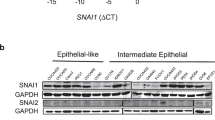
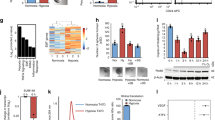
Abstract
The epithelial-to-mesenchymal transition (EMT) is a cellular process that functions during embryonic development and tissue regeneration, thought to be aberrantly activated in epithelial-derived cancer and has an important role in the process of metastasis. The transforming growth factor (TGF)-β signaling pathway is a key inducer of EMT and we have elucidated a posttranscriptional mechanism by which TGFβ modulates expression of select transcripts via the RNA-binding protein hnRNP E1 during EMT. One such transcript inhibin βA is a member of the TGFβ superfamily. Here, we show by polysome profiling that inhibin βA is translationally regulated by TGFβ via hnRNP E1. TGFβ treatment or knockdown of hnRNP E1 relieves silencing of the inhibin βA transcript, resulting in increased protein expression and secreted levels of the inhibin βA homodimer, activin A. Our data indicate that the translational upregulation of inhibin βA enhances the migration and invasion of cells that have undergone an EMT and promotes cancer progression in vivo.
This is a preview of subscription content, access via your institution
Access options
Subscribe to this journal
Receive 50 print issues and online access
$259.00 per year
only $5.18 per issue
Buy this article
- Purchase on Springer Link
- Instant access to full article PDF
Prices may be subject to local taxes which are calculated during checkout






Similar content being viewed by others
References
Radisky DC . Epithelial-mesenchymal transition. J Cell Sci 2005; 118: 4325–4326.
Polyak K, Weinberg RA . Transitions between epithelial and mesenchymal states: acquisition of malignant and stem cell traits. Nat Rev Cancer 2009; 9: 265–273.
Massagué J, Blain SW, Lo RS . TGFβ signaling in growth control, cancer, and heritable disorders. Cell 2000; 103: 295–309.
Scheel C, Eaton EN, Li SH, Chaffer CL, Reinhardt F, Kah KJ et al. Paracrine and autocrine signals induce and maintain mesenchymal and stem cell states in the breast. Cell 2011; 145: 926–940.
Thiery JP, Acloque H, Huang RY, Nieto MA . Epithelial-mesenchymal transitions in development and disease. Cell 2009; 139: 871–890.
Chaudhury A, Hussey GS, Ray PS, Jin G, Fox PL, Howe PH . TGF-[beta]-mediated phosphorylation of hnRNP E1 induces EMT via transcript-selective translational induction of Dab2 and ILEI. Nat Cell Biol 2010; 12: 286–293.
Hussey GS, Chaudhury A, Dawson AE, Lindner DJ, Knudsen CR, Wilce MC et al. Identification of an mRNP complex regulating tumorigenesis at the translational elongation step. Mol Cell 2011; 41: 419–431.
Hussey GS, Link LA, Brown AS, Howley BV, Chaudhury A, Howe PH . Establishment of a TGFβ-induced post-transcriptional EMT gene signature. PLoS One 2012; 7: e52624.
Li R, Phillips DM, Mather JP . Activin promotes ovarian follicle development in vitro. Endocrinology 1995; 136: 849–856.
Matzuk MM, Kumar TR, Vassalli A, Bickenbach JR, Roop DR, Jaenisch R et al. Functional analysis of activins during mammalian development. Nature 1995; 374: 354–357.
Ogawa Y, Schmidt DK, Nathan RM, Armstrong RM, Miller KL, Sawamura SJ et al. Bovine bone activin enhances bone morphogenetic protein-induced ectopic bone formation. J Biol Chem 1992; 267: 14233–14237.
Pearson S, Sroczynska P, Lacaud G, Kouskoff V . The stepwise specification of embryonic stem cells to hematopoietic fate is driven by sequential exposure to Bmp4, activin A, bFGF and VEGF. Development 2008; 135: 1525–1535.
Schmierer B, Hill CS . TGF[beta]-SMAD signal transduction: molecular specificity and functional flexibility. Nat Rev Mol Cell Biol 2007; 8: 970–982.
Le Bras GF, Loomans HA, Taylor CJ, Revetta FL, Andl CD . Activin A balance regulates epithelial invasiveness and tumorigenesis. Lab Invest 2014; 94: 1134–1146.
Burdette JE, Jeruss JS, Kurley SJ, Lee EJ, Woodruff TK . Activin A mediates growth inhibition and cell cycle arrest through smads in human breast cancer cells. Cancer Res 2005; 65: 7968–7975.
Lonardo E, Hermann PC, Mueller MT, Huber S, Balic A, Miranda-Lorenzo I et al. Nodal/activin signaling drives self-renewal and tumorigenicity of pancreatic cancer stem cells and provides a target for combined drug therapy. Cell Stem Cell 2011; 9: 433–446.
Reis FM, Cobellis L, Tameirão LC, Anania G, Luisi S, Silva ISB et al. Serum and tissue expression of activin A in postmenopausal women with breast cancer. J Clin Endocrinol Metab 2002; 87: 2277–2282.
Wildi S, Kleeff J, Maruyama H, Maurer CA, Büchler MW, Korc M . Overexpression of activin A in stage IV colorectal cancer. Gut 2001; 49: 409–417.
Wang Q, Wen YG, Li DP, Xia J, Zhou CZ, Yan DW et al. Upregulated INHBA expression is associated with poor survival in gastric cancer. Med Oncol 2012; 29: 77–83.
Leto G, Incorvaia L, Badalamenti G, Tumminello F, Gebbia N, Flandina C et al. Activin A circulating levels in patients with bone metastasis from breast or prostate cancer. Clin Exp Metastasis 2006; 23: 117–122.
Kang HY, Huang HY, Hsieh CY, Li CF, Shyr CR, Tsai MY et al. Activin A enhances prostate cancer cell migration through activation of androgen receptor and is overexpressed in metastatic prostate cancer. J Bone Miner Res 2009; 24: 1180–1193.
Kim H, Watkinson J, Varadan V, Anastassiou D . Multi-cancer computational analysis reveals invasion-associated variant of desmoplastic reaction involving INHBA, THBS2 and COL11A1. BMC Med Genomics 2010; 3: 51.
Antenos M, Stemler M, Boime I, Woodruff TK . N-linked oligosaccharides direct the differential assembly and secretion of inhibin α- and βA-subunit dimers. Mol Endocrinol 2007; 21: 1670–1684.
Piek E, Moustakas A, Kurisaki A, Heldin CH, ten Dijke P . TGF-(beta) type I receptor/ALK-5 and Smad proteins mediate epithelial to mesenchymal transdifferentiation in NMuMG breast epithelial cells. J Cell Sci 1999; 112: 4557–4568.
Gunasinghe NP, Wells A, Thompson EW, Hugo HJ . Mesenchymal–epithelial transition (MET) as a mechanism for metastatic colonisation in breast cancer. Cancer Metastasis Rev 2012; 31: 469–478.
Arai KY, Ono M, Kudo C, Fujioka A, Okamura R, Nomura Y et al. IL-1β stimulates activin βA mRNA expression in human skin fibroblasts through the MAPK pathways, the nuclear factor-κB pathway, and prostaglandin E2. Endocrinology 2011; 152: 3779–3790.
Wamsley JJ, Kumar M, Allison DF, Clift SH, Holzknecht CM, Szymura SJ et al. Activin upregulation by NF-κB is required to maintain mesenchymal features of cancer stem-like cells in non-small cell lung cancer. Cancer Res 2014; 75: 426–435.
Valcourt U, Kowanetz M, Niimi H, Heldin C-H, Moustakas A . TGF-β and the smad signaling pathway support transcriptomic reprogramming during epithelial-mesenchymal cell transition. Mol Biol Cell 2005; 16: 1987–2002.
Lee KL, Lim SK, Orlov YL, Yit LY, Yang H, Ang LT et al. Graded nodal/activin signaling titrates conversion of quantitative phospho-smad2 levels into qualitative embryonic stem cell fate decisions. PLoS Genet 2011; 7: e1002130.
Kim ES, Kim MS, Moon A . Transforming growth factor (TGF)-β in conjunction with H-ras activation promotes malignant progression of MCF10A breast epithelial cells. Cytokine 2005; 29: 84–91.
Seder CW, Hartojo W, Lin L, Silvers AL, Wang Z, Thomas DG et al. Upregulated INHBA expression may promote cell proliferation and is associated with poor survival in lung adenocarcinoma. Neoplasia 2009; 11: 388–396.
Walia V, Elble RC . Enrichment for breast cancer cells with stem/progenitor properties by differential adhesion. Stem Cells Dev 2010; 19: 1175–1182.
Acknowledgements
This work was supported by Grants CA055536 and CA154663 from the National Cancer Institute. BVH was supported by funding from the Abney Foundation. GSH was supported by an American Heart Association Pre-doctoral Fellowship 10PRE3870024. This study used the services of the MUSC Center for Oral Health Research (COHR), which is partially supported by the National Institute of General Medicine grant P30GM103331, the MUSC Flow Cytometry Facility which is supported by P30GM103342, the Cell & Molecular Imaging Shared Resource of MUSC supported by P30CA138313, the Biorepository & Tissue Analysis Shared Resource, Hollings Cancer Center, MUSC and the Gene Targeting and Knockout Shared Resource at MUSC. We would like to thank Maya El Sabban for assisting us with xenograft experiments.
Author information
Authors and Affiliations
Corresponding author
Ethics declarations
Competing interests
The authors declare no conflict of interest.
Additional information
Supplementary Information accompanies this paper on the Oncogene website
Supplementary information
Rights and permissions
About this article
Cite this article
Howley, B., Hussey, G., Link, L. et al. Translational regulation of inhibin βA by TGFβ via the RNA-binding protein hnRNP E1 enhances the invasiveness of epithelial-to-mesenchymal transitioned cells. Oncogene 35, 1725–1735 (2016). https://doi.org/10.1038/onc.2015.238
Received:
Revised:
Accepted:
Published:
Issue Date:
DOI: https://doi.org/10.1038/onc.2015.238
This article is cited by
-
The ubiquitin E3 ligase ARIH1 regulates hnRNP E1 protein stability, EMT and breast cancer progression
Oncogene (2022)
-
TGFβ promotes breast cancer stem cell self-renewal through an ILEI/LIFR signaling axis
Oncogene (2019)
-
A CREB3-regulated ER–Golgi trafficking signature promotes metastatic progression in breast cancer
Oncogene (2018)
-
Mutant HSPB1 causes loss of translational repression by binding to PCBP1, an RNA binding protein with a possible role in neurodegenerative disease
Acta Neuropathologica Communications (2017)
-
Activin signaling is an essential component of the TGF-β induced pro-metastatic phenotype in colorectal cancer
Scientific Reports (2017)