Abstract
β-Nucleosides and their analogs are dominant clinically-used antiviral and antitumor drugs. α-Nucleosides, the anomers of β-nucleosides, exist in nature and have significant potential as drugs or drug carriers. Currently, the most widely used methods for synthesizing β- and α-nucleosides are via N-glycosylation and pentose aminooxazoline, respectively. However, the stereoselectivities of both methods highly depend on the assisting group at the C2’ position. Herein, we report an additive-controlled stereodivergent iodocyclization method for the selective synthesis of α- or β-nucleosides. The stereoselectivity at the anomeric carbon is controlled by the additive (NaI for β-nucleosides; PPh3S for α-nucleosides). A series of β- and α-nucleosides are prepared in high yields (up to 95%) and stereoselectivities (β:α up to 66:1, α:β up to 70:1). Notably, the introduced iodine at the C2’ position of the nucleoside is readily functionalized, leading to multiple structurally diverse nucleoside analogs, including stavudine, an FDA-approved anti-HIV agent, and molnupiravir, an FDA-approved anti-SARS-CoV-2 agent.
Similar content being viewed by others
Introduction
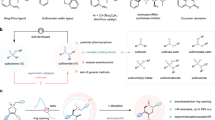
Nucleosides play vital roles in enzyme metabolism and regulation, cell signaling, and DNA and RNA synthesis1. Structurally, these molecules are composed of nucleobases covalently linked to five-membered sugars (ribose or deoxyribose) through glycosidic bonds at their C1’ positions (anomeric carbons) (Fig. 1a). A nucleoside whose nucleobase at C1’ is cis to the hydroxymethyl group at C4’ is referred to as a β-nucleoside, while the analogous trans-configured molecule is an α-nucleoside. These configurational differences lead to diverse biological functions and applications. Currently, β-nucleoside analogs are predominantly used to treat viruses and cancers, with almost half of the available antiviral medicines belonging to the β-nucleoside family2,3. In addition, 15 FDA-approved β-nucleoside analogs are currently used in clinical practice for anticancer chemotherapy4,5. In contrast, α-nucleosides exhibit intriguing physicochemical and biological activities, including high enzyme stabilities, unique parallel double-stranded structures, and inhibitory activities against tumors, bacteria, and plasmodia6. Selected β- and α-nucleosides are shown in Fig. 1a.
The coronavirus disease 2019 (COVID-19) pandemic caused by the severe acute respiratory syndrome coronavirus 2 (SARS-CoV-2) is the most devastating global crisis experienced in recent years. To date, the FDA has approved some medicines for the treatment of COVID-19, including remdesivir, molnupiravir, sotrovimab, paxlovid (nirmatrelvir and ritonavir), and others. Two of them, remdesivir (C-nucleoside) and molnupiravir (N-nucleoside), are nucleosidic drugs, while some other nucleoside candidates are being clinically assessed7,8,9,10,11. Because nucleoside analogs are crucial components of the arsenal used to battle COVID-19, the development of facile methods for the synthesis of nucleosides with high α/β selectivity is an urgent objective.
N-Glycosylation is the most widely used method for synthesizing nucleosides, especially β-nucleosides (Fig. 1b, blue arrow)12,13. The silyl-Hilbert-Johnson method developed by Vorbrüggen (Vorbrüggen glycosylation) is the dominant N-glycosylation protocol. It uses a strong Lewis acid to catalyze coupling between a per-acetylated sugar synthon and a per-trimethylsilylated nucleobase via a 1’, 2’-dioxolenium ion intermediate14,15,16,17. The α-face of the molecule is blocked to nucleophilic attack, which results in high β-selectivity. Yu et al. coined another representative strategy that involves the gold(I)-catalyzed N-glycosylation of a nucleobase with a glycosyl ortho-alkynylbenzoate via a glycosyl oxacarbenium intermediate18. β-Nucleosides are efficiently synthesized from various alkene- and alkyne-based sugars by Yu glycosylation and its developed methods18,19,20,21,22,23,24,25. In most glycosylation, the sugar synthons are fully protected. Hocek et al. reported the glycosylation of nucleobases with 5’-O-monoprotected ribose or C5’-modified ribose derivatives using modified Mitsunobu conditions to yield β-nucleosides26,27, whose anomeric selectivity highly depends on the hydroxy group at C2’ of the ribosyl donor. Together, the protecting groups of the sugar ring (substrate-dependent), especially the group at the C2’ position, determines the stereoselectivity at the anomeric C1’ carbon in these methods.
α-Nucleoside synthesis is relatively underexplored compared to its β-nucleoside counterpart. A pentose aminooxazoline usually serves as the key intermediate during the synthesis of an α-nucleoside, as it can be transformed into the pyrimidine α-nucleoside in several steps (Fig. 1b, blue arrow)6,28,29,30. However, poor α/β selectivity is a limitation of these current methods in the absence of a chimeric participating group at the C2’ position of the sugar synthon. The absence of an efficient method for the synthesis of α-nucleosides presents a major roadblock to the further exploration of α-nucleoside bioactivity.
In 2019, Trost et al. reported Pd-catalyzed iodoetherification chemistry for the enantioselective construction of pyrimidine-nucleoside analogs31. In particular, this method performed well during the stereoselective synthesis of nucleosides bearing seven-membered sugar rings. This pioneering study suggested that asymmetric sugar-ring formation is an efficient alternative nucleoside synthesis method (Fig. 1b, orange arrow). Asymmetric halocyclization of olefins is an important transformation to construct a wide spectrum of molecular structures32,33,34,35,36,37,38. However, their applications to the stereoselective synthesis of nucleosides are still underexplored.
Herein, we report a method for the synthesis of α- or β-nucleosides using additive-controlled asymmetric iodocyclization chemistry (Fig. 1c). Stereoselectivity is controlled by the additive (NaI or PPh3S), even while using the same chiral phosphine catalyst. Cyclic products bearing iodine at their C2’ positions are readily further functionalized, thereby providing an efficient and atom-economic approach to the synthesis of multifarious nucleosides.
Results and discussion
Our study commenced with the model combination of achiral alcohol 1a and N-iodosuccinimide (NIS). After catalysts and solvent screening, we found that chiral phosphoric acid C1 was the most effective catalyst for this transformation (see Supplementary Tables 6 and 7). Since the desired nucleosidic product 2a was generated along with a spot corresponding to the spontaneously cyclized product 3a observed by thin layer chromatography (TLC). To readily determine the yield and stereoselectivity of the reaction, the reaction solution was directly treated with 1,8-diazabicyclo [5.4.0] undec-7-ene (DBU) following iodoetherification, which completely converted the nucleosidic product 2a into the corresponding cyclic product 3a. Achiral alcohol 1a was treated with C1 (10 mol%), NIS (1.1 eq.), and Na2CO3 (0.1 eq.) in combination of PhMe and CHCl3 (1:1, v-v) at 0 °C, giving the product R−3a in 89% yield and 70% ee (Table 1, entry 1). We next, systematically screened the synthetic conditions for halogen sources. However, other halogen sources, such as I2, N-iodophthalimide (NIP), and 1,3-Diiodo-5,5-dimethylhydantoin (DIDMH), provided inferior results (entries 2–4). Further screening revealed that the additive influenced the conversion and enantioselectivity remarkably (see Supplementary Table 9), with NaI identified as the most effective additive for generating R−3a (93% yield, 96% ee; entry 6). The absolute configuration of R−3a was determined by single-crystal X-ray crystallography39. Unexpectedly, the S-enantiomer was mainly obtained when PPh3S was used as the additive (entry 7; 51% ee). The reaction was further improved when CH2Cl2 was used as the solvent, with S−3a formed in 90% ee and 94% yield (entry 8). We also examined other Lewis bases bearing bulky groups40, but found that PPh3S was the most effective S-selective additive (see Supplementary Table 9). Though the additive effect has been found in stereoselective glycosylations of six-membered-ring glycosyl donors41,42,43,44,45, to the best of our knowledge, no additive-controlled stereoselective method to synthesize nucleoside has been reported to date. Herein, we report an additive-controlled asymmetric iodocyclization methodology, with NaI as the R-directing (β-directing) additive and PPh3S as the S-directing (α-directing) additive.
With the optimized catalyst C1 and reaction conditions in hand, we next investigated the substrate scope for the R-selective process. In this study, we focused on pyrimidine as the nucleobase for nucleoside construction, since many pyrimidine-nucleoside analogs exhibit impressive bioactivities46,47,48, such as efficacy against COVID-19 (molnupiravir), human immunodeficiency virus (HIV, stavudine, zidovudine), herpes simplex virus (HSV, idoxuridine, trifluridine, brivudine), and hepatitis C virus (HCV, sofosbuvir). In addition, it is with worth that pyrimidine α-nucleoside analogs show intuitive bioactivities49,50. For instance, α-thymidine analogs inhibit plasmodium falciparum thymidylate kinase (PfTMPK), which is promising for the treatment of malaria in the clinic50.
The use of 5-ethylpyrimidine did not significantly affect the yield or selectivity, giving R−3b in high yield (92%) and ee (92%), as shown in Fig. 2. Thymine R−3c was also synthesized in excellent yield (89%) but with slightly lower ee (85%). Nucleobases bearing other substituents were also examined, with the desired products obtained in high yields (86–94%) and ees (90–99%), apart from R−3e, which was formed in slightly lower enantioselectivity (85% ee). The S-selective substrate scope was also investigated, with comparable reactivities and enantioselectivities to those obtained in the R-selective processes observed in most cases. However, particular examples, namely S−3c, S−3e, and S−3h, were synthesized in high yields and moderate enantioselectivities compared to the desired enantioselectivities of their R−3c, R−3e, and R−3h counterparts.
R-selective conditions (blue): 1a-i (0.1 mmol), C1 (0.01 mmol), NaI (0.01 mmol), NIS (0.11 mmol), and 4 Å MS (60 mg/mmol), PhMe: CHCl3 (1.2 mL: 1.2 mL), 0 °C for 8 h, then DBU (0.1 mmol), 0 °C for 30 min; S-selective conditions (red): 1a-i (0.1 mmol), C1 (0.01 mmol), PPh3S (0.01 mmol), NIS (0.11 mmol), and 4 Å MS (60 mg/mmol), CH2Cl2 (2.4 mL), 0 °C for 8 h, then DBU (0.1 mmol), 0 °C for 30 min.
The introduction of a functional group at the C4 position provides access to nucleoside-like structures that can be further transformed into β- or α-nucleosides. As shown in Scheme 2, we examined a chiral compound bearing a benzyl-ester at the C4 position under the described R-/S-selective conditions (referred to as β- or α-selective conditions, respectively), which led to 5a and 6a in excellent yields (93 and 94%, respectively) with β:α and α:β selectivities of 44:1 and 35:1, respectively. The absolute configurations of 5a and 6a were determined by single-crystal X-ray crystallography51. In the control reactions, products 5a and 6a were obtained in completely racemic forms in the absence of catalysis (neither NaI/C1 nor PPh3S/C1), which indicates that the chirality of the C4 position does not affect the stereoselectivity of the reaction.
We further investigated the selectivities of processes involving compounds with chiral C4 positions (Fig. 3). The methyl ester substituent was well tolerated, leading to a high yield of 5b and high β selectivity. In addition, a series of benzyl-ester-substituted alcohols, with various electron-donating or electron-withdrawing substituents on their pyrimidine moieties, exhibited excellent reactivities and stereoselectivities, with the desired β-nucleosides 5c–h obtained in high yields (up to 93%) and selectivities (β:α > 14:1). To closely mimic the nucleoside structure, we introduced a hydroxymethyl group with various protecting groups at the C4 position. All substrates performed well, with the exception of 5l, which was formed with relatively low stereoselectivity (β:α = 4:1). The phthalimide group was also well tolerated to give 5n in high yield (90%) and stereoselectivity (β:α = 19:1). The same substrates were also examined under selective conditions, with high yields and selectivities obtained. However, substrates bearing halo-substituents on the pyrimidine were exceptions; 6f–h were obtained in good yields (up to 83%) and moderate stereoselectivities (α:β ratios between 8:1 and 9:1). The absolute configurations of 5l and its diastereoisomer 6l were determined by single-crystal X-ray crystallography52.
To highlight the scalability of our method for nucleoside synthesis, β-nucleoside analog 5a and α-nucleoside analog 6a were synthesized at gram scale from 4a (Fig. 4). To our delight, yields and selectivities were maintained in these gram-scale reactions. The β-nucleoside analog 5a was obtained in high yield and β-selectivity (92% yield, β:α > 20:1), while the α-nucleoside analog 6a was prepared in 93% yield, with α:β > 20:1.
Next, we implemented the derivatizations of the mentioned β- and α-nucleoside analogs to demonstrate their usefulness as synthons (Figs. 5 and 6). As shown in Fig. 5, the iodine in 7 was readily reduced using radical chemistry (Bu3SnH/AIBN/PhMe) to give tetrahydrofuran derivative 8 in 85% yield. The benzyl carboxylate at the C4’ position in 8 was then reduced by NaBH4 to provide β-D-ddT 9, which is an anti-HIV agent candidate53. In addition, 5a was obtained by intramolecular cyclization when 7 was subjected to basic conditions. 3’-Deoxynucleoside analogs have been demonstrated as potential anticancer agents54. Thus, we performed the synthesis of 3’-deoxynucleoside 11 from 5a. NaBH4-mediated reduction (24 h) of 5a, in conjunction with ring opening, yielded compound 10 in 92% yield, whose primary alcohol was protected with a triphenylmethyl (Tr) group to generate the 3’-deoxynucleoside analog 11. When the NaBH4-mediated reduction of 5a was carried out in 30 min, only benzyl carboxylic group in 5a was reduced to yield alcohol intermediate, which was Tr-protected to give 5i in 84% yield over two steps. Compound 5i was added into aqueous sodium hydroxide and acetonitrile to give 11. With the key intermediate 11 in hand, we carried out the synthesis of others 3’-deoxynucleoside analogs 13, 14 and 15. Under the conditions of NMO/TPAP, 11 was oxidized to ketone 12 in 85% yield, which could be reversely reduced to 11. Ketone 12 was then reacted with allylmagnesium bromide to deliver 3’-deoxynucleoside 13 bearing a quaternary chiral carbon center at the C2’ position. Introduction of the azide group to 11 was accomplished by treatment with MsCl/Et3N/DMAP followed by NaN3, leading to 2’-azido-2’,3’-dideoxynucleoside 14 in 92% yield over two steps. Besides, via a two-step transformation, the β-amine group was introduced to 11 at C2’ position, yielding 2’-amino-2’,3’-dideoxynucleoside 15 in 89% yield. In addition, key tricyclic intermediate 5i could be constructed via the β-selective condition (Fig. 3) to synthesize stavudine, FDA-approved anti-HIV agent (Fig. 7). Ring opening and elimination of 5i with t-BuOK afforded the key intermediate 16. Stereospecifically, syn dihydroxylation of 16 with OsO4 followed by deprotection afforded 5-methyluridine (17), from which thymidine (18), a natural nucleoside, can be readily synthesized in three steps, as reported previously55.
Conditions: a. Bu3SnH, AIBN, PhMe, reflux, 85%; b. NaBH4, THF:H2O = 3:1, 0 °C to rt., 94%; c. DBU, PhMe:CHCl3 = 1:1, 0 °C, quant; d. NaBH4, THF:H2O = 3:1, rt., 92% of 10, 90% yield of 11 (12 to 11); e. TrCl, pyridine, DMAP, CH2Cl2, reflux; f. NaBH4, THF:H2O = 3:1, 0 °C, 84% yield over two step (5a to 5i); g. NaOH aq. (2.0 M), CH3CN, rt., quant; h. NMO, TPAP, CH2Cl2, rt., 85%; i. allylmagnesium bromide (1.0 M in THF), THF, –78 °C, 80%; j. MsCl, Et3N, DMAP, CH2Cl2, rt.; k. NaN3, DMF, 80 °C, 92% over two steps; l. PPh3, DEAD, phthalimide, THF, rt; m. N2H4·H2O, EtOH, 50 °C, 89% over two steps; n. t-BuOK, DMSO, rt., 82%; o. OsO4, NMO, acetone:H2O = 4:1, rt.; p. 80% AcOH aq., 50 °C, 88% over two steps.
Conditions: a. a. Bu3SnH, AIBN, PhMe, reflux; b. NaBH4, THF:H2O = 3:1, 0 °C to rt., 77% over two steps; c. DBU, CH2Cl2, 0 °C, quant; d. NaBH4, THF:H2O = 3:1, rt., 92%; e. NaBH4, THF:H2O = 3:1, 0 °C; f. TrCl, pyridine, DMAP, CH2Cl2, reflux; g. t-BuOK, DMSO, rt., 60% over three steps; h. 80% AcOH aq., 50 °C, 95%; i. OsO4, NMO, acetone:H2O = 4:1, rt., 62% yield of 24, 30% yield of 25.
a Synthesis of stavudine; b Synthesis of molnupiravir. Conditions: a. vinylmagnesium bromide (1 M in THF), CuI, THF, –78 °C, 1 h, quant; b. TBSCl, imidazole, CH2Cl2, 0 °C to rt., then O3, Et3N, –78 °C to rt., 91%; c. CrCl2, CHI3, THF, 0 °C, 82%; d. nucleobases (thymine for 4i, uracil for 4 m), CuTc, K3PO4, DMEDA, DMF, 75 °C, then TBAF, 79% yield of 4i, 78% yield of 4 m; e. C1, NIS, NaI, 4 Å MS, PhMe:CHCl3 = 1:1, then DBU, 89% of 5i (β:α = 13:1), 94% of 5 m (β:α = 40:1); f. t-BuOK, DMSO, rt., 85% of 16, 82% of 29; g. 80% AcOH aq., 50 °C, 97%; h. 1.0 M HCl aq., MeCN, rt; i. isobutyric anhydride, Et3N, DMAP, MeCN, 96% over two steps; j. OsO4, NMO, acetone:H2O = 4:1, rt., 94%; k. (NH2OH)2, H2SO4, NH4HSO4, imidazole, HMDS, 80 °C, 86%.
Furthermore, α-nucleoside 19 was also derivatized (Fig. 6); α-D-ddT (20) was synthesized from 19 in 77% yield over two steps. Treatment of 19 with DBU afforded 6a, which was transformed into 21 through slow reduction, while alkene 23 was readily obtained from 6a in a four-step sequence (reduction, protection of the alcohol, elimination, and deprotection). Moreover, the dihydroxylation of alkene 22 delivered a 2:1 ratio of diastereoisomers 24 and 25.
To further demonstrate the synthetic usefulness of our method, we applied it to the asymmetric synthesis of two FDA-approved agents, stavudine (anti-HIV) and molnupiravir (anti-SARS-Cov-2). As shown in Fig. 7a, stavudine was readily synthesized from the S-trityl glycidyl ether (26), a commercially available compound, in 7 steps and in 43% overall yield. Initially, the epoxide of 26 was subjected to ring opening with vinylmagnesium bromide to yield chiral alcohol 27. Then TBS-protection of alcohol group and oxidative cleavage of terminal alkene group were performed smoothly in one pot, affording aldehyde 28 in 91% yield. Iodination of 28 using CrCl2 and CHI3 gave the E-alkenyl iodide as the main product (E:Z > 10:1), from which the coupling with thymine and removal of the TBS group in one pot occurred to obtain the precursor of iodocyclization 4i in 65% yield over two steps. Then the β-selective iodocyclization was performed under optimized condition to generate β-nucleoside analog 5i in 89% yield (β:α = 13:1). Ring opening and elimination of 5i afforded 16 in the presence of t-BuOK in DMSO. After removal of the Tr group, stavudine was obtained. Besides, a ten-step route to synthesize molnupiravir in 35% overall yield was implemented (Fig. 7b). Precursor 4m was synthesized from 26 in 58% yield in four steps. Then the β-selective iodocyclization of 4m provided 5m in 94% yield with excellent β-selectivity (β:α = 40:1). The t-BuOK-mediated ring opening and elimination of 5m in one pot generated 29, which was coupled with isobutyric anhydride after deprotection, affording 30 in 79% yield over three steps (5m to 30). After subsequent dihydroxylation and hydroamination of 30, molnupiravir was successfully synthesized.
To gain a thorough understanding of the reaction mechanism, a series of control experiments using substrate 1a were performed as shown in Supplementary Table 12. To be specific, when the acidic site (-OH) of C1 was with a methoxy group (Supplementary Table 12, entries 6 and 20), the corresponding product 3a was nearly racemic (<5% ee). Besides, a chiral sodium phosphate C15 was prepared to catalyze the R-selective iodocyclization, which yielded R−3a in 88% yield with merely 35% ee (Supplementary Table 12, entry 7). Thus, the acidic site (-OH) of C1 is confirmed to play a critical role in the additive-controlled stereoselective iodocyclizations. When the R-selective reaction is performed in the absence of NIS (Supplementary Table 12, entry 2), no product was generated. Thus, NIS is determined to be the exclusive iodine source. When the R-selective reaction if performed in the absence of NaI (Supplementary Table 12, entry 3), R−3a was generated an 85% yield with only 20% ee. Combined with other NaI dosage screening experiments (Supplementary Table 12, entries 1 and 10–13), it is concluded that the additive NaI cooperates with C1, NIS and the substrate to catalyze R-selective iodocyclization in a unique manner benefitting stereoselectivity. To elucidate more details, density functional theory (DFT) studies were performed, using alkene 1a as model substrate (Fig. 8a). The R-selective iodocyclization starts favorably from Int-I rather than Int-I’, which is based on the calculated Gibbs energy difference of 24.1 kcal/mol between Int-I and Int-I’ (Int-I: –49.9 kcal/mol, Int-I’: –25.8 kcal/mol). It is worthwhile to mention that such intensely exothermic transformations are not observed without the NaI additive (–13.7 kcal/mol, Supplementary Fig. 7), in accordance with the conclusion that NaI is crucial in the catalytic system. Furthermore, we used interaction region indicator (IRI)56,57 and fuzzy bond order (FBO)58,59 to analyze the interactions between atoms of Int-I (IRI pic. of Int-I in Fig. 8a). Interestingly, NaI is found as a centered role that cooperates with C1, NIS and substrate 1a through LP…π interactions and Na-O interactions, providing an excellent stereoselective environment for R-selectivity. After electrophilic addition of the iodide anion to the alkene group of 1a, the Int-II is formed. The following nucleophilic cyclization occurs to generate P-R, with a reaction barrier of 9.6 kcal/mol (Int-II to TS-I).
In the S-selective iodocyclization, Lewis basic PPh3S was used as an essential additive (Supplementary Table 12, entries 16 and 17). Lewis bases have been reported to activate N-halosuccinimides through polarized covalent bonds or through noncovalent interactions40,60,61,62. To understand the interaction type of Lewis basic PPh3S and NIS in our S-selective iodocyclization, we monitored the course of the reaction by 31P NMR (Supplementary Fig. 6). It is suggested that PPh3S activates the electrophilicity of NIS through a weak noncovalent halogen bond, which promotes the forming of the transient halogen-bonded intermediate PInt-1. Meanwhile, computational studies of PPh3S-controlled stereoselective iodocyclizations are performed (Fig. 8b). In the first step, PPh3S enhances the reactivity of NIS via halogen bonding in Int-III, which is also supported by 31P NMR experiments (Fig. S1). Then the activated PInt-1 adds to the alkene group of 1a to form Int-IV and Int-IV’ releasing succinimide (NHS). Because of the large steric hindrance of C1 with PPh3S, Int-IV has lower energy than Int-IV’ (–12.0 vs –6.2 kcal/mol), facilitating the S-selectivity. The O-C bond between C1 and 1a is established smoothly to form Int-V, which is exothermic by 17.8 kcal/mol (Int-IV to Int-V) and requires only 1.7 kcal/mol of free energy to activate (Int-IV to TS-II). In the final step, the cyclization, which is an exothermic transformation (Int-V to P-S: –12.9 kcal/mol) via TS-III, occurs to produce P-S.
In summary, we developed an additive-controlled stereodivergent iodocyclization method for constructing β- and α-nucleoside analogs with remarkable yields and stereoselectivities that provides an efficient, stereodivergent, and versatile strategy for the synthesis of nucleosides. Meanwhile, pyrimidine-nucleoside products bearing iodine at their C2’ positions were shown to be crucial intermediates that can be further functionalized to yield structurally diverse nucleoside analogs. In addition, the anti-HIV drug stavudine and anti-SARS-Cov-2 drug molnupiravir were concisely synthesized using our method. To the best of our knowledge, few existing methods are capable of controlling stereoselectivity merely through using an additive, not to mention nucleoside synthesis. This work not only expands our fundamental chemical understanding. It also contributes to the battle against COVID-19 through facile nucleoside synthesis.
Methods
General procedure for R-selective (β-selective) reactions
Under an atmosphere of argon, 1a-i or 4a-n (0.1 mmol), C1 (0.01 mmol), NaI (0.01 mmol), and 4 Å MS (60 mg/mmol) were dissolved in anhydrous PhMe: CHCl3 (1.2 mL: 1.2 mL) and stirred at 0 °C for 15 min. After adding N-iodosuccinimide (NIS, 0.11 mmol), the reaction mixture was stirred at 0 °C for additional 8 h. After that, DBU (0.1 mmol) was added to the reaction mixture and stirred for 30 min. The solution was diluted with CHCl3 (2 mL) and saturated aqueous ammonium chloride (2 mL). The aqueous layer was extracted with CHCl3 (2 mL × 4). The combined organic layer was washed with brine (2 mL × 4), dried over Na2SO4 and filtered, concentrated in vacuo. The crude material was purified via silica gel column chromatography to obtain R−3a-i and 5a-n.
General procedure for S-selective (α-selective) reactions
Under an atmosphere of argon, 1a-i or 4a-n (0.1 mmol), C1 (0.01 mmol), PPh3S (0.01 mmol) and 4 Å MS (60 mg/mmol) were dissolved in anhydrous CH2Cl2 (2.4 mL) and stirred at 0 °C for 15 min. After adding NIS (0.11 mmol), the reaction mixture was stirred at 0 °C for 8 h. After that, DBU (0.1 mmol) was added to the reaction mixture and stirred for 30 min. The solution was diluted with CHCl3 (2 mL) and saturated aqueous ammonium chloride (2 mL). The aqueous layer was extracted with CHCl3 (2 mL × 4). The combined organic layer was washed with brine (2 mL × 4), dried over Na2SO4, and filtered, concentrated in vacuo. The crude material was purified via silica gel column chromatography to obtain S−3a-i and 6a-n.
Experimental data
For the experimental procedures and spectroscopic and physical data of compounds and the crystallographic data of compounds R−3a, 5a, 6a, 5l, and 6l, see Supplementary Methods. For optimization of the reaction conditions and mechanistic studies, see Supplementary Discussion. For NMR spectra of synthetic intermediates, see Supplementary Figs 9–299. For the comparisons of 1H and 13C NMR spectra of the known and synthetic β-D-ddT (9), stavudine, 5-methyluridine (17), and molnupiravir, see Supplementary Tables 15–21. For the HPLC analysis spectra of compounds R−3a-i, S−3a-i, 5a-n, and 6a-n, see Supplementary Figs. 300–322.
Data availability
The authors declare that the data supporting the findings of this study are available within the paper and its Supplementary Information files, and, also available from the corresponding author upon reasonable request. The Cartesian coordinates are shown in the Supplementary Data 1. Crystallographic data for compound R−3a (CCDC No. 2131198), https://www.ccdc.cam.ac.uk/mystructures/structuredetails/164e3c40-0367-ec11-96a5-00505695281c. Crystallographic data for compound 5a (CCDC No. 2131196), https://www.ccdc.cam.ac.uk/mystructures/structuredetails/c616fb23-0367-ec11-96a9-00505695f620. Crystallographic data for compound 6a (CCDC No. 2155961), https://www.ccdc.cam.ac.uk/mystructures/structuredetails/bab92800-3b9a-ec11-96aa-00505695281c. Crystallographic data for compound 5l (CCDC No. 2075890), https://www.ccdc.cam.ac.uk/mystructures/structuredetails/45dec435-0867-ec11-96a9-00505695f620. Crystallographic data for compound 6l (CCDC No. 2075734), https://www.ccdc.cam.ac.uk/mystructures/structuredetails/1bbd405a-0367-ec11-96a9-00505695f620.
References
Saenger, W. Principles of nucleic acid structure; Springer-Verlag New York, Incorporated: New York, NY, USA (1988).
Elzagheid, M. I. Nucleosides and nucleoside analogues as emerging antiviral drugs. Mini-Rev. Org. Chem. 18, 672–679 (2021).
Li, G., Yue, T., Zhang, P. & Gu, W. Drug discovery of nucleos(t)ide antiviral agents: dedicated to Prof. Dr. Erik De Clercq on occasion of his 80th Birthday. Molecules 26, 923 (2021).
Guinan, M., Benckendorff, C., Smith, M. & Miller, G. J. Recent advances in the chemical synthesis and evaluation of anticancer nucleoside analogues. Molecules 25, 2050 (2020).
Man, S., Lu, Y. & Yin, L. Potential and promising anticancer drugs from adenosine and its analogs. Drug Discov. Today 26, 1490–1500 (2021).
Ni, G. C., Du, Y. & Tang, F. Review of α-nucleosides: from discovery, synthesis to properties and potential applications. RSC Adv. 9, 14302–14320 (2019).
Nicola, B., Piccialli, G., Roviello, G. N. & Oliviero, G. Nucleoside analogs and nucleoside precursors as drugs in the fight against SARS-CoV-2 and other coronaviruses. Molecules 26, 986 (2021).
Jorgensen, S. C. J., Kebriaei, R. & Dresser, L. D. Remdesivir: review of pharmacology, pre-clinical data, and emerging clinical experience for COVID-19. Pharmacotherapy 40, 659–671 (2020).
Cully, M. A tale of two antiviral targets and the COVID-19 drugs that bind them. Nat. Rev. Drug Discov. 21, 3–5 (2021).
Tian, L. et al. RNA-dependent RNA poly-merase (RdRp) inhibitors: the current landscape and repurposing for the COVID-19 pandemic. Eur. J. Med. Chem. 213, 113201 (2021).
Tian, L. et al. Molnupiravir and its antiviral activity against COVID-19. Front. Immunol. 13, 855496 (2022).
Kaspar, F., Stone, M. R. L., Neubauer, P. & Kurreck, A. Route efficiency assessment and review of the synthesis of β-nucleosides via N-glycosylation of nucleobases. Green. Chem. 23, 37–50 (2021).
Wang, H., Zhong, Y., Xiao, Y. & Chen, F. Chemical and chemoenzymatic stereoselective synthesis of β-nucleosides and their analogues. Org. Chem. Front. 9, 1719−1741 (2022).
Niedballa, U. & Vorbrüggen, H. A general pyrimidine nucleosides. Angew. Chem. Int. Ed. 9, 461–462 (1970).
Niedballa, U. & Vorbrüggen, H. A general synthesis of N-glycosides. I. synthesis of pyrimidine nucleosides. J. Org. Chem. 39, 3654–3660 (1974).
Wang, Z., Prudhomme, D. R., Buck, J. R. & Park, M. Stereocontrolled syntheses of deoxyribonucleosides via photoinduced electron-transfer deoxygenation of benzoyl-protected ribo- and arabinonucleosides. J. Org. Chem. 65, 5969–5985 (2000).
Shen, B. & Jamison, T. F. Rapid continuous synthesis of 5’-deoxyribonucleosides in flow via Brønsted acid catalyzed glycosylation. Org. Lett. 14, 3348–3351 (2012).
Zhang, Q. & Yu, B. An efficient approach to the synthesis of nucleosides: gold(I)-catalyzed N-glycosylation of pyrimidines and purines with glycosyl ortho-alkynyl benzoates. Angew. Chem. Int. Ed. 50, 4933–4936 (2011).
Yang, F., Zhu, Y. & Yu, B. dramatic concentration effect on the stereoselectivity of N-glycosylation for the synthesis of 2’-deoxy-β-ribonucleosides. Chem. Commun. 48, 7097–7099 (2012).
Tang, Y., Li, J., Zhu, Y., Li, Y. & Yu, B. Mechanistic insights into the gold(I)-catalyzed activation of glycosyl ortho-alkynylbenzoates for glycosidation. J. Am. Chem. Soc. 135, 18396–18405 (2013).
Hu, Z., Tang, Y. & Yu, B. Glycosylation with 3,5-dimethyl-4-(2’-phenylethynyl-phenyl)phenyl (EPP) glycosides via a dearomative activation mechanism. J. Am. Chem. Soc. 141, 4806–4810 (2019).
Li, P. et al. Glycosyl ortho-(1-phenylvinyl)benzoates versatile glycosyl donors for highly efficient synthesis of both O-glycosides and nucleosides. Nat. Common. 11, 405 (2020).
Li, P. et al. Ortho-(1-phenylvinyl)benzyl glycosides: ether-type glycosyl donors for the efficient synthesis of both O-glycosides and nucleosides. Green. Synth. Catal. 1, 160–166 (2020).
Liu, R. et al. NIS/TMSOTf-promoted glycosidation of glycosyl orthohexynylben-zoates for versatile synthesis of O-glycosides and nucleosides. J. Org. Chem. 86, 4763–4778 (2021).
He, H. et al. An orthogonal and reactivity-based one-pot glycosylation strategy for both glycan and nucleoside synthesis: access to TMG-chitotriomycin, lipochitooligosaccharides and capuramycin. Chem. Sci. 12, 5143–5151 (2021).
Downey, A. M., Richter, C., Pohl, R., Mahrwald, R. & Hocek, M. Direct one-pot synthesis of nucleosides from unprotected or 5-O-monoprotected D-ribose. Org. Lett. 17, 4604–4607 (2015).
Downey, A. M., Pohl, R., Roithov, J. & Hocek, M. Synthesis of nucleosides through direct glycosylation of nucleobases with 5-O-monoprotected or 5-modified ribose: improved protocol, scope, and mechanism. Chem. Eur. J. 23, 3910–3917 (2017).
Shannahoff, D. H. & Sanchez, R. A. 2,2’-Anhydropyrimidine nucleosides. Novel syntheses and reactions. J. Org. Chem. 38, 593–598 (1973).
Sawai, H., Nakamura, A., Hayashi, H. & Shinozuka, K. Facile synthesis of α-anomeric pyrimidine nucleosides. Nucleosides Nucleotides Nucleic Acids 13, 1647–1654 (1994).
Shinozuka, K., Matsumoto, N. & Nakamura, A. Stereospecific synthesis of α-anomeric pyrimidine nucleoside. Nucleic Acids Symp. Ser. 44, 21–22 (2000).
Trost, B. M., Xu, S. & Sharif, E. U. New catalytic asymmetric formation of oxygen heterocycles bearing nucleoside bases at the anomeric carbon. J. Am. Chem. Soc. 141, 10199–10204 (2019).
Yan, J. et al. The applications of catalytic asymmetric halocyclization in natural product synthesis. Org. Chem. Front. 9, 499–516 (2022).
Huang, D. et al. Enantioselective bromocyclization of olefins catalyzed by chiral phosphoric acid. Org. Lett. 13, 6350–6353 (2011).
Tripathi, C. B. & Mukherjee, S. Catalytic enantioselective iodoetherification of oximes. Angew. Chem. Int. Ed. 52, 8450–8453 (2013).
Tay, D. W., Leung, G. Y. C. & Yeung, Y. Y. Desymmetrization of diolefinic diols by enantioselective amino-thiocarbamate-catalyzed bromoetherification: synthesis of chiral spirocycles. Angew. Chem. Int. Ed. 53, 5161–5164 (2014).
Shen, Z. et al. Chiral ion-pair organocatalyst promotes highly enantioselective 3-exo iodocycloetherification of allyl alcohols. Chem. Sci. 6, 6986–6990 (2015).
Xia, Z. et al. Enantioselective bromo-oxycyclization of silanol. Org. Lett. 18, 80–83 (2016).
Lu, Y., Nakatsuji, H., Okumura, Y. & Ishihara, K. Enantioselective halo-oxy- and halo-azacyclizations induced by chiral amidophosphate catalysts and halo-lewis acids. J. Am. Chem. Soc. 140, 6039–6043 (2018).
CCDC 2131198 (R-3a) contains the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Denmark, S. E. & Burk, M. T. Lewis base catalysis of bromo- and iodolactonization, and cycloetherification. PNAS 107, 20655–20660 (2010).
Geng, Y. & Ye, X. Additive-controlled stereoselective glycosylations of oxazolidinone- protected glucosamine and galactosamine thioglycoside donors based on preactivation protocol. Synlett 16, 2506–2512 (2010).
Wasonga, G., Zeng, Y. & Huang, X. Pre-activation based stereoselective glycosylations: stereochemical control by additives and solvent. Sci. China Chem. 54, 67–73 (2011).
Geng, Y., Qin, Q. & Ye, X. Lewis acids as α-directing additives in glycosylations by using 2,3-O-carbonate-protected glucose and galactose thioglycoside donors based on preactivation protocol. J. Org. Chem. 77, 5255–5270 (2012).
Qin, Q., Xiong, D. & Ye, X. Additive-controlled stereoselective glycosylations of 2,3-oxazolidinone protected glucosamine or galactosamine thioglycoside donors with phenols based on preactivation protocol. Carbohydr. Res. 403, 104–114 (2015).
Herman, S. O. & Jeroen, D. C. Reagent controlled stereoselective synthesis of α-glucans. J. Am. Chem. Soc. 140, 4632–4638 (2018).
Mengshetti, S. et al. Discovery of a series of 2’-α-fluoro, 2’-β-bromo-ribonucleosides and their phosphoramidate prodrugs as potent pan-genotypic inhibitors of hepatitis C virus. J. Med. Chem. 62, 1859–1874 (2019).
Yang, Q. et al. Synthesis and biological evaluation of 4-substituted fluoronucleoside analogs for the treatment of hepatitis B virus infection. J. Med. Chem. 58, 3693–3703 (2015).
Agarwal, H. K. et al. Synthesis and evaluation of thymidine kinase 1-targeting carboranyl pyrimidine nucleoside analogs for boron neutron capture therapy of cancer. Eur. J. Med. Chem. 100, 197–209 (2015).
Cui, H. et al. Synthesis and evaluation of α‑thymidine analogues as novel antimalarials. J. Med. Chem. 55, 10948–10957 (2010).
Daele, I. V., Froeyen, M. & Calenbergh, S. V. Rational design of 5’-thiourea-substituted α-thymidine analogues as thymidine monophosphate kinase inhibitors capable of inhibiting mycobacterial growth. J. Med. Chem. 50, 5281–5292 (2007).
CCDC 2131196 (5a) and CCDC 2155961 (6a) contain the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
CCDC 2075890 (5l) and CCDC 2075734 (6l) contain the supplementary crystallographic data for this paper. These data can be obtained free of charge from The Cambridge Crystallographic Data Centre via www.ccdc.cam.ac.uk/data_request/cif.
Martin, A., Souza, D. D., Feiertag, P. & Honig, H. A new concept for the preparation of β-L- and β-D-2′,3′-dideoxynucleoside analogues. Org. Lett. 4, 3251–3254 (2002).
Lin, T. S. et al. Synthesis and anticancer activity of various 3’-deoxy pyrimidine nucleoside analogues and crystal structure of l-(3-Deoxy-β-D-threo-pentofuranosy)cytosine. J. Med. Chem. 34, 693–701 (1991).
Moyroud, E., Baila, E. & Strazewski, P. Synthesis and enzymatic digestion of an RNA nonamer in both enantiomeric forms. Tetrahedron 56, 1475–1484 (2000).
Lu, T. & Chen, Q. Interaction region indicator: a simple real space function clearly revealing both chemical bonds and weak interactions. Chem. Methods 1, 231–239 (2021).
Lu, T. & Chen, F. Multiwfn: a multifunctional wavefunction analyzer. J. Comput. Chem. 33, 580–592 (2012).
Mayer, I. & Salvador, P. Overlap populations, bond orders and valences for ‘fuzzy’ atoms. Chem. Phys. Lett. 383, 368–375 (2004).
Matito, E., Poater, J., Sola‘, M., Duran, M. & Salvador, P. Comparison of the AIM delocalization index and the myer and fuzzy atom bond orders. J. Phys. Chem. A. 109, 9904–9910 (2005).
Bulfield, D. & Huber, S. M. Halogen bonding in organic synthesis and organocatalysis. Chem. Eur. J. 22, 14434–14450 (2016).
Guha, S., Kazi, I., Nandy, A. & Sekar, G. Role of lewis-base-coordinated halogen(I) intermediates in organic synthesis: the journey from unstable intermediates to versatile reagents. Eur. J. Org. Chem. 2017, 5497−5518 (2017).
Maddox, S. M., Dinh, A. N., Armenta, F., Um, J. & Gustafson, J. L. The catalyst-controlled regiodivergent chlorination of phenols. Org. Lett. 18, 5476–5479 (2016).
Acknowledgements
We are grateful for the financial support from the National Natural Science Foundation of China (Grant No. 22201192).
Author information
Authors and Affiliations
Contributions
F.C. and H.W. conceived the idea, guided the project. H.W. wrote the manuscript with feedbacks from other authors. Q.W. made the initial observations and analyzed the results. Q.W., J.M., Y.Z., Q.L., and Y.L. explored substrate scope and performed derivatizations. J.Z. performed the density functional theory calculations on the reaction mechanism. L.W. did experiment assistance and analysis for NMR.
Corresponding authors
Ethics declarations
Competing interests
The authors declare no competing interests.
Peer review
Peer review information
Nature Communications thanks Michael Downey, and the other, anonymous, reviewers for their contribution to the peer review of this work. Peer reviewer reports are available.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Wang, Q., Mu, J., Zeng, J. et al. Additive-controlled asymmetric iodocyclization enables enantioselective access to both α- and β-nucleosides. Nat Commun 14, 138 (2023). https://doi.org/10.1038/s41467-022-35610-w
Received:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41467-022-35610-w
Comments
By submitting a comment you agree to abide by our Terms and Community Guidelines. If you find something abusive or that does not comply with our terms or guidelines please flag it as inappropriate.