Abstract
Given that cardiovascular diseases remain a primary cause of mortality and morbidity, there is a need to consider preventative strategies to improve vascular function from early in life. The aims of this study were therefore to investigate which interventions may improve endothelial function, intima media thickness and arterial stiffness in children and young people and to assess whether these interventions differ in boys and girls. A systematic literature search of Science Direct, Pubmed, Google Scholar and the Cochrane Library by two independent reviewers was performed to source articles. Inclusion criteria were any studies including any child ≤18 years of age receiving an intervention, which measured vascular function other than blood pressure. Exclusion criteria were studies assessing children with chronic medical conditions. A total of 72 studies were identified, which met the inclusion criteria. A measurable change in outcome was more likely to be reported in studies investigating endothelial function (p = 0.03). Interventions which improved vascular function included physical activity and dietary programmes. Under 10% of studies considered sex differences. In conclusion, school-based physical activity interventions are most likely to result in improvements in vascular function. Endothelial function may be the first variable of vascular function to change secondary to an intervention. Standardisation of reporting of differences between the sexes is essential to be able to ensure interventions are equally effective for boys and girls.
Similar content being viewed by others
Introduction
Cardiovascular diseases remain the primary cause of mortality and a major cause of morbidity globally [1]. Risk factors such as obesity, hypertension and atherogenic lipid profiles are rising in children, with this rise being attributed to pregnancy complications, genetic inheritance and lifestyle and environment in childhood and adolescence [2]. Sexual dimorphism is seen in many important physiological mechanisms for the development of cardiovascular disease, although the underlying reasons behind why females and males should have these differences have not yet been fully elucidated. Mechanisms may include developmental programming, differences in sex steroid production, oxidative stress and calcium signalling [3]. There are now standardised ways to assess vascular function in children, resulting in international collaborations to establish reference ranges, which are age- and sex-matched and validated in large population datasets [4] and mitigation of cardiovascular risk in childhood has been shown to improve outcomes later in life [5,6,7]. As such, there is a need to assess which interventions are most effective in childhood and adolescence to improve vascular health, prior to their implementation through large-scale public health campaigns.
The primary aim of this study was, therefore, to investigate which interventions may improve endothelial function, intima media thickness (IMT) and arterial stiffness in healthy children and young people under the age of 18 years. The secondary aim was to assess whether these interventions differ in boys and girls. Endothelial function, IMT and arterial stiffness were chosen as they represent the most standardised and commonly used methods for assessing vascular structure and function in children and young people to date.
Methods
A systematic review was conducted following the Preferred Reporting Items for Systematic Reviews and Meta-Analyses (PRISMA) reporting guidance. Studies had to fulfil the following criteria as per the PICO principle to be eligible. These were determined at the start of the project:
-
P: Participant: Children and young people ≤18 years of age at time of recruitment
-
I: Intervention: Any
-
C: Comparison of effect to improve vascular function
-
O: Outcome: Improvement in arterial stiffness, endothelial function and IMT
A systematic computerised literature search of Science Direct (previously Embase), PubMed, Google Scholar and the Cochrane Library by two independent reviewers (SE, MF) was performed to source articles in October 2021. The following key terms were searched for: (Vascular OR carotid intima media thickness OR Atherosclerosis OR pulse wave OR flow mediated dilatation OR FMD OR endothelial OR arterial stiffness OR remodelling) AND (Child OR youth). Bibliographic references were manually searched for potentially relevant studies based on the inclusion and exclusion criteria.
Study inclusion
Two authors (SE, MF) independently reviewed the titles, abstracts and bodies of all studies identified by the search in a sequential fashion, to identify which were eligible for inclusion using the Covidence platform (www.covidence.org). After independent evaluation at each stage, the authors convened and discussed which articles should be included. Disagreements were evaluated by a third author (ALH).
The authors initially screened titles, abstracts and then full text to inform decisions on inclusion. For inclusion in the review, studies had to include participants who were aged ≤18 years of age. Studies were included that used an intervention of any kind with the expectation of improving vascular outcome, and which assessed an effect of any measure of vascular function. Only original research was included. Studies which assessed the effects of interventions within any chronic health condition e.g., type 1 diabetes were excluded. There were no language restrictions imposed.
Data extracted
Two authors (SE, MF) independently extracted the data from the studies that met the inclusion criteria and quality standards using the Covidence review software. Data extracted included; authors, year and location of studies, type of study, setting, number of participants (sex, age), type of intervention, follow up period and measures of vascular function.
Quality review
Assessment of bias was undertaken using the Cochrane tool, available on the Covidence platform, which assesses the methodological quality of the papers, as well as incomplete data, outcome reporting and other sources of bias. This was again performed independently by two reviewers (SE, MF). Disagreements were resolved by a third reviewer (ALH).
Results
Study selection
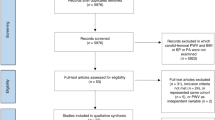
Figure 1 demonstrates the PRISMA flow diagram with the numbers of included and excluded studies at each step of the review. A high number of studies was initially identified (14,640) using the search strategy above after removal of duplicates. A total of 98% of these however did not meet the PICO eligibility criteria and as such were excluded. Thereafter, 257 full text articles were reviewed. Of the 184 (70%) excluded at this stage, 99 (54%) were due to irrelevant outcomes being reported; 38 (21%) were due to the wrong patient population (adult/chronic health condition) being included; 29 (16%) were because the protocol only had been published, with the results pending; 9 (5%) were because only conference abstracts with insufficient data were available and 9 (5%) were because they were review articles only and did not contain original data.
Characteristics of included studies
Of the 72 studies included (Table 1), the median (range) number of participants was 60 (11, 1826) and the median age of participants was 12.1 years (0.8, 21.6). The dates of publication ranged from 2004–2021 and they were published from 29 countries in 5 continents with a total of 15,757 children studied. In terms of study design, 39 (53%) were randomised controlled trials, whereas 26 (37%) were prospective cohort studies, 5 (7%) were non-randomised controlled studies and 3 (4%) reviewed retrospective data. The majority of the studies were assessed as having a ‘medium’ risk of bias (n = 48, 65%).
Of the 72 studies, 34 (46%) studies reported endothelial function (27 (79%) flow mediated dilatation (FMD); 5 (7%) reactive hyperaemia index; 2 (6%) plethysmography). For assessment of vascular structure, 29 (39%) examined IMT (27 (93%) CIMT and 1 (3%) aortic IMT). For assessment of arterial stiffness, 30 (41%) measured pulse wave velocity (PWV). Of these, only 7 (23%) measured carotid-femoral PWV. The median (range) number of methods of measuring vascular structure and function per study was 1 [1, 3]. Studies were more likely to identify a significant improvement in vascular structure and function when measuring endothelial function (n = 28, 82% of total) compared to IMT (n = 16, 53% of total) or arterial stiffness (n = 19, 63% of total) (p = 0.03).
The interventions examined were physical activity alone in 40 (55%); dietary intervention alone in 12 (16%); dietary supplements in 3 (4%); a combination of lifestyle measures in 9 (12.5%); and various methods of drug therapy in 9 (12.5%). The median duration of intervention was 0.5 years (0, 15). In general, there was no association between duration of intervention and likelihood of finding an improvement in vascular function (r2 = 0.01, p = 0.2).
Physical activity interventions
Physical activity interventions improved markers of vascular health in 29 out of 40 (74%) included studies (Table 1) and of these 29, 16 (55%) were school-based interventions. Although the interventions varied, 12 of the 29 studies (41%) included a structured exercise programme performed 3 times per week [8,9,10,11,12,13,14,15,16,17,18]. Fifteen of these 29 studies recruited only obese or overweight children and found improvements in terms of IMT and FMD [14,15,16,17,18,19,20,21,22] were consistent, but not in arterial stiffness or other markers of endothelial function [23,24,25,26,27]. In non-obese children, all 7 studies of exercise intervention, ranging from resistance training to active dance-based video game use, which measured FMD demonstrated significant increases in endothelial function [28,29,30,31,32,33]. Of the 9 studies [34,35,36,37,38,39,40,41,42] which measured CIMT, 2 (22%) found a decrease in IMT with exercise training, one with shuttle run training [34] and one finding that an increase in intensity and duration of any form of exercise resulted in improvements [35]. Results were inconsistent in the 12 studies, which measured PWV, with 6 (50%) [10, 12, 35, 43,44,45] finding reductions in arterial stiffness and 6 (50%) [39,40,41,42, 46, 47] finding no differences after the introduction of the exercise intervention. One study examined central retinal arteriolar and venular vessel diameters as early markers of vascular dysfunction and found that these reduced after an 18 month school-based physical activity programme [48].
Dietary interventions
Of the 12 studies (Table 1) which introduced dietary interventions, nutritional alterations included increasing fatty acids [49]; increasing casein [50]; increasing ketones [51]; and increasing protein intake [52, 53] with no significant differences found. The introduction of low calorie diets reduced IMT and arterial stiffness [54], but did not affect endothelial function [55]. The Dietary Interventions to Stop Hypertension (DASH) programme, consisting of face-to-face dietetic input and a specialised meal plan was found to improve endothelial function in adolescents [56, 57]. In comparison, the Special Turku Coronary Risk Factor Intervention Project for Children (STRIP) studies, incorporating 1042 participants found that counselling alone from dietetic professionals regarding low cholesterol/low saturated fat diets introduced in infancy and maintained at 3–12 monthly intervals until 20 years of age significantly increased FMD and decreased IMT [6, 58, 59], without specific meal plans. That said, the increase in FMD was only established in boys (p = 0.0034) and not in girls (p = 0.69) [59].
Drugs and dietary supplements
Single studies assessing the impact of alpha-lipoic acid [60], extract of citrus fruit peels [61], exenatide [62], folic acid [63] and vitamin C [64] found no statistically significant changes in vascular function (Table 2). One study of 67 obese adolescents (30% female) aged between 11–18 years reported that a 10 week prescription of orlistat 120 mg three times daily improved endothelial function, as measured by FMD, as well as reducing bodyweight, BMI and fasting total and LDL-cholesterol when combined with dietary control alone (% change in FMD 1.0 (0.5–1.6) vs 0.1 (−0.1–0.4, p < 0.001) [65]. Two studies examined the effects of vitamin D supplementation 2000 IU per day orally [66] and 120,000 IU per month via intramuscular (IM) injections [67]. There were no statistically significant differences reported in the IM group [67] but arterial stiffness was reduced in the high dose oral supplementation group (5.41 ± 0.73 m/sec at baseline vs 5.33 ± 0.79 m/sec after 16 weeks, p = 0.031) [66]. Two studies examined the effects of metformin; 1 measuring CIMT (850 mg once daily for 24 months) [68] and 1 performing echocardiography to measure arterial elasticity and systemic vascular resistance (500 mg twice daily for 22 months) [69]. Both recruited obese young people and found that metformin increased weight loss and resulted in improvements in their chosen measure of vascular function, however, both also included dietary and physical activity interventions as part of the programme.
Combined lifestyle modification
Of the 10 studies assessing a combined lifestyle approach to improve vascular function, interventions ranged from 6 weeks to 1 year of length in children between the ages of 6–18 years (Table 2). Two studies reported no significant differences when combining dietary advice with exercise interventions [70]. One examined CIMT after 6 weeks of exercise and dietary advice and the other [71] examined FMD and PWV after 6 months of intervention [72]. Seven studies found that various combinations of diet and exercise advice reduced CIMT [73, 74] and arterial stiffness [75, 76] and increased endothelial function [16, 73, 77, 78]. None of these studies reported sex differences.
Evidence of sexual dimorphism
Of the included studies, 6292 (40%) of the participants were female with 70 studies (95%) including both boys and girls in their study design. However, only 6 (8%) of these studies provided data for girls and boys separately, enabling assessment of sexual dimorphism in response to the intervention [28, 30, 33, 35, 38, 59]. In terms of sex differences, baseline artery diameter was reported to be different between boys and girls [33]. and an increase in FMD after moderate to vigorous physical activity was only detected when the absolute FMD value was adjusted for baseline diameter at rest. One study of 427 children and adolescents (14.0 ± 1.4 years) found that physical activity resulted in different effects in boys and girls, with increased training duration associated with increased IMT with increased arterial compliance and reduced arterial stiffness in boys, whereas only arterial compliance was affected in girls [35]. Of note, baseline arterial diameter was higher in boys compared to girls [35]. Bond et al. reported that high intensity exercise increased FMD and reduced oxidative stress, as well as providing high rates of enjoyment of the intervention in both girls and boys. Girls also demonstrated reductions in postprandial lipaemia, although this was not seen in boys [28]. Endothelial function, as measured by reactive hyperaemia index was also reported to be increased in girls compared to boys, with further increases observed in those who undertook more physical activity [30]. IMT was reported to be increased in overweight and obese patients at baseline in a group of 212 adolescents in Bohm et al. [38], but not when adjusted for sex and age. The introduction of a 4-week hospital based intervention did not improve IMT in boys or girls [37].
Discussion
Interest in interventions to improve vascular structure and function in children and young people is growing, as evidenced by the number of studies investigating interventions to improve this in children. Given it is well established that early modification of cardiovascular risk factors improves cardiovascular outcomes later in life [8, 79], there is a public health need to study these further.
Meta-analysis was not possible within this systematic review due to differences in study methodology and methods of assessing vascular function. For example, there are many different ways to measure PWV [80], although evidence suggests different devices are likely to produce similar results [81]. To date carotid-femoral PWV has been shown to link to risk factors for CVD [82]. However, under a quarter of studies reporting arterial stiffness measured carotid-femoral PWV, with others using alternative methods such as carotid-radial PWV. As such, even available PWV data are difficult to compare.
In addition, there was a preponderance of studies focussing on obesity. This review did not consider interventions on vascular function in particular chronic health conditions, as it is likely that due to differing underlying mechanisms of disease, that the success of interventions may not represent the likelihood of success in healthy children. Studies assessing the effects on obese children were included, due to the high and rising prevalence of children being overweight internationally [83]. For any population-based initiative to be effective, it will have to improve vascular function in children with obesity, as well as normal weight children.
Our review suggests, however, that exercise may be a consistent method of improving endothelial function in children and adolescents. This is in agreement with other recent systematic reviews demonstrating that physical activity interventions produce improvements in body weight and physical fitness in preschool age children [84] and that high intensity exercise at any stage in the lifespan can improve cardiometabolic parameters [85]. Whilst the studies generally reported good adherence with the exercise interventions, most of the interventions were of <6 months duration. It is not clear whether adherence would continue with longer-term interventions. The techniques used in the included studies remain as research tools, rather than part of standard clinical practice. As such, it is not clear whether any improvements in vascular function are clinically translatable. The studies did not report having run in/washout periods and it is currently also not known how long it is likely that any changes in IMT, FMD, or PWV would persist after the interventions. This would be an increasing focus for future work. Further studies should, therefore, investigate the optimal mode, duration and intensity of exercise, with the realisation that this may vary at different stages of childhood. Based on the limited data available, however, we would suggest initiation of school based physical activity interventions of medium-high intensity at least three times per week for 60 min for optimal vascular function in both boys and girls throughout childhood and adolescence. Physical activity is likely to increase endothelial function, reduce arterial stiffness, increase arterial compliance and slow the progression of lipid accumulation in the carotid and aortic intima, delaying vascular ageing and mitigating cardiovascular risk [86], although it is not clear whether the underlying mechanisms for these changes may differ between boys and girls.
In our review, studies investigating endothelial function were more likely to detect a difference between groups. The effects of intervention were measured at variable time points, ranging from immediately afterwards to 15 years later. FMD has been shown to reduce within 30 min of exercise, whereas it remains unclear how long it might take for changes in arterial stiffness or vascular remodelling to be evident and as such, study design is important when comparing the effects of these interventions [87].
The fact that <10% of studies in children and young people report data from boys and girls separately is perhaps surprising. Reference data for blood pressure, PWV and CIMT are different for boys and girls [88,89,90]. It is also been reported that girls have higher FMD than boys [91]. Boys are reported to have higher baseline arterial diameters compared to girls and therefore reduced capacity to dilate [92, 93]. The effects of oestrogens and androgens on the vasculature have been studied widely, with the consensus that both can affect the vasculature and regulation of blood pressure significantly [3, 94]. It is therefore likely that these will influence vascular function as children progress through puberty into adulthood. It is recommended that sex-specific data are represented for all health conditions [95], as it was realised that historically women have been excluded from major clinical trials [96]. As a result, the sexes should be considered separately in any study, but particularly those including children from the age of adolescence onwards.
In conclusion, school-based physical activity interventions are most likely to result in improvements in endothelial function. Endothelial function may be the first variable of vascular function to change secondary to an intervention and therefore should be considered in studies looking to assess the vasculature in children. The number of studies which considered the effects of interventions on vascular function in girls and boys separately is low. Standardisation of reporting of differences between the sexes is essential to be able to ensure interventions are equally effective for boys and girls.
Summary
What is known about this topic
-
Vascular function is different in boys and girls.
-
There are several different methods of assessing vascular structure and function in children including pulse wave velocity, carotid intima media thickness and flow mediated dilatation.
-
It is not clear how to improve cardiovascular function in children.
What this study adds
-
School-based physical activity interventions are most likely to result in improvements in vascular function.
-
Endothelial function may be the first variable of vascular function to change secondary to an intervention.
-
Standardisation of reporting of differences between the sexes is essential to be able to ensure interventions are equally effective for boys and girls.
Data availability
Data is available upon reasonable request.
References
Mensah GA, Roth GA, Fuster V. The global burden of cardiovascular diseases and risk factors: 2020 and beyond. American College of Cardiology Foundation Washington, DC, 2019:2529–32.
Benschop L, Schalekamp‐Timmermans S, Roeters van Lennep JE, Jaddoe VW, Steegers EA, Ikram MK. Cardiovascular risk factors track from mother to child. J Am Heart Assoc. 2018;7:e009536.
Connelly PJ, Casey H, Montezano AC, Touyz RM, Delles C. Sex steroids receptors, hypertension, and vascular ageing. J Hum Hypertension. 2022;36:120–5.
Fong TS, Urbina EM, Howden EJ, Wallace I, Park C, Gall S, et al. Youth Vascular Consortium (YVC) protocol: establishing reference intervals for vascular ageing in children, adolescents and young adults. Heart Lung Circulation. 2021;30:1710–5.
Laitinen TT, Pahkala K, Magnussen CG, Viikari JS, Oikonen M, Taittonen L, et al. Ideal cardiovascular health in childhood and cardiometabolic outcomes in adulthood: the Cardiovascular Risk in Young Finns Study. Circulation. 2012;125:1971–8.
Pahkala K, Hietalampi H, Laitinen TT, Viikari JSA, Rönnemaa T, Niinikoski H, et al. Ideal cardiovascular health in adolescence. Circulation. 2013;127:2088–96.
Vedanthan R, Bansilal S, Soto AV, Kovacic JC, Latina J, Jaslow R, et al. Family-based approaches to cardiovascular health promotion. J Am Coll Cardiol. 2016;67:1725–37.
Kallerman PH. Cardiovascular Health in Children and Adolescents with Obesity: Prevalence, Prediction and Supervised Exercise. Karolinska Institutet (Sweden), 2019.
Phillips L, Lundholm C, Kvist U, Almqvist C, Nordenskjöld A, Nordenvall AS. Increased androgen-related comorbidity in adolescents and adults born with hypospadias: a population-based study. Andrology. 2022;10:1376–86.
Karki S, Son WM, Seo KE, Park SY. Combined exercise training reduces blood pressure, arterial stiffness, and insulin resistance in obese prehypertensive adolescent girls. FASEB J. 2017;31:1012.14–1012.14.
Wong A, Sanchez-Gonzalez MA, Son W-M, Kwak Y-S, Park S-Y. The effects of a 12-week combined exercise training program on arterial stiffness, vasoactive substances, inflammatory markers, metabolic profile, and body composition in obese adolescent girls. Pediatr Exerc Sci. 2018;30:480–6.
Ketelhut S, Ketelhut SR, Ketelhut K. School-based exercise intervention improves blood pressure and parameters of arterial stiffness in children: a randomized controlled trial. Pediatr Exerc Sci. 2020;33:1–7.
Chuensiri N, Suksom D, Tanaka H. Effects of high-intensity intermittent training on vascular function in obese preadolescent boys. Child Obes. 2018;14:41–49.
Park J-H, Miyashita M, Kwon Y-C, Park H-T, Kim E-H, Park J-K, et al. A 12-week after-school physical activity programme improves endothelial cell function in overweight and obese children: a randomised controlled study. BMC Pediatrics. 2012;12:1–9.
Meyer AA, Kundt G, Lenschow U, Schuff-Werner P, Kienast W. Improvement of early vascular changes and cardiovascular risk factors in obese children after a six-month exercise program. J Am Coll Cardiol. 2006;48:1865–70.
Farpour-Lambert NJ, Aggoun Y, Marchand LM, Martin XE, Herrmann FR, Beghetti M. Physical activity reduces systemic blood pressure and improves early markers of atherosclerosis in pre-pubertal obese children. J Am Coll Cardiol. 2009;54:2396–406.
Watts K, Beye P, Siafarikas A, Davis EA, Jones TW, O’Driscoll G, et al. Exercise training normalizes vascular dysfunction and improves central adiposity in obese adolescents. J Am Coll Cardiol. 2004;43:1823–7.
Watts K, Beye P, Siafarikas A, O’Driscoll G, Jones TW, Davis EA, et al. Effects of exercise training on vascular function in obese children. J Pediatrics. 2004;144:620–5.
Tjønna AE, Stølen TO, Bye A, Volden M, Slørdahl SA, Ødegård R, et al. Aerobic interval training reduces cardiovascular risk factors more than a multitreatment approach in overweight adolescents. Clin Sci. 2009;116:317–26.
Jebeile H, Gow ML, Lister NB, Mosalman Haghighi M, Ayer J, Cowell CT, et al. Intermittent energy restriction is a feasible, effective, and acceptable intervention to treat adolescents with obesity. J Nutr. 2019;149:1189–97.
Kelly AS, Wetzsteon RJ, Kaiser DR, Steinberger J, Bank AJ, Dengel DR. Inflammation, insulin, and endothelial function in overweight children and adolescents: the role of exercise. J Pediatrics. 2004;145:731–6.
Poeta LS, Duarte MDFDS, Caramelli B, Jorge M, Giuliano IDCB. Effects of physical exercises and nutritional guidance on the cardiovascular risk profile of obese children. Rev da Associacao Med Brasileira. 2013;59:56–63.
Hansen PR, Andersen LJ, Rebelo AN, Brito J, Hornstrup T, Schmidt JF, et al. Cardiovascular effects of 3 months of football training in overweight children examined by comprehensive echocardiography: a pilot study. J sports Sci. 2013;31:1432–40.
Starkoff BE. Children active to stay healthy (CASH): exercise as a tool for reducing inflammation and cardiovascular risk in sedentary, pubescent adolescents with obesity: the Ohio State University, 2013.
Davis CL, Litwin SE, Pollock NK, Waller JL, Zhu H, Dong Y, et al. Exercise effects on arterial stiffness and heart health in children with excess weight: The SMART RCT. Int J Obes. 2020;44:1152–63.
Nourse SE, Olson I, Popat RA, Stauffer KJ, Vu CN, Berry S, et al. Live video diet and exercise intervention in overweight and obese youth: adherence and cardiovascular health. J Pediatrics. 2015;167:533–9.e1
Sung K-D, Pekas EJ, Scott SD, Son W-M, Park S-Y. The effects of a 12-week jump rope exercise program on abdominal adiposity, vasoactive substances, inflammation, and vascular function in adolescent girls with prehypertension. Eur J Appl Physiol. 2019;119:577–85.
Bond B. The influence of exercise intensity on vascular health outcomes in adolescents. 2015. Available online at: https://ore.exeter.ac.uk/repository/handle/10871/18057?show=full.
Bond B, Hind S, Williams CA, Barker AR. The acute effect of exercise intensity on vascular function in adolescents. Med Sci Sports Exerc. 2015;47:2628–35.
Mueller UM, Walther C, Adam J, Fikenzer K, Erbs S, Mende M, et al. Endothelial function in children and adolescents is mainly influenced by age, sex and physical activity—an analysis of reactive hyperemic peripheral artery tonometry. Circulation J. 2017;81:717–25.
Yu CC-W, McManus AM, So H-K, Chook P, Au C-T, Li AM, et al. Effects of resistance training on cardiovascular health in non-obese active adolescents. World J Clin Pediatrics. 2016;5:293.
Murphy EC, Carson L, Neal W, Baylis C, Donley D, Yeater R. Effects of an exercise intervention using Dance Dance Revolution on endothelial function and other risk factors in overweight children. Int J Pediatr Obes. 2009;4:205–14.
Bacauanu J. The relationship between objectively measured habitual physical activity in preschoolers and peripheral artery endothelial function in school-aged children. 2019.
Sheridan S. Cardiorespiratory fitness, physical activity, sedentary behaviour and vascular health in male adolescents. Dublin City University, 2016.
Baumgartner L, Weberruß H, Engl T, Schulz T, Oberhoffer-Fritz R. Exercise training duration and intensity are associated with thicker carotid intima-media thickness but improved arterial elasticity in active children and adolescents. Front Cardiovasc Med. 2021;8:652.
Gobbi R. Cardiometabolic risk in 10 to 11 year old children: the impacts of physical activity, cardiorespiratory fitness, body composition and lifestyle education: Liverpool John Moores University (United Kingdom), 2012.
Böhm B. Risk screening and exercise interventions in pediatric prevention of atherosclerotic cardiovascular disease: Links between carotid intima-media thickness and physical fitness. Technische Universität München, 2012.
Böhm B, Reiner N, Oberhoffer R. Increased carotid intima-media thickness and impaired sport motor fitness in obese adolescents: What are the effects of a short-term multidisciplinary intervention? Increased carotid intima-media thickness impaired sport motor fitness obese adolescents: what are effects a short-term multidisciplinary intervention? 2012;23:23.
Lee S, Libman I, Hughan KS, Kuk JL, Barinas-Mitchell E, Chung H, et al. Effects of exercise modality on body composition and cardiovascular disease risk factors in adolescents with obesity: a randomized clinical trial. Appl Physiol Nutr Metab. 2020;45:1377–86.
Heiss KI. Kardiovaskuläre Risikofaktoren im Kindesalter-Effekte eines sechsmonatigen schulischen Interventionsprogramms auf Stoffwechsel-und Entzündungsparameter. Technische Universität München, 2018.
Horner K, Barinas-Mitchell E, DeGroff C, Kuk JL, Drant S, Lee S. Effect of aerobic versus resistance exercise on pulse wave velocity, intima media thickness and left ventricular mass in obese adolescents. Pediatr Exerc Sci. 2015;27:494–502.
Johnson SC. The effects of a psychosocial behavioural intervention on arterial health in children. 2013. Available online at: https://core.ac.uk/display/62645761.
Ketelhut S, Kircher E, Ketelhut SR, Wehlan E, Ketelhut K. Effectiveness of multi-activity, high-intensity interval training in school-aged children. Int J Sports Med. 2020;41:227–32.
Van Biljon A. Identification and modification of cardiometabolic disease risk factors in South African urban primary school children. University of Zululand, 2018.
Melo X, Fernhall B, Santos DA, Pinto R, Pimenta NM, Sardinha LB, et al. The acute effect of maximal exercise on central and peripheral arterial stiffness indices and hemodynamics in children and adults. Appl Physiol Nutr Metab. 2016;41:266–76.
Hacke C, Ketelhut S, Wendt U, Müller G, Schlesner C, Ketelhut K. Effectiveness of a physical activity intervention in preschoolers: A cluster‐randomized controlled trial. Scand J Med Sci Sports. 2019;29:742–52.
Hiisijärvi H. Neljän viikon HIIT-intervention vaikutus 16-19-vuotiaiden nuorten kestävyyskuntoon ja valtimojäykkyyteen. Itä-Suomen yliopisto, 2019.
Siegrist M, Hanssen H, Lammel C, Haller B, Koch A-M, Stemp P, et al. Effects of a cluster-randomized school-based prevention program on physical activity and microvascular function (JuvenTUM 3). Atherosclerosis. 2018;278:73–81.
Ayer JG, Harmer JA, Xuan W, Toelle B, Webb K, Almqvist C, et al. Dietary supplementation with n-3 polyunsaturated fatty acids in early childhood: effects on blood pressure and arterial structure and function at age 8 y. Am J Clin Nutr. 2009;90:438–46.
Arnberg K, Larnkjær A, Michaelsen KF, Jensen SM, Hoppe C, Mølgaard C. Casein improves brachial and central aortic diastolic blood pressure in overweight adolescents: a randomised, controlled trial. J Nutr Sci. 2013;2:e43.
Kapetanakis M, Liuba P, Odermarsky M, Lundgren J, Hallböök T. Effects of ketogenic diet on vascular function. Eur J Paediatr Neurol. 2014;18:489–94.
Gruszfeld D, Weber M, Nowakowska-Rysz M, Janas R, Kozlik-Feldmann R, Xhonneux A, et al. Protein intake in infancy and carotid intima media thickness at 5 years-a secondary analysis from a randomized trial. Ann Nutr Metab. 2015;66:51–9.
Kinra S, Gregson J, Prabhakaran P, Gupta V, Walia GK, Bhogadi S, et al. Effect of supplemental nutrition in pregnancy on offspring’s risk of cardiovascular disease in young adulthood: Long-term follow-up of a cluster trial from India. PLoS Med. 2020;17:e1003183.
Iannuzzi A, Licenziati MR, Vacca M, De Marco D, Cinquegrana G, Laccetti M, et al. Comparison of two diets of varying glycemic index on carotid subclinical atherosclerosis in obese children. Heart Vessels. 2009;24:419.
Montero D, Walther G, Perez-Martin A, Roche E, Vinet A. Endothelial dysfunction, inflammation and oxidative stress in obese children and adolescents: markers of lifestyle intervention. Obes Rev. 2012;13:441–55.
Couch SC, Saelens BE, Khoury PR, Dart KB, Hinn K, Mitsnefes MM, et al. Dietary approaches to stop hypertension dietary intervention improves blood pressure and vascular health in youth with elevated blood pressure. Hypertension. 2021;77:241–51.
Thopy AJ. Effects of the DASH diet on brachial artery flow mediated dilation in adolescents with pre-hypertension and hypertension. University of Cincinnati, 2011.
Raitakari OT, Rönnemaa T, Järvisalo MJ, Kaitosaari T, Volanen I, Kallio K, et al. Endothelial function in healthy 11-year-old children after dietary intervention with onset in infancy: the Special Turku Coronary Risk Factor Intervention Project for children (STRIP). Circulation. 2005;112:3786–94.
Niinikoski H, Pahkala K, Viikari J, Rönnemaa T, Jula A, Lagström H, et al. The STRIP study: long-term impact of a low saturated fat/low cholesterol diet. Curr Cardiovascular Risk Rep. 2014;8:410.
Tromba L, Perla FM, Carbotta G, Chiesa C, Pacifico L. Effect of alpha-lipoic acid supplementation on endothelial function and cardiovascular risk factors in overweight/obese youths: a double-blind, placebo-controlled randomized trial. Nutrients. 2019;11:375.
Hashemi M, Khosravi E, Ghannadi A, Hashemipour M, Kelishadi R. Effect of the peels of two Citrus fruits on endothelium function in adolescents with excess weight: A triple-masked randomized trial. J Res Med Sci. 2015;20:721–6.
Kelly AS, Metzig AM, Rudser KD, Fitch AK, Fox CK, Nathan BM, et al. Exenatide as a weight-loss therapy in extreme pediatric obesity: a randomized, controlled pilot study. Obes (Silver Spring). 2012;20:364–70.
Peña AS, Wiltshire E, Gent R, Piotto L, Hirte C, Couper J. Folic acid does not improve endothelial function in obese children and adolescents. Diabetes Care. 2007;30:2122–7.
Sabri M, Ghaffari G, Hashemipour M, Mostofizadeh N, Koushki AM. Effect of long-term Vitamin C intake on vascular endothelial function in diabetic children and adolescents: A pilot study. J Res Med Sci. 2016;21:119.
Yu CC, Li AM, Chan KO, Chook P, Kam JT, Au CT, et al. Orlistat improves endothelial function in obese adolescents: a randomised trial. J Paediatr Child Health. 2013;49:969–75.
Dong Y, Stallmann-Jorgensen IS, Pollock NK, Harris RA, Keeton D, Huang Y, et al. A 16-week randomized clinical trial of 2000 international units daily vitamin D3 supplementation in black youth: 25-hydroxyvitamin D, adiposity, and arterial stiffness. J Clin Endocrinol Metab. 2010;95:4584–91.
Varshney S, Khadgawat R, Gahlot M, Khandelwal D, Oberoi AK, Yadav RK, et al. Effect of high-dose vitamin D supplementation on beta cell function in obese Asian-Indian children and adolescents: a randomized, double blind, active controlled study. Indian J Endocrinol Metab. 2019;23:545–51.
Bassols J, Martínez-Calcerrada JM, Osiniri I, Díaz-Roldán F, Xargay-Torrent S, Mas-Parés B, et al. Effects of metformin administration on endocrine-metabolic parameters, visceral adiposity and cardiovascular risk factors in children with obesity and risk markers for metabolic syndrome: A pilot study. PLoS One. 2019;14:e0226303.
Ho M, Gow M, Baur LA, Benitez-Aguirre PZ, Tam CS, Donaghue KC, et al. Effect of fat loss on arterial elasticity in obese adolescents with clinical insulin resistance: RESIST study. J Clin Endocrinol Metab. 2014;99:E1846–53.
Farpour-Lambert NJ, Martin XE, Della Torre SB, von Haller L, Ells LJ, Herrmann FR, Yacine A. Effectiveness of individual and group programmes to treat obesity and reduce cardiovascular disease risk factors in pre-pubertal children. Clin Obes. 2019;9:12335.
Demschar M. Die Intima-Media-Thickness der Arteria carotis communis bei adipösen Kindern/Auswirkungen eines sechswöchigen stationären Adipositasinterventionsprogramms. 2015. Available online at: https://mediatum.ub.tum.de/1207828.
Abu-Kishk I, Alumot-Yehoshua M, Reisler G, Efrati S, Kozer E, Doenyas-Barak K, et al. Lifestyle modifications in an adolescent dormitory: a clinical trial. Korean J Pediatr. 2014;57:520–5.
Woo KS, Chook P, Yu CW, Sung RY, Qiao M, Leung SS, et al. Effects of diet and exercise on obesity-related vascular dysfunction in children. Circulation. 2004;109:1981–6.
Genoni G, Menegon V, Monzani A, Archero F, Tagliaferri F, Mancioppi V, et al. Healthy lifestyle intervention and weight loss improve cardiovascular dysfunction in children with obesity. Nutrients. 2021;13:1301.
Khadilkar VV, Pandit DS, Khadilkar AV, Chiplonkar SA, Kinare AS. Diet and exercise intervention, with special reference to micronutrients, reduces cardiometabolic risk in overweight children. Indian J Endocrinol Metab. 2012;16:124–33.
Santiprabhob J, Limprayoon K, Aanpreung P, Charoensakdi R, Kalpravidh RW, Phonrat B, et al. Impact of a group-based treatment program on adipocytokines, oxidative status, inflammatory cytokines and arterial stiffness in obese children and adolescents. J Pediatr Endocrinol Metab. 2018;31:733–42.
Donghui T, Shuang B, Xulong L, Meng Y, Yujing G, Yujie H, et al. Improvement of microvascular endothelial dysfunction induced by exercise and diet is associated with microRNA-126 in obese adolescents. Microvascular Res. 2019;123:86–91.
Bruyndonckx L, Hoymans VY, De Guchtenaere A, Van Helvoirt M, Van Craenenbroeck EM, Frederix G, et al. Diet, exercise, and endothelial function in obese adolescents. Pediatrics. 2015;135:e653–61.
McCrindle BW. Cardiovascular consequences of childhood obesity. Can J Cardiol. 2015;31:124–30.
Segers P, Rietzschel ER, Chirinos JA. How to measure arterial stiffness in humans. Arteriosclerosis, Thrombosis, Vasc Biol. 2020;40:1034–43.
Kis E, Cseprekál O, Kerti A, Salvi P, Benetos A, Tisler A, et al. Measurement of pulse wave velocity in children and young adults: a comparative study using three different devices. Hypertension Res. 2011;34:1197–202.
Tillin T, Chambers J, Malik I, Coady E, Byrd S, Mayet J, et al. Measurement of pulse wave velocity: site matters. J Hypertens. 2007;25:383–9.
Garrido-Miguel M, Cavero-Redondo I, Álvarez-Bueno C, Rodríguez-Artalejo F, Moreno LA, Ruiz JR, et al. Prevalence and trends of overweight and obesity in European children from 1999 to 2016: a systematic review and meta-analysis. JAMA Pediatrics. 2019;173:e192430–e192430.
García-Hermoso A, Alonso-Martinez AM, Ramírez-Vélez R, Izquierdo M. Effects of exercise intervention on health-related physical fitness and blood pressure in preschool children: a systematic review and meta-analysis of randomized controlled trials. Sports Med. 2020;50:187–203.
Martland R, Mondelli V, Gaughran F, Stubbs B. Can high-intensity interval training improve physical and mental health outcomes? A meta-review of 33 systematic reviews across the lifespan. J Sports Sci. 2020;38:430–69.
Climie RE, Park C, Avolio A, Mynard JP, Kruger R, Bruno R-M. Vascular ageing in youth: a call to action. Heart Lung Circ. 2021;30:1613–26.
Dawson EA, Green DJ, Timothy Cable N, Thijssen DHJ. Effects of acute exercise on flow-mediated dilatation in healthy humans. J Appl Physiol. 2013;115:1589–98.
Diaz A, Bia D, Zócalo Y, Manterola H, Larrabide I, Lo Vercio L, et al. Carotid intima media thickness reference intervals for a healthy Argentinean population aged 11–81 years. Int J Hypertens. 2018;2018:2018:8086714
Reusz GS, Cseprekal O, Temmar M, Kis E, Cherif AB, Thaleb A, et al. Reference values of pulse wave velocity in healthy children and teenagers. Hypertension. 2010;56:217–24.
Flynn JT, Kaelber DC, Baker-Smith CM, Blowey D, Carroll AE, Daniels SR, et al. Clinical practice guideline for screening and management of high blood pressure in children and adolescents. Pediatrics. 2017;140:2017904.
Hopkins ND, Dengel DR, Stratton G, Kelly AS, Steinberger J, Zavala H, et al. Age and sex relationship with flow-mediated dilation in healthy children and adolescents. J Appl Physiol. 2015;119:926–33.
Atkinson G, Batterham AM. Allometric scaling of diameter change in the original flow-mediated dilation protocol. Atherosclerosis. 2013;226:425–7.
Celermajer DS, Sorensen KE, Gooch VM, Spiegelhalter DJ, Miller OI, Sullivan ID, et al. Non-invasive detection of endothelial dysfunction in children and adults at risk of atherosclerosis. Lancet. 1992;340:1111–5.
Reckelhoff JF. Sex differences in regulation of blood pressure. Sex Specif Anal Cardiovasc Funct. 2018;1065:139–51.
Clayton JA, Collins FS. Policy: NIH to balance sex in cell and animal studies. Nature. 2014;509:282–3.
Steinberg JR, Turner BE, Weeks BT, Magnani CJ, Wong BO, Rodriguez F, et al. Analysis of female enrollment and participant sex by burden of disease in US clinical trials between 2000 and 2020. JAMA Netw Open. 2021;4:e2113749–e2113749.
Funding
ALH is funded by an NES/CSO Clinical Lectureship.
Author information
Authors and Affiliations
Contributions
The authors confirm contribution to the paper as follows: study conception and design: SFA and ALH; literature search and data collection: SE and MF; quality assessment: SE and MF; manuscript drafting and editing: SE, MF, SFA, ALH. All authors reviewed the results and approved the final version of the manuscript.
Corresponding author
Ethics declarations
Competing interests
The authors declare no competing interests.
Additional information
Publisher’s note Springer Nature remains neutral with regard to jurisdictional claims in published maps and institutional affiliations.
Rights and permissions
Open Access This article is licensed under a Creative Commons Attribution 4.0 International License, which permits use, sharing, adaptation, distribution and reproduction in any medium or format, as long as you give appropriate credit to the original author(s) and the source, provide a link to the Creative Commons license, and indicate if changes were made. The images or other third party material in this article are included in the article’s Creative Commons license, unless indicated otherwise in a credit line to the material. If material is not included in the article’s Creative Commons license and your intended use is not permitted by statutory regulation or exceeds the permitted use, you will need to obtain permission directly from the copyright holder. To view a copy of this license, visit http://creativecommons.org/licenses/by/4.0/.
About this article
Cite this article
Edwards, S., Foster, M., Ahmed, S.F. et al. Preventative interventions that target cardiovascular dysfunction in children and young people: a systematic review of their effectiveness and an investigation of sexual dimorphism. J Hum Hypertens 37, 726–734 (2023). https://doi.org/10.1038/s41371-022-00780-z
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/s41371-022-00780-z
This article is cited by
-
Journal of Human Hypertension special issue on sex and gender differences in hypertension
Journal of Human Hypertension (2023)