Abstract
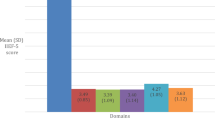
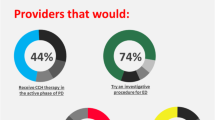
This retrospective descriptive analysis explores underrepresented minority men in our clinical trial for restorative therapy for erectile dysfunction and to identify strategies to promote diversity and inclusion in the study population. Demographic data were collected from all participants and the prevalence in our population was compared to the source population. The proportion of individuals taking part in our clinical trial was compared to the overall disease population using the participation to prevalence ratio. Among the 61 participants enrolled in the Platelet-Rich Plasma for Erectile Dysfunction trial, 72.1% were Hispanic compared to 39.9% in the national source population. There were 27.9% non-Hispanic participants, and 41.2% non-Hispanic men in the local South Florida population. The racial composition of our study shows 80.3% of PRP participants identify as White, 16.4% as Black, and 3.4% as Asian. In the national source population, 61.8% of patients were White, 27.5% are black, and 1.5% are Asian. Through the implementation of strategies such as having Hispanic team members on the clinical trial staff and providing education and outreach materials both in Spanish and English, we were able to overcome barriers to participation in Hispanic men and potentially improve health outcomes for underrepresented minority men with erectile dysfunction.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 8 print issues and online access
$259.00 per year
only $32.38 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout

Similar content being viewed by others
Data availability
The data that support the findings of this study are openly available in PUBMED at https://doi.org/10.1097/JU.0000000000003481. The authors also confirm that the data supporting the findings of this study are available within the article and its Supplementary Materials. Local and National epidemiology data also available publicly at URIDE and HCN networks.
References
United States Census Bureau. Census.gov. Demographic Turning Points for the United States: Population Projections for 2020 to 2060. 2023. https://www.census.gov/library/publications/2020/demo/p25-1144.html.
Fisher JA, Kalbaugh CA. Challenging assumptions about minority participation in US clinical research. Am J Public Health. 2011;101:2217–22.
Hamel LM, Penner LA, Albrecht TL, Heath E, Gwede CK, Eggly S. Barriers to clinical trial enrollment in racial and ethnic minority patients with cancer. Cancer Control. 2016;23:327–37.
Owens-Walton J, Williams C, Rompré-Brodeur A, Pinto PA, Ball MW. Minority enrollment in phase II and III clinical trials in urologic oncology. J Clin Oncol. 2022;40:1583–9.
Balakrishnan AS, Palmer NR, Fergus KB, Gaither TW, Baradaran N, Ndoye M, et al. Minority recruitment trends in phase III prostate cancer clinical trials (2003 to 2014): progress and critical areas for improvement. J Urol. 2019;201:259–67.
Burroughs VJ, Maxey RW, Levy RA. Racial and ethnic differences in response to medicines: towards individualized pharmaceutical treatment. J Natl Med Assoc. 2002;94:1–26.
Chen T, Li S, Eisenberg ML. Associations between race and erectile dysfunction treatment patterns. Urol Pract. 2022;9:423–30.
Taitt HE. Global trends and prostate cancer: a review of incidence, detection, and mortality as influenced by race, ethnicity, and geographic location. Am J Mens Health. 2018;12:1807–23.
Kessler A, Sollie S, Challacombe B, Briggs K, Van Hemelrijck M. The global prevalence of erectile dysfunction: a review: Global prevalence of erectile dysfunction. BJU Int. 2019;124:587–99.
Saltzman RG, Zucker I, Campbell K, Gandhi DA, Otiono K, Weber A, et al. An evaluation of race-based representation among men participating in clinical trials for prostate cancer and erectile dysfunction. Contemp Clin Trials Commun. 2022;29:100986.
Duma N, Vera Aguilera J, Paludo J, Haddox CL, Gonzalez Velez M, Wang Y, et al. Representation of minorities and women in oncology clinical trials: review of the past 14 years. J Oncol Pract. 2018;14:e1–10.
Occa A, Morgan SE, Potter JE. Underrepresentation of Hispanics and other minorities in clinical trials: recruiters’ perspectives. J Racial Ethn Health Disparities. 2018;5:322–32.
Friedman DB, Bergeron CD, Foster C, Tanner A, Kim SH. What do people really know and think about clinical trials? A comparison of rural and urban communities in the South. J Community Health. 2013;38:642–51.
Diehl KM, Green EM, Weinberg A, Frederick WA, Holmes DR, Green B, et al. Features associated with successful recruitment of diverse patients onto cancer clinical trials: report from the American College of Surgeons Oncology Group. Ann Surg Oncol. 2011;18:3544–50.
Javier-DesLoges J, Nelson TJ, Murphy JD, McKay RR, Stewart TF, Kader AK, et al. An evaluation of trends in the representation of patients by age, sex, and diverse race/ethnic groups in bladder and kidney cancer clinical trials. Urol Oncol Semin Orig Investig. 2022;40:199.e15–21.
Kupelian V, Link CL, Rosen RC, McKinlay JB. Socioeconomic status, not race/ethnicity, contributes to variation in the prevalence of erectile dysfunction: results from the Boston Area Community Health (BACH) Survey. J Sex Med. 2008;5:1325–33.
Akbar A, Liu K, Michos ED, Brubaker L, Markossian T, Bancks MP, et al. Racial differences in urinary incontinence prevalence, overactive bladder and associated bother among men: the Multi-Ethnic Study of Atherosclerosis. J Urol. 2021;205:524–31.
Squires A, Sadarangani T, Jones S. Strategies for overcoming language barriers in research. J Adv Nurs. 2020;76:706–14.
Funding
This work was supported by NIH Grant R01 DK130991 to RR.
Author information
Authors and Affiliations
Contributions
BRL, RR, JW, and RGS designed research; BRL and RGS performed research; BRL and RGS analyzed data; and BRL, JT, RGS, AG, AA, MCSA, and RR wrote the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Consent for publication
All authors have provided their consent for publication.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Springer Nature or its licensor (e.g. a society or other partner) holds exclusive rights to this article under a publishing agreement with the author(s) or other rightsholder(s); author self-archiving of the accepted manuscript version of this article is solely governed by the terms of such publishing agreement and applicable law.
About this article
Cite this article
Ledesma, B.R., Thomas, J., Ghomeshi, A. et al. Exploring representation of underrepresented minority men in a restorative therapy clinical trial for erectile dysfunction: addressing barriers and promoting inclusion. Int J Impot Res (2023). https://doi.org/10.1038/s41443-023-00747-9
Received:
Revised:
Accepted:
Published:
DOI: https://doi.org/10.1038/s41443-023-00747-9