Abstract
Poor vitamin D status has been associated with tuberculosis (TB); whether poor status is cause or consequence of disease is uncertain. We conducted a case-control study and two nested case-control studies to determine whether vitamin D levels were associated with active TB, tuberculin skin test (TST) conversion, and risk of progression to the active TB in prisoners in Brazil. In multivariable conditional logistic regression, subnormal vitamin D levels (OR, 3.77; 95% CI, 1.04–13.64) were more likely in prisoners with active TB. In contrast, vitamin D was not found to be a risk factor for either TST conversion (OR, 2.49; 95% CI, 0.64–9.66) or progression to active disease (OR, 0.59; 95% CI, 0.13–2.62). Black race (OR, 11.52; 95% CI, 2.01–63.36), less than 4 years of schooling (OR, 2.70; 95% CI, 0.90–8.16), cigarette smoking (OR, 0.23; 95% CI, 0.06–0.79) were identified as risk factors for TST conversion. Risk of progression to active TB was found to be associated with cigarette smoking (OR, 7.42; 95% CI, 1.23–44.70). Our findings in the prison population show that poor vitamin D status is more common in individuals with active TB, but is not a risk factor for acquisition of latent TB or progression to active TB.
Similar content being viewed by others
Introduction
Tuberculosis (TB) is a major global public health problem, causing approximately 1.4 million deaths annually1. The World Health Organization estimates that one-third of the world’s population has latent TB1, of which approximately 10% will develop active disease2. Understanding which individuals will progress to TB and which will maintain lifelong immune control or achieve clearance, remains an important scientific question with substantial public health implications. This high-risk group also provides the opportunity to examine host factors that currently lack complete understanding in the disease pathway for active TB.
One of the prominent host factors that has been implicated in TB risk is vitamin D levels3. Nutritional deficiency, low exposure to sunlight due to spending only short periods of time outdoors, and seasonal changes resulting in reduced ultraviolet radiation type B (UVB) may be associated with subnormal vitamin D levels4. Vitamin D levels of <30 ng/ml, which are characterized clinically as either insufficient (29–20 ng/ml) or deficient (<20 ng/ml). Vitamin D is composed of a group of secosteroid molecules that are predominantly synthesized in the skin following exposure to solar radiation, resulting in vitamin D3 (or cholecalciferol), or obtained via dietary intake, resulting in vitamin D2 (or ergocalciferol) or D35. Active vitamin D requires two hydroxylation reactions. The primary circulating form of vitamin D is 25(OH)D3 after vitamin D3 is hydroxylated in the liver. 25(OH)D3 is the precursor for the biologically active form of vitamin D, 1,25(OH)2D3, formed largely in the kidney. It is now known that immune cells (innate and adaptive), e.g., macrophages express enzymes to locally activate 25(OH)D3 to supra-physiological concentrations of 1,25(OH)2D3 for diverse functions. For example, it has been demonstrated that activated vitamin D is required for interferon-γ mediated antimicrobial activity of macrophages against Mycobacterium tuberculosis6.
Many studies have reported seasonality of TB disease incidence, leading to speculation that seasonal differences in vitamin D levels may play a role in TB infection or disease progression7. However, isolating the role of vitamin D from other seasonal confounders has been difficult. The majority of studies investigating the relationship between vitamin D levels and TB infection or disease risk have been cross-sectional in nature; as TB may cause micronutrient deficiencies, determining whether there is a causal relationship between vitamin D levels and TB risk remains challenging. One prospective study among household contacts in Spain found that vitamin D deficiency was associated with risk of TST conversion and incident disease among household contacts of TB cases8. Further evidence is needed to understand whether vitamin D levels are predictive of risk of incident TB infection and subsequent risk of disease.
Brazil is a medium TB-burden country, which has over 70,000 new cases TB diagnosed annually. Of the 526,000 new cases of TB reported between 2009 and 2014 in Brazil, 38,000 (7.3%) were diagnosed among prisoners9. In this population, the incidence of active TB is at least 30 times higher than that observed in the general population10. A number of structural and host determinants have been implicated in the high rates of TB infection and disease observed in prisons11. These include factors associated with increased transmissibility of the disease, such as overcrowding and ventilation11, as well host factors associated with both TB risk and incarceration, such as nutritional deficiency12, HIV infection, smoking13,14 and drug use15,16. Previous studies have identified low vitamin D levels among prisoners in correctional facilities, likely due to prolonged indoor incarceration with insufficient sunlight exposure or dietary sources of vitamin D7. Whether differences in vitamin D levels among prisoners are an important determinant of elevated TB risk is not understood.
The purpose of this study was to investigate the role of subnormal vitamin D in incident TB infection and disease risk among incarcerated individuals. We performed one case-control study and two, nested case-control studies among an observational cohort of Brazilian prisoners. We aimed to determine whether vitamin D levels were associated with three distinct features of TB disease: active TB, M. tuberculosis infection (tuberculin skin test (TST) conversion), and progression to active disease in this high-risk population.
Results
Risk factors associated with active TB
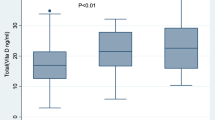
The frequency distribution of the variables and risk factors are shown in Table 1. We compared 24 TB cases and 48 matched controls without TB. The mean age of prisoners for the case-control study was 32 years (±7 years) among cases and 33 years (±8 years) among the controls (Table 2). Mean serum 25(OH)D3 level was significantly lower among the cases (27.7 ± 7.85 ng/mL) than controls (37.1 ± 8.94 ng/mL, p < 0.01; Table 2). Prisoners with active TB were more likely to have subnormal vitamin D levels (<30 ng/mL) when compared to healthy controls (18/24 [75%] vs 16/48 [33%], p = 0.004; Table 1). As shown in Table 1, there is a clear difference between cases and controls with active TB (p < 0.01). A greater difference existed among TB patients with vitamin D deficiency (3.5×), than those with vitamin D insufficiency (2.0×) when compared with controls. All cases had negative HIV serology and only two controls had positive HIV serology (Table 1).
Bivariable conditional logistic regression analyses were performed to assess the associations between active TB and thirteen potential predictor variables. These variables included drug use over the last year (OR, 4.88; 95% CI, 1.02–23.15), previous incarceration (OR, 6.82; 95% CI, 0.82–56.46), black race (OR, 3.71; 95% CI, 0.76–18.20), and serum 25(OH)D3 level <30 ng/mL (OR, 4.37; 95% CI, 1.56–12.25). In multivariable analysis, only subnormal serum 25(OH)D3 level (OR, 3.77; 95% CI, 1.04–13.64) was determined to be significantly associated with active TB after adjustment for drug use (OR, 2.87; 95% CI, 0.43–19.01), previous incarceration (OR, 3.12; 95% CI, 0.34–28.24) and black race (OR, 1.57; 95% CI, 0.26–9.71).
Risk factors associated with TST conversion
In the first nested case-control study, we selected individuals who screened negative for TB at baseline by symptoms and sputum culture, and compared those who converted their TST from negative to positive when retested at one year (converters) with those who remained negative upon repeat testing (non-converters). The mean age of the converters was 32 years (±6 years) and 31 years (±7 years) among the non-converters (Table 2). Vitamin D levels did not differ significantly between the TST converters (37.7 ± 11.93 ng/mL) and non-converters (34.5 ± 14.89 ng/mL, p = 0.19). Albumin levels differed significantly between cases (4.63 ± 0.43 g/dL) and controls (4.28 ± 1.00 g/dL, p < 0.01) but were in the normal range (>3.5–5.2 g/dL) (Table 2). All cases and controls had negative HIV serology (Table 1).
In bivariable analysis, black race (OR, 3.12; 95% CI, 0.98–9.95), less than 4 years of schooling (OR, 2.44; 95% CI, 1.01–5.94), cigarette smoking (OR, 0.44; 95% CI, 0.17–1.15), serum 25(OH)D3 level <30 ng/mL (OR, 0.79; 95% CI, 0.31–2.04) and serum albumin level (OR, 2.05; 95% CI, 0.97–4.32) were associated with risk of TST conversion. The multivariable analysis showed black race (OR, 11.52; 95% CI, 2.01–63.36), less than 4 years of schooling (OR, 2.70; 95% CI, 0.90–8.16), cigarette smoking (OR, 0.22; 95% CI, 0.06–0.79) and serum albumin level (OR, 0.09; 95% CI, 0.01–1.13) were associated with an increased risk of TST conversion (Table 3). Vitamin D levels, however, were not associated with risk of TST conversion (OR, 2.49; 95% CI, 0.64–9.66; Table 3).
Risk factors associated with progression to active TB
In the second nested case-control study, we compared baseline characteristics of individuals, who screened negative for active TB at baseline, according to whether they progressed to active TB over the subsequent year (“progressors”) or did not (“non-progressors”). Thirty-six percent of those who progressed and 50% of those who did not progress had positive TSTs at baseline. No significant difference in baseline serum 25(OH)D3 levels was identified between progressors (37.0 ± 14.83 ng/mL) and non-progressors (37.5 ± 11.04 ng/mL, p = 0.86; Table 2). All cases had negative HIV serology and only one control had positive HIV serology (Table 1).
In bivariable analysis, drug use during the last year (OR, 4.28; 95% CI, 0.90–20.30), cigarette smoking (OR, 7.77; 95% CI, 1.68–35.86) and black race (OR, 3.37; 95% CI, 1.01–11.18) were associated with risk of TB progression. However, in the multivariable analysis, only current smoker (OR, 7.42; 95% CI, 1.23–44.70) was associated with the development of active TB after adjustment for black race (OR, 3.43; 95% CI, 0.85–13.75), drug use during the last year (OR, 2.00; 95% CI, 0.38–10.44), and serum 25(OH)D3 level <30 ng/mL (OR, 0.59; 95% CI, 0.13–2.62; Table 3).
The average time to active TB diagnosis among progressors was 6.9 months. No statistical difference in serum 25(OH)D3 level at enrollment was observed among prisoners who progressed to active TB in the first 6 months compared to prisoners who had progressed after 6 months of follow-up (35.5 ng/mL ±12.01 and 38.4 ng/mL ±15.36, respectively; p = 0.46).
Discussion
Despite the low power, this is the largest study ever published that evaluate the association of vitamin D levels with progression to TST conversion and progression of active disease in the prison population. We found that prisoners with active TB had significantly lower vitamin D levels at the time of diagnosis compared with prisons without active TB. However, baseline vitamin D levels were not predictive of TST conversion or progression to active disease. Notably, while 75% of prisoners with active TB had subnormal vitamin D levels at TB diagnosis, less than 33% of all other groups of participants (cases and controls) presented subnormal levels. These findings provide support for a pattern of disrupted vitamin D metabolism that results from the presence of active TB disease, and do not provide evidence for subnormal vitamin increasing susceptibility to infection or disease.
Our primary findings of poor vitamin D status associated with active TB among prisoners were consistent with the results of case-control studies conducted in the general population8,17,18,19,20,21,22,23,24,25,26. Furthermore, previous cohort studies conducted among household contacts of TB cases have suggested that low levels of vitamin D may be associated with risk of subsequent M. tuberculosis infection27 and development of active TB28,29; we did not find these associations in this study when vitamin D levels were examined months prior to diagnosis and found to be largely normal. The number of subjects experiencing the outcome of interest in previous studies was limited, however, ranging from 3 to 18 over follow-up periods 2 to 4 years. These studies have been found to vary with study design, geographical location8, as well as ethnicities30 and gender24 of subjects. Thus, studies with larger sample sizes in different populations are needed to help clarify whether the association between vitamin D and susceptibility to TB infection or disease is causal in nature.
Notably, we previously identified high rates of conversion (28%) and incident active disease (1.39%) over one year in this cohort study10. These rates provided more robust sample sizes of 30 and 24 cases, respectively, for the nested case-control studies to examine the impact of vitamin D status at baseline. Surprisingly, the majority of healthy prisoners show normal vitamin D. In the individuals without active disease at enrollment, both diet and sun exposures in this incarceration setting appear to be adequate to sustain precursor vitamin D levels in healthy prisoners. Vitamin D status under these conditions appeared not to be associated with either the acquisition of latent infection, or the development of active disease in this prison population. It is possible that there was insufficient heterogeneity in vitamin D in this population and setting to detect such effects. Here, factors other than vitamin D levels (e.g. smoking status) appear to contribute risk along the TB disease pathway.
Previous studies have recognized that inflammatory process may lower 25(OH)D3 levels as the precursor supply for activated vitamin D31,32. The normal immune response against M. tuberculosis produces inflammatory cytokines that activate CYP27B1 (1α-hydroxylase), converting available 25(OH)D3 to its activated form 1,25(OH)2D331,33 and contributing to the measurement of low 25(OH)D3 levels34. Thus, the low levels of 25(OH)D3 (precursor) identified among prisoners with active TB in our case-control and among in several previous studies may have been associated with high conversion rates to 1,25(OH)2D3 caused by persistent disease. Notably, elevated plasma 1,25(OH)2D3 levels have been found in TB patients compared to healthy individuals35,36, suggesting hyperresponsive metabolism to form activated vitamin D may be associated with the disease in humans. However, vitamin D supplementation as part of randomized clinical trials during TB treatment has not been shown to reduce time to sputum culture conversion after 8 weeks37,38,39,40,41,42,43,44.
One of the major defense mechanisms involving vitamin D and M. tuberculosis is associated with Toll-like receptor (TLR)45. Mycobacterial antigens, especially lipoproteins, are recognized by TLR2 or associated with TLR1 or TLR6 which when activated participate in the expression of vitamin D receptor (VDR) and activation of 25(OH)D, assisting in the release of cathelicidin, a peptide capable of inhibiting the intracellular growth of M. tuberculosis, in vivo46 and in vitro45.
Vitamin D levels may be influenced by anti-TB drugs, especially rifampicin and isoniazid47, and if vitamin D measurement is performed after the initiation of treatment, these levels may be considerably lower among patients with TB17,48,49,50,51. One strength of our study was that all serum samples used to measure vitamin D levels were collected before treatment was initiated removing this potential factor impacting vitamin D.
Risk of TST conversion was not influenced by baseline vitamin D status in the prisoners, but other risk factors showed associations consistent with the previous reports. For example, previous studies reported an association between being a current smoker and TB52,53. Evidence suggests that smoking decreases TST reactivity54,55 but increases the risk of progression to active TB56 by directly impairing host immunity, especially the responses of T-cells and macrophages to M. tuberculosis57,58. An impaired immune response makes it more difficult to eliminate M. Tuberculosis and easier to develop the active disease52,57. While the variable cigarette smoking was found here to exert a protective effect on risk of TST conversion to latent infection, it dramatically increased risk of progression to active TB. TST conversion was also associated with black race, which increased the odds of infection by M. tuberculosis.
Evidence suggests that black race may increase risk of M. tuberculosis infection greater than other racial backgrounds59. Although the mechanism underlying this association has not been fully elucidated, it may be attributed to environmental factors, unfavorable socioeconomic conditions60,61, and genetic factors62,63. Black skin can impair vitamin D synthesis from UVB exposure, but notably, vitamin D status was found to be an independent risk factor for active TB when controlling for race in the case-control study. Although it is not yet clear why race is a risk factor for TST conversion, we believe that in addition to the polymorphisms vitamin D receptors, other genetic factors may be associated. In our study, in the final model, the level of Vitamin D was adjusted by race and even then, the serum level of vitamin D was not associated with TST conversion. Other polymorphisms associated with black race should be investigated.
Low levels of schooling also showed a tendency toward increased risk and may be directly related with incarceration mixing patterns of high risk individuals during incarceration64. The prisoners are combined in cell blocks of mixed socioeconomic conditions, thus increasing the probability of becoming infected with M. tuberculosis65,66. However, the fact that all prisoners were exposed to the same conditions during incarceration, reinforces the need for further studies to be conducted to explain the reasons behind the racial and education inequalities among prisoners with active TB.
We examined risk of incident TB among all prisoners rather than those who were TST positive at baseline. The majority of TB disease in this high transmission setting occurs due to recent infection rather that late reactivation. Indeed, the majority of incident TB cases over one year in this cohort occurred among individuals who were TST negative at baseline.
While studies remain challenged by power, this work represents the largest study ever conducted to evaluate TST conversion and progression of active disease. We used the TST conversion to assess TB infections and progression to active TB, however, some studies indicate that TST can cause false-positive results by BCG vaccination and exposure to non-tuberculosis mycobacteria (NTM). In Brazil, BCG vaccination usually performed in the first days of life that does not interfere with the TST result if carried out 10 years or more after vaccination67,68. In this study the average age was 32.1 (±6.87, p = 0.50) in TST conversion and 30.8 (±8.90, p = 0.63) in progression to active TB, it seems unlikely that BCG vaccination interfered with the TST results. As well as, exposure to non-tuberculosis mycobacteria (NTM) may interferes with TST results only in populations with a high prevalence of NTM sensitization and low TB infection, that does not happen in our study population, where the incidence of active TB is at least 30 times higher than that observed in the general population10. Interferon-ɤ release assays (IGRA) to determinate TST conversion may be more useful in populations where the BCG vaccine has not been administered or if the test was performed within 10 years of vaccination, thus, in our study, the TST and IGRAs likely would generate similar findings67.
Data suggest that seasonality69 and the incarceration time70 may favor a decrease in vitamin D levels. A strength of our design however, was that all prisoners were screened in the same months, and cases and controls were paired according to the prison cell block and incarceration time, having the same time of potential sun exposure and similar diet; these features help minimize confounding in our analysis, however is very difficult to perfectly match due to the prolonged and variable infectious period, variable social mixing, and frequent movement of inmates between cells. Overmatching among cases and controls, may have decreased the variability in vitamin D levels limiting the ability to identify important associations over the spectrum of vitamin D with the disease pathway. As a caveat, as many participants have sufficient vitamin D this may play a minor role in relation to other risk factors.
Conclusion
We identified that prisoners with active TB had poor vitamin D status; however, vitamin D levels were not identified to be a risk factor for TST conversion or the development of active TB over a follow-up period of one year in the evaluated prison population. Other risk factors such as black race and low education were found to be risk factors for TST conversion, while smoking was the only variable associated with disease progression. Our findings suggest active TB may alter vitamin D metabolism, leading to compromised precursor supply, but that vitamin D status is not necessarily a risk factor for susceptibility to infection or disease.
Methods
Population
In 2013, the state of Mato Grosso do Sul (MS), located in the west-central Brazil, had the country’s seventh largest prison population, including 14,904 prisoners, and the higher rate of national incarceration (568.9 prisoners per 100,000 inhabitants)71. A one-year prospective cohort study was conducted in 2013 in 12 prisons (8 male prisons, with 6,552 prisoners and 4 female prisons, with 669 prisoners) to identify the incidence of latent and active TB10,16. The case-study and two nested case-studies here are part of this larger cohort study.
Prisoners who were over 18 years old and consented to participate in the study were recruited from a randomly stratified sample at the 12 prisons. A structured questionnaire was administered and a (TST) (PPD RT23, Staten Serum Institute, Copenhagen) was performed. Two sputum samples were obtained from all participants reporting cough, and smear microscopy and culture, using the Ogawa-Kudoh technique, was performed. Although X-ray screening shows good sensitivity, and is an important method for screening suspected cases of TB, the specificity of X-ray screening is low. Additionally, chest radiography is not available in the study prisons. For these reasons, we used sputum culture to define TB cases. Individuals with at least one smear or culture result positive for M. tuberculosis were considered TB cases. A venous blood sample was collected, and plasma was frozen for assessment of vitamin D levels.
TST positivity was defined as an induration of ≥10 mm measured by a trained TST reader, 48 hours after injected. After one year, in 2014, a second TST was performed, and two sputum samples were collected for smear microscopy and culture from all participants reporting cough at the second time point. TST conversion was defined as TST ≥10 mm and an induration increase of at least 6 mm in an individual with a baseline TST <10 mm10.
Incident TB cases were defined as those occurring after the first screening time point, including those diagnosed between the two screening dates (day 0 and day 365) and those diagnosed on the second screening date. Those diagnosed between the screening dates were diagnosed through routine medical services provided by the prison, in which symptomatic individuals are clinically evaluated and sputum is sent for smear and/or culture as determined the prison clinician. These TB cases occurring between the screening dates were identified through prison medical records and the National Notifiable Disease database (Sistema de Informação de Agravos de Notificação National, SINAN).
A plasma sample for vitamin D levels was collected at the time of enrollment, which was prior to TB diagnosis, and therefore, prior to initiation of treatment. Due to the low rates of TB in the female prisoners, the vitamin D evaluation was conducted in only male prisoners10.
All eligible participants provided written informed consent prior to study participation. All methods described in this study were approved by Research Ethics Committee of the Federal University of Grande Dourados (Number 191,877 and 793,740) and were performed in accordance with the relevant guidelines and regulations. The study was authorized by the State Agency of Administration of the Penitentiary System (AGEPEN) who gave full support during the study period.
Risk factors associated with active TB
During enrollment in the cohort study, a total of 29 male prisoners from 2,861 were culture positive for M. tuberculosis and, therefore, had active TB16 (Fig. 1). Based on this finding, a case-control study was performed in which 25(OH)D levels were measured in serum samples obtained from 24 prisoners with active TB and compared with those from samples obtained from 48 controls, 25 of whom were TST-negative and 23 were TST positive, who were matched on the basis of age (±5 years), prison cell block and incarceration time (±6 months) (Fig. 1). Other risk factors of interest were age, marital status, education, resides in MS, smoking, drug use over the last year, alcohol, contact with a TB-positive individual and previous incarceration. The participant’s race/skin color (i.e., white, black, indigenous, asian or mixed) was self-reported. The presence of a Bacillus Calmette-Guérin (BCG) vaccine scar was determined by inspecting the participant’s arm. The body mass index (BMI) wasn’t included in evaluation of active TB because we do not know if cause or effect.
Risk factors associated with TST conversion
A nested case-control study was performed to identify risk factors associated with TST conversion. Thirty prisoners randomly who converted their TST from negative to positive (converters) over the course of 12 months were compared with sixty prisoners who had negative TST tests at baseline and repeat testing at 12 months (non-converters) (Fig. 1). Controls were matched based on age (±5 years), prison cell block and incarceration time (±6 months). Other risk factors assessed for TST conversion were age, marital status, education, resides in MS, smoking, drug use over the last year, alcohol, serum albumin, contact with a TB-positive individual, BMI and previous incarceration. The presence of a BCG vaccine scar was determined by inspecting the participant’s arm.
Risk factors associated with progression to active TB
A nested case-control study was conducted to investigate the association between vitamin D levels and risk of subsequent TB disease. Of the 1,235 prisoners who remained incarcerated after 1 year in the male prison system (Fig. 1), we selected 24 individuals without TB at baseline who were diagnosed with active TB over the course of 12 months (progressors) and compared them with 48 individuals who were TB negative throughout 12 months (non-progressors), as documented by active TB screening at baseline and 12 months. Controls were matched to cases on the basis of age (±5 years), prison cell block and incarceration time (±6 months) (Fig. 1). The risk factors assessed for progression to active TB were age, marital status, education, resides in MS, smoking, drug use over the last year, alcohol, serum albumin, contact with a TB-positive individual, BMI and previous incarceration. The presence of a BCG vaccine scar was determined by inspecting the participant’s arm.
Biochemical analyses
Vitamin D levels were measured in serum samples, which were stored away from light and at −20 °C until the time of analysis. Levels of 25(OH)D (ng/ml) were identified using the automated electrochemiluminescence immunoassay method and Cobas® e411 analyzer (Roche Diagnostics, Mannheim, Germany). Serum 25(OH)D levels were considered normal when values were ≥30 ng/mL72,73,74. Subnormal vitamin D was used to indicate combined groups for vitamin D insufficiency (29–20 ng/ml) and deficiency (<20 ng/ml). Serum albumin levels were measured in samples obtained from prisoners included the nested case-control studies using the Cobas Integra® 400 plus analyzer (Roche Diagnostics, Mannheim, Germany). All procedures were performed in the Laboratory of the University Hospital of the Federal University of Grande Dourados.
Data analysis
All questionnaires were double entered into the Research Electronic Data Capture database (REDCap). SAS version 9.2 (SAS Institute, Cary, NC, USA) was used to perform the analyses. We used the t-test for continuous variables, Chi-square test for categorical variables and bivariable conditional logistic regression analyses to examine the associations between dependent and independent variables. Vitamin D insufficient and all variables with p ≤ 0.10 in the bivariable analyses were included in multivariable conditional logistic regression analysis. P values ≤0.05 were considered statistically significant.
Availability of materials and data
The data will not be shared in order to protect the participants’ anonymity.
References
WHO. Global tuberculosis report. (World Health Organization, 2016).
Abel, L., El-Baghdadi, J., Bousfiha, A. A., Casanova, J. L. & Schurr, E. Human genetics of tuberculosis: a long and winding road. Philos Trans R Soc Lond B Biol Sci 369, 20130428, https://doi.org/10.1098/rstb.2013.0428 (2014).
Huang, S. J. et al. Vitamin D deficiency and the risk of tuberculosis: a meta-analysis. Drug Des Devel Ther 11, 91–102, https://doi.org/10.2147/DDDT.S79870 (2017).
Nwosu, B. U. et al. The vitamin D status of prison inmates. PLoS One 9, e90623, https://doi.org/10.1371/journal.pone.0090623 (2014).
Bender, D. A. In Bioquímica Ilustrada de Harper (eds V. W. Bender Rodwell, D. A. Botham, K. M. Kennelly, P. J. & Weil, P. A.) 546–560 (Ed. Amgh, 2016).
Fabri, M. et al. Vitamin D is required for IFN-gamma-mediated antimicrobial activity of human macrophages. Sci Transl Med 3, 104ra102, https://doi.org/10.1126/scitranslmed.3003045.
Santos, L. G., Pires, G. N., Azeredo Bittencourt, L. R., Tufik, S. & Andersen, M. L. Chronobiology: relevance for tuberculosis. Tuberculosis 92, 293–300, https://doi.org/10.1016/j.tube.2012.03.006 (2012).
Arnedo-Pena, A. et al. Latent tuberculosis infection, tuberculin skin test and vitamin D status in contacts of tuberculosis patients: a cross-sectional and case-control study. BMC Infect Dis 11, 349, https://doi.org/10.1186/1471-2334-11-349 (2011).
Bourdillon, P. M. et al. Increase in Tuberculosis Cases among Prisoners, Brazil, 2009–2014. Emerging infectious diseases 23, 496–499, https://doi.org/10.3201/eid2303.161006 (2017).
Paiao, D. S. et al. Impact of mass-screening on tuberculosis incidence in a prospective cohort of Brazilian prisoners. BMC Infect Dis 16, 533, https://doi.org/10.1186/s12879-016-1868-5 (2016).
Urrego, J. et al. The Impact of Ventilation and Early Diagnosis on Tuberculosis Transmission in Brazilian Prisons. Am J Trop Med Hyg 93, 739–746, https://doi.org/10.4269/ajtmh.15-0166 (2015).
Kalonji, G. M. et al. Prevalence of tuberculosis and associated risk factors in the Central Prison of Mbuji-Mayi, Democratic Republic of Congo. Trop Med Health 44, 30, https://doi.org/10.1186/s41182-016-0030-9 (2016).
Edge, C. L., King, E. J., Dolan, K. & McKee, M. Prisoners co-infected with tuberculosis and HIV: a systematic review. J Int AIDS Soc 19, 20960, https://doi.org/10.7448/IAS.19.1.20960 (2016).
Valenca, M. S. et al. Prevalence of tuberculosis in prisons: risk factors and molecular epidemiology. Int J Tuberc Lung Dis 19, 1182–1187, https://doi.org/10.5588/ijtld.15.0126 (2015).
Coninx, R., Maher, D., Reyes, H. & Grzemska, M. Tuberculosis in prisons in countries with high prevalence. BMJ 320, 440–442 (2000).
Carbone, A. S. S. et al. Active and latent tuberculosis in Brazilian correctional facilities: a cross-sectional study. BMC Infect Dis 15, 24, https://doi.org/10.1186/s12879-015-0764-8 (2015).
Sita-Lumsden, A., Lapthorn, G., Swaminathan, R. & Milburn, H. J. Reactivation of tuberculosis and vitamin D deficiency: the contribution of diet and exposure to sunlight. Thorax 62, 1003–1007, https://doi.org/10.1136/thx.2006.070060 (2007).
Iftikhar, R., Kamran, S. M., Qadir, A., Haider, E. & Bin Usman, H. Vitamin D deficiency in patients with tuberculosis. J Coll Physicians Surg Pak 23, 780–783, 11.2013/JCPSP.780783 (2013).
Kim, J. H. et al. Low serum 25-hydroxyvitamin D level: an independent risk factor for tuberculosis? Clin Nutr 33, 1081–1086, https://doi.org/10.1016/j.clnu.2013.11.014 (2014).
Hong, J. Y. et al. Association between vitamin D deficiency and tuberculosis in a Korean population. Int J Tuberc Lung Dis 18, 73–78, https://doi.org/10.5588/ijtld.13.0536 (2014).
Davies, P. D., Brown, R. C. & Woodhead, J. S. Serum concentrations of vitamin D metabolites in untreated tuberculosis. Thorax 40, 187–190 (1985).
Wilkinson, R. J. et al. Influence of vitamin D deficiency and vitamin D receptor polymorphisms on tuberculosis among Gujarati Asians in west London: a case-control study. Lancet 355, 618–621, https://doi.org/10.1016/S0140-6736(99)02301-6 (2000).
Wejse, C. et al. Serum 25-hydroxyvitamin D in a West African population of tuberculosis patients and unmatched healthy controls. The American journal of clinical nutrition 86, 1376–1383, http://ajcn.nutrition.org/content/86/5/1376.long (2007).
Ho-Pham, L. T. et al. Association between vitamin D insufficiency and tuberculosis in a Vietnamese population. BMC Infect Dis 10, 306, https://doi.org/10.1186/1471-2334-10-306 (2010).
Nielsen, N. O. et al. Both high and low serum vitamin D concentrations are associated with tuberculosis: a case-control study in Greenland. Br J Nutr 104, 1487–1491, https://doi.org/10.1017/S0007114510002333 (2010).
Oh, J. et al. Evaluation of vitamin status in patients with pulmonary tuberculosis. J Infect 74, 272–280, https://doi.org/10.1016/j.jinf.2016.10.009 (2017).
Arnedo-Pena, A. et al. Vitamin D status and incidence of tuberculosis infection conversion in contacts of pulmonary tuberculosis patients: a prospective cohort study. Epidemiol Infect 143, 1731–1741, https://doi.org/10.1017/S0950268814002386 (2015).
Arnedo-Pena, A. et al. Vitamin D status and incidence of tuberculosis among contacts of pulmonary tuberculosis patients. Int J Tuberc Lung Dis 19, 65–69, https://doi.org/10.5588/ijtld.14.0348 (2015).
Talat, N., Perry, S., Parsonnet, J., Dawood, G. & Hussain, R. Vitamin d deficiency and tuberculosis progression. Emerging infectious diseases 16, 853–855, https://doi.org/10.3201/eid1605.091693 (2010).
Pilarski, A., Penn, N., Ratnakumar, S., Barker, R. D. & Milburn, H. J. Variation in vitamin D deficiency among tuberculosis patients by ethnic group and geographical region of birth: evidence from a diverse south London population. Eur Respir J 48, 1507–1510, https://doi.org/10.1183/13993003.00057-2016 (2016).
Bikle, D. Nonclassic actions of vitamin D. J Clin Endocrinol Metab 94, 26–34, https://doi.org/10.1210/jc.2008-1454 (2009).
Tsiaras, W. G. & Weinstock, M. A. Factors influencing vitamin D status. Acta Derm Venereol 91, 115–124, https://doi.org/10.2340/00015555-0980 (2011).
Dusso, A. S. et al. gamma-Interferon-induced resistance to 1,25-(OH)2 D3 in human monocytes and macrophages: a mechanism for the hypercalcemia of various granulomatoses. J Clin Endocrinol Metab 82, 2222–2232, https://doi.org/10.1210/jcem.82.7.4074 (1997).
Edfeldt, K. et al. T-cell cytokines differentially control human monocyte antimicrobial responses by regulating vitamin D metabolism. Proc Natl Acad Sci USA 107, 22593–22598, https://doi.org/10.1073/pnas.1011624108 (2010).
Selvaraj, P., Harishankar, M. & Afsal, K. Vitamin D: Immuno-modulation and tuberculosis treatment. Can J Physiol Pharmacol 93, 377–384, https://doi.org/10.1139/cjpp-2014-0386 (2015).
Chang, J. M. et al. 1-alpha,25-Dihydroxyvitamin D3 regulates inducible nitric oxide synthase messenger RNA expression and nitric oxide release in macrophage-like RAW 264.7 cells. J Lab Clin Med 143, 14–22, https://doi.org/10.1016/S0022214303001884 (2004).
Ralph, A. P. et al. L-arginine and vitamin D adjunctive therapies in pulmonary tuberculosis: a randomised, double-blind, placebo-controlled trial. PLoS One 8, e70032, https://doi.org/10.1371/journal.pone.0070032 (2013).
Martineau, A. R. et al. High-dose vitamin D(3) during intensive-phase antimicrobial treatment of pulmonary tuberculosis: a double-blind randomised controlled trial. Lancet 377, 242–250, https://doi.org/10.1016/S0140-6736(10)61889-2 (2011).
Wejse, C. et al. Vitamin D as supplementary treatment for tuberculosis: a double-blind, randomized, placebo-controlled trial. Am J Respir Crit Care Med 179, 843–850, https://doi.org/10.1164/rccm.200804-567OC (2009).
Daley, P. et al. Adjunctive vitamin D for treatment of active tuberculosis in India: a randomised, double-blind, placebo-controlled trial. Lancet Infect Dis 15, 528–534, https://doi.org/10.1016/S1473-3099(15)70053-8 (2015).
Salahuddin, N. et al. Vitamin D accelerates clinical recovery from tuberculosis: results of the SUCCINCT Study (Supplementary Cholecalciferol in recovery from tuberculosis). A randomized, placebo-controlled, clinical trial of vitamin D supplementation in patients with pulmonary tuberculosis’. BMC Infect Dis 13, 22, https://doi.org/10.1186/1471-2334-13-22 (2013).
Tukvadze, N. et al. High-dose vitamin D3 in adults with pulmonary tuberculosis: a double-blind randomized controlled trial. The American journal of clinical nutrition 102, 1059–1069, https://doi.org/10.3945/ajcn.115.113886 (2015).
Mily, A. et al. Significant Effects of Oral Phenylbutyrate and Vitamin D3 Adjunctive Therapy in Pulmonary Tuberculosis: A Randomized Controlled Trial. PLoS One 10, e0138340, https://doi.org/10.1371/journal.pone.0138340 (2015).
Nursyam, E. W., Amin, Z. & Rumende, C. M. The effect of vitamin D as supplementary treatment in patients with moderately advanced pulmonary tuberculous lesion. Acta Med Indones 38, 3–5 (2006).
Liu, P. T. et al. Toll-like receptor triggering of a vitamin D-mediated human antimicrobial response. Science 311, 1770–1773, https://doi.org/10.1126/science.1123933 (2006).
Shin, D. M. et al. Mycobacterial lipoprotein activates autophagy via TLR2/1/CD14 and a functional vitamin D receptor signalling. Cell Microbiol 12, 1648–1665, https://doi.org/10.1111/j.1462-5822.2010.01497.x.
Brodie, M. J. et al. Effect of rifampicin and isoniazid on vitamin D metabolism. Clin Pharmacol Ther 32, 525–530, https://doi.org/10.1038/clpt.1982.197 (1982).
Brodie, M. J. et al. Rifampicin and vitamin D metabolism. Clin Pharmacol Ther 27, 810–814, https://doi.org/10.1038/clpt.1980.115 (1980).
Brodie, M. J. et al. Effect of isoniazid on vitamin D metabolism and hepatic monooxygenase activity. Clin Pharmacol Ther 30, 363–367, https://doi.org/10.1038/clpt.1981.173 (1981).
Keflie, T. S., Nolle, N., Lambert, C., Nohr, D. & Biesalski, H. K. Vitamin D deficiencies among tuberculosis patients in Africa: A systematic review. Nutrition 31, 1204–1212, https://doi.org/10.1016/j.nut.2015.05.003 (2015).
Sloan, D. J. et al. Vitamin D deficiency in Malawian adults with pulmonary tuberculosis: risk factors and treatment outcomes. Int J Tuberc Lung Dis 19, 904–911, https://doi.org/10.5588/ijtld.15.0071 (2015).
Den Boon, S. et al. Association between smoking and tuberculosis infection: a population survey in a high tuberculosis incidence area. Thorax 60, 555–557, https://doi.org/10.1136/thx.2004.030924 (2005).
Zhang, H. et al. A dose-response relationship of smoking with tuberculosis infection: A cross-sectional study among 21008 rural residents in China. PLoS One 12, e0175183, https://doi.org/10.1371/journal.pone.0175183 (2017).
Bothamley, G. H. Smoking and tuberculosis: a chance or causal association? Thorax 60, 527–528, https://doi.org/10.1136/thx.2004.036012 (2005).
Plant, A. J. et al. Predictors of tuberculin reactivity among prospective Vietnamese migrants: the effect of smoking. Epidemiol Infect 128, 37–45 (2002).
Maurya, V., Vijayan, V. K. & Shah, A. Smoking and tuberculosis: an association overlooked. Int J Tuberc Lung Dis 6, 942–951 (2002).
O’Leary, S. M. et al. Cigarette smoking impairs human pulmonary immunity to Mycobacterium tuberculosis. Am J Respir Crit Care Med 190, 1430–1436, https://doi.org/10.1164/rccm.201407-1385OC (2014).
Feng, Y. et al. Exposure to cigarette smoke inhibits the pulmonary T-cell response to influenza virus and Mycobacterium tuberculosis. Infect Immun 79, 229–237, https://doi.org/10.1128/IAI.00709-10 (2011).
Stead, W. W., Senner, J. W., Reddick, W. T. & Lofgren, J. P. Racial differences in susceptibility to infection by Mycobacterium tuberculosis. N Engl J Med 322, 422–427, https://doi.org/10.1056/NEJM199002153220702 (1990).
Chiavegatto Filho, A. D. & Laurenti, R. (Racial/ethnic disparities in self-rated health: a multilevel analysis of 2,697 individuals in 145 Brazilian municipalities). Cad Saude Publica 29, 1572–1582, http://ref.scielo./ds7x2f (2013).
Cantwell, M. F., McKenna, M. T., McCray, E. & Onorato, I. M. Tuberculosis and race/ethnicity in the United States: impact of socioeconomic status. Am J Respir Crit Care Med 157, 1016–1020, https://doi.org/10.1164/ajrccm.157.4.9704036 (1998).
Ness, R. B., Haggerty, C. L., Harger, G. & Ferrell, R. Differential distribution of allelic variants in cytokine genes among African Americans and White Americans. Am J Epidemiol 160, 1033–1038, https://doi.org/10.1093/aje/kwh325 (2004).
Hoffmann, S. C. et al. Ethnicity greatly influences cytokine gene polymorphism distribution. Am J Transplant 2, 560–567 (2002).
San Pedro, A. & Oliveira, R. M. Tuberculosis and socioeconomic indicators: systematic review of the literature. Rev Panam Salud Publica 33, 294–301, https://goo.gl/dA2QM8 (2013).
Kim, S. & Crittenden, K. S. Risk factors for tuberculosis among inmates: a retrospective analysis. Public Health Nurs 22, 108–118, https://doi.org/10.1111/j.0737-1209.2005.220204.x (2005).
Winetsky, D. E. et al. Prevalence, risk factors and social context of active pulmonary tuberculosis among prison inmates in Tajikistan. PLoS One 9, e86046, https://doi.org/10.1371/journal.pone.0086046 (2014).
Farhat, M., Greenaway, C., Pai, M. & Menzies, D. False-positive tuberculin skin tests: what is the absolute effect of BCG and non-tuberculous mycobacteria? Int J Tuberc Lung Dis 10, 1192–1204 (2006).
Brasil. Manual de recomendações para o controle da tuberculose no Brasil. Ministério da Saúde (2011).
Balcells, M. E. et al. Association of vitamin D deficiency, season of the year, and latent tuberculosis infection among household contacts. PLoS One 12, e0175400, https://doi.org/10.1371/journal.pone.0175400 (2017).
Jacobs, E. T. & Mullany, C. J. Vitamin D deficiency and inadequacy in a correctional population. Nutrition 31, 659–663, https://doi.org/10.1016/j.nut.2014.10.010 (2015).
Brasil. Ministério da Justiça. Departamento Penitenciário Nacional (2014).
Kiggundu, D. S. et al. Vitamin D deficiency and its characteristics among patients with acute stroke at a national referral hospital in Kampala Uganda. BMC Endocr Disord 15, 53, https://doi.org/10.1186/s12902-015-0053-y (2015).
Thacher, T. D. & Clarke, B. L. Vitamin D insufficiency. Mayo Clinic proceedings 86, 50–60, https://doi.org/10.4065/mcp.2010.0567 (2011).
Maeda, S. S. et al. Recommendations of the Brazilian Society of Endocrinology and Metabology (SBEM) for the diagnosis and treatment of hypovitaminosis D. Arq Bras Endocrinol Metabol 58, 411–433, https://doi.org/10.1590/0004-2730000003388 (2014).
Acknowledgements
Financial supports provided by the Foundation for Support and Development of Education, Science and Technology of the State of Mato Grosso do Sul (Fundação de Apoio ao Desenvolvimento do Ensino, Ciência e Tecnologia do Estado do Mato Grosso do Sul) [07/2015] and U.S. Department of Health & Human Services, NIH, National Institute of Allergy and Infectious Diseases (NIAID) [R01 AI130058-01] are gratefully appreciated.
Author information
Authors and Affiliations
Contributions
E.B.M. was involved in the study conception and design, data analysis and manuscript drafting. C.C.M.G., J.R.A., A.I.K. and C.W.Y. were involved in the study design and manuscript review. J.C. was involved in conception, design, data analysis, coordination of the study and review the manuscript. All authors contributed in writing of the manuscript and approved submission of the final manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Maceda, E.B., Gonçalves, C.C.M., Andrews, J.R. et al. Serum vitamin D levels and risk of prevalent tuberculosis, incident tuberculosis and tuberculin skin test conversion among prisoners. Sci Rep 8, 997 (2018). https://doi.org/10.1038/s41598-018-19589-3
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-018-19589-3
This article is cited by
-
Vitamin D and the risk of latent tuberculosis infection: a systematic review and meta-analysis
BMC Pulmonary Medicine (2022)
-
Hypovitaminosis D among newly diagnosed pulmonary TB patients and their household contacts in Uganda
Scientific Reports (2022)
-
Positive Correlation Between General Public Knowledge and Attitudes Regarding COVID-19 Outbreak 1 Month After First Cases Reported in Indonesia
Journal of Community Health (2021)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.



