Abstract
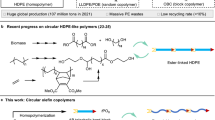
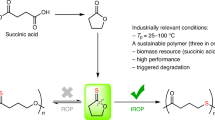
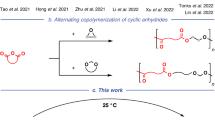
Geminal disubstitution of cyclic monomers is an effective strategy to enhance the chemical recyclability of their polymers, but it is utilized for that purpose alone and often at the expense of performance properties. Here we present synergistic use of gem-α,α-disubstitution of available at-scale, bio-based δ-valerolactones to yield gem-dialkyl-substituted valerolactones (\({\rm{VL}}^{{\rm{R}}_{2}}\)), which generate polymers that solve not only the poor chemical recyclability but also the low melting temperature and mechanical performance of the parent poly(δ-valerolactone); the gem-disubstituted polyesters (\({\rm{PVL}}^{{\rm{R}}_{2}}\)) therefore not only exhibit complete chemical recyclability but also thermal, mechanical and transport properties that rival or exceed those of polyethylene. Through a fundamental structure–property study that reveals intriguing impacts of the alkyl chain length on materials performance of \({\rm{PVL}}^{{\rm{R}}_{2}}\), this work establishes a simple circular, high-performance polyester platform based on \({\rm{VL}}^{{\rm{R}}_{2}}\) and highlights the importance of synergistic utilization of gem-disubstitution for enhancing both chemical recyclability and materials performance of sustainable polyesters.

This is a preview of subscription content, access via your institution
Access options
Access Nature and 54 other Nature Portfolio journals
Get Nature+, our best-value online-access subscription
$29.99 / 30 days
cancel any time
Subscribe to this journal
Receive 12 print issues and online access
$259.00 per year
only $21.58 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
Data availability
All of the data necessary to support the conclusions of this paper are provided in the paper and its Supplementary Information.
References
Shi, C. et al. Design principles for intrinsically circular polymers with tunable properties. Chem 7, 2896–2912 (2021).
Jambeck, J. R. et al. Plastic waste inputs from land into the ocean. Science 347, 768–771 (2015).
Geyer, R., Jambeck, J. R. & Law, K. L. Production, use, and fate of all plastics ever made. Sci. Adv. 3, e1700782 (2017).
The New Plastics Economy: Rethinking the Future of Plastics (World Economic forum, Ellen MacArthur Foundation, McKinsey and Company, 2016); www.ellenmacarthurfoundation.org/publications/the-new-plasticseconomy-rethinking-the-future-of-plastics
Borrelle, S. B. et al. Predicted growth in plastic waste exceeds efforts to mitigate plastic pollution. Science 369, 1515–1518 (2020).
Coates, G. W. & Getzler, Y. D. Y. L. Chemical recycling to monomer for an ideal, circular polymer economy. Nat. Rev. Mater. 5, 501–516 (2020).
Worch, J. C. & Dove, A. P. 100th anniversary of macromolecular science viewpoint: toward catalytic chemical recycling of waste (and future) plastics. ACS Macro Lett. 9, 1494–1506 (2020).
Kumar, A. et al. Hydrogenative depolymerization of nylons. J. Am. Chem. Soc. 142, 14267–14275 (2020).
Zhang, F. et al. Polyethylene upcycling to long-chain alkylaromaticsby tandem hydrogenolysis/aromatization. Science 370, 437–441 (2020).
Jehanno, C., Pérez-Madrigal, M. M., Demarteau, J., Sardon, H. & Dove, A. P. Organocatalysis for depolymerisation. Polym. Chem. 10, 172–186 (2019).
Fagnani, D. E. et al. 100th anniversary of macromolecular science viewpoint: redefining sustainable polymers. ACS Macro Lett. 10, 41–53 (2020).
Lau, W. W. Y. et al. Evaluating scenarios toward zero plastic pollution. Science 369, 1455–1461 (2020).
Lu, X.-B., Liu, Y. & Zhou, H. Learning nature: recyclable monomers and polymers. Chem. Eur. J. 24, 11255–11266 (2018).
Rahimi, A. & García, J. M. Chemical recycling of waste plastics for new materials production. Nat. Rev. Chem. 1, 0046 (2017).
Kaitz, J. A., Lee, O. P. & Moore, J. S. Depolymerizable polymers: preparation, applications, and future outlook. MRS Commun. 5, 191–204 (2015).
Hong, M. & Chen, E. Y. X. Chemically recyclable polymers: a circular economy approach to sustainability. Green Chem. 19, 3692–3706 (2017).
Hong, M. & Chen, E. Y. X. Future directions for sustainable polymers. Trends Chem. 1, 148–151 (2019).
Tang, X. & Chen, E. Y. X. Toward infinitely recyclable plastics derived from renewable cyclic esters. Chem 5, 284–312 (2019).
Zhang, X., Fevre, M., Jones, G. O. & Waymouth, R. M. Catalysis as an enabling science for sustainable polymers. Chem. Rev. 118, 839–885 (2018).
Schneiderman, D. K. & Hillmyer, M. A. 50th anniversary perspective: there is a great future in sustainable polymers. Macromolecules 50, 3733–3749 (2017).
Nishida, H. et al. Poly(tetramethyl glycolide) from renewable carbon, a racemization-free and controlled depolymerizable polyester. Macromolecules 44, 12–13 (2011).
Fahnhorst, G. W., Hoe, G. X. D., Hillmyer, M. A. & Hoye, T. R. 4-Carboalkoxylated polyvalerolactones from malic acid: tough and degradable polyesters. Macromolecules 53, 3194–3201 (2020).
Fahnhorst, G. W. & Hoye, T. R. A. Carbomethoxylated polyvalerolactone from malic acid: synthesis and divergent chemical recycling. ACS Macro Lett. 7, 143–147 (2018).
Cederholm, L., Olsén, P., Hakkarainen, M. & Odelius, K. Turning natural δ-lactones to thermodynamically stable polymers with triggered recyclability. Polym. Chem. 11, 4883–4894 (2020).
MacDonald, J. P. & Shaver, M. P. An aromatic/aliphatic polyester prepared via ring-opening polymerisation and its remarkably selective and cyclable depolymerisation to monomer. Polym. Chem. 7, 553–559 (2016).
Hong, M. & Chen, E. Y.-X. Completely recyclable biopolymers with linear and cyclic topologies via ring-opening polymerization of γ-butyrolactone. Nat. Chem. 8, 42–49 (2016).
Hong, M. & Chen, E. Y.-X. Towards truly sustainable polymers: a metal-free recyclable polyester from biorenewable non-strained γ-butyrolactone. Angew. Chem. Int. Ed. 55, 4188–4193 (2016).
Zhu, J.-B., Watson, E. M., Tang, J. & Chen, E. Y.-X. A synthetic polymer system with repeatable chemical recyclability. Science 360, 398–403 (2018).
Zhu, J. B. & Chen, E. Y.-X. Catalyst-sidearm-induced stereoselectivity switching in polymerization of a racemic lactone for stereocomplexed crystalline polymer with a circular life cycle. Angew. Chem. Int. Ed. 58, 1178–1182 (2019).
Cywar, R. M., Zhu, J.-B. & Chen, E. Y.-X. Selective or living organopolymerization of a six-five bicyclic lactone to produce fully recyclable polyesters. Polym. Chem. 10, 3097–3106 (2019).
Sangroniz, A. et al. Packaging materials with desired mechanical and barrier properties and full chemical recyclability. Nat. Commun. 10, 3559 (2019).
Xiong, W. et al. Geminal dimethyl substitution enables controlled polymerization of penicillamine-derived β-thiolactones and reversed depolymerization. Chem 6, 1831–1843 (2020).
Shi, C. et al. Hybrid monomer design for unifying conflicting polymerizability, recyclability, and performance properties. Chem 7, 670–685 (2021).
Shi, C. et al. High-performance pan-tactic polythioesters with intrinsic crystallinity and chemical recyclability. Sci. Adv. 6, eabc0495 (2020).
Yuan, J. et al. 4-Hydroxyproline-derived sustainable polythioesters: controlled ring-opening polymerization, complete recyclability, and facile functionalization. J. Am. Chem. Soc. 141, 4928–4935 (2019).
Yu, Y., Fang, L.-M., Liu, Y. & Lu, X.-B. Chemical synthesis of CO2-based polymers with enhanced thermal stability and unexpected recyclability from biosourced monomers. ACS Catal. 11, 8349–8357 (2021).
Liu, Y., Zhou, H., Guo, J.-Z., Ren, W.-M. & Lu, X.-B. Completely recyclable monomers and polycarbonate: approach to sustainable polymers. Angew. Chem. Int. Ed. 56, 4862–4866 (2017).
Ellis, W. C. et al. Copolymerization of CO2 and meso epoxides using enantioselective β-diiminate catalysts: a route to highly isotactic polycarbonates. Chem. Sci. 5, 4004–4011 (2014).
Saxon, D. J., Gormong, E. A., Shah, V. M. & Reineke, T. M. Rapid synthesis of chemically recyclable polycarbonates from renewable feedstocks. ACS Macro Lett. 10, 98–103 (2021).
Abell, B. A., Snyderl, R. L. & Coates, G. W. Chemically recyclable thermoplastics from reversible-deactivation polymerization of cyclic acetals. Science 373, 783–789 (2021).
Schneiderman, D. K. et al. Chemically recyclable biobased polyurethanes. ACS Macro Lett. 5, 515–518 (2016).
Lloyd, E. M. et al. Fully recyclable metastable polymers and composites. Chem. Mater. 31, 398–406 (2019).
Diesendruck, C. E. et al. Mechanically triggered heterolytic unzipping of a low-ceiling-temperature polymer. Nat. Chem. 6, 623–628 (2014).
Beromi, M. M. et al. Iron-catalysed synthesis and chemical recycling of telechelic 1,3-enchained oligocyclobutanes. Nat. Chem. 13, 156–162 (2021).
Sathe, D. et al. Olefin metathesis-based chemically recyclable polymers enabled by fused-ring monomers. Nat. Chem. 13, 743–750 (2021).
Chen, H., Shi, Z., Hsu, T.-G. & Wang, J. Overcoming the low driving force in forming depolymerizable polymers through monomer isomerization. Angew. Chem. Int. Ed. 60, 25493–25498 (2021).
Jung, M. E. & Piizzi, G. Gem-disubstituent effect: theoretical basis and synthetic applications. Chem. Rev. 105, 1735–1766 (2005).
Bachrach, S. M. The Gem-dimethyl effect revisited. J. Org. Chem. 73, 2466–2468 (2008).
Zhou, J., Sathe, D. & Wang, J. Understanding the structure–polymerization thermodynamics relationships of fused-ring cyclooctenes for developing chemically recyclable polymers. J. Am. Chem. Soc. 144, 928–934 (2022).
Save, M., Schappacher, M. & Soum, A. Controlled ring-opening polymerization of lactones and lactides initiated by lanthanum isopropoxide, 1 general aspects and kinetics. Macromol. Chem. Phys. 203, 889–899 (2002).
Larrañaga, A. & Lizundia, E. A review on the thermomechanical properties and biodegradation behaviour of polyesters. Eur. Polym. J. 121, 109296 (2019).
Rabnawaz, M., Wyman, I., Auras, R. & Cheng, S. A roadmap towards green packaging: the current status and future outlook for polyesters in the packaging industry. Green Chem. 19, 4737–4753 (2017).
Reinišová, L. & Hermanová, S. Poly(trimethylene carbonate-co-valerolactone) copolymers are materials with tailorable properties: from soft to thermoplastic elastomers. RSC Adv. 10, 44111–44120 (2020).
Schneiderman, D. K. & Hillmyer, M. A. Aliphatic polyester block polymer design. Macromolecules 49, 2419–2428 (2016).
Olsén, P., Odelius, K. & Albertsson, A.-C. Thermodynamic presynthetic considerations for ring-opening polymerization. Biomacromolecules 17, 699–709 (2016).
Tang, X. et al. Biodegradable polyhydroxyalkanoates by stereoselective copolymerization of racemic diolides: stereocontrol mechanism and polyolefin-like properties. Angew. Chem. Int. Ed. 59, 7881–7890 (2020).
Tang, X., Westlie, A. H., Watson, E. M. & Chen, E. Y.-X. Stereosequenced crystalline polyhydroxyalkanoates from diastereomeric monomer mixtures. Science 366, 754–758 (2019).
Häußler, M., Eck, M., Rothauer, D. & Mecking, S. Closed-loop recycling of polyethylene-like materials. Nature 590, 423–427 (2021).
Munnuri, S. et al. Catalyst-controlled diastereoselective synthesis of cyclic amines via C–H functionalization. J. Am. Chem. Soc. 139, 18288–18294 (2017).
Du, A. & Kowalski, A. in Handbook of Ring-Opening Polymerization (eds Dubois, P., Coulembier, O. & Raquez, J.-M.) Ch. 1 (Wiley-VCH, 2009).
Wang, Y. & Xu, T. Topology-controlled ring-opening polymerization of o-carboxyanhydride. Macromolecules 53, 8829–8836 (2020).
Acknowledgements
The work performed at Dalian University of Technology was supported by the National Natural Science Foundation of China (grant no. 21774017) and the Fundamental Research Funds for the Central Universities (grant no. DUT20LK35). The work performed at Colorado State University was supported by RePLACE (Redesigning Polymers to Leverage A Circular Economy) funded by the Office of Science of the US Department of Energy via award no. DE-SC0022290.
Author information
Authors and Affiliations
Contributions
X.-L.L., T.-Q. X. and E.Y.-X.C. conceived the idea and designed the experiments. X.-L.L., R.W.C. and J.-Y.J. performed the experiments, and analysed and processed the data. All authors co-wrote the manuscript and participated in data analyses and discussions.
Corresponding authors
Ethics declarations
Competing interests
T.X. and X.L. are named inventors on a Chinese patent application submitted by Dalian University of Technology that covers the polyester based on α,α-disubstituted valerolactones as well as the their preparation method and degradation. E.Y.-X.C. is a named inventor on a US patent application submitted by Colorado State University Research Foundation, which covers chemically circular semi-crystalline polyesters. The other authors declare no competing interests.
Peer review
Peer review information
Nature Chemistry thanks Joshua Worch and the other, anonymous, reviewer(s) for their contribution to the peer review of this work.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Supplementary information
Supplementary Information
Supplementary Figs. 1–114 and Tables 1–15.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Li, XL., Clarke, R.W., Jiang, JY. et al. A circular polyester platform based on simple gem-disubstituted valerolactones. Nat. Chem. 15, 278–285 (2023). https://doi.org/10.1038/s41557-022-01077-x
Received:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41557-022-01077-x
This article is cited by
-
Closed-loop recycling of sulfur-rich polymers with tunable properties spanning thermoplastics, elastomers, and vitrimers
Nature Communications (2024)
-
Hydrolysis properties of polyglycolide fiber mats mixed with a hyperbranched polymer as a degradation promoter
Polymer Journal (2024)
-
Proton-triggered topological transformation in superbase-mediated selective polymerization enables access to ultrahigh-molar-mass cyclic polymers
Nature Chemistry (2024)
-
Cyclic-acyclic monomers metathesis polymerization to access photodegradable polydicyclopentadiene and polyethylene-like materials
Science China Chemistry (2024)
-
Insights into substitution strategy towards thermodynamic and property regulation of chemically recyclable polymers
Nature Communications (2023)