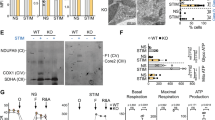
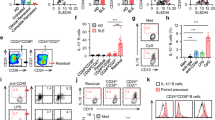
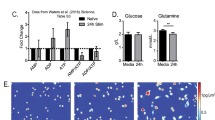
Abstract
B cells are activated by two temporally distinct signals, the first provided by the binding of antigen to the B cell antigen receptor (BCR), and the second provided by helper T cells. Here we found that B cells responded to antigen by rapidly increasing their metabolic activity, including both oxidative phosphorylation and glycolysis. In the absence of a second signal, B cells progressively lost mitochondrial function and glycolytic capacity, which led to apoptosis. Mitochondrial dysfunction was a result of the gradual accumulation of intracellular calcium through calcium response–activated calcium channels that, for approximately 9 h after the binding of B cell antigens, was preventable by either helper T cells or signaling via the receptor TLR9. Thus, BCR signaling seems to activate a metabolic program that imposes a limited time frame during which B cells either receive a second signal and survive or are eliminated.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout








Similar content being viewed by others
References
Scharenberg, A. M., Humphries, L. A. & Rawlings, D. J. Calcium signalling and cell-fate choice in B cells. Nat. Rev. Immunol. 7, 778–789 (2007).
De Silva, N. S. & Klein, U. Dynamics of B cells in germinal centres. Nat. Rev. Immunol. 15, 137–148 (2015).
Yuseff, M. I., Pierobon, P., Reversat, A. & Lennon-Duménil, A. M. How B cells capture, process and present antigens: a crucial role for cell polarity. Nat. Rev. Immunol. 13, 475–486 (2013).
Roche, P. A. & Furuta, K. The ins and outs of MHC class II-mediated antigen processing and presentation. Nat. Rev. Immunol. 15, 203–216 (2015).
Elgueta, R. et al. Molecular mechanism and function of CD40/CD40L engagement in the immune system. Immunol. Rev. 229, 152–172 (2009).
Ruprecht, C. R. & Lanzavecchia, A. Toll-like receptor stimulation as a third signal required for activation of human naive B cells. Eur. J. Immunol. 36, 810–816 (2006).
Chaturvedi, A. & Pierce, S. K. How location governs toll-like receptor signaling. Traffic 10, 621–628 (2009).
Rawlings, D. J., Schwartz, M. A., Jackson, S. W. & Meyer-Bahlburg, A. Integration of B cell responses through Toll-like receptors and antigen receptors. Nat. Rev. Immunol. 12, 282–294 (2012).
Akkaya, M. et al. B cells produce type 1 IFNs in response to the TLR9 agonist CpG-A conjugated to cationic lipids. J. Immunol. 199, 931–940 (2017).
Caro-Maldonado, A. et al. Metabolic reprogramming is required for antibody production that is suppressed in anergic but exaggerated in chronically BAFF-exposed B cells. J. Immunol. 192, 3626–3636 (2014).
Caro-Maldonado, A., Gerriets, V.A. & Rathmell, J.C. Matched and mismatched metabolic fuels in lymphocyte function. Semin. Immunol. 24, 405–413 (2012).
Lam, W. Y. et al. Mitochondrial pyruvate import promotes long-term survival of antibody-secreting plasma cells. Immunity 45, 60–73 (2016).
Doughty, C. A. et al. Antigen receptor-mediated changes in glucose metabolism in B lymphocytes: role of phosphatidylinositol 3-kinase signaling in the glycolytic control of growth. Blood 107, 4458–4465 (2006).
Xiao, G. et al. B-cell-specific diversion of glucose carbon utilization reveals a unique vulnerability in B cell malignancies. Cell 173, 470–484 (2018).
Traba, J., Miozzo, P., Akkaya, B., Pierce, S. K. & Akkaya, M. An optimized protocol to analyze glycolysis and mitochondrial respiration in lymphocytes. J. Vis. Exp. 117, e54918 (2016).
Akkaya, M. et al. Toll-like receptor 9 antagonizes antibody affinity maturation. Nat. Immunol. 19, 255–266 (2018).
Fleire, S. J. et al. B cell ligand discrimination through a spreading and contraction response. Science 312, 738–741 (2006).
Lemasters, J. J.., & Holmuhamedov, E.. Voltage-dependent anion channel (VDAC) as mitochondrial governator-thinking outside the box. Biochim. Biophys. Acta 1762, 181–190 (2006).
Li, Y., Park, J. S., Deng, J. H. & Bai, Y. Cytochrome c oxidase subunit IV is essential for assembly and respiratory function of the enzyme complex. J. Bioenerg. Biomembr. 38, 283–291 (2006).
Larsson, N. G. et al. Mitochondrial transcription factor A is necessary for mtDNA maintenance and embryogenesis in mice. Nat. Genet. 18, 231–236 (1998).
Onesto, E. et al. Gene-specific mitochondria dysfunctions in human TARDBP and C9ORF72 fibroblasts. Acta Neuropathol. Commun. 4, 47 (2016).
Graves, J. A. et al. Mitochondrial structure, function and dynamics are temporally controlled by c-Myc. PLoS One 7, e37699 (2012).
Sgarbi, G. et al. Mitochondria hyperfusion and elevated autophagic activity are key mechanisms for cellular bioenergetic preservation in centenarians. Aging 6, 296–310 (2014).
Mitra, K., Wunder, C., Roysam, B., Lin, G. & Lippincott-Schwartz, J. A hyperfused mitochondrial state achieved at G1-S regulates cyclin E buildup and entry into S phase. Proc. Natl Acad. Sci. USA 106, 11960–11965 (2009).
Agnello, M., Morici, G. & Rinaldi, A. M. A method for measuring mitochondrial mass and activity. Cytotechnology 56, 145–149 (2008).
Yu, J. et al. Inflammasome activation leads to Caspase-1-dependent mitochondrial damage and block of mitophagy. Proc. Natl Acad. Sci. USA 111, 15514–15519 (2014).
Keij, J. F., Bell-Prince, C. & Steinkamp, J. A. Staining of mitochondrial membranes with 10-nonyl acridine orange, MitoFluor Green, and MitoTracker Green is affected by mitochondrial membrane potential altering drugs. Cytometry 39, 203–210 (2000).
Contreras, L., Drago, I., Zampese, E. & Pozzan, T. Mitochondria: the calcium connection. Biochim. Biophys. Acta 1797, 607–618 (2010).
Celsi, F. et al. Mitochondria, calcium and cell death: a deadly triad in neurodegeneration. Biochim. Biophys. Acta 1787, 335–344 (2009).
Giorgi, C. et al. Mitochondrial Ca2+ and apoptosis. Cell Calcium 52, 36–43 (2012).
Görlach, A., Bertram, K., Hudecova, S. & Krizanova, O. Calcium and ROS: A mutual interplay. Redox Biol. 6, 260–271 (2015).
Mbaya, E. et al. Calcium signalling-dependent mitochondrial dysfunction and bioenergetics regulation in respiratory chain complex II deficiency. Cell Death Differ. 17, 1855–1866 (2010).
Choi, H., Yang, Z. & Weisshaar, J. C. Single-cell, real-time detection of oxidative stress induced in Escherichia coli by the antimicrobial peptide CM15. Proc. Natl Acad. Sci. USA 112, E303–E310 (2015).
Robinson, K. M. et al. Selective fluorescent imaging of superoxide in vivo using ethidium-based probes. Proc. Natl Acad. Sci. USA 103, 15038–15043 (2006).
Santo-Domingo, J. & Demaurex, N. Calcium uptake mechanisms of mitochondria. Biochim. Biophys. Acta 1797, 907–912 (2010).
Natkanski, E. et al. B cells use mechanical energy to discriminate antigen affinities. Science 340, 1587–1590 (2013).
Batista, F. D., Iber, D. & Neuberger, M. S. B cells acquire antigen from target cells after synapse formation. Nature 411, 489–494 (2001).
Tze, L. E., Baness, E. A., Hippen, K. L. & Behrens, T. W. Ig light chain receptor editing in anergic B cells. J. Immunol. 165, 6796–6802 (2000).
Batista, F. D. & Neuberger, M. S. B cells extract and present immobilized antigen: implications for affinity discrimination. EMBO J. 19, 513–520 (2000).
Aucher, A., Magdeleine, E., Joly, E. & Hudrisier, D. Capture of plasma membrane fragments from target cells by trogocytosis requires signaling in T cells but not in B cells. Blood 111, 5621–5628 (2008).
Traba, J. et al. Prolonged fasting suppresses mitochondrial NLRP3 inflammasome assembly and activation via SIRT3-mediated activation of superoxide dismutase 2. J. Biol. Chem. 292, 12153–12164 (2017).
Gees, M., Colsoul, B. & Nilius, B. The role of transient receptor potential cation channels in Ca2+ signaling. Cold Spring Harb. Perspect. Biol. 2, a003962 (2010).
Rae, M. G., Hilton, J. & Sharkey, J. Putative TRP channel antagonists, SKF 96365, flufenamic acid and 2-APB, are non-competitive antagonists at recombinant human α1β2γ2 GABAA receptors. Neurochem. Int. 60, 543–554 (2012).
Ohga, K., Takezawa, R., Arakida, Y., Shimizu, Y. & Ishikawa, J. Characterization of YM-58483/BTP2, a novel store-operated Ca2+ entry blocker, on T cell-mediated immune responses in vivo. Int. Immunopharmacol. 8, 1787–1792 (2008).
Donjerković, D. & Scott, D. W. Activation-induced cell death in B lymphocytes. Cell Res. 10, 179–192 (2000).
Rathmell, J. C. et al. CD95 (Fas)-dependent elimination of self-reactive B cells upon interaction with CD4+ T cells. Nature 376, 181–184 (1995).
Rothstein, T. L. et al. Protection against Fas-dependent Th1-mediated apoptosis by antigen receptor engagement in B cells. Nature 374, 163–165 (1995).
Lagresle, C., Mondière, P., Bella, C., Krammer, P. H. & Defrance, T. Concurrent engagement of CD40 and the antigen receptor protects naive and memory human B cells from APO-1/Fas-mediated apoptosis. J. Exp. Med. 183, 1377–1388 (1996).
Akkaya, B. et al. Ex-vivo iTreg differentiation revisited: convenient alternatives to existing strategies. J. Immunol. Methods 441, 67–71 (2017).
Akkaya, B. et al. A simple, versatile antibody-based barcoding method for flow cytometry. J. Immunol. 197, 2027–2038 (2016).
Traba, J., Del Arco, A., Duchen, M. R., Szabadkai, G. & Satrústegui, J. SCaMC-1 promotes cancer cell survival by desensitizing mitochondrial permeability transition via ATP/ADP-mediated matrix Ca2+ buffering. Cell Death Differ. 19, 650–660 (2012).
Sohn, H. W., Tolar, P., Brzostowski, J. & Pierce, S. K. A method for analyzing protein-protein interactions in the plasma membrane of live B cells by fluorescence resonance energy transfer imaging as acquired by total internal reflection fluorescence microscopy. Methods Mol. Biol. 591, 159–183 (2010).
Liu, W., Meckel, T., Tolar, P., Sohn, H. W. & Pierce, S. K. Antigen affinity discrimination is an intrinsic function of the B cell receptor. J. Exp. Med. 207, 1095–1111 (2010).
Akkaya, M., Aknin, M. L., Akkaya, B. & Barclay, A. N. Dissection of agonistic and blocking effects of CD200 receptor antibodies. PLoS One 8, e63325 (2013).
Offerdahl, D. K., Dorward, D. W., Hansen, B. T. & Bloom, M. E. A three-dimensional comparison of tick-borne flavivirus infection in mammalian and tick cell lines. PLoS One 7, e47912 (2012).
Mastronarde, D. N. Automated electron microscope tomography using robust prediction of specimen movements. J. Struct. Biol. 152, 36–51 (2005).
Acknowledgements
We thank S. Bolland (National Institutes of Health) for TLR9-deficient mice; I. Gery (National Institutes of Health) for 3A9mice; O. Voss (National Institutes of Health) for the NIH3T3 mouse fibroblast cell line; P. Allen (Washington University) for the 3A9 mouse T cell hybridoma line; R. Kissinger for preparing the illustration in Supplementary Fig. 2; and P.W. Sheehan, T. Leto, J. Brzostowski and J. Manzella-Lapeira for assistance and advice in various experiments. Supported by the National Institutes of Health Intramural Research Program, National Institute of Allergy and Infectious Diseases and National Heart, Lung, Blood Institute.
Author information
Authors and Affiliations
Contributions
M.A. wrote the manuscript; M.A., J.T., A.S.R.,P.M., B.A., B.P.T., H.S., M.P., M.S. and D.W.D. carried out experiments; M.A., J.T., A.S.R., P.M., B.A., B.P.T., H.S., J.K., E.D. and J.S. analyzed data; M.A., J.T., A.S.R., P.M., B.P.T. and S.K.P. designed the experiments; M.A. and S.K.P. conceived of the project; M.N.S. and S.K.P. edited the manuscript; and S.K.P. secured funding;
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note: Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Integrated supplementary information
Supplementary Figure 1 Early changes in B cell metabolism following B cell stimulation do not require cellular remodeling.
a-j) Purified mouse B cells were stimulated with 1 μM CpG and/or 5 μg/ml anti-IgM or left unstimulated (media only). The flow cytometry plots (a,c) and MFI graphs (b,d) of TMRM alone, TMRM + Oligomycin, TMRM + FCCP at 1 h (a,b) and 4 h (c,d) post stimulation; flow cytometry plots (e,f) and MFI graphs (g) of GLUT 1 and 3 at 4 h post stimulation; change in MFI of 2NBDG (added to the culture at 10 μM) between 0–2 h (h) post stimulation and representative flow cytometry plot showing the 2NBDG levels at 2 h (i) and expression levels of TOM20 at 4 h (j) post stimulation are shown. Data represents three independent experiments each done with triplicates. Bars indicate the mean of the triplicates and error bars represent the standard deviation. (P > 0.05 = n.s.) (one sided two-way ANOVA).
Supplementary Figure 2
Schematic illustration, depicting the metabolic functions of genes that are transcriptionally regulated in response to B cell activation through TLR9 and /or BCR.
Supplementary Figure 3 The roles of glycolysis and oxidative phosphorylation in B cell functionality and survival.
a) Representative flow cytometry plots for the experiment outlined in Fig. 1i. b-c) Purified mouse splenic B cells were stained with e450 proliferation dye and then cultured in growth media alone or media supplemented with 1 μM CpG and/or 5 μg/ml anti-IgM. Flow cytometry plots (b) and bar graphs (c) demonstrating the total proliferating cells and cells that have proliferated at least two times at 24 h and 48 h post stimulation are shown. Bars and error bars indicate mean and standard deviation respectively. Data is representative of three independent experiments. d) Graphs representing the changes in the fold expression of CD69 in the experiment outlined in Fig. 1j are shown. (P > 0.05 = n.s.; P ≤ 0.0001 = ****) (One-way ANOVA with Tukey’s adjustment).
Supplementary Figure 4 Long term cellular and metabolic changes following B cell stimulation.
a,b) Unstimulated B cells and B cells stimulated with anti-IgM (5 μg/ml) were harvested at 0 (unstimulated only) 3, 8 or 24 h post stimulation and the percentages of live, early apoptotic, late apoptotic and necrotic cells were determined by staining the cells with both 7AAD and VAD (FAM-FLICA). Representative flow cytometry plots (a) and quantification of each population in triplicates for each time point (b). Bars and error bars represent mean and standard deviation respectively. Data are representative of two independent experiments. c) Representative flow cytometry plots showing the GLUT 1 expression 24 h post stimulation of WT and TLR9 KO B cells in the experiment outlined in Fig. 2i. d) Representative flow cytometry plot showing the 2NBDG staining at 24 h (120 min after addition of 2NBDG) in the experiment outlined in Fig. 2j e) Representative flow cytometry plots for the experiment outlined in Fig. 3a
Supplementary Figure 5 BCR activation induced mitochondrial changes in B cells.
a) B cells were purified from WT mice and cultured in growth media alone or media supplemented with 1 μM CpG and/or 5 μg/ml anti-IgM for 24 h. Cells were then harvested and stained with Live/DEAD stain and MitoTracker Green. Stained cells were immobilized in chambers coated with Poly-L-lysine and imaged under confocal microscope. Representative images showing the MitoTracker Green staining in viable B cells for each stimulation condition are shown. b-e) Representative flow cytometry plots for experiments outlined in Fig. 4c. (b), Fig. 4d (c), Fig. 4i (d) and Fig. 4k (e) are shown.
Supplementary Figure 6 Outline of the adoptive transfer strategy.
a) Depiction of the experimental design described in Fig. 5. Purified B cells from the spleens of WT (CD45.1) and MD4 (CD45.2) mice were mixed 1:1, stained with e450 and adoptively transferred into WT (CD45.2) recipient mice (4.5 × 106 cells per mouse). Mice were injected i.v. 24 h post transfer with 200 μl PBS alone, PBS containing HEL (100 μg HEL/mouse), PBS containing CpG (100 μg CpG) or PBS containing HEL and CpG, 24 h later mice were euthanized and splenocytes were harvested. b) Adoptively transferred B cells from WT and MD4 mice were identified in the spleens of the recipient mouse using the gating strategy shown. Gating of singlets and live cells carried out prior to this step are not shown.
Supplementary Figure 7 BCR stimulation mediated changes in mitochondrial function are linked to the intracellular Calcium levels.
a) Representative flow cytometry plot for the experiment outlined in Fig. 6a. b) Histogram overlay demonstrating the expression levels of recombinant proteins consisting of rCD4 attached to WT or mutant HEL or DEL proteins on the surfaces of NIH3T3 cells as detected by anti rCD4 antibody. c-f) Representative flow cytometry plots for the experiment outlined in Fig. 8 h-k respectively.
Supplementary Information
Supplementary Figures
Supplementary Figures 1–7
Supplementary Text
Supplementary Table 1
Rights and permissions
About this article
Cite this article
Akkaya, M., Traba, J., Roesler, A.S. et al. Second signals rescue B cells from activation-induced mitochondrial dysfunction and death. Nat Immunol 19, 871–884 (2018). https://doi.org/10.1038/s41590-018-0156-5
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41590-018-0156-5
This article is cited by
-
RNA modifications in cellular metabolism: implications for metabolism-targeted therapy and immunotherapy
Signal Transduction and Targeted Therapy (2024)
-
New insights into the ontogeny, diversity, maturation and survival of long-lived plasma cells
Nature Reviews Immunology (2024)
-
Single-B cell analysis correlates high-lactate secretion with stress and increased apoptosis
Scientific Reports (2024)
-
Dynamic mitochondrial transcription and translation in B cells control germinal center entry and lymphomagenesis
Nature Immunology (2023)
-
Expression of TRAF6 in peripheral blood B cells of patients with myasthenia gravis
BMC Neurology (2022)