Abstract
The objective of the present study was to determine whether there was an association between single nucleotide polymorphisms (SNPs) in ABCG2 and gout. We recruited 333 participants including 210 patients with gout and 123 controls and genotyped 45 SNPs in both cohorts. We found that 24 SNPs in ABCG2 are susceptibility loci associated with gout. Haplotype analysis revealed five blocks across the ABCG2 locus were associated with an increased risk of gout with odds ratios (ORs) from 2.59–3.17 (all P < 0.0001). A novel finding in the present study was the identification of rs3114018 in block 3 and its association with increased gout risk. We found that the rs2231142T allele in block 2 and the rs3114018C-rs3109823T (C-T) risk haplotype in block 3 conferred the greatest evidence of association to gout risk (P = 1.19 × 10−12 and P = 9.20 × 10−11, respectively). Our study provides an improved understanding of ABCG2 variations in patients with gout and, as shown by haplotype analysis, that ABCG2 may have a role in gout susceptibility.
Similar content being viewed by others
Introduction
Gout is an arthritis that is characterized by elevated serum uric acid level, recurrent acute arthritis, and chronic tophaceous gout1,2,3. Epidemiological studies from several countries have found that the incidence and prevalence of gout may be increasing1,2,3,4,5. Moreover, women comprise approximately 5% of all patients with gout, but the incidence of gout in women has doubled in the past 20 years3,4,5. An increased serum uric acid concentration is because of either overproduction or under excretion of uric acid1. In over 90 percent of patients, gout is caused by the under excretion of uric acid1. Genome-wide association studies (GWAS) that scan the genome for common genetic variants associated with gout have greatly advanced our medical knowledge2, 6. The majority of genes associated with serum urate levels or gout are involved in the renal urate-transport system.
Gout is a complex disease with a multifactorial etiology involving genetic and environmental factors4, 5. Several molecules are associated with gout and hyperuricemia in various populations7,8,9,10,11,12,13,14,15,16,17,18,19,20. Moreover, several GWASs on gout and hyperuricemia have been performed to date, and more than 50 loci have been identified2, 7, 9, 14, 18,19,20. Recent GWAS have identified substantial associations between SNPs in ABCG2 and uric acid concentration and gout in different ethnic groups2, 7. ABCG2 (also known as BCRP) is located at a gout-susceptibility locus on chromosome 4q22, which was previously identified in several genome-wide linkage studies of gout7, 12, 20. ABCG2 mediates urate secretion in proximal renal tubule cells, the intestine, and the liver2, 8, 9. Furthermore, several studies have proposed that variations in ABCG2 may be important in the etiology of gout2, 7,8,9,10,11,12,13,14,15,16,17, 21. To verify further the impact of polymorphisms in genes related to gout, we studied common genetic variability in ABCG2 using a case-control study to clarify the association between SNPs or haplotypes at ABCG2 with the risk of gout in a Chinese population.
Methods
Study population
All enrolled patients were recruited at the Chang Gung Memorial Hospital (CGMH) at Tao-Yuan County (Taiwan) from February 2013 to March 2016. The study was approved by the local ethics committee and Institutional Review Board of Chang Gung Memorial Hospital (IRB 101-4659A3, 101-2636A3, and CMRPG3C1421-3). All participants provided written informed consent documents before entering the study. The methods carried out in accordance with the approved study protocol. A diagnosis of gout was based on the 1977 American College of Rheumatology diagnostic criteria22. All blood specimens were sent to the clinical laboratory at our hospital, which is certified by the College of American Pathologists (CAP) from the United States. External quality control for laboratory data was assessed by participation in the CAP’s international survey proficiency testing program and the National Quality Control Program conducted by the Taiwanese government.
SNP identification and genotyping
DNA from peripheral blood was isolated from 333 participants including 210 patients with gout and 123 individuals who are gout-free (controls). DNA were extracted from venous blood using standard procedures, including lysis of blood cells, protein hydrolysis using proteinase K, DNA purification by extraction with phenol-chloroform, and DNA precipitation with ethanol. Genomic DNA was isolated from lymphocytes of each participant using a QIAamp DNA Blood Mini Kit and the standard protocol of the manufacturer (Qiagen, Valencia, CA, USA) according to the manufacturer’s instructions. We followed strict quality control procedure. Forty-six SNPs were selected from a small scale preliminary study to identify gout-associated variants by targeted next-generation sequencing of ABCG2 gene23. Forty-six SNPs in ABCG2 on chromosome 4q22 were genotyped in our 210 cases and 123 controls using the Sequenom Mass-ARRAY platform and the standard protocol recommended by the manufacturer (Sequenom, San Diego, CA, USA). The call rate was ≥99.4% for all SNPs. During quality control review of genotyping data, we excluded one SNP (rs386677040) from further analysis as it was out of Hardy–Weinberg equilibrium (HWE; P < 0.05) in controls. Ultimately, the 45 SNPs that were in HWE (P > 0.05) were tested in our study cohorts.
Fine mapping of ABCG2 and haplotype analysis
We calculated linkage disequilibrium (LD) coefficients and constructed haplotypes using Haploview version 4.2 (Mark Daly’s Laboratory, Massachusetts Institute of Technology/Harvard Broad Institute, Cambridge, MA, USA)24. For haplotype construction, genotype data from both case and control groups were used to estimate intermarker LD by measuring pairwise D′ and r2, and to define LD blocks24, 25.
Statistical analysis
Categorical variables were expressed as percentages and were analyzed by chi-square (χ 2) test or Fisher’s exact test, as appropriate. Continuous variables were expressed as mean ± SD. All P values in this study were two sided, and P < 0.05 was considered statistically significant. SNP frequencies were tested for departure from HWE using an exact test in control subjects. Allele and genotype frequencies for each SNP were compared between patient and control cohorts using the χ 2 test. Odds ratios (ORs) and 95% confidence intervals (CIs) were calculated using logistic-regression analysis. In addition to obtaining nominal P values, empirical P values were generated by running 10,000 permutations using the Max (T) permutation procedure implemented in PLINK v1.0726. In addition, we applied Bonferroni correction and set the significance threshold for these analyses at α = 1.1 × 10−3, which corresponds to a stringent Bonferroni correction for testing 45 independent markers. All statistical analyses were performed using SPSS 20.0 (IBM Corp., Armonk, NY, USA). Each marker was tested for association using PLINK v1.07 (http://pngu.mgh.harvard.edu/purcell/plink/)26. Haploview (v4.2) was used for assessing LD patterns and haplotype association statistics24. Haplotype blocks were determined using the algorithm of Gabriel et al.27. An omnibus (or global) test of haplotype association was performed using PLINK. ORs and 95% CIs for haplotype-specific risks were calculated using VassarStats (http://vassarstats.net/).
Results
Characteristics of study subjects
Our study consisted of 333 participants of which 210 were patients with gout and 123 were controls. Detailed information of study participants is shown in Table 1. The mean age of affected individuals was 52.4 ± 12.9 years (range 20–85 years) with a male-to-female ratio of 201:9 (approximately 22.3:1), while the mean age of controls was 51.9 ± 11.83 years (range 27–81 years) with a male-to-female ratio of 107:16 (approximately 6.7:1) (Table 1). As gout primarily affects males, fewer females than males participated in this study. However, we found that there was no significant difference between cohorts in terms of age distribution (P = 0.588).
ABCG2 SNP analysis
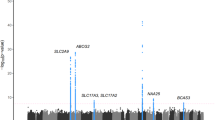
Forty-six SNPs were genotyped in patients (n = 210) and controls (n = 123). We calculated HWE for all SNPs and found that all were in HWE (P > 0.05) with the exception of rs386677040, which was excluded from further analysis. Detailed information of the 45 SNPs and the results of our association analysis with gout in the present study are presented in Table 2. Genomic position, nucleic acid composition, allele frequencies, summary OR, 95% CI, and significance level of these 45 SNPs are summarized in Table 2. Twenty-four SNPs (rs2231156, rs4148157, rs4693924, rs76979899, rs2725263, rs2054576, rs2622621, rs1481012, rs45499402, rs149027545, rs2231142, rs4148155, rs3114018, rs3109823, rs2725246, rs2725245, rs2622624, rs145778965, rs2725239, rs4148162, rs3841115, rs2622606, rs2622608, and rs2622609) were positively associated with gout risk. ORs of these 24 SNPs ranged from 2.32 to 3.29 (P values ranged from 1.70 × 10−7 to 1.82 × 10−12) (Table 2). The greatest evidence of association was found between the minor T allele of rs2231142 and an increased risk of gout, with a frequency of 0.586 in cases and 0.301 in controls (P = 1.19 × 10−12; Bonferroni corrected P = 5.36 × 10−11; OR = 3.29; 95% CI = 2.36–4.60). In contrast, 21 SNPs (rs1448784, rs4148160, rs2231164, rs34455506, rs2231148, rs12505410, rs200184409, rs5860118, rs397994425, rs45557042, rs11935697, rs3109824, rs2725256, rs2231138, rs3114017, rs2725254, rs12641369, rs4148152, rs2231137, rs1564481, and rs4148149) were associated with a decreased risk of gout (ORs ranged from 0.31 to 0.67, all P < 0.05). We found that rs3109824, rs2725256, rs3114017, rs2725254, and rs1564481 were not associated with gout in our affected cohort (P > 0.05). In addition, following adjustments for Bonferroni correction for testing 45 independent tests (α < 0.0011), we found that all SNPs with the exception of eight SNPs (rs45557042, rs11935697, rs3109824, rs2725256, rs2231138, rs3114017, rs2725254, and rs1564481) remained significantly associated with gout. We found that the inclusion of age and gender as covariates in logistic regression models did not substantially change the significance of the observed associations (data not shown).
Linkage disequilibrium plot and haplotype analysis
Using Haploview v4.2, we generated an LD plot of the 45 genotyped SNPs in ABCG2 in our affected cohort (Fig. 1), and found that more than half of the SNPs tested in the present study were highly correlated with each other (r2 ≥ 0.80), of which 220 out of 990 (22.2%) pairs revealed perfect linkage disequilibrium (D′ = 1). Moreover, the haplotype block structure spanning ABCG2 as derived by Haploview is shown in Fig. 1b with haplotype frequencies shown in both Fig. 1b and in Table 3. We found that there are five haplotype blocks across the ABCG2 locus. Based on the model of Gabriel et al.26, we identified a total of 25 common haplotypes in these five blocks (Table 3), which span approximately 16 kb, 33 kb, <1 kb, 15 kb, and 1 kb derived from 5, 11, 3, 4, and 2 SNPs, respectively (Fig. 1a). Haploview predicted 34 possible connections of haplotypes for recombination between blocks at a frequency >1%. In addition, calculating the frequency of recombination between blocks as a value of a multiallelic D′ coefficient, we found values of 0.85 between blocks 1 and 2, 0.91 between blocks 2 and 3, 0.84 between blocks 3 and 4, and 0.97 between blocks 4 and 5 (Fig. 1b and Table 3). Among the identified 25 common haplotypes, five haplotypes were associated with an increased risk for gout (all permuted P < 0.0001) and seven haplotypes were associated with a decreased risk for gout (Table 3). The five haplotypes associated with an increased risk of gout following correction with 10,000 permutations were the A-C-C-A-A-A-G-T-C-T haplotype in block 1 (OR = 2.67; 95% CI = 1.87–3.81; P < 0.0001), the G-T-G-T-A-A-G-G-C-C-A-T-A-T-T-G-C-C-G-T-C-C-T haplotype in block 2 (OR = 2.59; 95% CI = 1.81–3.69; P < 0.0001), the C-T haplotype in block 3 (OR = 3.16; 95% CI = 2.21–4.50; P < 0.0001), the A-A-C-C-A-A-A-T haplotype in block 4 (OR = 2.76; 95% CI = 1.89–4.04; P < 0.0001), and the T-C haplotype in block 5 (OR = 2.94; 95% CI = 2.05–4.22; P < 0.0001) (Table 3). The haplotype that conferred the greatest risk was the rs3114018C-rs3109823T haplotype (C-T) in block 3, with a haplotype frequency of 0.816 in affected individuals and 0.585 in controls (P = 9.20 × 10−11; OR = 3.16; 95% CI = 2.21–4.50; Table 3).
Linkage disequilibrium (LD) plots for ABCG2 and haplotype block structure across the ABCG2 locus. (a) Haploview plot defining haplotype block structure of the ABCG2 locus. The white horizontal bar in the upper diagram illustrates the location of each SNP on a physical scale. Each box provides estimated statistics of the coefficient of determination (r2). The diamond without a number corresponds to a D′ of 1. (b) Haplotypes in the haplotype blocks spanning the ABCG2 locus. There are five haplotype blocks across the region. Haplotype frequencies are shown to the right of each haplotype. SNP numbers across the top of the haplotypes correspond to those shown in the Haploview plot. A multiallelic D′ statistic, which indicates the level of recombination between two blocks, is shown in the crossing area. Connections from one block to the next are shown for haplotypes with a frequency >10% as thick lines and a frequency >1% as thin lines.
Discussion
Gout is an increasing global health problem caused by multiple genetic and environmental factors. In recent years, many variants in a growing number of genes involved in the pathogenesis of gout and hyperuricemia have been identified2, 9. ABCG2 is located at a gout-susceptibility locus on chromosome 4q22, which was previously identified in several genome-wide linkage studies of gout7, 12, 20. ABCG2, which is also known as breast cancer resistance protein (BCRP), is a high-capacity urate exporter, the dysfunction of which increases the risk of gout and hyperuricemia8, 28. ABCG2 mediates renal urate secretion as a urate efflux transporter in the brush-border membrane on the luminal surface of kidney proximal tubule cells2, 8, 9. In addition, ABCG2 is expressed at high levels in the intestine and liver27 and functions as an efflux transporter for many drugs and molecule substrates, including anticancer agents, antibiotics, antivirals, HMG-CoA reductase inhibitors, flavonoids, allopurinol, and uric acid28,29,30,31,32,33,34.
Using haplotype analyses, we found five blocks of LD that were significantly associated with gout. Moreover, an LD plot of ABCG2 demonstrated extensive correlation among 45 SNPs. Based on measures of r2, perfect linkage (r2 = 1) was detected in 220 out of the 990 pairs (D′ = 1) and strong LD (1 > |D′| ≥ 0.8) was detected in more than half of variant pairs. A novel finding in the present study was the identification of rs3114018 in block 3 and its association with increased gout risk. In addition, the minor T allele of rs2231142 in the second block of ABCG2 was associated with an increased risk of gout (OR = 3.29; 95% CI = 2.36–4.60), a finding that was similarly reported in previous studies in other populations7, 10,11,12, 17, 21, 33, 35,36,37.
Two independent functional studies of ABCG2 found that the Q141K (rs2231142) polymorphism occurs in a highly conserved region of the gene and is a loss-of-function mutation8, 33. These studies found that the rs2231142 risk allele resulted in a urate secretory transporter with a 53% reduced ability to transport urate in Xenopus oocytes8 and HEK293 membrane vesicles33. Moreover, Abcg2-knockout mice had increased serum uric acid levels and renal urate excretion, and decreased intestinal urate excretion28. Furthermore, Woodward et al.37 demonstrated the utility of using small molecules to correct the Q141K defect in expression and function as a potential therapeutic approach for hyperuricemia and gout. The association between the rs2231142T allele and the risk of gout has been replicated in many diverse study populations including Caucasian7, 11, 12, 33, African7, Japanese20, 33, 35, Mexican-American12, Native American12, Han Chinese10, 17, 36, and New Zealand Pacific Island ancestry11. These findings indicate that ABCG2 may have specific and important functions in the pathology of gout. However, an association between rs2231142 and gout has not been found in Maori populations11 and some studies in the Chinese population21, 38. The reason for this discrepancy is not known, but the difference may be because of either differences in gene structure or sampling bias13. Furthermore, an additional confounding factor is that the etiology of gout is linked to various genetic and environmental factors such as lifestyle and diet1, 5, 35, 39. However, the baseline socioeconomic status and diet habit were not available in the database, so we were unable to perform the analysis.
In this study, we thoroughly captured common genetic variation across ABCB2 and performed a comprehensive evaluation of common SNPs at ABCB2 associated with gout risk. Using haplotype analysis, we found five haplotype blocks that were associated with an increased risk of gout: block 1 with an OR of 2.67 (95% CI = 1.87–3.81), block 2 with an OR of 2.59 (95% CI = 1.81–3.69), block 3 with an OR of 3.16 (95% CI = 2.21–4.50), block 4 with an OR of 2.76 (95% CI = 1.89–4.04), and block 5 with an OR of 2.94 (95% CI = 2.05–4.22; all P < 0.0001) (Table 3). Our results, combined with those from previous studies, suggest that genetic variation in ABCG2 may influence gout susceptibility in the Han Chinese population. Consistent with the genetic susceptibility identified in patients with gout in several other populations, we observed that the minor allele of rs2231142 was associated with an increased risk for gout7, 10,11,12, 17, 20, 33, 35,36,37, while we found other SNPs in the present study that may confer a protective effect on susceptibility to gout. This finding is consistent with the hypothesis of two functional polymorphisms near the SNPs evaluated in this study, one that increases the risk of developing gout whereas the second confers a protective effect40. In addition, considering that the genomic regions of the five SNP haplotype blocks are characterized by high LD, we postulate that such SNPs are likely to tag any hitherto unidentified common SNPs in the candidate gene. For example, two recent studies from northwest China21, 38 found a significant difference in mean serum urate levels between a novel SNP, rs3114018, in ABCG2 and gout risk, which is consistent with the findings of the present study. In addition to rs2231142 in block 2, the greatest evidence of association in the present study was between the C-T risk haplotype of rs3114018 and rs3109823 in block 3. To the best of our knowledge, the relationships demonstrated in the present study between SNPs in blocks 1, 3, 4, and 5 with gout, such as the novel SNP rs3114018 in block 3 have not been previously observed until recently21, 38. Of note, rs2231137 is located in the same block with rs2231142 (block 2), resulting in a V12M substitution (p = 1.4 × 10−9). Our findings and previous studies10, 17 indicated that V12M substitution was associated with a decreased risk of hyperuricemia and/or gout. However, in vitro functional assays showed that V12M substitution did not result in any changes in protein expression and risk to phenotypes such as serum urate levels and gout33, 41. Further studies are required to elucidate the functional contributions of these novel SNPs in these genomic regions or blocks that confer increased risk for gout.
The present study had the following limitations. First, although we could identify genetic associations with gout, we could not elucidate the underlying causal mechanisms. Nonetheless, our findings with rs2231142 and rs3114018 are consistent with those of studies of other populations, which highlight their robustness and support for a role in gout. Second, considering the marked difference in SNP minor allele frequencies among populations, ethnic differences may exist, which would confound the identification of genetic risk factors for gout2, 9, 42. Future studies should incorporate larger sample sizes to verify present findings across more populations. Finally, the biological functions of other SNPs in ABCG2 have not been fully characterized, and therefore, the findings from the present study require functional confirmation by future expression studies.
In conclusion, this large-scale thorough evaluation of SNPs has identified common genetic variants in ABCG2 that are associated with gout risk. None of the tested SNPs, with the exception of rs2231142, which were identified as significant in this study were listed among the most significant results of three recently conducted GWAS on gout7, 12, 20. In addition to rs2231142, haplotype analysis of polymorphisms in ABCG2 revealed SNP-derived haplotypes associated with gout risk. Further identification of the functional and causal variant(s) in ABCG2 will lead to a better understanding of the mechanism underlying the development of gout pathologies.
References
Choi, H. K., Mount, D. B. & Reginato, A. M. Pathogenesis of gout. Ann Intern Med 143, 499–516 (2005).
Reginato, A. M., Mount, D. B., Yang, I. & Choi, H. K. The genetics of hyperuricaemia and gout. Nat Rev Rheumatol 8, 610–621 (2102).
Zhu, Y., Pandya, B. J. & Choi, H. K. Prevalence of gout and hyperuricemia in the US general population the National Health and Nutrition Examination Survey 2007–2008. Arthritis Rheum 63, 3136–3141 (2011).
Kuo, C. F., Grainge, M. J., Zhang, W. & Doherty, M. Global epidemiology of gout: prevalence, incidence and risk factors. Nat Rev Rheumatol 11, 649–662 (2015).
Kuo, C. F., Grainge, M. J. & See, L. C. et al. Familial aggregation of gout and relative genetic and environmental contributions: a nationwide population study in Taiwan. Ann Rheum Dis 74, 369–374 (2015).
Clarke, A. & Vyse, T. J. Genetics of rheumatic disease. Arthritis Res Ther 11, 248 (2009).
Dehghan, A., Köttgen, A. & Yang, Q. et al. Association of three genetic loci with uric acid concentration and risk of gout: a genome-wide association study. Lancet 372, 1953–1961 (2008).
Woodward, O. M. et al. Identification of a urate transporter, ABCG2, with a common functional polymorphism causing gout. Proc Natl Acad Sci USA 106, 10338–10342 (2009).
Merriman, T. R. An update on the genetic architecture of hyperuricemia and gout. Arthritis Res Ther 17, 98 (2105).
Zhou, D. et al. Functional polymorphisms of the ABCG2 gene are associated with gout disease in the Chinese Han male population. Int J Mol Sci 15, 9149–9159 (2104).
Phipps-Green, A. J., Hollis-Moffatt, J. E. & Dalbeth, N. et al. A strong role for the ABCG2 gene in susceptibility to gout in New Zealand Pacific Island and Caucasian, but not Māori, case and control sample sets. Hum Mol Genet 19, 4813–4819 (2010).
Zhang, L., Spencer, K. L. & Voruganti, V. S. et al. Association of functional polymorphism rs2231142 (Q141K) in the ABCG2 gene with serum uric acid and gout in 4 US populations: the PAGE Study. Am J Epidemiol 177, 923–932 (2003).
Lv, X., Zhang, Y. & Zeng, F. et al. The association between the polymorphism rs2231142 in the ABCG2 gene and gout risk: a meta-analysis. Clin Rheumatol 33, 1801–1805 (2104).
Anzai, N., Jutabha, P., Amonpatumrat-Takahashi, S. & Sakurai, H. Recent advances in renal urate transport: characterization of candidate transporters indicated by genome-wide association studies. Clin Exp Nephrol 16, 89–95 (2012).
Matsuo, H., Takada, T. & Ichida, K. et al. Identification of ABCG2 dysfunction as a major factor contributing to gout. Nucleosides Nucleotides Nucleic Acids 30, 1098–1104 (2011).
Matsuo, H., Ichida, K. & Takada, T. et al. Common dysfunctional variants in ABCG2 are a major cause of early-onset gout. Sci Rep 3, 2014 (2013).
Tu, H. P., Ko, A. M. & Chiang, S. L. et al. Joint effects of alcohol consumption and ABCG2 Q141K on chronic tophaceous gout risk. J Rheumatol 41, 749–758 (2104).
Kolz, M., Johnson, T. & Sanna, S. et al. Meta-analysis of 28,141 individuals identifies common variants within five new loci that influence uric acid concentrations. PLoS Genet 5, e1000504 (2009).
Köttgen, A., Albrecht, E. & Teumer, A. et al. Genome-wide association analyses identify 18 new loci associated with serum urate concentrations. Nat Genet 45, 145–154 (2013).
Matsuo, H. et al. Genome-wide association study of clinically defined gout identifies multiple risk loci and its association with clinical subtypes. Ann Rheum Dis 75, 652–659 (2016).
Jiri, M. et al. Genetic variation in the ABCG2 gene is associated with gout risk in the Chinese Han population. Clin Rheumatol 35, 159–63 (2016).
Wallace, S. et al. Preliminary criteria for the classification of the acute arthritis of primary gout. Arthritis Rheum 20, 895–900 (1997).
Li, J. et al. Identification of high-quality cancer prognostic markers and metastasis network modules. Nat Commun 1, 34 (2010).
Barrett, J. C., Fry, B., Maller, J. & Daly, M. J. Haploview: analysis and visualization of LD and haplotype maps. Bioinformatics 21, 263–5 (2005).
Jurinke, C., van den Boom, D., Cantor, C. R. & Koster, H. The use of massarray technology for high throughput genotyping. Adv Biochem Eng Biotechnol 77, 57–74 (2002).
Purcell, S. et al. PLINK: a tool set for whole-genome association and population-based linkage analyses. Am J Hum Genet 81, 559–75 (2007).
Gabriel, S. B. et al. The structure of haplotype blocks in the human genome. Science 296, 2225–2229 (2002).
Ichida, K. et al. Decreased extra-renal urate excretion is a common cause of hyperuricemia. Nat Commun 3, 764 (2012).
Woodward, O. M., Köttgen, A. & Köttgen, M. ABCG transporters and disease. FEBS J 278, 3215–3225 (2011).
Horsey, A. J., Cox, M. H., Sarwat, S. & Kerr, I. D. The multidrug transporter ABCG2: still more questions than answers. Biochem Soc Trans 44, 824–830 (2016).
Hosomi, A., Nakanishi, T., Fujita, T. & Tamai, I. Extra-renal elimination of uric acid via intestinal efflux transporter BCRP/ABCG2. PLoS One 7, e30456 (2102).
Wen, C. C. et al. Genome-wide association study identifies ABCG2 (BCRP) as an allopurinol transporter and a determinant of drug response. Clin Pharmacol Ther 97, 518–525 (2015).
Matsuo, H., Takada, T. & Ichida, K. et al. Common defects of ABCG2, a high-capacity urate exporter, cause gout: A function-based genetic analysis in a Japanese population. Sci Transl Med 1, 5ra11 (2009).
Tamura, A. et al. Re-evaluation and functional classification of non-synonymous single nucleotide polymorphisms of the human ATP-binding cassette transporter ABCG2. Cancer Sci 98, 231–239 (2007).
Yamagishi, K., Tanigawa, T. & Kitamura, A. et al. The rs2231142 variant of the ABCG2 gene is associated with uric acid levels and gout among Japanese people. Rheumatology 49, 1461–1465 (2010).
Wang, B. et al. Genetic analysis of ABCG2 gene C421A polymorphism with gout disease in Chinese Han male population. Hum Genet 127, 245–246 (2010).
Woodward, O. M. et al. Gout-causing Q141K mutation in ABCG2 leads to instability of the nucleotide-binding domain and can be corrected with small molecules. Proc Natl Acad Sci USA 110, 5223–5228 (2013).
Ren, Y. C. et al. PDK2 and ABCG2 genes polymorphisms are correlated with blood glucose levels and uric acid in Tibetan gout patients. Genet Mol Res 15, gmr.15017447 (2016).
Krishnan, E., Lienesch, D. & Kwoh, C. K. Gout in ambulatory care settings in the United States. J Rheumatol 35, 498–501 (2008).
Sokolova, E. A. et al. Association of SNPs of CD40 gene with multiple sclerosis in Russians. PLoS One 8, e61032 (2013).
Cleophas, M. C. et al. ABCG2 polymorphisms in gout: insights into disease susceptibility and treatment approaches. Pharmgenomics Pers Med 10, 129–142 (2017).
Maekawa, K. et al. Genetic variation and haplotype structure of the ABC transporter gene ABCG2 in a Japanese population. Drug Metabolism and Pharmacokinetics 21, 109–121 (2006).
Acknowledgements
We thank the Chang Gung Memorial Hospital (projects CMRPG3C1421-3 and CMRPG3C0361) for financial support. The sponsor of the study had no role in its design and conduct; the collection, management, analysis, and interpretation of the data; or the preparation, review, or approval of the manuscript for publication.
Author information
Authors and Affiliations
Contributions
K.H.Y., S.C.C., and J.J.L. conceived and designed the methodology used in the manuscript and coordination of the analyses, K.H.Y. and F.S.L. collected the data, P.Y.C., S.C.C., L.A.W. performed the experiments, K.H.Y. and J.J.L. wrote the draft manuscript, and all authors interpretation of the results and critically reviewed and revised the manuscript.
Corresponding author
Ethics declarations
Competing Interests
The authors declare that they have no competing interests.
Additional information
Publisher's note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Yu, KH., Chang, PY., Chang, SC. et al. A comprehensive analysis of the association of common variants of ABCG2 with gout. Sci Rep 7, 9988 (2017). https://doi.org/10.1038/s41598-017-10196-2
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41598-017-10196-2
This article is cited by
-
Genetic assessment of hyperuricemia and gout in Asian, Native Hawaiian, and Pacific Islander subgroups of pregnant women: biospecimens repository cross-sectional study
BMC Rheumatology (2022)
-
Multidrug efflux transporter ABCG2: expression and regulation
Cellular and Molecular Life Sciences (2021)
-
The association between genetic polymorphisms in ABCG2 and SLC2A9 and urate: an updated systematic review and meta-analysis
BMC Medical Genetics (2020)
-
Cellular expression and function of naturally occurring variants of the human ABCG2 multidrug transporter
Cellular and Molecular Life Sciences (2020)
-
Integrative Genome-Wide Association Studies of eQTL and GWAS Data for Gout Disease Susceptibility
Scientific Reports (2019)
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.