because we wanted to include one organ with low baseline IL-17D expression (spleen;20 and unpublished observations) and one with high baseline IL-17D expression (lung;20 and unpublished observations). Similar to MCMV infection after 24 h, neutrophils and monocytes were recruited to spleen and lung five days after infection in WT mice (Suppl Fig. S5b,c). In contrast to early MCMV infection, adaptive CD8+ T cells were now also recruited to the lung (Suppl Fig. S5d). Moreover, we observed an increase in Ly49H+ NK cells and CD69 expression on NK cells after MCMV infection (Suppl Fig. S4b,c). However, as expected from the results in Suppl Fig. S5a, there was no difference in immune cell recruitment or NK cell activation in MCMV-infected Il17d−/− compared to WT mice (Suppl Figs S4b,c, S5b–d). We conclude that early innate immune cell recruitment to peritoneal MCMV infection is mediated by IL-17D, while the recruitment of immune cells to systemically infected organs at later time points depends on mechanisms distinct from IL-17D. This might explain why the systemic infection severity of Il17d−/− mice is only slightly increased compared to WT mice (Fig. 1a,b). Additionally, we cannot exclude that the observed effect of IL-17D on immune cell recruitment is peritoneum-specific.
IL-17D expression is beneficial during MCMV infection in the absence of adaptive immunity
To examine the role of IL-17D in the absence of adaptive immunity, we crossed the Il17d−/− mice to Rag2−/− mice that lack adaptive immunity23 and infected double deficient progeny with MCMV. Similar to what has been published for Rag1−/− mice24, Rag2−/− mice were not able to recover from MCMV infection (Fig. 2a). Il17d/Rag2 double deficient mice were more susceptible to MCMV infection than Il17d or Rag2 single deficient mice, reflected by an earlier increase in weight loss, a decreased recovery (Fig. 2a) and a higher viral burden in most infected organs (Fig. 2b). We conclude that the early local recruitment of innate immune cells by IL-17D is enough to render mice slightly more resistant to MCMV infection, but that adaptive immunity is required for efficient systemic MCMV clearance at later time points.
Il17d/Rag2 double deficient mice are more susceptible to MCMV infection than Il17d or Rag2 single deficient mice. (a) Weight of MCMV-infected Il17d−/−, Rag2−/− and Il17d−/−/ Rag2−/− mice. (b) Viral burden as determined by qPCR of transcript of the viral gene gB in spleen, lung, heart, liver and kidney 5 days after infection, expressed relative to housekeeping gene. Data are combination of two independent experiments with n = 5 mice per group. Data are represented as mean ± SEM. Stars in (a) represent significant differences between Rag2−/− and Il17d−/−/Rag2−/− (days 2–4) and between Il17d−/− and Il17d−/−/Rag2−/− mice (days 7–11).
Nrf2- deficient mice are more susceptible to MCMV infection
We have previously shown that IL-17D expression is regulated by the transcription factor Nrf2 in cancer cells15. We now sought to explore the role of Nrf2 during MCMV infection and whether it plays a role for IL-17D induction. We therefore infected Nrf2−/− mice with MCMV. As hypothesized, Nrf2−/− mice were highly susceptible to MCMV infection as observed by a 23% decrease in weight, a worsened ability to recover (Fig. 3a), a significantly decreased survival rate (Fig. 3b), and an increase in viral burden of infected organs (Fig. 3c). Interestingly, the phenotype was more profound than that of Il17d−/− mice, suggesting that Nrf2 likely regulates other mediators of resistance to virus infection.
Nrf2−/− mice are more susceptible to MCMV infection.Weight (a) and survival (b) of MCMV-infected WT and Nrf2−/− mice. (c) Viral burden as determined by qPCR of transcript of the viral gene gB in spleen, lung, heart, liver and kidney 5 days after infection. gB transcript expression is expressed as fold change relative to expression in MCMV-infected WT mice for each organ.Representative of two independent experiments with n = 5 or 6 mice per group. Data are represented as mean ± SEM.
MCMV-induced Il17d expression depends on Nrf2
Since MCMV infection induced Il17d (Fig. 1c,d) and we have shown that Nrf2 regulates Il17d in cancer15, we next analyzed whether Nrf2 was induced after MCMV infection and whether Il17d expression depended on Nrf2. Indeed, we found Nrf2 to be upregulated 24 h after in vivo i.p. infection (Fig. 4a). Like shown in Fig. 1d, MCMV infection also induced Il17d and Ccl2 but Il17d upregulation was completely abolished in Nrf2−/− mice, indicating that MCMV-induced Il17d expression requires Nrf2. Similar to Il17d−/− mice (Fig. 1d), Ccl2 induction was modestly, but not significantly, induced in Nrf2−/− mice, suggesting that additional mechanisms control virus-induced Ccl2 expression.
MCMV infection induces Nrf2; and the recruitment of innate immune cells to the site of MCMV entry is reduced in Nrf2−/− mice. (a) Expression of Nrf2 (left), Il17d (middle) and Ccl2 (right) determined by qPCR 24 h after i.p. MCMV infection in WT and Nrf2−/− mice. Gene expression is expressed as fold change relative to expression in mock-infected mice. Combination of two independent experiments with n = 3 or 4 mice per group. (b) Total numbers of NK cells (7AAD−/CD45+/CD3−/NK1.1+), neutrophils (7AAD−/CD45+/CD11b+/Ly6C+/Ly6Ghigh) and monocytes/macrophage precursors (7AAD−/CD45+/CD11b+/Ly6C+/Ly6Glow) in the peritoneal lavage of mock or MCMV i.p. infected mice after 24 h. Representative of two independent experiments with n = 5 mice in each group.Data are represented as mean ± SEM.
MCMV infection recruits innate immune cells into the peritoneum dependent on Nrf2
We next examined immune cell recruitment 24 h after peritoneal MCMV infection in WT and Nrf2−/− animals. As already shown in Fig. 1e, MCMV infection increased the number of innate immune cells (NK cells, neutrophils, monocytes) compared to mock infection. Notably, Nrf2 deficiency reduced the numbers of these cells recruited to the MCMV-infected peritoneum, similar to Il17d deficiency (Fig. 4b).
To exclude that Nrf2−/− mice have baseline differences in the composition of their immune cell subtypes, we immune-phenotyped the peritoneum, spleen and lung of these mice (Suppl Fig. S6). To our knowledge, this has not been done before although anemia has been reported in Nrf2−/− mice25. The only significant differences observed were a decrease in macrophages in the peritoneum as well as a decrease of neutrophils in the lungs of Nrf2−/− mice. We conclude that Nrf2−/− mice have a slightly different composition of their peritoneum and lung-resident immune cells. We cannot exlude that these differences influence immune responses during MCMV infection. Further research is needed to clarify whether the reduced percentage of peritoneal macrophages in Nrf2−/− mice affects the recruitment of immune cells into this compartment due to induction of other cytokines. However, we consider it unlikely that it affects the IL-17D-mediated recruitment since IL-17D expression is absent in peritoneal cells from Nrf2−/− mice (Fig. 4a).
tBHQ protects from MCMV infection
We have recently published that topical application of the Nrf2 activator tBHQ on tumors induces IL-17D, which leads to NK cell recruitment and subsequent tumor rejection15. We next investigated if tBHQ could have a similar protective effect during viral infection. We found that injection of tBHQ protected WT mice from MCMV infection, resulting in significantly less weight loss and lower viral burden in most infected organs (Fig. 5a). Surprisingly, MCMV-infected Nrf2−/− mice treated with tBHQ were significantly less susceptible to infection than their DMSO-injected counterparts (Fig. 5b). However, tBHQ was not able to rescue from MCMV infection to a level as observed in WT mice. This suggests that tBHQ activates other protective pathways independent of Nrf2. We obtained similar results in Il17d−/− mice treated with tBHQ in that tBHQ still partly rescued the MCMV-induced weight loss and viral burden (Fig. 5c), although not significantly and not to WT levels.
tBHQ injection decreases susceptibility to MCMV.WT (a), Nrf2−/− (b) or Il17d−/− (c) mice were i.p. infected with MCMV and injected with 50 mM tBHQ/DMSO (or PBS/DMSO), and monitored daily for weight loss (left). Viral burden was determined by qPCR of the viral gene gB in spleen, lung, heart, liver and kidney (right) and expressed as fold change relative to expression in DMSO-injected mice.Representative of three (a,c) or two (b) independent experiments with n = 5 mice per group. Data are represented as mean ± SEM.
tBHQ locally induces Nrf2 activity, Il17d and Ccl2 expression and recruits innate immune cells
We sought to further analyze the mechanism of tBHQ-mediated protection against MCMV infection. We first tested whether Nrf2 is activated by tBHQ in this particular model of i.p. injection and whether it induces IL-17D. Indeed, we observed an upregulation of the Nrf2 target gene Hmox-1 in WT, but not Nrf2−/− mice (Fig. 6a). Moreover, tBHQ treatment induced Il17d in WT, but not Nrf2−/− mice, showing again that IL-17D expression is regulated by Nrf2. The immune cell-recruiting chemokine Ccl2 was also highly upregulated by tBHQ injection. In contrast to Hmox-1 and Il17d, however, Ccl2 was still induced in both Nrf2−/− and Il17d−/− animals, suggesting that tBHQ activates other pathways apart from Nrf2 and IL-17D. This is in contrast to its application in cancer where we have shown that it is not effective if Nrf2 or IL-17D are missing15. We next analyzed immune cell recruitment into the peritoneum after local tBHQ injection. Notably, tBHQ mobilized innate immune cell infiltration after 24 h (Fig. 6b). Interestingly, these cells were still recruited in Nrf2−/− mice, although not significantly and, in the case of monocytes, less profoundly. Likewise, neutrophils and monocytes still infiltrated in high numbers into the tBHQ-injected peritoneum even in the absence of IL-17D. Similar to the absence of Nrf2, the increase in number was not significant for neutrophils and monocytes and much lower for monocytes. NK cells were no longer recruited after tBHQ injection into the peritoneum of Il17d−/− mice. This shows that although other mechanisms apart from Nrf2 and IL-17D regulate neutrophil and, in part, monocyte recruitment after tBHQ injection, the recruitment of NK cells solely depends on IL-17D. We conclude that other mechanisms besides Nrf2 and IL-17D could regulate certain innate immune cell infiltration after tBHQ injection. Other studies have found that tBHQ can also activate Akt26 and its downstream target endothelial nitric oxide synthase (eNOS)27. It is possible that tBHQ acts through one of these or another yet unknown pathway in our system. It remains open how NK cell recruitment in Il17d−/− mice is not induced although Ccl2 is still upregulated. We speculate that IL-17D induces other NK cell-recruiting chemokines after tBHQ activation. Either way, tBHQ bears potential to serve as immune therapy or prophylaxis not only for cancer15, but also for viral infection.
tBHQ injection induces Hmox-1, Il17d and Ccl2 expression and the recruitment of innate immune cells. (a) Expression of Nrf2 target gene Hmox-1 (left), Il17d (middle) and Ccl2 (right) determined by qPCR 24 h after i.p. injection of 50 mM tBHQ/DMSO (or PBS/DMSO) in WT, Nrf2−/− or Il17d−/− mice. Gene expression is expressed as fold change relative to expression in DMSO-injected mice. Combination of three (two for Nrf2−/−) independent experiments with n = 5 or 6 (n = 3 or 4 for Nrf2−/−) mice per group. (b) Numbers of NK cells (7AAD−/CD45+/CD3−/NK1.1+) (left), neutrophils (7AAD− /CD45+/ CD11b+/ Ly6C+/ Ly6Ghigh) (middle) and monocytes/macrophage precursors (7AAD−/CD45+/CD11b+/Ly6C+/Ly6Glow) (right) in the peritoneal lavage of PBS/DMSO or tBHQ/DMSO i.p. injected WT, Nrf2−/− or Il17d−/− mice after 24 h. Representative of three (two for Nrf2−/−) independent experiments with n = 5 or 6 (n = 3 or 4 for Nrf2−/−) mice per group.Data are represented as mean ± SEM.
Poly (dA:dT) induces Il17d dependent on Nrf2
Infections are usually sensed by Toll-like receptors (TLRs) that bind to extracellular or intracellular pathogen-associated molecular patterns (PAMPs). For MCMV, previous studies have found a role for TLR3 and TLR9 in mediating responses28. Therefore, we examined whether agonists for TLR3 or TLR9 were sufficient to induce Il17d in uninfected cells. We did not observe an upregulation of Il17d expression after stimulation with the TLR3 ligand poly inosine-cytidylic acid (I:C) or the TLR9 ligand CpG oligodeoxynucleotides (ODN) (Fig. 7a–c). We also did not detect induction of Il17d by intracellular delivery of poly (I:C), which is sensed by the pattern recognition receptor Retinoic acid-inducible gene (RIG)-I/ Melanoma- differentiation- associated gene (MDA)-529 (Fig. 7a,b). We next investigated whether the MCMV-induced upregulation of Il17d might go through cytosolic DNA sensors such as the Absent in melanoma (AIM)2 inflammasome, recently shown to be important for MCMV infection30, or the cyclic GMP-AMP synthase (cGAS)/Stimulator of Interferon genes (STING) pathway, recently shown to be important for HCMV infection31. Indeed, the stimulator of these pathways poly deoxyadenylic-deoxythymidylic (dA:dT) did induce Il17d in in vitro stimulated peritoneal cells (Fig. 7d). This induction was prevented in Nrf2−/− mice. To confirm these data in a different in vitro system, we made use of previously in-house created B16 melanoma cell lines that feature an shRNA-mediated stable knockdown of Nrf2 expression15. In B16 sh_ctrl cell lines, Il17d was induced with different concentrations of poly (dA:dT), which was prevented when Nrf2 was knocked down (Fig. 7d). To sum up, Il17d was induced by poly (dA:dT) stimulation and this depended on Nrf2 in different kinds of cells. We suggest that the AIM2 inflammasome or the cGAS/STING pathway might be the MCMV-sensing complexes inducing the Nrf2/IL-17D pathway in our system. Nrf2 has already been shown to be required for AIM2 inflammasome activation32. More research is needed to clarify the role of inflammasomes for IL-17D biology.
Poly (dA:dT), but not poly (I:C) or ODNs, induces Il17d expression dependent on Nrf2. (a–c) Expression of Il17d in freshly isolated peritoneal cells determined by qPCR 24 h after intracellular (left) or extracellular (right) stimulation with poly (I:C) HMW (a), LMW (b) or different kinds of ODNs (c). Gene expression is expressed as fold change relative to expression in vehicle stimulated controls. (d) Expression of Il17d in freshly isolated WT or Nrf2−/− peritoneal cells (left) or B16 melanoma cells bearing a stable Nrf2 knockdown (right) determined by qPCR 24 h after stimulation with poly (dA:dT).Gene expression is expressed as fold change relative to expression in vehicle stimulated or sh_ctrl cells.Representative of three (two for Nrf2−/−) independent experiments with n = 3 replicates per group. Data are represented as mean ± SEM. HMW = high molecular weight, LMW = low molecular weight, ODN = oligodeoxynucleotide.
To sum up, we here document a role for IL-17D and Nrf2 in effective immune cell recruitment to peritoneal viral infection. We have previously established an important function for IL-17D and its regulator Nrf2 during cancer immune surveillance and even proposed the Nrf2/IL-17D/innate immunity pathway as a target for cancer immune therapy15. Here, we show in more detail that the same pathway is also involved in optimal immunity against viral infection. Mice deficient in IL-17D or Nrf2 featured higher susceptibility to MCMV infection likely caused by a defective ability to recruit innate immune cells to the site of viral entry. Although MCMV has been shown to be able to travel freely after i.p. infection22, we suggest that an innate peritoneal immune response could limit infection by eliminating free virus or lysing the cells that are peritoneally infected.
The ancient family of IL-17 cytokines has been described as important for anti-pathogen defense17,33,34. The most studied members IL-17A and F, which are expressed by Th17 cells, have been implicated in host immune responses against bacteria and fungi by recruitment of neutrophils16,35,36,37. IL-17C is expressed by epithelial cells and activates the autocrine production of antibacterial peptides and pro-inflammatory molecules, mediating immunity to extracellular pathogens, similar to IL-17A38,39. The roles for IL-17B and E are less clear but IL-17E has been studied in anti-helminth immunity and recruitment of eosinophils40. Less is known about IL-17 involvement in virus infections. Both pro- and anti-viral roles for IL-17A/F have been suggested for a number of viruses, including VV41,42, influenza43,44, Hepatitis B virus (HBV) (reviewed in45,46), Hepatitis C virus (HCV) (reviewed in47), Dengue virus (reviewed in48), respiratory syncytial virus (RSV) (reviewed in49), and Theiler’s murine encephalomyelitis virus (TMEV)50,51,52. Most of these publications assign the function of IL-17 to its expression in adaptive Th17 cells and subsequent induction of cytokine secretion, while only few include a leukocyte recruitment phenotype. To our knowledge, IL-17B, C or E have not been implicated in viral infections thus far. Our studies now solidify a role for IL-17D in antiviral responses. Given the ancient role of other IL-17 family members during infection and IL-17D’s function during local innate immune responses against viruses, we propose that it evolved early to mediate challenges caused by intracellular pathogens, maybe even before the evolution of adaptive immunity. Indeed, the finding that Il17d/Rag2 double deficient mice are even more susceptible to MCMV than single deficient mice documents a role for IL-17D in the absence of adaptive immunity, perhaps as an early local sentinel of infection.
Nrf2 is known to be required for effective antitumor responses, but paradoxically has both pro- and anti-cancer functions due to its induction of a host-protective antioxidant program (reviewed in53). Similarly, both promoting and inhibitory roles have been reported for Nrf2 in inflammatory responses and bacterial infections54,55,56,57. Research on Nrf2 in virus infections is emerging, suggesting an involvement of Nrf2 in a number of in vitro viral infections, e.g. HBV58, HCV59,60, influenza61, Dengue virus62 and HIV63,64, mainly attributed to its antioxidant activities. Some papers also use in vivo infections of RSV65 and influenza66 to show that Nrf2−/− mice are more susceptible. To our knowledge, no study has observed increased mortality of Nrf2−/− mice in response to viral infection, although one study demonstrated decreased survival after influenza infection, but only after the additional stimulus of cigarette smoke66. No study thus far has analyzed Nrf2 during MCMV infection although one paper showed that Nrf2 was upregulated after in vitro HCMV infection67, suggesting a role for Nrf2 during CMV infections. Here, we show that Nrf2−/− mice are highly susceptible to MCMV infection and we propose that this is in part due to a reduced recruitment of innate immune cells. We do not exclude that Nrf2’s antioxidant functions contribute to resistance to MCMV in our model but we suggest that the infiltration of innate leukocytes accounts for part of the phenotype. Since we have shown that Nrf2 regulates IL-17D15, we suggest that Nrf2 mediates the recruitment of immune cells by inducing IL-17D. However, additional mechanisms likely contribute to the reduced resistance to MCMV because Nrf2−/− mice were far more susceptible to infection than Il17d−/− mice. We do demonstrate, though, that IL-17D induction solely goes through Nrf2 because it was completely abrogated in Nrf2−/− mice after MCMV infection.
Conclusions
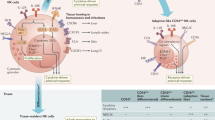
In conclusion, we present a heretofore-unknown function of Nrf2 and IL-17D during MCMV infection. Deficiency in either protein reduced early infiltration of innate leukocytes into the peritoneum (Fig. 8), leading to slightly increased disease progression. Moreover, activating Nrf2 using an agonist led to protective effects. We suggest that application of the Nrf2 activator tBHQ could serve as a viral immune therapy or prevention through IL-17D-mediated recruitment of NK and other innate immune effector cells. Researching molecular mechanisms of immune cell recruitment in the well-established experimental model of MCMV infection might also produce generalizable data that could be applied to other latent herpesvirus infections.
Nrf2 and IL-17D mediate recruitment of innate immune cells to the peritoneum after MCMV infection. Nrf2, IL-17D and CCL2 are locally induced after MCMV infection, resulting in innate immune cell recruitment into the peritoneum and partial viral clearance. Artificially activating Nrf2 with tBHQ has similar effects and could be used as antiviral immune therapy.
Materials and Methods
All methods were performed in accordance with the relevant guidelines and regulations.
Cell Lines
B16F10 melanoma and 3T3 cells were cultured in complete RPMI 1640 (Gibco) supplemented with 10% FCS (Atlanta Biologics). shRNA mediated knockdown of Nrf2 in B16 cells was achieved as previously described15.
Antibodies
Directly fluorophore-coupled antibodies used for flow cytometry in this work were: α-B220 (clone RA3-6B2), α-CD3 (clone 17 A.2), α-CD4 (clone GK1.5), α-CD8a (clone 53-6.7), α-CD11b (clone M1/70), α-CD45 (clone 30-F11), α-CD69 (H1.2F3), α-F4/80 (clone BM8), α-Ly6C (clone HK1.4), α-Ly6G (clone 1A8), α-Ly6H (clone 3D10), α-NK1.1 (PK136), and their respective isotype control antibodies (all from BioLegend).
Mice
All mouse experiments were approved by the UCSD Institutional Animal Care and Use Committee (IACUC protocol #S06201) and were performed in accordance with the relevant guidelines and regulations. In-house bred mice of C57BL/6, BALB/C, Rag2tm1.1Cgn (Rag2−/−), Nfe2l2tm1Ywk/J (Nrf2−/−) (in-house backcrossed to C57BL/6) (all from Jackson) and Il17dtm1Lex/Mmucd (Il17d−/−) (in-house backcrossed to C57BL/6) (UC Davis MMRC) and Rag2−/−/Il17d−/− double deficient mice (in-house crossed) were used in this work. All viral infection experiments shown in the figures were performed with mice on a C57BL/6 background. Salivary gland (SG) virus preparations were derived from BALB/C mice.
Viral Preparations
MCMV Smith Strain was kindly donated by Dr. Elina Zuniga (UCSD) and viral stocks were prepared from SG homogenates of BALB/C mice intraperitoneally (i.p.) infected with 1 × 104 pfu/mouse. Homogenates were prepared in sterile PBS, centrifuged at 1800g for 10 min and supernatant was used for subsequent experiments. MCMV Smith ‘tissue culture’ (TC)- derived stocks were derived from the BAC-cloned strain containing a wild-type viral MCK-2 gene68. The Smith BAC was first electroporated into 3T3 cells, replication was allowed to proceed until 100% cytopathogenic effect (cpe) was achieved and viral supernatants were harvested. Supernatants were then passaged 4 additional times, infecting at an MOI of ~0.1 and letting infection proceed to 100% cpe, allowing for excision of the BAC cassette from the replicating viral genome69. This was done 3 independent times, and comparisons of resulting virus showed similar genomic RFLP patterns, undetectable BAC-cassette sequences and similar replication levels in C57BL/6 and Balb/c mice at day 4 in spleen and liver and day 12 in salivary glands. One of these stocks was chosen as a ‘1° stock’, which was then passaged twice more in 3T3 cells to generate a ‘seed-stock’ and then a ‘working stock’, which was used for the described experiments. Viral titers were determined in vitro by plaque assays on 3T3 cells as described28. For some experiments, SG virus was heat- inactivated by incubation at 56 °C for 30 min.
Viral Infections
In vitro, freshly lavaged peritoneal cells were infected with 1 × 105 pfu/4 × 105 cells for 2 h, washed and lysed in PureLinkTM RNA mini kit (Ambion) lysis buffer after 24 h.
In vivo, age and sex-matched eight to twelve week old mice were infected with 3 × 105 pfu/mouse i.p., weighed and monitored daily for disease progression. For some experiments, mice were infected with 1 × 106 pfu/mouse of TC- derived MCMV. Immune cell recruitment into the peritoneum was determined 24 h after infection by lavaging the peritoneal cavity twice with 10 ml of PBS/3 mM EDTA, counting the total number of cells in the lavage and subsequent flow cytometer analysis70. Spleen and lung were processed 5 days after infection by mincing and subsequent collagenase (Sigma-Aldrich) digestion (only for lung), straining through a 70 μm filter followed by erythrocyte lysis with red blood cell lysis buffer (BioLegend). Viral burden in spleen, lung, liver, heart and kidney homogenates was determined by plaque assays on 3T3 cells as described28 or by qPCR analysis of the MCMV gene transcript Glycoprotein B (gB), determining the expression of transcribed virus RNA relevant to housekeeping RNA, directly correlating to disease severity and plaque assays. For absolute quantification of viral DNA copies, we performed qPCR of the MCMV Immediate early 1 (IE1) gene and calculated the number of target gene copies using a standard curve (see below under “Quantitative PCR”). Both qPCR methods are directly and quantitatively correlated to viral plaque assay measurements18,19. For some experiments, mice were i.p. injected with 50 mM tBHQ in PBS:DMSO (4:1).
FACS analysis
Single cell suspensions from peritoneal lavage, spleen and lung were incubated with 1 μg/100 μl of αCD16/CD32 (clone 2.4G2, BD Biosciences) FC-receptor block before staining the cells with 1 μg/100 μl of the respective antibodies. Samples were analyzed on a FACS CANTO II (BD Biosciences). We defined 7-aminoactinomycin D (7AAD)−/CD45+ immune subtypes in the peritoneal lavage, spleen and lung as follows: CD4+ T cells (CD3+/CD4+), CD8+ T cells (CD3+/CD8+), B cells (B220+), macrophages (F4/80+), NK cells (CD3−/NK1.1+), neutrophils (CD11b+/Ly6C+/Ly6Ghigh) and monocytes/macrophage precursors (CD11b+/Ly6C+/Ly6Glow) (gating strategy in Suppl Fig. S2).
Quantitative PCR
Whole tissues were either directly used or frozen at −80 °C in Trizol (Ambion) for no longer than one week. RNA was isolated according to the manufacturer’s manual without modifications. RNA from macrophages or peritoneal lavage cells was directly isolated using the PureLinkTM RNA mini kit (Ambion) according to the manufacturer’s manual without modifications. Concentrations and A260/280 absorbances were measured with NanoDrop 2000 Spectrophotometer (Thermo Scientific). Samples with A260/280 ~2.0 were considered pure RNA and used for cDNA generation. 2 μg of RNA was converted into cDNA with the High-Capacity cDNA Reverse Transcription Kit (Thermo Fisher Scientific, product ID 4368814) according to the manufacturer’s manual and concentrations and A260/280 were measured with NanoDrop, typically yielding DNA with A260/280 of 1.8. qPCR reactions were prepared with SYBR Green PCR Master Mix (Thermo Fisher Scientific, product ID 4309155), which includes AmpliTaq Gold DNA Polymerase, according to the manufacturer’s manual with 2 μM each of forward and reverse primers and performed on a Bio-Rad CFX96 machine. Each sample was run in triplicates and the reaction conditions were as follows: initial denaturation at 95 °C for 10 minutes followed by 95 °C for 10 seconds and 60 °C for 1 min for 40 cycles. The no template control (NTC) gave CT values of >40 for each experiment. Hprt was used as the housekeeping reference gene because it did not produce significant variations in CT values in any of the tested organs or conditions. Relative quantification of gene expression was performed using the 2−ΔΔCT method71, where gene expression was expressed as fold change relative to gene expression of the respective control relative to housekeeping reference in each experiment. The control for each experiment is indicated in the figure legends. We used this method to determine inductions of Il17d, Ccl2, Nrf2 and Hmox-1 as well as to detect transcribed MCMV gB relative to housekeeping gene. For absolute quantification of MCMV target gene copies in infected tissues, tissue was homogenized in PBS and frozen at −80 °C for no longer than one week. DNA was isolated with the QIAamp® DNA Mini kit (Qiagen) according to the manufacturer’s manual without modifications. qPCR of the MCMV gene IE1 was performed in triplicates as described in detail in19,72. This method determines absolute target gene copy numbers by relating the qPCR signal to a standard curve generated from plotting average CT values against the logarithm of target template molecules obtained from a control plasmid, followed by a sum of least squares regression analysis19. The following primer sequences were used: Ccl2 (fw- TAAAAACCTGGATCGGAACCAAA; rv- GCATTAGCTTCAGATTTACGGGT), gB (fw- GAAGATCCGCATGTCCTTCAG; rv- AATCCGTCCAACATCTTGTCG), Hmox-1 (fw-TGAAGGAGGCCACCAAGGAGG; rv- AGAGGTCACCCAGGTAGCGGG), Hprt (fw- GCTTGCTGGTGAAAAGGACCTCTCGAAG; rv- CCCTGAAGTACTCATTATAGTCAAGGGCAT), IE1 (fw- TCGCCCATCGTTTCGAGA; rv- TCTCGTAGGTCCACTGACCGA), Il17d (fw- AGCTTGTCCATGCTGGAGTT; rv- CTCTACGGGGAGGAGGACTT), and Nrf2 (fw CAGTCTTCACTGCCCCTCAT; rv- TCTGTCAGTGTGGCTTCTGG). Primers for Ccl2 (NM_011333.3,73), gB (NC_004065,74), Hmox-1 (NM_010442,75), Hprt (NM_013556,76), IE1 (GenBank L07320.1,72) and Nrf2 (NM_010902.4,77) were previously used and validated in other publications and additionally BLASTed for target specificity before use. Primers for gB were additionally validated by giving strong signals in MCMV-infected tissues, but no signals in mock-infected tissues. Additionally, the 2−ΔCT value relative to housekeeping gene directly correlated with disease severity in MCMV-infected animals. Primers for Il17d (NM_145837.3) were previously designed by our group15 and validated by producing the correctly sized product in an agarose gel, giving robust signals in tissues from WT mice, but giving no detectable signal in tissues from Il17d−/− mice. Additionally, we tested that these primers give reliably high signals in IL-17D-overexpressing cells, but reliably low signals in cells known to express low levels of IL-17D16.
Elisa
Enzyme-linked immunosorbent assay (ELISA) for CCL2 was performed according to the manufacturer’s manual (R&D Systems). ELISA for IL-17D was performed using the same protocol with rat-α-mIL-17D (R&D Systems, clone #312724) as capture antibody at 200 ng/ml and biotinylated goat α-mIL-17D (R&D Systems, immunogen accession #Q8K4C4) as detection antibody at 50 ng/ml.
Poly (I:C), poly (dA:dT) and oligodeoxynucleotide stimulations
Freshly lavaged peritoneal cells were stimulated with naked poly (I:C) or ODNs 1585, 1826, or 2395 (all invivogen) for 24 h and lysed in RNA lysing buffer (Ambion). For intracellular stimulation, cells were transfected with complexes between poly (I:C) or poly (dA:dT) (invivogen) and the transfection reagent LyoVecTM according to the manufacturer’s manual.
Statistical Analysis
Statistical significance was determined by the Mann-Whitney test with the Prism 6.0 software (GraphPad Software, Inc.). For survival curves, statistical significance was determined by the Log-rank (Mantel-Cox) test. Error bars are depicted using SEM. All experiments were repeated at least twice. *P < 0.05, **P < 0.01, ***P < 0.001, or no asterisks for not significant, in all data shown.
Data Availability
All data generated or analyzed during this study are included in this published article (and its Supplementary Information files). If additional details are desired, they are available from the corresponding author on request.
References
Boeckh, M. & Geballe, A. P. Cytomegalovirus: pathogen, paradigm, and puzzle. J Clin Invest 121, 1673–1680, https://doi.org/10.1172/JCI45449 (2011).
Bancroft, G. J., Shellam, G. R. & Chalmer, J. E. Genetic influences on the augmentation of natural killer (NK) cells during murine cytomegalovirus infection: correlation with patterns of resistance. J Immunol 126, 988–994 (1981).
Reddehase, M. J., Mutter, W., Munch, K., Buhring, H. J. & Koszinowski, U. H. CD8-positive T lymphocytes specific for murine cytomegalovirus immediate-early antigens mediate protective immunity. J Virol 61, 3102–3108 (1987).
Jonjic, S., Mutter, W., Weiland, F., Reddehase, M. J. & Koszinowski, U. H. Site-restricted persistent cytomegalovirus infection after selective long-term depletion of CD4 + T lymphocytes. J Exp Med 169, 1199–1212 (1989).
Andoniou, C. E. et al. Interaction between conventional dendritic cells and natural killer cells is integral to the activation of effective antiviral immunity. Nat Immunol 6, 1011–1019, https://doi.org/10.1038/ni1244 (2005).
Crane, M. J., Hokeness-Antonelli, K. L. & Salazar-Mather, T. P. Regulation of inflammatory monocyte/macrophage recruitment from the bone marrow during murine cytomegalovirus infection: role for type I interferons in localized induction of CCR2 ligands. J Immunol 183, 2810–2817, https://doi.org/10.4049/jimmunol.0900205 (2009).
Stacey, M. A. et al. Neutrophils recruited by IL-22 in peripheral tissues function as TRAIL-dependent antiviral effectors against MCMV. Cell Host Microbe 15, 471–483, https://doi.org/10.1016/j.chom.2014.03.003 (2014).
Masha, F.-H. Immune Responses to CMV and Vaccine Development. (2015).
Daniels, K. A. et al. Murine cytomegalovirus is regulated by a discrete subset of natural killer cells reactive with monoclonal antibody to Ly49H. J Exp Med 194, 29–44 (2001).
Salazar-Mather, T. P., Lewis, C. A. & Biron, C. A. Type I interferons regulate inflammatory cell trafficking and macrophage inflammatory protein 1alpha delivery to the liver. J Clin Invest 110, 321–330, https://doi.org/10.1172/JCI15376 (2002).
Hokeness, K. L. et al. CXCR3-dependent recruitment of antigen-specific T lymphocytes to the liver during murine cytomegalovirus infection. J Virol 81, 1241–1250, https://doi.org/10.1128/JVI.01937-06 (2007).
Cheeran, M. C. et al. Intracerebral infection with murine cytomegalovirus induces CXCL10 and is restricted by adoptive transfer of splenocytes. J Neurovirol 10, 152–162, https://doi.org/10.1080/13550280490441130 (2004).
Deshmane, S. L., Kremlev, S., Amini, S. & Sawaya, B. E. Monocyte chemoattractant protein-1 (MCP-1): an overview. J Interferon Cytokine Res 29, 313–326, https://doi.org/10.1089/jir.2008.0027 (2009).
Hokeness, K. L., Kuziel, W. A., Biron, C. A. & Salazar-Mather, T. P. Monocyte chemoattractant protein-1 and CCR2 interactions are required for IFN-alpha/beta-induced inflammatory responses and antiviral defense in liver. J Immunol 174, 1549–1556 (2005).
Saddawi-Konefka, R. et al. Nrf2 Induces IL-17D to Mediate Tumor and Virus Surveillance. Cell Rep 16, 2348–2358, https://doi.org/10.1016/j.celrep.2016.07.075 (2016).
O’Sullivan, T. et al. Interleukin-17D mediates tumor rejection through recruitment of natural killer cells. Cell Rep 7, 989–998, https://doi.org/10.1016/j.celrep.2014.03.073 (2014).
Iwakura, Y., Ishigame, H., Saijo, S. & Nakae, S. Functional specialization of interleukin-17 family members. Immunity 34, 149–162, https://doi.org/10.1016/j.immuni.2011.02.012 (2011).
Kawasaki, H., Kosugi, I., Arai, Y., Iwashita, T. & Tsutsui, Y. Mouse embryonic stem cells inhibit murine cytomegalovirus infection through a multi-step process. PLoS One 6, e17492, https://doi.org/10.1371/journal.pone.0017492 (2011).
Wheat, R. L., Clark, P. Y. & Brown, M. G. Quantitative measurement of infectious murine cytomegalovirus genomes in real-time PCR. J Virol Methods 112, 107–113 (2003).
Starnes, T., Broxmeyer, H. E., Robertson, M. J. & Hromas, R. Cutting edge: IL-17D, a novel member of the IL-17 family, stimulates cytokine production and inhibits hemopoiesis. J Immunol 169, 642–646 (2002).
Ray, A. & Dittel, B. N. Isolation of mouse peritoneal cavity cells. J Vis Exp. https://doi.org/10.3791/1488 (2010).
Hsu, K. M., Pratt, J. R., Akers, W. J., Achilefu, S. I. & Yokoyama, W. M. Murine cytomegalovirus displays selective infection of cells within hours after systemic administration. J Gen Virol 90, 33–43, https://doi.org/10.1099/vir.0.006668-0 (2009).
Shinkai, Y. et al. RAG-2-deficient mice lack mature lymphocytes owing to inability to initiate V(D)J rearrangement. Cell 68, 855–867 (1992).
Sell, S. et al. Control of murine cytomegalovirus infection by gammadelta T cells. PLoS Pathog 11, e1004481, https://doi.org/10.1371/journal.ppat.1004481 (2015).
Lee, J. M., Chan, K., Kan, Y. W. & Johnson, J. A. Targeted disruption of Nrf2 causes regenerative immune-mediated hemolytic anemia. Proc Natl Acad Sci USA 101, 9751–9756, https://doi.org/10.1073/pnas.0403620101 (2004).
Zhang, Y. et al. The antioxidant compound tert-butylhydroquinone activates Akt in myocardium, suppresses apoptosis and ameliorates pressure overload-induced cardiac dysfunction. Sci Rep 5, 13005, https://doi.org/10.1038/srep13005 (2015).
Xu, B. C., Long, H. B. & Luo, K. Q. Tert-butylhydroquinone lowers blood pressure in AngII-induced hypertension in mice via proteasome-PTEN-Akt-eNOS pathway. Sci Rep 6, 29589, https://doi.org/10.1038/srep29589 (2016).
Tabeta, K. et al. Toll-like receptors 9 and 3 as essential components of innate immune defense against mouse cytomegalovirus infection. Proc Natl Acad Sci USA 101, 3516–3521, https://doi.org/10.1073/pnas.0400525101 (2004).
Kato, H. et al. Differential roles of MDA5 and RIG-I helicases in the recognition of RNA viruses. Nature 441, 101–105, https://doi.org/10.1038/nature04734 (2006).
Rathinam, V. A. et al. The AIM2 inflammasome is essential for host defense against cytosolic bacteria and DNA viruses. Nat Immunol 11, 395–402, https://doi.org/10.1038/ni.1864 (2010).
Lio, C. W. et al. cGAS-STING Signaling Regulates Initial Innate Control of Cytomegalovirus Infection. J Virol 90, 7789–7797, https://doi.org/10.1128/JVI.01040-16 (2016).
Zhao, C., Gillette, D. D., Li, X., Zhang, Z. & Wen, H. Nuclear factor E2-related factor-2 (Nrf2) is required for NLRP3 and AIM2 inflammasome activation. J Biol Chem 289, 17020–17029, https://doi.org/10.1074/jbc.M114.563114 (2014).
Gaffen, S. L. Recent advances in the IL-17 cytokine family. Curr Opin Immunol 23, 613–619, https://doi.org/10.1016/j.coi.2011.07.006 (2011).
Seelige, R., Washington, A. Jr. & Bui, J. D. The ancient cytokine IL-17D is regulated by Nrf2 and mediates tumor and virus surveillance. Cytokine 91, 10–12, https://doi.org/10.1016/j.cyto.2016.11.017 (2016).
Joshi, A. D. et al. Interleukin-17-mediated immunopathogenesis in experimental hypersensitivity pneumonitis. Am J Respir Crit Care Med 179, 705–716, https://doi.org/10.1164/rccm.200811-1700OC (2009).
Ye, P. et al. Requirement of interleukin 17 receptor signaling for lung CXC chemokine and granulocyte colony-stimulating factor expression, neutrophil recruitment, and host defense. J Exp Med 194, 519–527 (2001).
Gladiator, A., Wangler, N., Trautwein-Weidner, K. & LeibundGut-Landmann, S. Cutting edge: IL-17-secreting innate lymphoid cells are essential for host defense against fungal infection. J Immunol 190, 521–525, https://doi.org/10.4049/jimmunol.1202924 (2013).
Ramirez-Carrozzi, V. et al. IL-17C regulates the innate immune function of epithelial cells in an autocrine manner. Nat Immunol 12, 1159–1166, https://doi.org/10.1038/ni.2156 (2011).
Song, X. et al. IL-17RE is the functional receptor for IL-17C and mediates mucosal immunity to infection with intestinal pathogens. Nat Immunol 12, 1151–1158, https://doi.org/10.1038/ni.2155 (2011).
Owyang, A. M. et al. Interleukin 25 regulates type 2 cytokine-dependent immunity and limits chronic inflammation in the gastrointestinal tract. J Exp Med 203, 843–849, https://doi.org/10.1084/jem.20051496 (2006).
Kohyama, S. et al. IL-23 enhances host defense against vaccinia virus infection via a mechanism partly involving IL-17. J Immunol 179, 3917–3925 (2007).
Patera, A. C., Pesnicak, L., Bertin, J. & Cohen, J. I. Interleukin 17 modulates the immune response to vaccinia virus infection. Virology 299, 56–63 (2002).
Wang, X. et al. IL-17A Promotes Pulmonary B-1a Cell Differentiation via Induction of Blimp-1 Expression during Influenza Virus Infection. PLoS Pathog 12, e1005367, https://doi.org/10.1371/journal.ppat.1005367 (2016).
Crowe, C. R. et al. Critical role of IL-17RA in immunopathology of influenza infection. J Immunol 183, 5301–5310, https://doi.org/10.4049/jimmunol.0900995 (2009).
Huang, Z., van Velkinburgh, J. C., Ni, B. & Wu, Y. Pivotal roles of the interleukin-23/T helper 17 cell axis in hepatitis B. Liver Int 32, 894–901, https://doi.org/10.1111/j.1478-3231.2012.02764.x (2012).
Arababadi, M. K., Bidaki, M. Z. & Kennedy, D. IL-17A in hepatitis B infection: friend or foe? Arch Virol 159, 1883–1888, https://doi.org/10.1007/s00705-014-2002-x (2014).
Balanescu, P. et al. Th17 and IL-17 immunity in chronic hepatitis C infection. Rom J Intern Med 50, 13–18 (2012).
Guabiraba, R. & Ryffel, B. Dengue virus infection: current concepts in immune mechanisms and lessons from murine models. Immunology 141, 143–156, https://doi.org/10.1111/imm.12188 (2014).
Bystrom, J., Al-Adhoubi, N., Al-Bogami, M., Jawad, A. S. & Mageed, R. A. Th17 lymphocytes in respiratory syncytial virus infection. Viruses 5, 777–791, https://doi.org/10.3390/v5030777 (2013).
Hou, W., Kang, H. S. & Kim, B. S. Th17 cells enhance viral persistence and inhibit T cell cytotoxicity in a model of chronic virus infection. J Exp Med 206, 313–328, https://doi.org/10.1084/jem.20082030 (2009).
Bowen, J. L. & Olson, J. K. Innate immune CD11b + Gr-1 + cells, suppressor cells, affect the immune response during Theiler’s virus-induced demyelinating disease. J Immunol 183, 6971–6980, https://doi.org/10.4049/jimmunol.0902193 (2009).
Hou, W., Jin, Y. H., Kang, H. S. & Kim, B. S. Interleukin-6 (IL-6) and IL-17 synergistically promote viral persistence by inhibiting cellular apoptosis and cytotoxic T cell function. J Virol 88, 8479–8489, https://doi.org/10.1128/JVI.00724-14 (2014).
Ma, Q. & He, X. Molecular basis of electrophilic and oxidative defense: promises and perils of Nrf2. Pharmacol Rev 64, 1055–1081, https://doi.org/10.1124/pr.110.004333 (2012).
Kobayashi, E. H. et al. Nrf2 suppresses macrophage inflammatory response by blocking proinflammatory cytokine transcription. Nat Commun 7, 11624, https://doi.org/10.1038/ncomms11624 (2016).
Thimmulappa, R. K. et al. Nrf2 is a critical regulator of the innate immune response and survival during experimental sepsis. J Clin Invest 116, 984–995, https://doi.org/10.1172/JCI25790 (2006).
Reddy, N. M. et al. Innate immunity against bacterial infection following hyperoxia exposure is impaired in NRF2-deficient mice. J Immunol 183, 4601–4608, https://doi.org/10.4049/jimmunol.0901754 (2009).
Nagai, N. et al. Nrf2 is a critical modulator of the innate immune response in a model of uveitis. Free Radic Biol Med 47, 300–306, https://doi.org/10.1016/j.freeradbiomed.2009.04.033 (2009).
Schaedler, S. et al. Hepatitis B virus induces expression of antioxidant response element-regulated genes by activation of Nrf2. J Biol Chem 285, 41074–41086, https://doi.org/10.1074/jbc.M110.145862 (2010).
Burdette, D., Olivarez, M. & Waris, G. Activation of transcription factor Nrf2 by hepatitis C virus induces the cell-survival pathway. J Gen Virol 91, 681–690, https://doi.org/10.1099/vir.0.014340-0 (2010).
Ivanov, A. V. et al. Hepatitis C virus proteins activate NRF2/ARE pathway by distinct ROS-dependent and independent mechanisms in HUH7 cells. PLoS One 6, e24957, https://doi.org/10.1371/journal.pone.0024957 (2011).
Kesic, M. J., Simmons, S. O., Bauer, R. & Jaspers, I. Nrf2 expression modifies influenza A entry and replication in nasal epithelial cells. Free Radic Biol Med 51, 444–453, https://doi.org/10.1016/j.freeradbiomed.2011.04.027 (2011).
Cheng, Y. L. et al. Activation of Nrf2 by the dengue virus causes an increase in CLEC5A, which enhances TNF-alpha production by mononuclear phagocytes. Sci Rep 6, 32000, https://doi.org/10.1038/srep32000 (2016).
Zhang, H. S., Li, H. Y., Zhou, Y., Wu, M. R. & Zhou, H. S. Nrf2 is involved in inhibiting Tat-induced HIV-1 long terminal repeat transactivation. Free Radic Biol Med 47, 261–268, https://doi.org/10.1016/j.freeradbiomed.2009.04.028 (2009).
Furuya, A. K. et al. Sulforaphane Inhibits HIV Infection of Macrophages through Nrf2. PLoS Pathog 12, e1005581, https://doi.org/10.1371/journal.ppat.1005581 (2016).
Cho, H. Y. et al. Antiviral activity of Nrf2 in a murine model of respiratory syncytial virus disease. Am J Respir Crit Care Med 179, 138–150, https://doi.org/10.1164/rccm.200804-535OC (2009).
Yageta, Y. et al. Role of Nrf2 in host defense against influenza virus in cigarette smoke-exposed mice. J Virol 85, 4679–4690, https://doi.org/10.1128/JVI.02456-10 (2011).
Lee, J., Koh, K., Kim, Y. E., Ahn, J. H. & Kim, S. Upregulation of Nrf2 expression by human cytomegalovirus infection protects host cells from oxidative stress. J Gen Virol 94, 1658–1668, https://doi.org/10.1099/vir.0.052142-0 (2013).
Jordan, S. et al. Virus progeny of murine cytomegalovirus bacterial artificial chromosome pSM3fr show reduced growth in salivary Glands due to a fixed mutation of MCK-2. J Virol 85, 10346–10353, https://doi.org/10.1128/JVI.00545-11 (2011).
Wagner, M., Jonjic, S., Koszinowski, U. H. & Messerle, M. Systematic excision of vector sequences from the BAC-cloned herpesvirus genome during virus reconstitution. J Virol 73, 7056–7060 (1999).
Seelige, R. et al. Cutting edge: Endothelial-specific gene ablation of CD99L2 impairs leukocyte extravasation in vivo. J Immunol 190, 892–896, https://doi.org/10.4049/jimmunol.1202721 (2013).
Livak, K. J. & Schmittgen, T. D. Analysis of relative gene expression data using real-time quantitative PCR and the 2(-Delta Delta C(T)) Method. Methods 25, 402–408, https://doi.org/10.1006/meth.2001.1262 (2001).
Johnson, L. R., Weizman, O. E., Rapp, M., Way, S. S. & Sun, J. C. Epitope-Specific Vaccination Limits Clonal Expansion of Heterologous Naive T Cells during Viral Challenge. Cell Rep 17, 636–644, https://doi.org/10.1016/j.celrep.2016.09.019 (2016).
Wen, A. Y. et al. Increased abscess formation and defective chemokine regulation in CREB transgenic mice. PLoS One 8, e55866, https://doi.org/10.1371/journal.pone.0055866 (2013).
Simon, C. O., Seckert, C. K., Dreis, D., Reddehase, M. J. & Grzimek, N. K. Role for tumor necrosis factor alpha in murine cytomegalovirus transcriptional reactivation in latently infected lungs. J Virol 79, 326–340, https://doi.org/10.1128/JVI.79.1.326-340.2005 (2005).
DeNicola, G. M. et al. Oncogene-induced Nrf2 transcription promotes ROS detoxification and tumorigenesis. Nature 475, 106–109, https://doi.org/10.1038/nature10189 (2011).
Mamo, S., Gal, A. B., Bodo, S. & Dinnyes, A. Quantitative evaluation and selection of reference genes in mouse oocytes and embryos cultured in vivo and in vitro. BMC Dev Biol 7, 14, https://doi.org/10.1186/1471-213X-7-14 (2007).
Francis, S. P. et al. Celastrol inhibits aminoglycoside-induced ototoxicity via heat shock protein 32. Cell Death Dis 2, e195, https://doi.org/10.1038/cddis.2011.76 (2011).
Acknowledgements
J.D.B. was supported by grants from the NCI (CA157885) and The Hartwell Foundation. R.S. was additionally supported by the DFG (SE-2418/1-1). N.M.A. was supported by NIH Medical Scientist Training Program grant T32GM07739 to the Weill Cornell/ Rockefeller/ Sloan Kettering Tri-Institutional MD-PhD Program. G.P. and C.A.B were supported by NIH grants AI101423 and AI113349. J.C.S. was supported by the Burroughs Wellcome Fund and the NIH grant AI100874. We thank Dr. Ellen Wehrens for support with plaque assay measurements.
Author information
Authors and Affiliations
Contributions
R.S. and J.D.B. designed the experiments. R.S. performed and analyzed the experiments. R.S.-K., N.M.A. and J.C.S. contributed scientific advice, experimental expertise and performance. C.A.B. and G.P. provided reagents and experimental advice. J.D.B. supervised the research. R.S. and J.D.B. wrote the paper.
Corresponding author
Ethics declarations
Competing Interests
The authors declare no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Electronic supplementary material
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Seelige, R., Saddawi-Konefka, R., Adams, N.M. et al. Interleukin-17D and Nrf2 mediate initial innate immune cell recruitment and restrict MCMV infection. Sci Rep 8, 13670 (2018). https://doi.org/10.1038/s41598-018-32011-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-32011-2
Keywords
This article is cited by
-
A state-of-the-art review on the NRF2 in Hepatitis virus-associated liver cancer
Cell Communication and Signaling (2023)
-
Upregulation of nuclear factor E2-related factor 2 (Nrf2) represses the replication of herpes simplex virus type 1
Virology Journal (2022)
-
Single-cell RNA-sequencing of herpes simplex virus 1-infected cells connects NRF2 activation to an antiviral program
Nature Communications (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.