Abstract
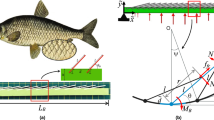
Living organisms use stingers that vary in length L over eight orders of magnitude, from a few tens of nanometres to several metres, across a wide array of biological taxa. Despite the extreme variation in size, their structures are strikingly similar. However, the mechanism responsible for this remarkable morphological convergence remains unknown. Using basic physical arguments and biomimetic experiments, we reveal an optimal design strategy that links their length, base diameter d0, Young’s modulus E and friction force per unit area μp0. This principle can be framed simply as \({d}_{0} \approx {(\mu {p}_{0}/E)}^{1/3}L\). Existing data from measurements on viruses, algae, marine invertebrates, terrestrial invertebrates, plants, terrestrial vertebrates, marine vertebrates—as well as man-made objects such as nails, needles and weapons—are consistent with our predictions. Our results highlight the evolutionary adaptation of mechanical traits to the constraints imposed by friction, elastic stability and cost.
This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$209.00 per year
only $17.42 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout




Similar content being viewed by others
Data availability
Source data are available in Supplementary Table 1. All other data that support the plots within this paper and other findings of this study are available from the corresponding author upon reasonable request.
Change history
15 February 2021
A Correction to this paper has been published: https://doi.org/10.1038/s41567-020-0996-4
References
van Tol, H. M., Irwin, A. J. & Finkel, Z. V. Macroevolutionary trends in silicoflagellate skeletal morphology: the costs and benefits of silicification. Paleobiology 38, 391–402 (2012).
Mershon, J. P., Becker, M. & Bickford, C. P. Linkage between trichome morphology and leaf optical properties in New Zealand alpine Pachycladon (Brassicaceae). New Zealand J. Botany 53, 175–182 (2015).
Emlen, D. J. The evolution of animal weapons. Annu. Rev. Ecol. Evol. Syst. 39, 387–413 (2008).
Kodric-Brown, A., Sibly, R. M. & Brown, J. H. The allometry of ornaments and weapons. Proc. Natl Acad. Sci. USA 103, 8733–8738 (2006).
Gould, S. J. Allometry and size in ontogeny and phylogeny. Biol. Rev. 41, 587–638 (1966).
Weigend, M., Mustafa, A. & Ensikat, H.-J. Calcium phosphate in plant trichomes: the overlooked biomineral. Planta 247, 277–285 (2018).
Farmer, E. E. Leaf Defence (Oxford Univ. Press, 2014).
Bartual, S. G. et al. Structure of the bacteriophage T4 long tail fiber receptor-binding tip. Proc. Natl Acad. Sci. USA 107, 20287–20292 (2010).
Uriz, M.-J., Turon, X., Becerro, M. A. & Agell, G. Siliceous spicules and skeleton frameworks in sponges: origin, diversity, ultrastructural patterns and biological functions. Microsc. Res. Tech. 62, 279–299 (2003).
Silverman, H. & Dunbar, M. Aggressive tusk use by the narwhal (Monodon monoceros L.). Nature 284, 57–58 (1980).
Wells, T. Nail chronology: the use of technologically derived features. Hist. Archaeol. 32, 78–99 (1998).
Gill, H. S. & Prausnitz, M. R. Does needle size matter? J. Diabetes Sci. Technol. 1, 725–729 (2007).
Markle, M. M. III The Macedonian sarissa, spear and related armor. Am. J. Archaeol. 81, 323–339 (1977).
DeVries, K. & Smith, R. D. Medieval Weapons: An Illustrated History of their Impact (ABC-CLIO, 2007).
Williams, A., Edge, D., Capwell, T. & Tschegg, S. A technical note on the armour and equipment for jousting. Gladius 32, 139–184 (2012).
Walters, D. Fortress Plant: How to Survive when Everything Wants to Eat You (Oxford Univ. Press, 2017).
Heemstra, P. C. & Heemstra, E. Coastal Fishes of Southern Africa (NISC, 2004).
Hu, B., Margolin, W., Molineux, I. J. & Liu, J. Structural remodeling of bacteriophage T4 and host membranes during infection initiation. Proc. Natl Acad. Sci. USA 112, E4919–E4928 (2015).
McMahon, T. A. & Bonner, J. T. On Size and Life (Scientific American Library, 1983).
Timoshenko, S. P. & Gere, J. M. Theory of Elastic Stability (McGraw-Hill, 1988).
Keller, J. B. The shape of the strongest column. Arch. Rational Mech. Anal. 5, 275–285 (1960).
Monn, M. A. & Kesari, H. A new structure–property connection in the skeletal elements of the marine sponge Tethya aurantia that guards against buckling instability. Sci. Rep. 7, 39547 (2017).
Keller, J. B. & Niordson, F. I. The tallest column. J. Math. Mech. 16, 433–446 (1966).
Wei, Z., Mandre, S. & Mahadevan, L. The branch with the furthest reach. Europhys. Lett. 97, 14005 (2012).
von Karman, T. & Biot, M. A. Mathematical Methods in Engineering (McGraw Hill, 1940).
Johnston, I., McCluskey, D., Tan, C. & Tracey, M. Mechanical characterization of bulk Sylgard 184 for microfluidics and microengineering. J. Micromech. Microeng. 24, 035017 (2014).
Brzinski, T. A. III, Mayor, P. & Durian, D. J. Depth-dependent resistance of granular media to vertical penetration. Phys. Rev. Lett. 111, 168002 (2013).
Roesthuis, R. J., Van Veen, Y. R., Jahya, A. & Misra, S. Mechanics of needle-tissue interaction. In 2011 IEEE/RSJ International Conference on Intelligent Robots and Systems (IROS) 2557–2563 (IEEE, 2011).
Davis, S. P., Landis, B. J., Adams, Z. H., Allen, M. G. & Prausnitz, M. R. Insertion of microneedles into skin: measurement and prediction of insertion force and needle fracture force. J. Biomech. 37, 1155–1163 (2004).
Ling, J. et al. Insertion and pull behavior of worker honeybee stinger. J. Bionic Eng. 13, 303–311 (2016).
van Gerwen, D. J., Dankelman, J. & van den Dobbelsteen, J. J. Needle-tissue interaction forces—a survey of experimental data. Med. Eng. Phys. 34, 665–680 (2012).
Kim, W. & Bush, J. W. Natural drinking strategies. J. Fluid Mech. 705, 7–25 (2012).
Montel, F. et al. Stress clamp experiments on multicellular tumor spheroids. Phys. Rev. Lett. 107, 188102 (2011).
Broders-Bondon, F., Ho-Bouldoires, T. H. N., Fernandez-Sanchez, M.-E. & Farge, E. Mechanotransduction in tumor progression: the dark side of the force. J. Cell Biol. 217, 1571–1587 (2018).
Pailler-Mattei, C., Bec, S. & Zahouani, H. In vivo measurements of the elastic mechanical properties of human skin by indentation tests. Med. Eng. Phys. 30, 599–606 (2008).
Jewel, R., Panaitescu, A. & Kudrolli, A. Micromechanics of intruder motion in wet granular medium. Phys. Rev. Fluids 3, 084303 (2018).
Quicke, D., Fitton, M., Tunstead, J., Ingram, S. & Gaitens, P. Ovipositor structure and relationships within the Hymenoptera, with special reference to the Ichneumonoidea. J. Nat. Hist. 28, 635–682 (1994).
Gibson, L. J. The hierarchical structure and mechanics of plant materials. J. R. Soc. Interface 9, 2749–2766 (2012).
McCartney, K., Witkowski, J. & Harwood, D. M. Early evolution of the silicoflagellates during the cretaceous. Marine Micropaleontol. 77, 83–100 (2010).
Kellenberger, E., Stauffer, E., Häner, M., Lustig, A. & Karamata, D. Mechanism of the long tail-fiber deployment of bacteriophages T-even and its role in adsorption, infection and sedimentation. Biophys. Chem. 59, 41–59 (1996).
Præstmark Juul, K. A. et al. Influence of hypodermic needle dimensions on subcutaneous injection delivery—a pig study of injection deposition evaluated by CT scanning, histology and backflow. Skin Res. Technol. 18, 447–455 (2012).
Zhu, J. et al. Gelatin methacryloyl microneedle patches for minimally invasive extraction of skin interstitial fluid. Small 16, 1905910 (2020).
Backholm, M. & Bäumchen, O. Micropipette force sensors for in vivo force measurements on single cells and multicellular microorganisms. Nat. Protoc. 14, 594–615 (2019).
Hooke, R. Micrographia, or Some Physiological Descriptions of Minute Bodies Made by Magnifying Glasses, with Observations and Inquiries Thereupon (Royal Society, 1665).
Acknowledgements
This work was supported by two research grants (17587 and 13166) from Villum Fonden.
Author information
Authors and Affiliations
Contributions
K.H.J. designed the research. A.H.C. and K.H.J. derived the model. K.S.H., K.P. and J.K. performed experiments. K.H.J., K.P. and J.K. collected and analysed data. K.H.J. wrote the manuscript with support from K.P. and J.K.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Peer review information Nature Physics thanks Douglas Holmes, Hamed Rajabi and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Fig. 1, Table 1 and refs. 1–55.
Rights and permissions
About this article
Cite this article
Jensen, K.H., Knoblauch, J., Christensen, A.H. et al. Universal elastic mechanism for stinger design. Nat. Phys. 16, 1074–1078 (2020). https://doi.org/10.1038/s41567-020-0930-9
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41567-020-0930-9
This article is cited by
-
Investigation of the rate-mediated form-function relationship in biological puncture
Scientific Reports (2023)