Abstract
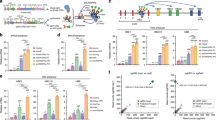
Epigenetic editing is an emerging technology that uses artificial transcription factors (aTFs) to regulate expression of a target gene. Although human genes can be robustly upregulated by targeting aTFs to promoters, the activation induced by directing aTFs to distal transcriptional enhancers is substantially less robust and consistent. Here we show that long-range activation using CRISPR-based aTFs in human cells can be made more efficient and reliable by concurrently targeting an aTF to the target gene promoter. We used this strategy to direct target gene choice for enhancers capable of regulating more than one promoter and to achieve allele-selective activation of human genes by targeting aTFs to single-nucleotide polymorphisms embedded in distally located sequences. Our results broaden the potential applications of the epigenetic editing toolbox for research and therapeutics.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Datasets from amplicon sequencing have been deposited with the NCBI Sequence Read Archive (PRJNA578485). Datasets from ChIP–seq, RNA-seq and ATAC–seq experiments have been deposited with the Gene Expression Omnibus repository under accession GSE139190. The GO database used in this study can be downloaded from https://bioportal.bioontology.org/ontologies/GO/. Source data are provided with this paper.
Change history
27 June 2022
A Correction to this paper has been published: https://doi.org/10.1038/s41592-022-01561-9
References
Pickar-Oliver, A. & Gersbach, C. A. The next generation of CRISPR–Cas technologies and applications. Nat. Rev. Mol. Cell Biol. 20, 490–507 (2019).
Thakore, P. I., Black, J. B., Hilton, I. B. & Gersbach, C. A. Editing the epigenome: technologies for programmable transcription and epigenetic modulation. Nat. Methods 13, 127–137 (2016).
Wang, H., La Russa, M. & Qi, L. S. CRISPR–Cas9 in genome editing and beyond. Annu. Rev. Biochem. 85, 227–264 (2016).
Maurano, M. T. et al. Systematic localization of common disease-associated variation in regulatory DNA. Science 337, 1190–1195 (2012).
Ernst, J. et al. Mapping and analysis of chromatin state dynamics in nine human cell types. Nature 473, 43–49 (2011).
Nasser, J. et al. Genome-wide enhancer maps link risk variants to disease genes. Nature 593, 238–243 (2021).
Gilbert, L. A. et al. Genome-scale CRISPR-mediated control of gene repression and activation. Cell 159, 647–661 (2014).
Gao, X. et al. Comparison of TALE designer transcription factors and the CRISPR–dCas9 in regulation of gene expression by targeting enhancers. Nucleic Acids Res. 42, e155 (2014).
Hilton, I. B. et al. Epigenome editing by a CRISPR–Cas9-based acetyltransferase activates genes from promoters and enhancers. Nat. Biotechnol. 33, 510–517 (2015).
Kuscu, C. et al. Temporal and spatial epigenome editing allows precise gene regulation in mammalian cells. J. Mol. Biol. 431, 111–121 (2019).
Li, K. et al. Interrogation of enhancer function by enhancer-targeting CRISPR epigenetic editing. Nat. Commun. 11, 485 (2020).
Klann, T. S. et al. CRISPR–Cas9 epigenome editing enables high-throughput screening for functional regulatory elements in the human genome. Nat. Biotechnol. 35, 561–568 (2017).
Mumbach, M. R. et al. Enhancer connectome in primary human cells identifies target genes of disease-associated DNA elements. Nat. Genet. 49, 1602–1612 (2017).
Simeonov, D. R. et al. Discovery of stimulation-responsive immune enhancers with CRISPR activation. Nature 549, 111–115 (2017).
Tak, Y. E. et al. Inducible and multiplex gene regulation using CRISPR–Cpf1-based transcription factors. Nat. Methods 14, 1163–1166 (2017).
Laguna, T. et al. New insights on the transcriptional regulation of CD69 gene through a potent enhancer located in the conserved noncoding sequence 2. Mol. Immunol. 66, 171–179 (2015).
Chen, J. C. J. & Goldhamer, D. J. The core enhancer is essential for proper timing of MyoD activation in limb buds and branchial arches. Dev. Biol. 265, 502–512 (2004).
Wienert, B., Martyn, G. E., Funnell, A. P. W., Quinlan, K. G. R. & Crossley, M. Wake-up sleepy gene: reactivating fetal globin for β-hemoglobinopathies. Trends Genet. 34, 927–940 (2018).
Diepstraten, S. T. & Hart, A. H. Modelling human haemoglobin switching. Blood Rev. 33, 11–23 (2019).
Sankaran, V. G. & Orkin, S. H. The switch from fetal to adult hemoglobin. Cold Spring Harb. Perspect. Med. 3, a011643 (2013).
Sankaran, V. G. et al. Human fetal hemoglobin expression is regulated by the developmental stage-specific repressor BCL11A. Science 322, 1839–1842 (2008).
Li, Q., Harju, S. & Peterson, K. R. Locus control regions: coming of age at a decade plus. Trends Genet. 15, 403–408 (1999).
Zannis, V. I., Kan, H. Y., Kritis, A., Zanni, E. E. & Kardassis, D. Transcriptional regulatory mechanisms of the human apolipoprotein genes in vitro and in vivo. Curr. Opin. Lipidol. 12, 181–207 (2001).
Cavalli, M. et al. Allele-specific transcription factor binding to common and rare variants associated with disease and gene expression. Hum. Genet. 135, 485–497 (2016).
Spisák, S. et al. CAUSEL: an epigenome- and genome-editing pipeline for establishing function of noncoding GWAS variants. Nat. Med. 21, 1357–1363 (2015).
Bailey, S. D. et al. ZNF143 provides sequence specificity to secure chromatin interactions at gene promoters. Nat. Commun. 2, 6186 (2015).
Tapscott, S. J., Lassar, A. B. & Weintraub, H. A novel myoblast enhancer element mediates MyoD transcription. Mol. Cell. Biol. 12, 4994–5003 (1992).
Jost, M. et al. Titrating gene expression using libraries of systematically attenuated CRISPR guide RNAs. Nat. Biotechnol. 38, 355–364 (2020).
Andersson, R. et al. An atlas of active enhancers across human cell types and tissues. Nature 507, 455–461 (2014).
ENCODE Project Consortium. Expanded encyclopaedias of DNA elements in the human and mouse genomes. Nature 583, 699–710 (2020).
Zhou, D., Pawlik, K. M., Ren, J., Sun, C.-W. & Townes, T. M. Differential binding of erythroid Krupple-like factor to embryonic/fetal globin gene promoters during development. J. Biol. Chem. 281, 16052–16057 (2006).
Liu, N. et al. Direct promoter repression by BCL11A controls the fetal to adult hemoglobin switch. Cell 173, 430–442 (2018).
Liu, N. et al. Transcription factor competition at the γ-globin promoters controls hemoglobin switching. Nat. Genet. 53, 511–520 (2021).
Lek, M. et al. Analysis of protein-coding genetic variation in 60,706 humans. Nature 536, 285–291 (2016).
Cooper, D. N., Krawczak, M., Polychronakos, C., Tyler-Smith, C. & Kehrer-Sawatzki, H. Where genotype is not predictive of phenotype: towards an understanding of the molecular basis of reduced penetrance in human inherited disease. Hum. Genet. 132, 1077–1130 (2013).
Veitia, R. A., Caburet, S. & Birchler, J. A. Mechanisms of Mendelian dominance. Clin. Genet. 93, 419–428 (2018).
Matharu, N. et al. CRISPR-mediated activation of a promoter or enhancer rescues obesity caused by haploinsufficiency. Science 363, eaau0629 (2019).
Dang, V. T., Kassahn, K. S., Marcos, A. E. & Ragan, M. A. Identification of human haploinsufficient genes and their genomic proximity to segmental duplications. Eur. J. Hum. Genet. 16, 1350–1357 (2008).
Inoue, K. & Fry, E. A. Haploinsufficient tumor suppressor genes. Adv. Med. Biol. 118, 83–122 (2017).
Pochampally, R. R., Horwitz, E. M., DiGirolamo, C. M., Stokes, D. S. & Prockop, D. J. Correction of a mineralization defect by overexpression of a wild-type cDNA for COL1A1 in marrow stromal cells (MSCs) from a patient with osteogenesis imperfecta: a strategy for rescuing mutations that produce dominant-negative protein defects. Gene Ther. 12, 1119–1125 (2005).
Liang, J. R., Lingeman, E., Ahmed, S. & Corn, J. E. Atlastins remodel the endoplasmic reticulum for selective autophagy. J. Cell Biol. 217, 3354–3367 (2018).
Bernstein, B. E. et al. Genomic maps and comparative analysis of histone modifications in human and mouse. Cell 120, 169–181 (2005).
Rohland, N. & Reich, D. Cost-effective, high-throughput DNA sequencing libraries for multiplexed target capture. Genome Res. 22, 939–946 (2012).
Li, H. & Durbin, R. Fast and accurate short read alignment with Burrows–Wheeler transform. Bioinformatics 25, 1754–1760 (2009).
Thorvaldsdóttir, H., Robinson, J. T. & Mesirov, J. P. Integrative Genomics Viewer: high-performance genomics data visualization and exploration. Brief. Bioinform. 14, 178–192 (2013).
Quinlan, A. R. & Hall, I. M. BEDTools: a flexible suite of utilities for comparing genomic features. Bioinformatics 26, 841–842 (2010).
Trapnell, C. et al. Transcript assembly and quantification by RNA-seq reveals unannotated transcripts and isoform switching during cell differentiation. Nat. Biotechnol. 28, 511–515 (2010).
Love, M. I., Huber, W. & Anders, S. Moderated estimation of fold change and dispersion for RNA-seq data with DESeq2. Genome Biol. 15, 550 (2014).
Mi, H. et al. Protocol update for large-scale genome and gene function analysis with the PANTHER classification system (v14.0). Nat. Protoc. 14, 703–721 (2019).
Corces, M. R. et al. An improved ATAC–seq protocol reduces background and enables interrogation of frozen tissues. Nat. Methods 14, 959–962 (2017).
Buenrostro, J. D., Giresi, P. G., Zaba, L. C., Chang, H. Y. & Greenleaf, W. J. Transposition of native chromatin for fast and sensitive epigenomic profiling of open chromatin, DNA-binding proteins and nucleosome position. Nat. Methods 10, 1213–1218 (2013).
Ktistaki, E., Lacorte, J.-M., Katrakili, N., Zannis, V. I. & Talianidis, I. Transcriptional regulation of the apolipoprotein A-IV gene involves synergism between a proximal orphan receptor response element and a distant enhancer located in the upstream promoter region of the apolipoprotein C-III gene. Nucleic Acids Res. 22, 4689–4696 (1994).
Clement, K. et al. CRISPResso2 provides accurate and rapid genome editing sequence analysis. Nat. Biotechnol. 37, 224–226 (2019).
Li, B., Kadura, I., Fu, D.-J. & Watson, D. E. Genotyping with TaqMAMA. Genomics 83, 311–320 (2004).
Durand, N. C. et al. Juicer provides a one-click system for analyzing loop-resolution Hi-C experiments. Cell Syst. 3, 95–98 (2016).
Wang, Y. et al. SPIN reveals genome-wide landscape of nuclear compartmentalization. Genome Biol. 22, 36 (2021).
Acknowledgements
J.K.J. was supported by grants from the National Institutes of Health (R35 GM118158, R01 CA211707, RM1 HG009490 and R01 CA204954), a St. Jude Children’s Research Hospital Collaborative Research Consortium award, a Massachusetts General Hospital (MGH) Collaborative Center for X-Linked Dystonia-Parkinsonism grant, and the Desmond and Ann Heathwood MGH Research Scholar Award. L.P. was supported by grants from the National Institute of Health (R00 HG008399 and R35 HG010717). We thank B. Kleinstiver (MGH) for providing the BPK1179, BPK880, BPK617 and BPK1160 plasmids. We thank M. Freedman, J. Seo and C. Lareau for assistance with important pilot experiments. We thank P. Farnham, M. Rivera and L. P. Pottenplackel for comments on the manuscript.
Author information
Authors and Affiliations
Contributions
Y.E.T. and J.K.J. conceived of and designed experiments. Y.E.T., J.E.H., N.T.P. and H.T.S. performed experiments. S.I. designed and made the computational pipeline for RNA-seq, ChIP–seq and ATAC–seq. L.P. and Q.Y. analyzed SNP density at regulatory elements. M.J.A. and L.S.Z. analyzed Hi-C data and performed SPIN analysis. Y.E.T. and J.K.J. wrote the manuscript with input from all the authors.
Corresponding author
Ethics declarations
Competing interests
J.K.J. has financial interests in Beam Therapeutics, Chroma Medicine (formerly YKY, Inc.), Editas Medicine, Excelsior Genomics, Pairwise Plants, Poseida Therapeutics, SeQure Dx, Transposagen Biopharmaceuticals and Verve Therapeutics (formerly Endcadia). L.P. has financial interests in Excelsior Genomics, Edilytics and SeQure Dx. M.J.A. has financial interests in Excelsior Genomics and SeQure Dx. The interests of J.K.J., L.P. and M.J.A. were reviewed and are managed by MGH and Partners HealthCare in accordance with their conflict-of-interest policies. S.I. is an employee of Verve Therapeutics. Y.E.T. and J.K.J. are inventors on patent applications that cover epigenetic editing technologies. All other authors have no competing interests.
Additional information
Peer review information Nature Methods thanks Fyodor Urnov and the other, anonymous, reviewers for their contribution to the peer review of this work. Lei Tang was the primary editor on this article and managed its editorial process and peer review in collaboration with the rest of the editorial team.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Extended data
Extended Data Fig. 1 Chromatin status at IL2RA, CD69, and MYOD1 determined by ATAC–seq and H3K27Ac ChIP-seq.
a, IL2RA promoter was closed and inactive in all cell types, IL2RA enhancer regions were closed and inactive in HEK293 and K562 cells, but open and active in U2OS and HepG2 cells. E1, E2, E3, E4: IL2RA enhancer gRNA target sites, P: IL2RA promoter gRNA target site. The RBM17 locus which was open (transposase accessible chromatin) and active (enriched in H3K27Ac marks) in all cell types is shown for comparison. b, CD69 promoter was closed and inactive in all cell types, CD69 enhancer regions were closed in all cell types. E1, E2: CD69 enhancer gRNA target sites, P: CD69 promoter gRNA target site. c, Chromatin at MYOD1 promoter was open in U2OS and HEK293 cells but not in HepG2 and K562 cells. E1, E2, E3, E4: MYOD1 enhancer gRNA target sites, P: MYOD1 promoter gRNA target site.
Extended Data Fig. 2 Chromatin status at the β-globin locus determined by ATAC-seq and H3K27Ac ChIP-seq.
All promoters at the β-globin locus showed closed and inactive chromatin states in all cell types. HS2 enhancer region showed closed and inactive chromatin features in HEK293 cells, but open and active chromatin features U2OS and HepG2 cells. E: HS2 enhancer gRNA target site, PE: HBE1 promoter gRNA target site, PG: HBG1/2 promoter gRNA target site, PB: HBB promoter gRNA target site.
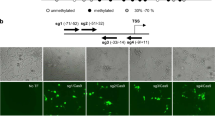
Extended Data Fig. 3 Total activation of APOA4 and APOC3 and orthogonal confirmation of allele-selective expression via allele-specific RT-qPCR.
a-b, Total expression of APOA4 and APOC3 in HEK293 cells by bi-partite p65 aTF targeting the promoter (P) and various sites on the enhancers including SNP regions (E1 to E6) and non-SNP region (E0) determined by RT-qPCR, normalized to HPRT1 levels, calculated relative to sample with non-targeting gRNA (None). Open circles indicate biological replicates (n = 3), bars the mean of replicates and error bars the s.e.m. c, Schematic of the location of RT-qPCR primers used for APOC3 allele-selective expression. Allele-specific primers detecting a SNP in APOC3 exon 3 have a common forward primer (PF) which spans the exon 2 and exon 3 junction, and two different reverse primers which are specific for allele 1 (T at rs4520, PR_1) or for allele 2 (C at rs4520, PR_2) in exon 3, with a ‘T’ mismatch in the penultimate base at the 3’ for both primers. d, Allele-selective expression of APOC3 in HEK293 cells by bi-partite p65 aTF targeting the promoter (P) and various sites on the enhancers including SNP regions (E1 to E6) and non-SNP region (E0) determined by RT-qPCR using the primers described in c, normalized to HPRT1 levels, calculated relative to sample with non-targeting gRNA. Open circles indicate biological replicates (n = 3), bars the mean of replicates and error bars the s.e.m. The apparent difference in allele-specific expression levels when compared to total expression in b is potentially due to the amplification of a smaller fragment of cDNA in the allele-specific reaction. e, Validation of the specificity of allele-specific RT-qPCR primers used in d, with U2OS cells in which the variant T nucleotide is absent at rs4520 (only the C nucleotide is present at the same position). APOC3 expression was measured by RT-qPCR using the allele-specific primers used in d, in U2OS cells co-expressing the bi-partite p65 aTF and gRNAs targeting the promoter or non-SNP region of the enhancer (E0). Open circles indicate biological replicates (n = 3), bars the mean of replicates and error bars the s.e.m.
Extended Data Fig. 4 Haplotype of potential APOA4 and APOC3 enhancer regions and allele ratios of target SNPs.
a, The potential enhancer region was identified by its open and active chromatin features which are similar to the known enhancer, based on the DNase-seq and H3K27Ac data (UCSC genome browser) from HepG2 cells in which APOC3 is highly expressed. Genomic locations of SNPs in SpCas9 PAMs identified in the potential enhancer are shown. SNPs in exon 2 of APOA4 and exon 3 of APOC3 are shown. b, Sanger sequencing traces from TOPO cloned amplicons showing the SNPs in the potential enhancer and exonic regions of APOA4 and APOC3 in HEK293 cells. E1 to E6 are gRNA binding sites in the potential enhancer region which has SNPs in the PAM sequence. SNPs are exclusively associated with one another in two unique haplotypes. c, Allele ratios of target SNPs in the genomic DNA of HEK293 cells were determined by targeted genomic DNA amplicon sequencing and indicate a 1:1 ratio.
Extended Data Fig. 5 Binding of bi-partite p65 aTF to APOA4 and APOC3 promoter and enhancer target sites in HEK293 cells.
a, Genomic locations of the enhancer gRNAs, APOA4 promoter and APOC3 promoter gRNA. The regions amplified in ChIP-qPCR assays are shown as boxes. b, Binding activity of bi-partite p65 aTF at each gRNA target region in APOA4 and APOC3 loci determined by Cas9 ChIP-qPCR, expressed as a percentage of input DNA. Two sets of primers designed to amplify the human genome at locations other than the APOC3 and APOA4 loci were used as negative controls. Open circles indicate biological replicates (n = 3), bars the mean of replicates and error bars the s.e.m. c, Binding of the bi-partite p65 aTF to the potential upstream enhancer sequence in the presence of the E1-E6 gRNAs. E1, E2, and E4 are expected to bind selectively to Allele 1 (yellow); E3, E5, and E6 to Allele 2 (orange). Relative quantification (percent next-generation sequencing reads) of the two alleles in the DNA from ChIP experiments performed with an anti-Cas9 antibody are shown. Open circles indicate biological replicates (n = 3), bars the mean of replicates and error bars the s.e.m. In, Input DNA; Ch, Cas9 ChIP DNA.
Extended Data Fig. 6 Allele-selective upregulation of HBB genes in U2OS and K562 cells using heterotopic activation of enhancer.
a, Schematic of HBB gene and the three alleles present in U2OS cells. P indicates the binding site for the gRNA targeting the HBB promoter. E1-E4 indicate binding sites for gRNAs in the HS4 putative enhancer region, which are expected to target either all alleles (E1), selectively target one allele (E2, E3) or two alleles (E4) based on the PAM of the target site (black bold indicates a base that maintains an intact PAM site and gray bold indicates a base that is expected to disrupt the PAM). A SNP in exon 1 of HBB distinguishes between allele 1 (light purple) and allele 2/3 (pink). b, Total expression of HBB in U2OS cells when the bi-partite p65 aTF was co-expressed with a gRNA targeting the promoter (P) and/or with one or more gRNAs targeting the HS4 enhancer region (E1-E4) was determined by RT-qPCR, normalized to HPRT1 levels, and calculated relative to sample with non-targeting gRNA (None). Open circles indicate biological replicates (n = 3), bars the mean of replicates and error bars the s.e.m. c, Relative quantification (percent next-generation sequencing reads of cDNA) of the three alleles of HBB mRNA when the bi-partite p65 aTF was co-expressed with a gRNA targeting the promoter (P) alone or with one or more gRNAs targeting the HS4 enhancer region (E1-E4). Open circles indicate biological replicates (n = 3), bars the mean of replicates and error bars the s.e.m.
Extended Data Fig. 7 Allele-selective upregulation of MYOD1 genes in U2OS and K562 cells, respectively, using heterotopic activation of enhancer.
a, Schematic of MYOD1 gene and the three alleles present in K562 cells. P indicates the binding site for the gRNA targeting the MYOD1 promoter. E1-E4 indicate binding sites for gRNAs in the known enhancer region termed the distal regulatory region (DRR), which are expected to selectively target allele 1 (E1, E3, E4) or all alleles (E2) based on the PAM of the target site (black bold indicates a base that maintains an intact PAM site and gray bold indicates a base that is expected to disrupt the PAM). A SNP in exon 3 of MYOD1 distinguishes between allele 1 and allele 2/3. b, Total expression of MYOD1 in K562 cells when the bi-partite p65 aTF was co-expressed with a gRNA targeting the promoter (P) and/or with one or more gRNAs targeting the DRR enhancer region (E1-E4) was determined by RT-qPCR, normalized to HPRT1 levels, and calculated relative to sample with non-targeting gRNA (None). Open circles indicate biological replicates (n = 3), bars the mean of replicates and error bars the s.e.m. c, Relative quantification (percent next-generation sequencing reads of cDNA) of the three alleles of MYOD1 mRNA when the bi-partite p65 aTF was co-expressed with a gRNA targeting the promoter (P) alone or with one or more gRNAs expected to target the DRR enhancer region (E1-E4). Open circles indicate biological replicates (n = 3), bars the mean of replicates and error bars the s.e.m.
Extended Data Fig. 8 Distribution of SNP densities that create or disrupt NGG PAM sequences at putative enhancers and promoters.
The density of SNPs is the number of SNPs divided by the base pair size of each regulatory element (promoter or enhancer) identified from the 1000 Genomes Project using DHS data from 83 different cell lines from ENCODE/Roadmap project. center line, median; box limits, upper and lower quartiles; whiskers, 1.5x interquartile range. *** indicates p < 0.001 (Mann-Whitney U test (two-sided) with Bonferroni test for multiple comparison).
Supplementary information
Supplementary Information
Supplementary Note and Figs. 1–6.
Supplementary Data
Source data for supplementary figures.
Source data
Source Data Fig. 1
Guide IDs and qPCR data.
Source Data Fig. 2
Guide IDs and qPCR data.
Source Data Fig. 3
Guide IDs and qPCR data.
Source Data Fig. 4
Guide IDs, qPCR data and amplicon sequencing read counts.
Source Data Extended Data Fig. 3
Guide IDs and qPCR data.
Source Data Extended Data Fig. 4
Amplicon sequencing read counts.
Source Data Extended Data Fig. 5
Guide IDs, qPCR data and amplicon sequencing read counts.
Source Data Extended Data Fig. 6
Guide IDs, qPCR data and amplicon sequencing read counts.
Source Data Extended Data Fig. 7
Guide IDs, qPCR data and amplicon sequencing read counts.
Source Data Extended Data Fig. 8
SNP density at each category (promoter, enhancer, PAM creation, PAM disruption and mixed).
Rights and permissions
About this article
Cite this article
Tak, Y.E., Horng, J.E., Perry, N.T. et al. Augmenting and directing long-range CRISPR-mediated activation in human cells. Nat Methods 18, 1075–1081 (2021). https://doi.org/10.1038/s41592-021-01224-1
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41592-021-01224-1
This article is cited by
-
tgCRISPRi: efficient gene knock-down using truncated gRNAs and catalytically active Cas9
Nature Communications (2023)
-
Promising therapeutic aspects in human genetic imprinting disorders
Clinical Epigenetics (2022)
-
Gene activation guided by nascent RNA-bound transcription factors
Nature Communications (2022)