Abstract
Sleep is assumed to subserve homeostatic processes in the brain; however, the set point around which sleep tunes circuit computations is unknown. Slow-wave activity (SWA) is commonly used to reflect the homeostatic aspect of sleep; although it can indicate sleep pressure, it does not explain why animals need sleep. This study aimed to assess whether criticality may be the computational set point of sleep. By recording cortical neuron activity continuously for 10–14 d in freely behaving rats, we show that normal waking experience progressively disrupts criticality and that sleep functions to restore critical dynamics. Criticality is perturbed in a context-dependent manner, and waking experience is causal in driving these effects. The degree of deviation from criticality predicts future sleep/wake behavior more accurately than SWA, behavioral history or other neural measures. Our results demonstrate that perturbation and recovery of criticality is a network homeostatic mechanism consistent with the core, restorative function of sleep.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
The raw datasets generated in this study constitute >10 terabytes of raw neural broadband. The data are stored in a cost-efficient manner not immediately accessible to the Internet. Data are available upon request (khengen@wustl.edu). Source data are provided with this paper.
Code availability
Code is available on GitHub (https://github.com/hengenlab/Sleep_restores_criticality).
References
Cirelli, C. & Tononi, G. Is sleep essential? PLoS Biol. 6, e216 (2008).
Lim, J. & Dinges, D. F. A meta-analysis of the impact of short-term sleep deprivation on cognitive variables. Psychol. Bull. 136, 375–389 (2010).
Buzsáki, G. Hippocampal sharp wave–ripple: a cognitive biomarker for episodic memory and planning. Hippocampus 25, 1073–1188 (2015).
Majde, J. A. & Krueger, J. M. Links between the innate immune system and sleep. J. Allergy Clin. Immunol. 116, 1188–1198 (2005).
McDermott, C. M. et al. Sleep deprivation causes behavioral, synaptic, and membrane excitability alterations in hippocampal neurons. J. Neurosci. 23, 9687–9695 (2003).
Borbély, A. A., Daan, S., Wirz-Justice, A. & Deboer, T. The two-process model of sleep regulation: a reappraisal. J. Sleep Res. 25, 131–143 (2016).
Dijk, D. J. & Czeisler, C. A. Contribution of the circadian pacemaker and the sleep homeostat to sleep propensity, sleep structure, electroencephalographic slow waves, and sleep spindle activity in humans. J. Neurosci. 15, 3526–3538 (1995).
Franken, P., Chollet, D. & Tafti, M. The homeostatic regulation of sleep need is under genetic control. J. Neurosci. 21, 2610–2621 (2001).
Thomas, C. W. et al. Global sleep homeostasis reflects temporally and spatially integrated local cortical neuronal activity. eLife 9, e54148 (2020).
Borbély, A. A. From slow waves to sleep homeostasis: new perspectives. Arch. Ital. Biol. 139, 53–61 (2001).
Hubbard, J. et al. Rapid fast-delta decay following prolonged wakefulness marks a phase of wake-inertia in NREM sleep. Nat. Commun. 11, 3130 (2020).
Adam, K. in Progress in Brain Research Vol. 53 (eds McConnell, P. S. et al.) 289–305 (Elsevier, 1980).
Seibt, J. et al. Protein synthesis during sleep consolidates cortical plasticity in vivo. Curr. Biol. 22, 676–682 (2012).
O’Connor, T. Emergent properties. Am. Philos. Q. 31, 91–104 (1994).
Abásolo, D., Simons, S., Morgado da Silva, R., Tononi, G. & Vyazovskiy, V. V. Lempel–Ziv complexity of cortical activity during sleep and waking in rats. J. Neurophysiol. 113, 2742–2752 (2015).
Huber, R., Felice Ghilardi, M., Massimini, M. & Tononi, G. Local sleep and learning. Nature 430, 78–81 (2004).
Milinski, L. et al. Waking experience modulates sleep need in mice. BMC Biol. 19, 65 (2021).
Tsao, A. et al. Integrating time from experience in the lateral entorhinal cortex. Nature 561, 57–62 (2018).
Shew, W. L. & Plenz, D. The functional benefits of criticality in the cortex. Neuroscientist 19, 88–100 (2013).
Beggs, J. M. & Plenz, D. Neuronal avalanches in neocortical circuits. J. Neurosci. 23, 11167–11177 (2003).
O’Byrne, J. & Jerbi, K. How critical is brain criticality? Trends Neurosci. 45, 820–837 (2022).
Cramer, B. et al. Control of criticality and computation in spiking neuromorphic networks with plasticity. Nat. Commun. 11, 2853 (2020).
Langton, C. G. Computation at the edge of chaos: phase transitions and emergent computation. Physica D 42, 12–37 (1990).
Massobrio, P., de Arcangelis, L., Pasquale, V., Jensen, H. J. & Plenz, D. Criticality as a signature of healthy neural systems. Front. Syst. Neurosci. 9, 22 (2015).
Heynen, A. J. et al. Molecular mechanism for loss of visual cortical responsiveness following brief monocular deprivation. Nat. Neurosci. 6, 854–862 (2003).
Ma, Z., Turrigiano, G. G., Wessel, R. & Hengen, K. B. Cortical circuit dynamics are homeostatically tuned to criticality in vivo. Neuron 104, 655–664 (2019).
Turrigiano, G. G. & Nelson, S. B. Homeostatic plasticity in the developing nervous system. Nat. Rev. Neurosci. 5, 97–107 (2004).
Pearlmutter, B. A. & Houghton, C. J. A new hypothesis for sleep: tuning for criticality. Neural Comput. 21, 1622–1641 (2009).
Meisel, C., Klaus, A., Vyazovskiy, V. V. & Plenz, D. The interplay between long- and short-range temporal correlations shapes cortex dynamics across vigilance states. J. Neurosci. 37, 10114–10124 (2017).
Meisel, C., Olbrich, E., Shriki, O. & Achermann, P. Fading signatures of critical brain dynamics during sustained wakefulness in humans. J. Neurosci. 33, 17363–17372 (2013).
Longordo, F., Kopp, C. & Lüthi, A. Consequences of sleep deprivation on neurotransmitter receptor expression and function. Eur. J. Neurosci. 29, 1810–1819 (2009).
Rechtschaffen, A., Gilliland, M. A., Bergmann, B. M. & Winter, J. B. Physiological correlates of prolonged sleep deprivation in rats. Science 221, 182–184 (1983).
Cardin, J. A., Palmer, L. A. & Contreras, D. Stimulus feature selectivity in excitatory and inhibitory neurons in primary visual cortex. J. Neurosci. 27, 10333–10344 (2007).
Hengen, K. B., Lambo, M. E., Van Hooser, S. D., Katz, D. B. & Turrigiano, G. G. Firing rate homeostasis in visual cortex of freely behaving rodents. Neuron 80, 335–342 (2013).
Hengen, K. B., Pacheco, A. T., McGregor, J. N., Van Hooser, S. D. & Turrigiano, G. G. Neuronal firing rate homeostasis is inhibited by sleep and promoted by wake. Cell 165, 180–191 (2016).
Niell, C. M. & Stryker, M. P. Highly selective receptive fields in mouse visual cortex. J. Neurosci. 28, 7520–7536 (2008).
Watson, B. O., Levenstein, D., Greene, J. P., Gelinas, J. N. & Buzsaki, G. Network homeostasis and state dynamics of neocortical sleep. Neuron 90, 839–852 (2016).
Touboul, J. & Destexhe, A. Power-law statistics and universal scaling in the absence of criticality. Phys. Rev. E 95, 012413 (2017).
Vyazovskiy, V. V. et al. Cortical firing and sleep homeostasis. Neuron 63, 865–878 (2009).
Kim, J., Gulati, T. & Ganguly, K. Competing roles of slow oscillations and delta waves in memory consolidation versus forgetting. Cell 179, 514–526 (2019).
Vyazovskiy, V., Borbély, A. A. & Tobler, I. Unilateral vibrissae stimulation during waking induces interhemispheric EEG asymmetry during subsequent sleep in the rat. J. Sleep Res. 9, 367–371 (2000).
Saleem, A. B., Ayaz, A., Jeffery, K. J., Harris, K. D. & Carandini, M. Integration of visual motion and locomotion in mouse visual cortex. Nat. Neurosci. 16, 1864–1869 (2013).
Guitchounts, G., Masís, J., Wolff, S. B. E. & Cox, D. Encoding of 3D head orienting movements in the primary visual cortex. Neuron 108, 512–525 (2020).
Wilting, J. & Priesemann, V. Inferring collective dynamical states from widely unobserved systems. Nat. Commun. 9, 2325 (2018).
Havekes, R. & Aton, S. J. Impacts of sleep loss versus waking experience on brain plasticity: parallel or orthogonal? Trends Neurosci. 43, 385–393 (2020).
Scarpetta, S. et al. Criticality of neuronal avalanches in human sleep and their relationship with sleep macro- and micro-architecture. iScience 26, 107840 (2023).
Ponce-Alvarez, A., Jouary, A., Privat, M., Deco, G. & Sumbre, G. Whole-brain neuronal activity displays crackling noise dynamics. Neuron 100, 1446–1459 (2018).
Shew, W. L. et al. Adaptation to sensory input tunes visual cortex to criticality. Nat. Phys. 11, 659–663 (2015).
Achermann, P. & Borbély, A. A. Mathematical models of sleep regulation. Front. Biosci. 8, s683–s693 (2003).
Bridi, M. C. D. et al. Daily oscillation of the excitation–inhibition balance in visual cortical circuits. Neuron 105, 621–629 (2020).
Hensch, T. K. Critical period plasticity in local cortical circuits. Nat. Rev. Neurosci. 6, 877–888 (2005).
Tononi, G. & Cirelli, C. Sleep and the price of plasticity: from synaptic and cellular homeostasis to memory consolidation and integration. Neuron 81, 12–34 (2014).
Cirelli, C. Sleep, synaptic homeostasis and neuronal firing rates. Curr. Opin. Neurobiol. 44, 72–79 (2017).
de Vivo, L. et al. Ultrastructural evidence for synaptic scaling across the wake/sleep cycle. Science 355, 507–510 (2017).
Aton, S. J. et al. Mechanisms of sleep-dependent consolidation of cortical plasticity. Neuron 61, 454–466 (2009).
Frank, M. G. & Cantera, R. Sleep, clocks, and synaptic plasticity. Trends Neurosci. 37, 491–501 (2014).
Morrell, M. C., Nemenman, I. & Sederberg, A. J. Avalanche scaling in large neural populations with distributed coupling to multiple dynamical latent variables. Preprint at arXiv https://doi.org/10.48550/arXiv.2301.00759 (2023).
Stepp, N., Plenz, D. & Srinivasa, N. Synaptic plasticity enables adaptive self-tuning critical networks. PLoS Comput. Biol. 11, e1004043 (2015).
Zeraati, R., Priesemann, V. & Levina, A. Self-organization toward criticality by synaptic plasticity. Front. Phys. https://doi.org/10.3389/fphy.2021.619661 (2021).
Severin, D. et al. All-or-none disconnection of pyramidal inputs onto parvalbumin-positive interneurons gates ocular dominance plasticity. Proc. Natl Acad. Sci. USA 118, e2105388118 (2021).
Ponce-Alvarez, A., Kringelbach, M. L. & Deco, G. Critical scaling of whole-brain resting-state dynamics. Commun. Biol. 6, 627 (2023).
Sulaman, B. A., Wang, S., Tyan, J. & Eban-Rothschild, A. Neuro-orchestration of sleep and wakefulness. Nat. Neurosci. 26, 196–212 (2023).
Buccino, A. P. et al. SpikeInterface, a unified framework for spike sorting. eLife 9, e61834 (2020).
Chung, J. E. et al. A fully automated approach to spike sorting. Neuron 95, 1381–1394 (2017).
Mathis, A. et al. DeepLabCut: markerless pose estimation of user-defined body parts with deep learning. Nat. Neurosci. 21, 1281–1289 (2018).
dos Santos Lima, G. Z. et al. Hippocampal and cortical communication around micro-arousals in slow-wave sleep. Sci. Rep. 9, 5876 (2019).
Miyawaki, H., Billeh, Y. N. & Diba, K. Low activity microstates during sleep. Sleep 40, zsx066 (2017).
Poil, S.-S., Hardstone, R., Mansvelder, H. D. & Linkenkaer-Hansen, K. Critical-state dynamics of avalanches and oscillations jointly emerge from balanced excitation/inhibition in neuronal networks. J. Neurosci. 32, 9817–9823 (2012).
Marshall, N. et al. Analysis of power laws, shape collapses, and neural complexity: new techniques and MATLAB support via the NCC toolbox. Front. Physiol. 7, 250 (2016).
Fosque, L. J., Williams-Garcia, R. V., Beggs, J. M. & Ortiz, G. Evidence for quasicritical brain dynamics. Phys. Rev. Lett. 126, 098101 (2021).
Sela, Y., Vyazovskiy, V. V., Cirelli, C., Tononi, G. & Nir, Y. Responses in rat core auditory cortex are preserved during sleep spindle oscillations. Sleep 39, 1069–1082 (2016).
Spano, G. M. et al. Sleep deprivation by exposure to novel objects increases synapse density and axon–spine interface in the hippocampal CA1 region of adolescent mice. J. Neurosci. 39, 6613–6625 (2019).
Spitzner, F. P. et al. MR. Estimator, a toolbox to determine intrinsic timescales from subsampled spiking activity. PLoS ONE 16, e0249447 (2021).
Chen, T. & Guestrin, C. XGBoost: a scalable tree boosting system. In Proc. 22nd ACM SIGKDD International Conference on Knowledge Discovery and Data Mining 785–794 (Association for Computing Machinery, 2016); https://doi.org/10.1145/2939672.2939785
Acknowledgements
We would like to thank V. Priesemann and M. Frank for helpful discussions. This project was supported by National Institutes of Health BRAIN (Brain Research through Advancing Innovative Neurotechnologies) Initiative award R01NS118442 (K.B.H.) and the Incubator for Transdisciplinary Futures, an Arts & Sciences signature initiative at Washington University in St. Louis (K.B.H.).
Author information
Authors and Affiliations
Contributions
Y.X. developed code, ran analyses, performed surgeries, collected data and contributed to the writing and figure production. A.S. built the model for sleep/wake prediction. R.W. provided mentorship and intellectual and technical consultation. K.B.H. led, directed and envisioned the project, edited figures and wrote the paper.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Neuroscience thanks Gustavo Deco, Vladyslav Vyazovskiy and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Variability within animals over days in the distribution of wake and sleep substates.
a. Example of sleep scoring shows local field potential recorded across cortical layers (green to red heat map) as well as 15 Hz measurement of motor output (gray). Scoring (semi-supervised) is shown along the top in colored blocks. Black arrows denote microarousals. In this preparation (V1, array of microelectrodes), NREM sleep is characterized by high delta power (0.1–4 Hz), low theta (6–8 Hz), and small muscle movements. REM sleep is characterized by low delta, and obvious theta. Wake is consistently demarcated by low delta and increased motor signal. b. Young rats spend significantly more time asleep in the light than the dark, although this is highly variable (n = 8 animals). All box-and-whisker plots in b-f show the median, the first and third quartiles, minimum and maximum, and outliers of the data. c-f. 5 d of data from a single animal showing the percentage of each hour spent in each of four states: NREM (c, blue), REM (d, orange), active wake (e, green), and quiet wake (f, pink). The total time (across 8 animals) in each state as a function of light/dark is shown to the right of each panel. Statistics for b-f, linear mixed effects: % time sleep ~ condition + (1|animal). g. Example raw data traces from a single tetrode (4 channels). Spikes with high signal-to-noise ratio are readily apparent. h. Normalized single unit firing rate considered by state. Each unit is normalized to its own mean rate during REM. Linear mixed effects: FR ~ Behavior + (1|animal). i. Mean single unit coefficient of variation by state. Data in h and i are presented as mean ± s.e.m. across 8 animals.
Extended Data Fig. 2 Avalanche statistics, and DCC by ZT and light/dark.
a. Two seconds of raw voltage data during wake from four channels show clear action potentials (top). Binarized spike counts are extracted (middle), and the integrated network activity (bottom) shows fluctuations. Neuronal ‘avalanches’ start when network activity crosses above a threshold (dashed pink) and stop when it drops below. Avalanches are measured in terms of their size (total number of spiking neurons) and duration. b. Avalanches can include activity from a variable number of recorded neurons. Plotted is a kernel density estimate of the number of neurons activated in an avalanche. c. A given neuron may contribute to many or few avalanches. Plotted is a histogram of the proportion of avalanches that individual neurons contribute to. d. An example of an animal’s avalanche rate (Hz) across 5 d shows stability when binned at 1 h. e. Avalanche rate as a function of behavioral state and light/dark condition. f. Variability in DCC is not explained by time of day. DCC data from 8 animals shown in 2 h bins across the 24 h cycle. g. DCC is not significantly different in light than in dark (n = 8 animals). P = 0.165, Linear mixed effects: DCC ~ Condition + (1|animal). All box-and-whisker plots in e-g show the median, the first and third quartiles, minimum and maximum, and outliers of the data.
Extended Data Fig. 3 Effects of behavior and environmental conditions on neural dynamics.
a. In both light and dark, there is a significant negative correlation between time spent asleep and DCC (measured across the entire 4 h window). Note that this differs from the data in Fig. 3d by virtue of examining DCC across the entire 4 h window, inclusive of both sleep and wake. In this approach, each point is 4 h of data, but contains mixed states. In Fig. 3d, DCC is calculated only during the subset of the window that is spent asleep, and thus has variable time but constant state. The remaining measurements (b-f) are calculated only within a given state, consistent with Fig. 3. b. Time spent in active wake (locomotion) in the dark significantly correlates with DCC. Time spent in quiet waking in the dark has no correlation with DCC. c. Time spent asleep has no correlation with normalized firing rates during sleep in either light or dark. d. Similarly, time spent awake has no correlation with normalized firing rates during waking in light or dark. e, f. Same as c and d but coefficient of variation (CV) of interspike intervals. Statistics of a-f are derived from the default linear regression fit in the Scipy Package (Python).
Extended Data Fig. 4 Extended waking data.
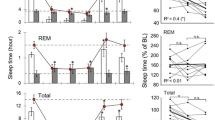
a. Process S is traditionally quantified by changes in slow-wave activity (SWA), especially in the context of sleep deprivation8 (Franken et al.). To confirm that our extended waking protocol is consistent with prior work, we quantified SWA (absolute power) throughout an extended waking epoch and the recovery period. 90 min of extended wake (teal) is sufficient to drive elevated SWA at the onset of NREM (blue) during recovery sleep. SWA progressively declines during NREM sleep. SWA was calculated in 5 mins bins (with a median filter of 4 s sliding windows in each epoch, similar to Franken et al.8). b. Normalized firing rate (FR) does not vary as a function of time spent in extended waking. FR is significantly lower in the recovery phase, the majority of which is NREM sleep (consistent with Extended Data Fig. 1h). Data in b–f are presented as mean ± s.e.m. c. FR at the start and end of extended waking, divided by light and dark phases. d. The interspike interval coefficient of variation (CV) does not vary significantly across the extended waking protocol or recovery period. e. Same as in c but for CV. f. The impact of extended waking on DCC is a significant increase between the start and end of the protocol. The magnitude of this change does not differ when comparing the first half of the 24 h period (light blue) to the second half (dark blue). P = 2.4e−16 for the first half; P = 6.7e−4 for the second half; Linear mixed effects: DCC ~ stage of extended wake + (1|animal). g. Quantification of data in f (P = 0.549, Linear mixed effects). The box-and-whisker plots show the median, the first and third quartiles, minimum and maximum, and outliers of the data. Data in b-f are from 8 animals. *** P < 0.001.
Extended Data Fig. 5 Prior behavior influences DCC within wake.
a. 1 h wake-dense epochs are identified (window B, red) in which features of neural activity are calculated. The amount of sleep in the preceding 2 h window (A, black) is calculated as the recent behavioral history. b. Despite the ongoing effects of wake on DCC within each wake-dense block, there is a significant negative correlation in DCC during 1 h of wake and time spent asleep in the prior 2 h. c. In the same data, no relationship is observed between sleep in the 2 h prior and normalized single unit firing rate (left) or coefficient of variation (right) in current wake-dense window. Statistics are derived from the default linear regression fit in the Scipy Package (Python).
Supplementary information
Supplementary Information
Supplementary Fig. 1.
Source data
Source Data Fig. 1
Statistical source data.
Source Data Fig. 2
Statistical source data.
Source Data Fig. 3
Statistical source data.
Source Data Fig. 4
Statistical source data.
Source Data Fig. 5
Statistical source data.
Source Data Fig. 6
Statistical source data.
Source Data Extended Data Fig. 1
Statistical source data.
Source Data Extended Data Fig. 2
Statistical source data.
Source Data Extended Data Fig. 3
Statistical source data.
Source Data Extended Data Fig. 4
Statistical source data.
Source Data Extended Data Fig. 5
Statistical source data.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Xu, Y., Schneider, A., Wessel, R. et al. Sleep restores an optimal computational regime in cortical networks. Nat Neurosci 27, 328–338 (2024). https://doi.org/10.1038/s41593-023-01536-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41593-023-01536-9
This article is cited by
-
Linking activity dyshomeostasis and sleep disturbances in Alzheimer disease
Nature Reviews Neuroscience (2024)